Abstract
One pathogenic mechanism of ethanol-induced liver injury is the excessive production of reactive oxygen species (ROS), which may result in alcoholic liver disease (ALD) characterized by cell death due to necrosis and apoptosis. Taurine was proved to protect against liver damage. However, whether taurine attenuates ethanol-induced hepatic apoptosis remains unknown. The present study aims to elucidate this effect and its underlying mechanism. Taurine was administered to ALD rats and an in vitro experiment in which taurine was added to primary rat hepatocytes cultured with ethanol was conducted. Mitochondrial function and anti-oxidative capacity of the liver were tested. TUNEL and AO-EB double staining were conducted to detect apoptosis of liver cells. Expressions of factors and proteins involved in mitochondrial and death receptor pathways were detected by RT-PCR and Western-blot. The results showed that taurine inhibited the decline of cell functions and apoptosis in hepatocytes cultured with ethanol. Furthermore, increased malondialdehyde (MDA) and reduced superoxide dismutase (SOD), glutathione peroxidase (GSH-Px), total antioxidant capacity (T-AOC), cytochrome c oxidase (COX) and NADH dehydrogenase (ND) in ALD rats were mediated by taurine. RT-PCR and western-blot results revealed that taurine down-regulated expression of Bax, Fas, Fas ligand (FasL), caspase 3 and caspase 9 while up-regulating the expression of Bcl-2 in ethanol-cultured hepatocytes. In summary, taurine inhibit ethanol-induced hepatic apoptosis by regulating mitochondrial or death receptor pathways.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Long-term and excessive alcohol intake causes alcoholic liver disease (ALD), which is a major complication and a cause of morbidity and mortality due to alcohol abuse (Mcclain et al. 2004). Researchers have determined that alcohol consumption causes apoptosis, a major cellular event, in the liver, and ethanol-induced liver damage has been observed both in animals with alcohol-induced injuries and ALD patients (Allam et al. 2013; Ma et al. 2015). The metabolic processes initiated by ethanol and its metabolite aldehyde generate large quantities of reactive oxygen species (ROS) that lead to oxidative stress in the mitochondria of the liver and finally induce intrinsic apoptosis among hepatocytes, a process called the mitochondrial pathway (Revollo and Li 2013). An additional extrinsic apoptosis pathway mediated by death receptors is associated with ALD-induced liver injury (Lambert et al. 2003; Liu 2004; Morio et al. 2013; Zhou et al. 2001). Previous work demonstrated that apoptosis caused by alcohol intake involves both the intrinsic and extrinsic pathways, partially in response to ethanol-induced increases in ROS (Hoek and Pastorino 2002). Thus, the identification of a functional food or medicine to combat oxidative stress and apoptosis may be an effective preventative or treatment strategy for ALD.
Taurine, a type of β-amino acid that is widely distributed in the organs of animals and humans, is primarily synthesized in the liver, which is often damaged by ethanol and its metabolites. Taurine reportedly exerts effects on oxidative stress caused by different factors, including liver or hepatocyte damage (Basaran-Kucukgergin et al. 2016; Li et al. 2017; Nagai et al. 2016; Reeta et al. 2017). Taurine also exerts protective effects during ethanol-induced oxidative stress and liver injury (Balkan et al. 2002; Chen et al. 2009). Our previous study demonstrated that taurine increases antioxidative capacity and decreases lipid peroxidation in the livers of ALD rats (Wu et al. 2009). Other researchers found that taurine inhibits apoptosis in different organs (Aly and Khafagy 2014; Aydin et al. 2016; Li et al. 2017; Yang et al. 2015), particularly in liver-damaged animals (Das et al. 2010; Lakshmi Devi and Anuradha 2010; Yalcinkaya et al. 2009). In addition, taurine was found to be safe at doses up to 3 g/day in healthy adults or 1000 mg/kg body weight per day according to the European Food Safety Authority (Shao and Hathcock 2008). Therefore, taurine is safe for use as a functional food or medicinal substance to prevent or cure ALD as an antioxidant or anti-apoptotic agent, but the mechanisms underlying its effects remain unclear.
This study established both an ALD rat model and ethanol-injured hepatocyte model, and taurine was administered in vitro or in vivo. Antioxidative indexes, apoptosis and apoptotic pathways were detected to evaluate the antioxidant and anti-apoptotic effects of taurine in ALD rats.
Materials and methods
Animals
Ninety male SPF Wistar rats (180 ± 20 g) obtained from Chang Sheng Biotechnology Co., LTD, Liaoning Province, China, were given free access to standard feed and tap water at a temperature of 22 ± 2 °C and a 12-h light/dark cycle. All procedures using animals were conducted according to the requirements of the Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals. The procedures were approved by the Animal Care and Use Committee of Shenyang Agricultural University (No. 20141013).
Animal studies
The experiments included preventive trial and curative trial. In the preventive trial, taurine was administered at the same time of ethanol exposure to determine the preventive effect of taurine on liver deficiency induced by ethanol. Rats in the ALD model group (M) and the taurine preventive group (M + T) were both intragastrically treated with 40% ethanol (15 mL/kg/d) and pyrazole (24 mg/kg/d), and were given free access to tap water or 2% taurine, respectively. Rats in the normal control group (N) and taurine control group (T) were intragastrically administered with saline, and were given free access to tap water or 2% taurine, respectively. In the curative trial, taurine were administered after ALD model establishment which was the same to M group of the preventive trial. Then, ALD model rats were divided into the taurine curative group (Tc) and automatic recovery group (A), which drank 2% taurine or water, respectively. The preventive and curative trials lasted for 12 and 4 weeks, respectively. Serum and liver samples were collected for further analysis.
Antioxidant indexes and mitochondrial function analysis
Antioxidant indexes including glutathione peroxidase (GSH-Px), superoxide dismutase (SOD), total antioxidant capacity (T-AOC) and malondialdehyde (MDA) were assayed with commercial kits (Nanjing Jiancheng Bioengineering Institute, China) according to the manufacturers’ protocols. GSH-Px and T-AOC were assayed using colorimetric method under 412 or 520 nm. SOD was assayed using hydroxylamine method under 550 nm, MDA was assayed using thiobarbituric acid (TBA) method under 532 nm.
Mitochondrial was extracted from the liver using traditional dounce homogenization to mince the fresh tissues on ice in mitochondria isolation buffer, followed by 800g for 5 min centrifugation at 4 °C. The supernatant was collected and centrifuged at 15,000g for 10 min at 4 °C. Collect the pellet and resuspend in mitochondria isolation buffer followed by centrifugation at 15,000g for 10 min at 4 °C. Resuspend the pellet in mitochondria storage buffer and kept it on ice. Mitochondrial function were assessed by the measurement of mitochondria cytochrome c oxidase (COX) (My BioSource, USA, MBS 700786) and NADH dehydrogenase (ND) (Biomatik, USA, EKU06144) activities using commercial ELISA kits by the quantitative sandwich enzyme immunoassay technique. Finally, the intensity of the color change is measured at 450 nm.
Immunohistochemistry
Apoptosis in rat liver tissue was detected in 5-μm-thick deparaffinized sections using a TdT-mediated dUTP nick-end labeling (TUNEL) apoptosis detection kit (Roche Molecular Biochemicals, Germany, No. 11684817910) according to the instructions. Apoptotic hepatocytes were identified by positive TUNEL staining and morphology. Data are presented as the percentage of TUNEL-positive hepatocyte nuclei per total nuclei in six different random fields from 5 sections for each group.
Hepatocyte isolation and treatment
Rat primary hepatocytes were isolated from male Wistar rats by in situ perfusion. The portal vein was cannulated in situ and initially perfused with HEPES buffer, followed by incubation in 0.025% collagenase IV at 37 °C for 40–60 min (Sigma, USA). Viable hepatocytes were separated and purified by centrifugation at 50g, 5 min at 4 °C for three times. Cell viability was observed to exceed 90% in a trypan blue exclusion test. Subsequently, hepatocytes were plated in Dulbecco’s modified Eagle’s medium (DMEM) with high glucose (BioInd, Israel, 11-055-1K) supplemented with 100 mL/L bovine serum (BioInd, Israel, 04-010-1A). After incubation for 24 h, the medium was replaced with DMEM (high glucose) containing 10% bovine serum, 0.5 mg/L insulin, 100 nmol/L dexamethasone, and 1% penicillin–streptomycin solution (BioInd, Israel, 03-031-1B). The cells were cultured at 37 °C for 24 h, and then periodic acid–Schiff (PAS) staining was carried out to identify hepatocytes. The hepatocytes were incubated in a 96-well plate with DMEM (high glucose) supplemented with either 75 mmol/L ethanol or 10 mmol/L taurine for 48 h. Then, the cells and medium were collected for further analysis.
Cell viability assay
Cell viability was determined by a methylthiazol tetrazolium (MTT) assay. Briefly, 20 µL of MTT solution (Sigma, USA, M5655) was applied to cells, which were incubated for 4 h at 37 °C; then, 150 µL of medium was replaced with 150 µL of DMSO. 20 min later, optical density was measured at A570 nm, and cell viability was expressed as the percentage of absorbance in the control group, for which cell viability was set as 100%.
Liver function index analysis
Alanine aminotransferase (ALT), aspartate aminotransferase (AST) and glutamyl transpeptidase (GGT) activities in cell culture medium were measured with commercial kits (Nanjing Jiancheng Bioengineering Institute, China) according to the manufacturer’s protocols.
Cell apoptosis assay
Hepatocyte apoptosis was identified and quantified by acridine orange (AO) and ethidium bromide (EB) double staining (Vijayalakshmi and Sindhu 2017) using kits (Solarbio Life Sciences, China, CA1140). AO can penetrate intact cell membrane and embedded into nuclei DNA to exhibit bright green fluorescence, while EB can only penetrate through damaged cell membrane and embedded to nuclei DNA to exhibit orange fluorescence. The cells were observed under a fluorescence microscope (Leica 4000B, Germany) with the magnification of 400, apoptotic hepatocytes were identified by positive EB staining and morphology. The apoptosis rate was calculated as the percentage of EB-positive hepatocyte nuclei per total nuclei in six different random fields. Triplicate tests were carried out for each group. Images were taken at a magnification of 200, scale bar = 100 μm.
Real-time PCR analysis
Total RNA from hepatocytes were extracted using Trizol (Takara, Dalian, China, 9109), and reverse transcription was conducted using a commercial kit (Takara, Dalian, China). Real-time PCR was carried out in a total volume of 20 μL using a SYBR® green kit (ABI, USA, No.4472908) on a Bio-Rad iQTM5 detection system (Bio-Rad, Hercules, USA) with the following program: 95 °C for 30 s, followed by 40 cycles of 0.05 s at 95 °C and 30 s at 58.8 °C for Bcl-2, 60 °C for Bax, 58.8 °C for Fas, 58.8 °C for FasL, 57.8 °C for caspase 3, and 63.7 °C for caspase 9; finally, a melting curve analysis was run for 30 s at 55 °C. The following primers were used: Bcl-2 (forward, TGCAGAGATGTCCAGTCAGC, and reverse, TCAAAGAAGGCCACAATCCT), Bax (forward, ACGCATCCACCAAGAAGC, and reverse, GCCACACGGAAGAAGACCT), Fas (forward, GGGCATGGTTTAGAAGTGGA, and reverse, CCACACGAGGTGCAGTGATA), FasL (forward, GCAGAGGCAAAGAGAAGGAA, and reverse, CAAGTAGACCCACCCTGGAA), caspase 3 (forward, ACGGGACTTGGAAAGCATC, and reverse, TAAGGAAGCCTGGAGCACAG), caspase 9 (forward, CCACTGCCTCATCATCAACA, and reverse, TCGTTCTTCACCTCCACCAT), and β-actin (forward, GGAGATTACTGCCCTGGCTCCTA, and reverse, GACTCATCGTACTCCTGCTTGCTG). Each reaction was repeated three times, and the melting curve was analyzed. Relative gene expression was normalized to the expression of β-actin, which served as the internal control, and calculated by the 2−△△Ct method. Data are expressed as a fold-change relative to the control group.
Western blotting analysis
Proteins were extracted from hepatocytes and measured with a protein extraction kit (Beyotime, China) and a BCA protein assay kit (Beyotime, China, P0012S). SDS-PAGE electrophoresis was used to separate the proteins, which were then transferred onto polyvinylidene difluoride (PVDF) membranes (Bio-Rad, USA). After blocking the membranes with 5% (W/V) non-fat milk in Tris buffer solution (TBS) (Applygen, China, B1009) for 2 h at room temperature (RT), the membranes were first incubated with antibodies against β-actin (anti mouse β-actin, Abcam, Hong Kong, ab8226, dilution: 1:10000, 42KD), GAPDH (anti mouse GAPDH, Abcam, Hong Kong, ab8245, dilution:1:10000, 37KD), Bcl-2 (anti rabbit Bcl-2, Abcam, Hong Kong, ab59348, dilution: 1:1000, 26KD), Bax (anti rabbit Bax, Abcam, Hong Kong, ab32503; dilution: 1:10000, 21KD), Fas (anti rabbit Fas, Abcam, Hong Kong, ab82419; dilution: 1:1000, 45KD), FasL (anti rabbit FasL, Abcam, Hong Kong, ab15285; dilution: 1:1000; 31 KD), active cleaved-caspase 3 (anti rabbit cleaved-caspase 3, Abcam, Hong Kong, ab49822, dilution: 1:500; 17 KD) and cleaved-caspase 9 (anti rabbit cleaved-caspase 9, CST, USA, #9507, dilution: 1:1000, 37KD) at 4 °C overnight. The membranes were then washed in Tris buffer solution Tween (TBST) for 1 h and incubated with the appropriate horseradish peroxidase (HRP)-conjugated secondary antibody (1:10000, Proteintech, USA, SA00001-1, SA00001-2) for 1 h at RT. Protein bands were visualized with a Super ECL kit (Beyotime, China, P0018), recorded with a DNR bio-imaging system (Microchemi 4.2, Israel) and quantified with GelQuant v12.3. Relative expression was normalized to β-actin or GAPDH.
Statistical analysis
All values are expressed as the mean ± SEM. Data were analyzed by one-way ANOVA, followed by LSD and Duncan’s multiple range tests using SPSS 17.0. GraphPad Prism 5.0 was used to create the artwork. A difference at the p < 0.05 or p < 0.01 level was considered statistically significant or extremely significant, respectively.
Results
Taurine increased antioxidant capacity in the livers of ALD rats
Oxidative stress and decreased antioxidative capacity are thought to contribute to hepatocyte injury and apoptosis in response to alcohol exposure. Figure 1 showed that in animal experiments, hepatic GSH-Px, SOD and T-AOC in the M group dropped markedly, while MDA content increased (p < 0.01) compared to the values for the N and T groups in the preventive trial. Those changes were ameliorated by taurine in the M + T group (p < 0.01). In the curative trial, hepatic GSH-Px, SOD and T-AOC in the A group were much lower than that in the N and Tc groups (p < 0.01), whereas hepatic MDA content was higher (p < 0.01 or p < 0.05). The results showed that taurine administered preventively or curatively increases antioxidant activity in the livers of ALD rats.
Effects of taurine on antioxidant activity in the livers of ALD rats. a Hepatic MDA concentration; b hepatic GSH-Px concentration; c hepatic SOD concentration; d hepatic T-AOC concentration. N, normal control group; T, taurine control group; M + T, taurine preventive group; M, ALD model group; Tc, taurine curative group; A, automatic recovery group. All data are expressed as the mean ± SE and were analyzed by one-way ANOVA followed by LSD and Duncan’s multiple comparisons post hoc test (n = 5). * indicates p < 0.05, ** indicates p < 0.01
Taurine-protected mitochondria in the livers of ALD rats
Changes in COX and ND activities reflect mitochondrial function in the liver. As shown in Fig. 2, the activity of these two enzymes obviously decreased in the M group in the preventive trial and the A group of the curative trial compared to that in the N group (p < 0.01). In the M + T and Tc groups, COX and ND activities in the liver were much higher than in the M and A groups (p < 0.01), suggesting there is a protective role for taurine in the liver mitochondria of ALD rats.
Protective effects of taurine on mitochondria in the livers of ALD rats. a COX activity; b ND activity. N, normal control group; T, taurine control group; M + T, taurine preventive group; M, ALD model group; Tc, taurine curative group; A, automatic recovery group. All data are expressed as the mean ± SE and were analyzed by one-way ANOVA followed by LSD and Duncan’s multiple comparisons post hoc test (n = 5). * indicates p < 0.05, ** indicates p < 0.01
Taurine-inhibited apoptosis of liver cells induced by ethanol
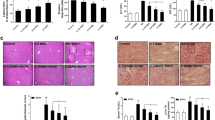
The liver apoptosis rates of the animals are illustrated in Fig. 3, with apoptotic DNA fragmentation shown in brown. In the preventive trial, apoptosis increased significantly in the M group compared to that in the N, T and M + T groups (p < 0.01). In the curative trial, apoptosis was highest in the A group, while apoptosis in the Tc group was obviously lower than that in the A group (p < 0.01). The results showed that taurine protects the liver from ethanol-induced apoptosis.
Effects of taurine on apoptosis in the livers of ALD rats. a TUNEL staining of liver paraffin sections; b apoptosis rate in the livers of rats. N, normal control group; T, taurine control group; M + T, taurine preventive group; M, ALD model group; Tc, taurine curative group; A, automatic recovery group. All data are expressed as the mean ± SE and were analyzed by one-way ANOVA followed by LSD and Duncan’s multiple comparisons post hoc test (n = 5). * indicates p < 0.05, ** indicates p < 0.01
The apoptosis rate of hepatocytes cultured in vitro is shown in Fig. 4 the cell apoptosis rate in the M group increased significantly (p < 0.01) compared to that in the N and T groups, while the apoptosis rate was reduced with taurine treatment in the M + T group (p < 0.01).
Effects of taurine on cell apoptosis in hepatocytes exposed to ethanol. a AO-EB staining; b cell apoptosis rate. N, normal control group in which cells were cultured with DMEM; T, taurine group in which cells were cultured with DMEM + taurine; M + T, taurine preventive group in which cells were cultured with DMEM + ethanol + taurine; M, model group in which cells were cultured with ethanol. AO can penetrate intact cell membrane and embedded into nuclei DNA to exhibit bright green fluorescence, while EB can only penetrate through damaged cell membrane and embedded to nuclei DNA to exhibit orange fluorescence. All data are expressed as the mean ± SE and were analyzed by one-way ANOVA followed by LSD and Duncan’s multiple comparisons post hoc test (n = 6). * indicates p < 0.05, ** indicates p < 0.01
Taurine-protected hepatocytes from ethanol-induced injury
As shown in Fig. 5, cell viability in the M group declined significantly compared to that in the N and T groups, which indicates that ethanol administration can seriously damage hepatocytes (p < 0.01). Cell viability in the M + T group obviously increased compared to that in the M group, indicating that taurine administration in the cell culture medium inhibits cell death caused by ethanol (p < 0.01).
Protective effects of taurine on hepatocytes exposed to ethanol. a Cell viability of hepatocytes; b ALT activity in culture medium; c AST activity in culture medium; d. GGT activity in culture medium. N, normal control group in which cells were cultured with DMEM; T, taurine group in which cells were cultured with DMEM + taurine; M + T; taurine preventive treatment group in which cells were cultured with DMEM + ethanol + taurine; M, model group in which cells were cultured with ethanol. All data are expressed as the mean ± SE and were analyzed by one-way ANOVA followed LSD and Duncan’s multiple comparisons post hoc test (n = 6). * indicates p < 0.05, ** indicates p < 0.01
Increased liver enzymes in cell culture medium are an indicator of hepatocyte injury. ALT, AST and GGT activities in the cell culture medium were obviously higher in the M group than in the other three groups, indicating significant cell injury caused by ethanol (p < 0.01). However, the activities of these enzymes were significantly lower in the M + T group (p < 0.01) than in the M group, suggesting a protective effect of taurine in hepatocytes (Fig. 5).
Taurine regulates the expression of key factors involved in apoptotic pathways
Several regulator proteins are involved in the regulation of cell apoptosis. In the present experiment, the mRNA and protein expression of regulator proteins in cultured hepatocytes was detected. As shown in Fig. 6, the mRNA expression of Bax, Fas, FasL, caspase 3 and caspase 9 increased significantly (p < 0.01), whereas mRNA expression of Bcl-2 in the M group decreased significantly (p < 0.01). In the M + T group, mRNA expression of Bax, Fas, FasL, caspase 3 and caspase 9 was lower and Bcl-2 mRNA expression was much higher compared to that in the M group (p < 0.01). Figure 7 illustrated that in the M group, protein expression of Bax, Fas, FasL, cleaved-caspase 3 and cleaved-caspase 9 were raised significantly (p < 0.05 or p < 0.01), whereas protein expression of Bcl-2 were reduced significantly (p < 0.01) compared to the other three groups. The results indicated that taurine administration up-regulates anti-apoptotic proteins and down-regulates pro-apoptotic proteins.
Effects of taurine on the mRNA expression of key factors involved in the apoptotic pathways. a Bcl-2 mRNA expression; b Bax mRNA expression; c Fas mRNA expression; d FasL mRNA expression; e Caspase-3 mRNA expression; f Caspase-9 mRNA expression. N, normal control group in which cells were cultured with DMEM; T, taurine group in which cells were cultured with DMEM + taurine; M + T, taurine preventive group in which cells were cultured with DMEM + ethanol + taurine; M, model group in which cells were cultured with ethanol. All data are expressed as the mean ± SE and were analyzed by one-way ANOVA followed by LSD and Duncan’s multiple comparisons post hoc test (n = 6). * indicates p < 0.05, ** indicates p < 0.01
Effects of taurine on the protein expression of key factors involved in the apoptotic pathways. a Bcl-2 protein expression; b Bax protein expression; c Fas protein expression; d FasL protein expression; e cleaved-caspase-3 protein expression; f cleaved-caspase-9 protein expression. N, normal control group in which cells were cultured with DMEM; T, taurine group in which cells were cultured with DMEM + taurine; M + T, taurine preventive group in which cells were cultured with DMEM + ethanol + taurine; M, model group in which cells were cultured with ethanol. All data are expressed as the mean ± SE and were analyzed by one-way ANOVA followed by LSD and Duncan’s multiple comparisons post hoc test (n = 6). * indicates p < 0.05, ** indicates p < 0.01
Discussion
Chronic and excessive ethanol exposure seriously damages the liver and induces ALD. During ethanol metabolism, large quantities of ROS are generated and attack the mitochondria in liver cells, which further induces apoptosis and necrosis (Liu et al. 2016). Taurine possesses antioxidant and anti-apoptotic properties in the liver (Basaran-Kucukgergin et al. 2016; Lakshmi Devi and Anuradha 2010). This study aims to verify the hypothesis that taurine administration reduces oxidative stress and apoptosis induced by ethanol. We found that taurine effectively decreases ethanol-induced liver injury and lipid accumulation, increases antioxidant capacity, inhibits apoptosis and regulates the mitochondrial and death receptor apoptotic pathways.
Liver enzymes are primarily synthesized inside liver cells, and increases in these enzymes in the extracellular medium are indicators of hepatocyte injury. AST and ALT, two main aminotransferases inside the hepatic cells under normal conditions, are released from damaged hepatocytes into the blood to obviously increase serum ALT and AST, the concentrations of which are main sensitive indexes of liver damage. GGT is present in many tissues including kidneys, bile duct, pancreas, gallbladder, spleen, heart, brain and seminal vesicles. Under normal conditions, serum GGT mainly comes from the liver, in which GGT was mainly expressed in the hepatic cytoplasm and bile duct epithelium, and has a sensitivity for the diagnosis and prognosis of liver disease due to its function in the control of redox status and is also a well-known marker of alcohol excess (Dillon and Miller 2016). Enhanced ALT, AST and GGT activities were observed in culture medium, suggesting there was hepatocyte injury under ethanol exposure. Taurine added to the medium along with ethanol prevented the increased liver enzyme activity, indicating taurine has a protective role against ethanol-induced hepatocyte injury, which were in accordance to other reports that taurine was shown to protect the liver from injury by decreasing blood ALT, AST and GGT levels (Devi et al. 2009; Erman et al. 2004; Lee and Ko 2016).
Ethanol has been shown to elevate the formation of lipid radicals and consequently cause oxidative stress in the liver (Livero and Acco 2016). As shown in the current results, ethanol intake led to an increase in hepatic MDA and a decrease in hepatic SOD, GSH-Px and T-AOC in the M group. However, these parameters were reversed by taurine, which was given preventively or curatively. The ability of ethanol to promote oxidative damage and the role of free radicals in alcoholic liver injury have been widely studied because antioxidants such as N-acetylcysteine (NAC) or Vitamin E are therapeutically applied to treat ALD (Lee et al. 2013; Ronis et al. 2005). Taurine also acts as an antioxidant during organ or cellular injury, accompanied by lower MDA and higher SOD, GSH-Px and T-AOC activities, which was confirmed in the present study (Reeta et al. 2017; Yang et al. 2015; Yildirim et al. 2007).
Mitochondrion is a vital organ that maintains biological functions of a cell by providing energy. Defective mitochondria result in shortage in ATP, leakage of ROS, accumulation of fat, and activation of intrinsic apoptotic cell death pathway, the dysfunction of which has been reported to be the earliest event in the development of ALD (Sun et al. 2017). Mitochondria function in the present study was assayed by the activation of COX and ND, which are key enzymes of the mitochondrial respirator chain. COX is the last enzyme in the respiratory electron transport chain located in the mitochondrial membrane. It receives an electron from each of four cytochrome c molecules, and transfers them to one oxygen molecule, converting molecular oxygen to two molecules of water. ND, also referred to as complex I, is the first enzyme of the mitochondrial electron transport chain that translocates four protons across the inner membrane per molecule of oxidized NADH, helping to build the electrochemical potential difference used to produce ATP. Deficiency in the activities of COX and ND can decrease the ATP level and destroys respiratory function of mitochondria which further damages the liver cells. Chronic ethanol consumption have been proved to results in decreased mitochondrial protein synthesis, impairment of the oxidative phosphorylation system, depression in ATP synthesis, leakage of ROS, and further cause mitochondrial depolarization and Δψm alteration that were proved to induce the activation of intrinsic apoptotic pathway (Sun et al. 2017; Xie et al. 2018). Taurine was reported to diminish the increase in mitochondrial ROS, resulting in the recovery of COX activity in the hippocampus (Jia et al. 2016), whereas a lack of taurine may underlie mitochondrial dysfunction. This dysfunction manifests in the form of decreased COX in rat islets (Lee et al. 2011) and down-regulates ND in cardiomyocytes (Jong et al. 2012). The results are similar to those in the present study in ALD rats, which showed that decreased COX and ND activities in the liver of ALD rats, indicating that the respiratory electron transport chain was imbalanced by ethanol, while taurine administration can prevent or reverse mitochondrial dysfunction.
Furthermore, accumulating evidence suggests oxidative stress may frequently initiate cell injury mediated by mitochondria or death receptor pathways (Liu et al. 2016). Alcoholic mitochondrial damage induced by redundant ROS initiates the mitochondrial membrane permeability transition pore (MPTP) opening (Xie et al. 2018), and the translocation of Bax to the mitochondria from the cytosol and promotes the release of cytochrome C, which can associate with pro-caspase 9 and Apaf-1(Heaton et al. 2011). The complex processes pro-caspase 9 to cleave and further active caspase 3, which cleaves and initiate a caspase cascade leading to apoptosis (Mo et al. 2017). Meanwhile, the up-regulation of FasL has been reported as another mechanism connected to hepatocyte apoptosis induced by alcohol (Darwish et al. 2012). The combination of Fas and FasL initiates signal transduction, followed by the activation of caspase proteases, which is known as the death-receptor-mediated extrinsic pathway leading to apoptosis (McVicker et al. 2006). In the present study, a TUNEL assay and AO-EB staining indicted a substantial increase in apoptosis compared to that in hepatocytes cultured with ethanol in vitro and in the livers of ALD rats. The present results showed increased gene expression of Bax, Caspase9, Caspase3, Fas and FasL and reduced mRNA expression of Bcl-2 in ethanol-treated hepatocytes. Western-blot results further proved that protein expression of Bax, cleaved-caspase 9, cleaved-caspase 3, Fas and FasL were lowered, while protein expression of Bcl-2 were raised by ethanol. Taurine decreases apoptosis when administered both in vivo and in vitro. Furthermore, down-regulation of the expression of pro-apoptotic factors, including Bax, cleaved-caspase 9, cleaved-caspase 3, Fas and FasL, and up-regulation of the anti-apoptotic factor Bcl-2 in hepatocytes have been observed in groups treated with taurine. Similar anti-apoptotic effects of taurine were found in 2,5-hexanedione-treated PC12 cells and rats with iron-potentiated alcoholic liver fibrosis, and the opening of MPTP in taurine deficiency animals were found to induce the release of cytochrome c from the mitochondria and the onset of the apoptotic cascade (Jong et al. 2011), indicating that treatment with taurine may prevent apoptosis by inhibiting a mitochondria-dependent pathway (Lakshmi Devi and Anuradha 2010; Li et al. 2017). Meanwhile, caspase 3, caspase 9 and apoptosis levels were found to increase in taurine-deficient mice (Jong et al. 2017), and taurine administration can decrease mRNA expression of Fas and Bax in acute hepatic damaged animals (Nagai et al. 2016). Based on these results, taurine exerts anti-apoptotic effects on ethanol-induced hepatocyte apoptosis through the mitochondrial and death receptor pathways.
Conclusions
In conclusion, our results show that taurine protects liver cells from ethanol injury by improving the antioxidant capacity of hepatocytes and decreasing the apoptosis of hepatocytes exposed to ethanol through the regulation of mitochondrial or death receptor pathways.
References
Allam RM et al (2013) Hepatoprotective effects of Astragalus kahiricus root extract against ethanol-induced liver apoptosis in rats. Chin J Nat Med 11:354–361
Aly HA, Khafagy RM (2014) Taurine reverses endosulfan-induced oxidative stress and apoptosis in adult rat testis. Food Chem Toxicol 64:1–9
Aydin AF, Coban J, Dogan-Ekici I, Betul-Kalaz E, Dogru-Abbasoglu S, Uysal M (2016) Carnosine and taurine treatments diminished brain oxidative stress and apoptosis in d-galactose aging model. Metab Brain Dis 31:337–345
Balkan J, Kanbagli O, Aykac-Toker G, Uysal M (2002) Taurine treatment reduces hepatic lipids and oxidative stress in chronically ethanol-treated rats. Biol Pharm Bull 25:1231–1233
Basaran-Kucukgergin C, Bingul I, Tekkesin MS, Olgac V, Dogru-Abbasoglu S, Uysal M (2016) Effects of carnosine, taurine, and betaine pretreatments on diethylnitrosamine-induced oxidative stress and tissue injury in rat liver. Toxicol Ind Health 32:1405–1413
Chen X, Sebastian BM, Tang H, McMullen MM, Axhemi A, Jacobsen DW, Nagy LE (2009) Taurine supplementation prevents ethanol-induced decrease in serum adiponectin and reduces hepatic steatosis in rats. Hepatology 49:1554–1562
Darwish HA, Abd Raboh NR, Mahdy A (2012) Camel’s milk alleviates alcohol-induced liver injury in rats. Food Chem Toxicol 50:1377–1383
Das J, Ghosh J, Manna P, Sil PC (2010) Protective role of taurine against arsenic-induced mitochondria-dependent hepatic apoptosis via the inhibition of PKCdelta-JNK pathway. PLoS One 5:e12602. https://doi.org/10.1371/journal.pone.0012602
Devi SL, Viswanathan P, Anuradha CV (2009) Taurine enhances the metabolism and detoxification of ethanol and prevents hepatic fibrosis in rats treated with iron and alcohol. Environ Toxicol Pharmacol 27:120–126
Dillon JF, Miller MH (2016) Gamma glutamyl transferase ‘To be or not to be’ a liver function test? Ann Clin Biochem 53:629–631
Erman F, Balkan J, Cevikbas U, Kocak-Toker N, Uysal M (2004) Betaine or taurine administration prevents fibrosis and lipid peroxidation induced by rat liver by ethanol plus carbon tetrachloride intoxication. Amino Acids 27:199–205
Heaton MB, Paiva M, Siler-Marsiglio K (2011) Ethanol influences on Bax translocation, mitochondrial membrane potential, and reactive oxygen species generation are modulated by vitamin E and brain-derived neurotrophic factor. Alcohol Clin Exp Res 35:1122–1133
Hoek JB, Pastorino JG (2002) Ethanol, oxidative stress, and cytokine-induced liver cell injury. Alcohol 27:63–68
Jia N, Sun Q, Su Q, Dang S, Chen G (2016) Taurine promotes cognitive function in prenatally stressed juvenile rats via activating the Akt-CREB-PGC1alpha pathway. Redox Biol 10((C)):179–190
Jong CJ, Azuma J, Schaffer SW (2011) Role of mitochondrial permeability transition in taurine deficiency-induced apoptosis. Exp Clin Cardiol 16:125–128
Jong CJ, Azuma J, Schaffer S (2012) Mechanism underlying the antioxidant activity of taurine: prevention of mitochondrial oxidant production. Amino Acids 42:2223–2232
Jong CJ, Ito T, Prentice H, Wu JY, Schaffer SW (2017) Role of mitochondria and endoplasmic reticulum in taurine-deficiency-mediated apoptosis. Nutrients. https://doi.org/10.3390/nu9080795
Lakshmi Devi S, Anuradha CV (2010) Mitochondrial damage, cytotoxicity and apoptosis in iron-potentiated alcoholic liver fibrosis: amelioration by taurine. Amino Acids 38:869–879
Lambert JC, Zhou Z, Kang YJ (2003) Suppression of Fas-mediated signaling pathway is involved in zinc inhibition of ethanol-induced liver apoptosis. Exp Biol Med (Maywood) 228:406–412
Lee SY, Ko KS (2016) Effects of S-adenosylmethionine and its combinations with taurine and/or betaine on glutathione homeostasis in ethanol-induced acute hepatotoxicity. J Cancer Prev 21:164–172
Lee YY et al (2011) Taurine supplementation restored the changes in pancreatic islet mitochondria in the fetal protein-malnourished rat. Br J Nutr 106:1198–1206
Lee SJ, Kim SY, Min H (2013) Effects of vitamin C and E supplementation on oxidative stress and liver toxicity in rats fed a low-fat ethanol diet. Nutr Res Pract 7:109–114
Li S et al (2017) Taurine inhibits 2,5-hexanedione-induced oxidative stress and mitochondria-dependent apoptosis in PC12 cells. Ind Health 55:108–118
Liu J (2004) Fas-mediated signaling pathway in ethanol-induced liver apoptosis: inhibition by zinc. Exp Biol Med (Maywood) 229:365–366
Liu J, Zhou J, Wu Z, Wang X, Liu L, Yao C (2016) Cyanidin 3-O-β-glucoside ameliorates ethanol-induced acute liver injury by attenuating oxidative stress and apoptosis: the role of SIRT1/FOXO1 signaling. Alcohol Clin Exp Res 40:457–466
Livero FA, Acco A (2016) Molecular basis of alcoholic fatty liver disease: from incidence to treatment. Hepatol Res 46:111–123
Ma Z, Hou T, Shi W, Liu W, He H (2015) Inhibition of hepatocyte apoptosis: an important mechanism of corn peptides attenuating Liver injury induced by ethanol. Int J Mol Sci 16:22062–22080
Mcclain CJ, Song Z, Barve SS, Hill DB, Deaciuc I (2004) Recent advances in alcoholic liver disease. IV. Dysregulated cytokine metabolism in alcoholic liver disease. Am J Physiol Gastrointest Liver Physiol 287:497–502
McVicker BL, Tuma DJ, Kubik JL, Tuma PL, Casey CA (2006) Ethanol-induced apoptosis in polarized hepatic cells possibly through regulation of the Fas pathway. Alcohol Clin Exp Res 30:1906–1915
Mo XQ et al (2017) Molecular mechanisms of apoptosis in hepatocellular carcinoma cells induced by ethanol extracts of Solanum lyratum Thumb through the mitochondrial pathway. World J Gastroenterol 23:1010–1017
Morio Y et al (2013) Ethanol-induced apoptosis in human liver adenocarcinoma cells (SK-Hep1): fas- and mitochondria-mediated pathways and interaction with MAPK signaling system. Toxicol In Vitro 27:1820–1829
Nagai K, Fukuno S, Oda A, Konishi H (2016) Protective effects of taurine on doxorubicin-induced acute hepatotoxicity through suppression of oxidative stress and apoptotic responses. Anticancer Drugs 27:17–23
Reeta KH, Singh D, Gupta YK (2017) Chronic treatment with taurine after intracerebroventricular streptozotocin injection improves cognitive dysfunction in rats by modulating oxidative stress, cholinergic functions and neuroinflammation. Neurochem Int 108:146–156
Revollo JR, Li X (2013) The ways and means that fine tune Sirt1 activity. Trends Biochem Sci 38:160
Ronis MJ et al (2005) Effects of N-acetylcysteine on ethanol-induced hepatotoxicity in rats fed via total enteral nutrition. Free Radic Biol Med 39:619–630
Shao A, Hathcock JN (2008) Risk assessment for the amino acids taurine, l-glutamine and l-arginine. Regul Toxicol Pharmacol 50:376–399
Sun Q, Zhang W, Zhong W, Sun X, Zhou Z (2017) Pharmacological inhibition of NOX4 ameliorates alcohol-induced liver injury in mice through improving oxidative stress and mitochondrial function. Biochim Biophys Acta 1861:2912–2921
Vijayalakshmi A, Sindhu G (2017) Umbelliferone arrest cell cycle at G0/G1 phase and induces apoptosis in human oral carcinoma (KB) cells possibly via oxidative DNA damage. Biomed Pharmacother 92:661–671
Wu G, Yang J, Sun C, Luan X, Shi J, Hu J (2009) Effect of taurine on alcoholic liver disease in rats. Amino Acids 36:457–464
Xie X, Xu X, Sun C, Yu Z (2018) Protective effects of cilostazol on ethanol-induced damage in primary cultured hepatocytes. Cell Stress Chaperones 23:203–211
Yalcinkaya S, Unlucerci Y, Giris M, Olgac V, Dogru-Abbasoglu S, Uysal M (2009) Oxidative and nitrosative stress and apoptosis in the liver of rats fed on high methionine diet: protective effect of taurine. Nutrition 25:436–444
Yang J, Zong X, Wu G, Lin S, Feng Y, Hu J (2015) Taurine increases testicular function in aged rats by inhibiting oxidative stress and apoptosis. Amino Acids 47:1549–1558
Yildirim Z, Kilic N, Ozer C, Babul A, Take G, Erdogan D (2007) Effects of taurine in cellular responses to oxidative stress in young and middle-aged rat liver. Ann N Y Acad Sci 1100:553–561
Zhou Z, Sun X, Kang YJ (2001) Ethanol-induced apoptosis in mouse liver: fas- and cytochrome c-mediated caspase-3 activation pathway. Am J Pathol 159:329–338
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Nos. 31772694, 31302051 and 31172285).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
Research involving human participants and/or animals
All procedures using animals were conducted according to the requirements of the Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals. The procedures were approved by the Animal Care and Use Committee of Shenyang Agricultural University (No. 20141013).
Rights and permissions
About this article
Cite this article
Wu, G., Yang, J., Lv, H. et al. Taurine prevents ethanol-induced apoptosis mediated by mitochondrial or death receptor pathways in liver cells. Amino Acids 50, 863–875 (2018). https://doi.org/10.1007/s00726-018-2561-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00726-018-2561-3