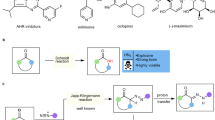
Abstract
A simple method has been developed for homocoupling of terminal alkynes bearing different functional groups by reaction with CuI/tribromoisocyanuric acid/piperidine in acetonitrile at room temperature. A telescoped approach based on Hunsdiecker/Cadiot–Chodkiewicz reactions for C(sp)-C(sp) cross-coupling was also presented.
Graphic abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Polyynes are a unique class of compounds in organic chemistry [1]. A subset of this large group, the 1,3-diyne moiety is fairly common in a variety naturally occurring molecules with biological properties [2, 3]. In addition to being useful synthetic building blocks, especially in polymer and supramolecular chemistry [4], these compounds have been used for the preparation of a wide range of other functionalities [5, 6]. Traditional preparation of 1,3-diynes involves the oxidative Glaser [7] and related [8,9,10] coupling reactions of terminal alkynes through copper acetylides [11], as shown in Scheme 1.

Due to the importance of carbon–carbon bond formation, many attempts have been made to improve the Glaser coupling reaction [12]. Solvent-free reactions [13] or environmentally benign solvents [14] gained considerable attention and have been increasingly reported. Other interesting approaches involve the use of different oxidants (e.g. I2 [15], NBS [16], α,α-dibromo-β-dicarbonyl compounds [17], 3,4-dihalo-2(5H)-furanones [18], among others), different metal catalysts, ligands and also utilization of alternative substrates [19].
Tribromoisocyanuric acid (1,3,5-tribromo-1,3,5-triazine-2,4,6-(1H,3H,5H)-trione, TBCA, Fig. 1) is an effective and stable electrophilic brominating reagent that can be easily prepared from readily accessible material (cyanuric acid, KBr, Oxone) [20]. From the green chemistry point of view, it presents a higher atom economy as being able to transfer up to three bromine atoms to a substrate corresponding to 66% of its mass [20]. In addition, in reactions involving TBCA, cyanuric acid by-product can be reused to produce more of trihaloisocyanuric acid [21].
Continuing our interest on the chemistry of tribromoisocyanuric acid [22,23,24,25], we wish to report here its use as a novel oxidant for the Glaser coupling reaction.
Results and discussion
Optimization studies were performed using ethynylbenzene as a model substrate for the Glaser homocoupling reaction using TBCA/CuI and different bases. The reactions were carried out in a 1 mmol scale using a molar ratio of 1.0:0.5:0.17 (base/CuI/TBCA) and led to 1,4-diphenylbuta-1,3-diyne (1a). The results, summarized in Table 1, clearly indicate that inorganic bases were ineffective for the Glaser homocoupling in the presence of CuI and tribromoisocyanuric acid, providing only trace of the product, whilst secondary cyclic amines are more effective for this transformation. The reaction proceeded smoothly at room temperature and the best results were obtained when piperidine or pyrrolidine was used as bases, which led to a quantitative conversion of ethynylbenzene to the corresponding 1,3-diyne. On the other hand, a reaction performed without any oxidative reagent gave a low yield (< 20%) of the diyne after 24 h in the presence of triethylamine as base.
Based on the above results, the optimized conditions were extended to different terminal alkynes and the scope of this protocol is shown in Table 2. Alkynes with different functional groups reacted in up to 40 min under mild conditions to give the corresponding symmetrical 1,3-diynes 1a–1g in good to excellent yields, including less acidic substrates (e.g. trimethylsilyl and alkyl terminal alkynes) and tertiary propargylic alcohols. Interestingly, the homocoupling reaction of 2-methylbut-3-yn-2-ol was also effective using water as solvent, but the corresponding diyne was obtained in a lower yield (87 vs. 70%). Although tribromoisocyanuric acid has been extensively reported as an efficient electrophilic brominating reagent for alkynes [25] and arenes [26], none of these brominated compounds were detected as side products by the analytical techniques employed. Rearrangements of tertiary propargylic alcohols were not observed too [27].
Further, we decided to investigate this new system to promote a cross-coupling of two different terminal alkynes. When equimolar quantities of the corresponding alkynes were used, a mixture of all possible three coupling products (two homocoupling and one cross-coupling) was obtained. In all cases, the cross-coupling products were the major constituents, but significant amounts of both homocoupling products were obtained (independently of the nature of substrate), which turned the purification of desired cross-coupling product difficult (Table 3).
Unfortunately, the chemoselectivity in Glaser cross-coupling reaction remains a challenge and effective methodologies for the preparation of unsymmetrical 1,3-diynes suffer from drawbacks, especially from the standpoint of starting materials, expensive catalysts or use of a large excess of the alkynes [28,29,30]. However, the selectivity towards unsymmetrical 1,3-diynes can be improved using the Cadiot–Chodkiewicz coupling reaction [31], wherein a terminal alkyne reacts with an alkynyl halide in the presence of a Cu(I), an organic base, and a reductive agent (frequently hydroxylamine hydrochloride). Therefore, we decided to investigate a telescoped approach (i.e. execution of multiple transformations, quenches and workup procedures without the isolation of intermediates [32]) for a one-pot halogenation and Cadiot–Chodkiewicz-type reaction to promote the cross-coupling of terminal alkynes with 1-bromoalkynes.
Previously, we showed that trihaloisocyanuric acids are effective reagents for the Hunsdiecker reaction (halodecaboxylation of carboxylic acids) and prepared styryl halides from cynnamic acids [33]. Initially, we investigated the bromodecarboxylation of phenylpropiolic acid mediated by TBCA to produce (bromoethynyl)benzene. The reaction was conducted upon treatment with TBCA (1 equiv) and a base (1 equiv). Organic bases (piperidine, DBU, and pyrrolidine) were ineffective to promote this reaction, producing a mixture of (bromoethynyl)benzene and ethynylbenzene in equal proportions. However, using Na2CO3 as base in the presence of a mixture of MeCN/H2O (1:1) produced (bromoethynyl)benzene (2) in 87% yield without further need of purification (Scheme 2).

With these results in hand, we decided to perform a telescoped reaction for the cross-coupling of terminal alkynes with 1-bromoalkynes generated in situ. Therefore, phenylpropiolic acid was treated with TBCA and Na2CO3 in aqueous acetonitrile (1:1) at room temperature. Further addition of terminal alkynes, BuNH2 (6 mol equiv), CuI (20 mmol%), and NH2OH∙HCl led to the desired cross-coupling product in high selectivity (Table 4).
The mechanism of the reaction is not clearly understood, however based on previous works [34,35,36], a plausible proposal involves two simultaneous pathways (Scheme 3). Initially, a copper–acetylene π-complex would activate the alkyne toward deprotonation. In pathway A, an oxidative addition promoted by TBCA would lead to an intermediate that collapses through a reductive elimination to give the 1,3-diyne. On the other hand, in pathway B, alkyne deprotonation would lead to 1-bromoalkyne via intermediacy of tribromoisocyanuric acid following a Cadiot–Chodkiewicz-type reaction (in an independent experiment, ethynylbenzene produced (bromoethynyl)benzene by reaction with TBCA in the presence of base). Therefore, in pathway A, tribromoisocyanuric acid may act as an oxidant, while in pathway B, it acts as an electrophilic brominating reagent.

Conclusion
In summary, we developed an efficient protocol for the high-yield synthesis of symmetrical 1,3-diynes using CuI/piperidine/TBCA system as a new approach to the Glaser coupling reaction. Broad substrate scope proved that these conditions are well tolerated by diverse functional groups. In addition, an alternative telescoped approach for a one-pot Hunsdiecker/Cadiot–Chodkiewicz-type reaction to promote the cross-coupling of terminal alkynes with 1-bromoalkynes was introduced. Experimental simplicity, mild conditions, short-time reactions, and readily available starting materials turn this method attractive for a wide range of applications in organic synthesis, supramolecular chemistry and material chemistry. Furthermore, to the best of our knowledge, our methodology is the first report of a C(sp)–C(sp) bond formation promoted by a trihaloisocyanuric acid.
Experimental
All chemicals and solvents were used as received. Tribromoisocyanuric acid was prepared as described [37]. NMR spectra were recorded on Bruker spectrometers using CDCl3 or acetone-d6 as solvents. IR spectra were recorded on a Nicolet 740 FT-IR spectrometers. GC–MS analyses were performed on a Shimadzu GC-MS-QP2010S gas chromatograph with electron impact (70 eV) using a 30 m DB-5 silica capillary column with 0.25 mm internal diameter and 0.25 μm phase thickness. Melting points were determined on a Laboratory Device Mel-Temp II and are corrected.
Typical procedure for homocoupling of terminal alkynes
To 1 mmol alkyne dissolved in 10 cm3 MeCN, 95 mg CuI (0.5 mmol), 85 mg piperidine (1 mmol), and 62 mg tribromoisocyanuric acid (0.17 mmol) were successively added. The mixture was stirred at room temperature and TLC monitored progress of the reaction. After completion of the reaction, the resulting mixture was filtered, 10 cm3 H2O was added and then the solution was extracted with 3 × 10 cm3 Et2O. The combined organic layers were washed with 10 cm3 water, 2 × 10 cm3 brine and dried over Na2SO4. The solvent was removed under reduced pressure, and the residue obtained was purified by column chromatography using EtOAc-hexanes as eluent to give the corresponding 1,3-diyne pure.
1,4-Diphenylbuta-1,3-diyne (1a)
Yield: 94%; m.p.: 83–84 °C (Ref [38]. 83–85 °C); the analytical data were found to be identical with those described in [39].
Dodeca-5,7-diyne (1b)
Yield: 97%; colorless oil; the analytical data were found to be identical with those described in [40].
1,4-Bis(trimethylsilyl)buta-1,3-diyne (1c)
Yield: 76%; m.p.: 106–107 °C (Ref [39]. 107–109 °C); the analytical data were found to be identical with those described in [39].
2,7-Dimethylocta-3,5-diyne-2,7-diol (1d)
Yield: 87%; m.p.: 127–129 °C (Ref [39]. 130–132 °C); the analytical data were found to be identical with those described in [41].
1,1′-(Buta-1,3-diyne-1,4-diyl)dicyclohexanol (1e)
Yield: 81%; m.p.: 173–174 °C (Ref [42]. 173–175 °C); the analytical data were found to be identical with those described in [41].
1,1′-(Buta-1,3-diyne-1,4-diyl)dicyclopentanol (1f)
Yield: 97%; m.p.: 133–134 °C (Ref [43]. 127–129 °C); the analytical data were found to be identical with those described in [41].
Hexa-2,4-diyne-1,6-diyldibenzoate (1g)
Yield: 90%; m.p.: 69–71 °C (Ref [44]. 74.8 °C); the analytical data were found to be identical with those described in [44].
Hunsdiecker reaction of phenylpropiolic acid: preparation of (bromoethynyl)benzene (2)
To 0.146 g phenylpropiolic acid (1 mmol) and 0.105 g Na2CO3 (1 mmol) dissolved in 10 cm3 MeCN/H2O (1:1), 0.125 g TBCA (0.34 mmol) was added. After 1 h stirring at room temperature, the reaction mixture was filtered, 5 cm3 H2O was added and the solution was extracted with 3 × 10 cm3 pentane. The combined organic layers were washed with 10 cm3 water, 2 × 10 cm3 brine and dried over Na2SO4. The solvent was removed under reduced pressure to give (bromoethynyl)benzene pure. Yield: 87%; colorless liquid; the analytical data were found to be identical with those described in [45].
References
Shun ALKS, Tykwinski RR (2006) Angew Chem Int Ed 45:1034
Zhou Z-F, Menna M, Cai Y-S, Guo Y-W (2015) Chem Rev 115:1543
Mao J, Li S, Zhong J, Wang B, Jin J, Gao Z, Yang H, Bian Q (2016) Tetrahedron Asymmetry 27:69
Shi W, Lei A (2014) Tetrahedron Lett 55:2763
Andrade CB, Carvalho DB, Trefzger OS, Kassab NM, Guerrero PG, Barbosa SL, Shiguemoto CYK, Baroni ACM (2019) Eur J Org Chem 696
Ötvös SB, Georgiádes A, Ozsvár D, Fülöp F (2019) RSC Adv 9:8197
Glaser C (1869) Ber Dtsch Chem Ges 2:422
Eglinton G, Galbraith AR (1956) Chem Ind 737
Hay AS (1962) J Org Chem 27:3320
Chodkiewicz W, Cadiot C (1955) C R Hebd Seances Acad Sci 241:1055
For a review on copper acetylides in organic synthesis, see: Adeleke AF, Brown APN, Cheng LJ, Mosleh KAM, Cordier CJ (2017) Synthesis 49:790
For a review on the Glaser coupling reaction, see: Sindhu KS, Anilkumar G (2014) RSC Adv 4:27867
Bettanin L, Botteselle GV, Godoi M, Braga AL (2014) Green Chem Lett Rev 7:105
Kusuda A, Xu X-H, Wang X, Tokunaga E, Shibate N (2011) Green Chem 13:843
Li D, Yin K, Li J, Jia X (2008) Tetrahedron Lett 49:5918
Li L, Wang J, Zhang G, Liu Q (2009) Tetrahedron Lett 50:4033
Fan X, Li N, Shen T, Cui X-M, Lv H, Zhu H-B, Guan Y-H (2014) Tetrahedron 70:256
Li JX, Liang HR, Wang ZY, Fu J-H (2011) Monatsh Chem 142:507
Kaldhi D, Vodnala N, Arup RG, Kabi K, Nayak S, Malakar CC (2020) Tetrahedron Lett 61:151775 (and references cited therein)
For a review on the chemistry of tribromoisocyanuric acid, see: de Almeida LS, Esteves PM, de Mattos MCS (2014) Curr Green Chem 1:94
Tozetti SDF, de Almeida LS, Esteves PM, de Mattos MCS (2007) J Braz Chem Soc 18:675
Sindra HC, dos Santos CVP, de Mattos MCS (2020) Lett Org Chem 17:586
de Andrade VSC, de Mattos MCS (2020) Tetrahedron Lett 61:152164
Crespo LTC, Senra MR, Esteves PM, de Mattos MCS (2019) Lett Org Chem 16:627
Sanabria CM, Costa BBS, Viana GM, de Aguiar LCS, de Mattos MCS (2018) Synthesis 50:1359
de Almeida LS, Esteves PM, de Mattos MCS (2006) Synthesis 221
Waminathan S, Narayanan KV (1971) Chem Rev 71:429
Yin W, He C, Chen M, Zhang H, Lei A (2008) Org Lett 11:709
Balaraman K, Kesavan V (2010) Synthesis 3461
Su L, Dong J, Liu L, Sun M, Qiu R, Zhou Y, Yin S-F (2016) J Am Chem Soc 138:12348
For a recent review on the Cadiot-Chodkiewicz reaction, see: Radhika S, Harry NA, Neetha M, Anilkumar G (2019) Org Biomol Chem 17:9081
Hayashi Y (2016) Chem Sci 7:866
Sodré LR, Esteves PM, de Mattos MCS (2013) J Braz Chem Soc 24:212
Rossi R, Carpita A, Bigelli C (1985) Tetrahedron Lett 26:523
Bohlmann F, Schönowsky H, Inhoffen E, Grau G (1964) Chem Ber 97:794
Jover J, Spuhler P, Zhao L, McArdle C, Maseras F (2014) Catal Sci Technol 4:4200
de Almeida LS, Esteves PM, de Mattos MCS (2006) Synlett 1515
Susanto W, Chu C-Y, Ang WJ, Chou TC, Lo L-C, Lam Y (2012) J Org Chem 77:2729
Tang S, Li L, Ren X, Li J, Yang G, Li H, Yuan B (2019) Green Chem 21:2899
Mo G, Tian Z, Li J, Wen G, Yang X (2015) Appl Organomet Chem 29:231
Kusuda A, Xu X-H, Wang X, Tokunaga E, Shibata N (2011) Green Chem 13:843
Li J-H, Liang Y, Zhang X-D (2005) Tetrahedron 61:1903
Adimurthy S, Malakar CC, Beifuss U (2009) J Org Chem 74:5648
Xu R, Gramlich V, Frauenrath H (2006) J Am Chem Soc 128:5541
Li M, Li Y, Zhao B, Liang F, Jin L-Y (2014) RSC Adv 4:30046
Acknowledgements
We thank CNPq and CAPES for financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
de Andrade, V.S.C., de Mattos, M.C.S. Tribromoisocyanuric acid as a useful oxidant for the synthesis of 1,3-diynes via Glaser coupling. Monatsh Chem 151, 1403–1408 (2020). https://doi.org/10.1007/s00706-020-02673-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00706-020-02673-8