Abstract
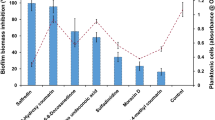
Candida albicans is considered one of the most important opportunistic fungi due to the large arsenal of virulence factors that help throughout the progress of the infection. In this sense, antimicrobial peptides (AMPs) appear as an alternative, with great antifungal action. Among these, aurein 1.2 has been widely explored, becoming the basis for the discovery of new AMPs, such as K-aurein (K-au). Thus, this study evaluated the anti-C. albicans potential of K-au against virulence factors, planktonic growth, and biofilm formation of clinical isolates. Firstly, K-au antifungal activity was determined by the microdilution method and time-kill curve. The inhibition of hydrolytic enzyme secretion (proteinase, phospholipase, and hemolysin) and germ tube formation was tested. Then, the antibiofilm potential of K-au was verified through biomass quantification and scanning electron microscopy (SEM). All tests were compared with the classical antifungal drug, amphotericin B (AmB). The outcomes showed fungicidal action of K-au at 62.50 µg mL−1 for all isolates, with a time of action around 150–180 min, determined by the time-kill curve. K-au-treated cells decreased by around 40% of the germinative tube compared to the control. Additionally, K-au inhibited the biofilm formation by more than 90% compared to AmB and the control group. SEM images show apparent cellular disaggregation without the formation of filamentous structures. Therefore, the findings suggest a promising anti-C. albicans effect of K-au due to its fungicidal activity against planktonic cells, or its ability to inhibit important virulence factors like germ tube and biofilm formation. Thus, this peptide could be explored as a useful compound against C. albicans-related infection.





Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the article or its supplementary materials.
References
Alder-Moore J, Lewis RE, Brüggemann RJM, Rijnders BJA, Groll AH, Walsh T (2019) Preclinical safety, tolerability, pharmacokinetics, pharmacodynamics, and antifungal activity of liposomal amphotericin B. Clin Infect Dis 68:s244–s259. https://doi.org/10.1093/2Fcid/2Fciz064
Boparai JK, Sharma PK (2020) Mini review on antimicrobial peptides, sources, mechanism and recent applications. Protein Pept Lett 27:4–16. https://doi.org/10.2174/0929866526666190822165812
Braunsdorf C, Leibundgut-Landmann S (2018) Modulation of the fungal-host interaction by the intra-species diversity of C. albicans. Pathogens 7:1–12. https://doi.org/10.3390/2Fpathogens7010011
Brazilian Committee on Antimicrobial Susceptibility Testing, BrCAST. Cut-off tables for the interpretation of antifungal MICs. Version 10.0. February 2020.
Canela HM, Cardoso B, Vitali LH, Coelho HC, Martinez R, Ferreira ME (2018) Prevalence, virulence factors and antifungal susceptibility of Candida spp. isolated from bloodstream infections in a tertiary care hospital in Brazil. Mycoses 61:11–21. https://doi.org/10.1111/myc.12695
Cardoso P, Glossop H, Meikle TG, Aburto-Medina A, Conn CE, Sarojini V, Valery C (2021) Molecular engineering of antimicrobial peptides: microbial targets, peptide motifs and translation opportunities. Biophys Rev 13:35–69. https://doi.org/10.1007/s12551-021-00784-y
Carneiro VA, Oliveira ST, Silva RL, Duarte HS, Silva ML, Matos MNC, Cavalcante RMB, Figueira CS, Lorenzón EN, Cilli EM, Cunha RMS (2020) Antimicrobial and antibiofilm activity of Lys-[Trp6]hy-a1 combined with ciprofloxacin against Gram-negative bacteria. Protein Pept Lett 27:1–8. https://doi.org/10.2174/0929866527666200416145549
Clinical and Laboratory Standards Institute (CLSI). References methods for broth dilution antifungal susceptibility testing of yeasts; Approved standard—Fourth Edition. CLSI document M27-A3. Wayne, PA. 2017.
Costa NCS, Piccoli JP, Santos-Filho NA, Clementino LC, Fusco-Almeida AM, De Annunzio SR et al (2020) Antimicrobial activity of RP-1 peptide conjugate with ferrocene group. PLoS One 15(3):e0228740. https://doi.org/10.1371/journal.pone.0228740
Crusca E Jr, Rezende AA, Marchetto R, Mendes-Giannini MJ, Fontes W, Castro MS, Cilli EM (2011) Influence of N-terminus modifications on the biological activity, membrane interaction, and secondary structure of the antimicrobial peptide hylin-a1. Biopolymers 96:41–48. https://doi.org/10.1002/bip.21454
Dadar M, Tiwari R, Karthik K, Chakraborty S, Shahali Y, Dhama K (2018) Candida albicans—biology, molecular characterization, pathogenicity, and advances in diagnosis and control—an update. Microb Pathog 117:128–138. https://doi.org/10.1016/j.micpath.2018.02.028
Deslouches S, Di PY (2017) Antimicrobial peptides: a potential therapeutic option for surgical site infections. Clin Surg 1740:1–8
Erbani SN, Madanchi H, Far HA, Rostamian M, Rahmati S, Shabani AA (2021) First report of antifungal activity of CecropinA-Magenin2 (CE-MA) hybrid peptide and its truncated derivatives. Biochem Biophys Res Commun 549:157–163. https://doi.org/10.1016/j.bbrc.2021.02.106
Fernandez DI, Sani M, Miles AJ, Wallace BA, Separovic F (2013) Membrane defects enhance the interaction of antimicrobial peptides, aurein 1.2 versus caerin 1.1. Biochim Biophys Acta 1828:1863–1872. https://doi.org/10.1016/j.bbamem.2013.03.010
Freitas LM, Lorenzón EN, Santos-Filho NA, Zago LHP, Uliana MP, Oliveira KT, Cilli EM, Fontana CR (2018) Antimicrobial photodynamic therapy enhanced by the peptide aurein 1.2. Sci Rep 8:1–15. https://doi.org/10.1038/s41598-018-22687-x
Galdiero E, de Alteriis E, de Natale A, D’Alterio A, Siciliano A, Guida M, Lombardi L, Falanga A, Galdiero S (2020) Eradication of Candida albicans persister cell bioflm by the membranotropic peptide gH625. Sci Rep 10:1–12. https://doi.org/10.1038/2Fs41598-020-62746-w
Graham CE, Cruz MR, Garsin DA, Lorenz MC (2017) Enterococcus faecalis bacteriocin EntV inhibits hyphal morphogenesis, biofilm formation, and virulence of Candida albicans. PNAS 114:4507–4512. https://doi.org/10.1073/pnas.1620432114
Guirao-Abad JP, González-Párraga P, Argüelles J (2015) Strong correlation between the antifungal effect of amphotericin B and its inhibitory action on germ-tube formation in a Candida albicans URA+ strain. Int Microbiol 18:25–31. https://doi.org/10.2436/20.1501.01.231
Hacioglu M, Tan ASB, Dosler S, Inan N, Otuk G (2018) In vitro activities of antifungals alone and in combination with tigecycline against Candida albicans biofilms. PeerJ 3:1–17. https://doi.org/10.7717/peerj.5263
Henriques M, Willians D (2020) Pathogenesis and Virulence of Candida albicans and Candida glabrata. Pathogens 9:1–3. https://doi.org/10.3390/2Fpathogens9090752
Kornitzer D (2019) Regulation of Candida albicans hyphal morphogenesis by endogenous signals. J Fungi 5:1–15. https://doi.org/10.3390/jof5010021
Kumar A, Jah A (2017) Chapter 7 - Drug Development Strategies. Anticandidal agents. Academic Press, Chhattisgarh, pp 63–71. https://doi.org/10.1016/B978-0-12-811311-0.00007-7
Lemos JA, Costa CR, De Araújo CR, Sousa LKH, Silva MRR (2009) Susceptibility testing of Candida albicans isolated from oropharyngeal mucosa of HIV+ patients to fluconazole, amphotericin b and caspofungin. Killing kinetics of caspofungin and amphotericin b against fluconazole resistant and susceptible isolates. Braz J Microbiol 40:163–168. https://doi.org/10.1590/S1517-838220090001000028
Li WS, Chen YC, Kuo SF, Chen FJ, Lee CH (2018) The impact of biofilm formation on the persistence of candidemia. Front Microbiol 4:1–10. https://doi.org/10.3389/fmicb.2018.01196
Lorenzón EN, Sanches PRS, Nogueira LG, Bauab TM, Cilli EM (2013) Dimerization of aurein 1.2: effects in structure, antimicrobial activity and aggregation of Candida albicans cells. Amino Acids 44:1521–1528. https://doi.org/10.1007/s00726-013-1475-3
Lorenzón EN, Riske KA, Troiano GF, Da Hora GCA, Soares TA, Cilli EM (2016) Effect of dimerization on the mechanism of action of aurein 1.2. Biochim Biophys Acta Biomembr 1858:1129–1138. https://doi.org/10.1016/j.bbamem.2016.02.010
Loshe MB, Gulati M, Johnson A, Nobile CJ (2018) Development and regulation of single- and multi-species Candida albicans biofilms. Nat Rev Microbiol 16:19–31. https://doi.org/10.1038/nrmicro.2017.107
Madanchi H, Khalaj V, Jang S, Shabani AA, Kiasari RE, Mousavi SJS, Sealani SK, Sardari S (2019) AurH1: a new heptapeptide derived from Aurein1.2 antimicrobial peptide with specific and exclusive fungicidal activity. J Pept Sci 25:1–9. https://doi.org/10.1002/psc.3175
Mahlapuu M, Björn C, Ekblom J (2020) Antimicrobial peptides as therapeutic agents: opportunities and challenges. Crit Rev Biotechnol 40:978–992. https://doi.org/10.1080/07388551.2020.1796576
Mayer FL, Wilson D, Hube B (2013) Candida albicans pathogenicity mechanisms. Virulence 4:119–128. https://doi.org/10.4161/2Fviru.22913
Mól AR, Castro MS, Fontes W (2018) NetWheels: a web application to create high quality peptide helical wheel and net projections. bioRxiv 416347:1–8. https://doi.org/10.1101/416347
Nascimento Dias J, De Souza Silva C, De Araújo AR, Souza JMT, De Holanda Veloso Júnior PH, Cabral WF, Da Glória da Silva M, Eaton P, De Souzade Almei da Leite JR, Nicola AM, Albuquerque P, Silva-Pereira I (2020) Mechanisms of action of antimicrobial peptides ToAP2 and NDBP-5.7 against Candida albicans planktonic and biofilm cells. Sci Rep 10:1–14. https://doi.org/10.1038/s41598-020-67041-2
O’toole GA, Kolter R (1998) Initiation of biofilm formation in Pseudomonas fluorescens WCS365 proceeds via multiple, convergente signalling pathways: a genetic analysis. Mol Microbiol 28:449–461. https://doi.org/10.1046/j.1365-2958.1998.00797.x
Oliveira VMC, Santos SSF, Silva CRG, Jorge AOC, Leão MVP (2016) Lactobacillus is able to alter the virulence and the sensitivity profile of Candida albicans. J Appl Microbiol 121:1737–1744. https://doi.org/10.1111/jam.13289
Pellon A, Nasab SDS, Moyes DL (2020) New insights in Candida albicans innate immunity at the mucosa: toxins, epithelium, metabolism, and beyond. Front Cell Infect Microbiol 10:1–14. https://doi.org/10.3389/fcimb.2020.00081
Pereira R, dos Santos Fontenelle RO, de Brito EHS, de Morais SM (2021) Biofilm of Candida albicans: formation, regulation and resistance. J Appl Microbiol 131:11–22. https://doi.org/10.1111/jam.14949
Pinalto KM, Alspaugh JA (2016) New horizons in antifungal therapy. J Fungi 2:1–24. https://doi.org/10.3390/jof2040026
Ponde NO, Lortal L, Ramage G, Naglik JR, Richardson JP (2021) Candida albicans biofilms and polymicrobial interactions. Crit Rev Microbiol 47:91–111. https://doi.org/10.1080/1040841x.2020.1843400
Poulain D (2013) Candida albicans, plasticity and pathogenesis. Crit Rev Microbiol 41:208–217. https://doi.org/10.3109/1040841x.2013.813904
Price MF, Wilkinson ID, Gentry LO (1982) Plate method for detection of phospholipase activity in Candida albicans. Sabouraudia 20(1):7–14. https://doi.org/10.1080/00362178285380031
Qian S, Zolnierczuk PA (2022) Interaction of a short antimicrobial peptide on charged lipid bilayer: a case study on aurein 1.2 peptide. BBA Adv 2:100045. https://doi.org/10.1016/j.bbadva.2022.100045
Rai DK, Quian S (2017) Interaction of the antimicrobial peptide aurein 1.2 and charged lipid bilayer. Sci Rep 7:1–10. https://doi.org/10.1038/s41598-017-03795-6
Ramezanzadeh M, Saeedi N, Mesbahfar E, Farrokh P, Salimi F, Ramezanzadeh M, Saeedi N, Mesbahfar E, Farrokh P, Salimi F, Rezaei A (2021) Design and characterization of new antimicrobial peptides derived from aurein 1.2 with enhanced antibacterial activity. Biochime 181:42–51. https://doi.org/10.1016/j.biochi.2020.11.020
Rodriguez DL, Quail MM, Hernday AD, Nobile CJ (2020) Transcriptional circuits regulating developmental processes in Candida albicans. Front Cell Infect Microbiol 10:1–20. https://doi.org/10.3389/fcimb.2020.605711
Rozek T, Wegener KL, Bowie JH, Olver IN, Carver JA, Wallace JC, Tyler MJ (2000) The antibiotic and anticancer active aurein peptides from the Australian Bell Frogs Litoria aurea and Litoria raniformis the solution structure of aurein 1.2. Eur J Biochem 267:5330–5341. https://doi.org/10.1046/j.1432-1327.2000.01536.x
Sajjadiyan Z, Cheraghi N, Mohammadinejad S, Hassani L (2017) Interaction of aurein 1.2 and its analogue with DPPC lipid bilayer. J Biol Phys 43:127–137. https://doi.org/10.1007/s10867-016-9438-z
Sanches PRS, Carneiro BM, Batista MN, Braga ACS, Lorenzón EN, Rahal P, Cilli EM (2015) A conjugate of the lytic peptide Hecate and gallic acid: structure, activity against cervical cancer, and toxicity. Amino Acids 47:1433–1443. https://doi.org/10.1007/s00726-015-1980-7
Santos R, Costa C, Mil-Homens D, Romão D, De Carvalho CCCR, Pais P, Mira NP, Fialho AM, Teixeira MC (2016) The multidrug resistance transporters CgTpo1_1 and CgTpo1_2 play a role in virulence and biofilm formation in the human pathogen Candida glabrata. Cell Microbiol 19:1–43. https://doi.org/10.1111/cmi.12686
Santos-Filho NA, Righetto GM, Pereira MR, Piccoli JP, Almeida LMT, Leal TC, Camargo ILBC, Cilli EM (2022) Effect of C-terminal and N-terminal dimerization and alanine scanning on antibacterial activity of the analogs of the peptide p-BthTX-I. Pept Sci 114:e24243. https://doi.org/10.1002/pep2.24243
Shahmiri M, Enciso M, Mechler A (2015) Controls and constrains of the membrane disrupting action of aurein 1.2. Sci Rep 5:1–14. https://doi.org/10.1038/srep16378
Shareck J, Belhumeur P (2011) Modulation of morphogenesis in Candida albicans by various small molecules. Eukaryot Cell 10:1004–1012. https://doi.org/10.1128/2FEC.05030-11
Silva ACB, de Cassia Orlandi Sardi J, de Oliveira DGL, de Oliveira CFR, dos Santos HF, dos Santos EL, Crusca E Jr, Cardoso MH, Franco LO, Macedo (2020) Development of a novel anti-biofilm peptide derived from profilin of Spodoptera frugiperda. Biofouling 36:516–527. https://doi.org/10.1080/08927014.2020.1776857
Strandberg E, Bentz D, Wadhwani P, Bürck J, Ulrich AS (2020) Terminal charges modulate the pore forming activity of cationic amphipathic helices. Biochim Biophys Acta Biomembr 1862:1–10. https://doi.org/10.1016/j.bbamem.2020.183243
Struyfs C, Cammue BPA, Thevissen K (2021) Membrane-interacting antifungal peptides. Front Cell Dev Biol 9:1–17. https://doi.org/10.3389/fcell.2021.649875
Szerencsés B, Gácser A, Endre G, Domonkos I, Tiricz H, Vágvölgyi C, Szolomajer J, Howan DHO, Tóth GK, Pfeiffer I, Kondorosi E (2021) Symbiotic NCR peptide fragments affect the viability, morphology and biofilm formation of Candida species. Int J Mol Sci 22:1–20. https://doi.org/10.3390/2Fijms22073666
Taff HT, Mitchell KF, Edward JA, Andes DR (2013) Mechanisms of Candida biofilm drug resistance. Future Microbiol 8:1325–1337. https://doi.org/10.2217/fmb.13.101
Talapko J, Juzbašić M, Matijević T, Pustijanac E, Bekić S, Kotris I, Škrlec I (2021) Candida albicans—The virulence factors and clinical manifestations of infection. J Fungi 22:2–19. https://doi.org/10.3390/jof7020079
Tossi A, Sandri L, Giangaspero A (2000) Amphipathic, alpha-helical antimicrobial peptides. Biopolymers 55:4–30. https://doi.org/10.1002/1097-0282(2000)55:1%3C4::aid-bip30%3E3.0.co;2-m
Tumbarello M, Prosteraro B, Trecarichi EM, Fiori B, Rossi M, Porta R, de Gaetano DK, La Sorda M, Spanu T, Fadda G, Cauda R, Sanguinetti M (2007) Biofilm production by Candida species and inadequate antifungal therapy as predictors of mortality for patients with candidemia. J Clin Microbiol 45:1843–1850. https://doi.org/10.1128/jcm.00131-07
Ullivarri MF, Arbulu S, Garcia-Gutierrez E, Cotter PD (2020) Antifungal peptides as therapeutic agents. Front Cell Infect Microbiol 10:1–22. https://doi.org/10.3389/fcimb.2020.00105
Vila T, Sultan AS, Montelongo-Jauregui D, Jabra-Rizk M (2020) Oral candidiasis: a disease of opportunity. J Fungi 6:1–28. https://doi.org/10.3390/jof6010015
Yadav MK, Go YY, Chae S, Song J (2015) The small molecule DAM inhibitor, pyrimidinedione, disrupts Streptococcus pneumoniae biofilm growth in vitro. PLoS One 10:1–35. https://doi.org/10.1371/2Fjournal.pone.0139238
Zheng M, Wang R, Chen SI, Zou Y, Yan L, Zhao L, Li X (2021) Design, synthesis and antifungal activity of stapled aurein 1.2 peptides. Antibiotics (basel) 10:1–8. https://doi.org/10.3390/2Fantibiotics10080956
Acknowledgements
The LaBAM team is grateful for the Center for Analytical Microscopy's collaboration with the Federal University of Ceará (UFC) for assistance with SEM images, which improved the manuscript significantly.
Funding
This work was funded by the Coordination for the Improvement of Higher Education Personnel (CAPES). Eduardo M. Cilli is the recipient of a Research Productivity Scholarship from the National Council for Research and Development (CNPq) and supported by FAPESP (#2013/07600-3).
Author information
Authors and Affiliations
Contributions
Methodology, data curation, investigation, and writing––first draft: MLS, RMBC, MNC, JAPG. Formal analysis and investigation: ROSF, ENL. Funding acquisition: EMC. Conceptualization, data curation, supervision, and writing—review and edition: VAC.
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported.
Ethical approval
The data collection and experimental procedures using clinical isolates were approved by the Ethics Committee of the State University Vale do Acaraú (UVA), under protocol number 644.765, guaranteeing the confidentiality and privacy of the research participants.
Additional information
Handling editor: P. Fechter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Silva, M.L., Carneiro, M.N., Cavalcante, R.M.B. et al. K-aurein: A notable aurein 1.2-derived peptide that modulates Candida albicans filamentation and reduces biofilm biomass. Amino Acids 55, 1003–1012 (2023). https://doi.org/10.1007/s00726-023-03288-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-023-03288-z