Abstract
Background
To examine the factors contributing to persistent and recurrent hemifacial spasms (HFS) following a microvascular decompression (MVD) procedure and to suggest technical improvements to prevent such failures.
Methods
A retrospective review was conducted on fifty-two cases of repeat surgery. The extent of the previous craniotomy and the location of neurovascular compression (NVC) were investigated. The operative findings were categorized into two groups: “Missing Compression” and “Teflon Contact”. The analysis included long-term outcomes and operative complications after repeat MVD procedures.
Results
Missing compression was identified in 29 patients (56%), while Teflon contact was observed in 23 patients (44%). Patients with missing compression were more likely to experience improper craniotomy (66%) compared to those with Teflon contact (48%). Medially located NVC was a frequent finding in both groups, mainly due to compression by the anterior inferior cerebellar artery. In the missing compression group, during the repeat MVD, Teflon sling retraction was utilized in 79% of cases, while in the Teflon contact group, the most common procedure involved removing the Teflon in contact (65%). After the repeat MVD procedure, immediate spasm relief was achieved in 42 patients (81%), with six (12%) experiencing delayed relief. After a median follow-up of 54 months, 96% of patients were free from spasms. Delayed facial palsy, facial weakness, and hearing impairment were more frequently observed in the Teflon contact group.
Conclusions
A proper craniotomy that provides adequate exposure around the REZ is crucial to prevent missing the culprit vessel during the initial MVD procedure. Teflon contact on the REZ should be avoided, as it poses a potential risk of procedure failure and recurrence.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Microvascular decompression (MVD) is a surgical procedure that has been shown to be a potential curative treatment for hemifacial spasms (HFS) [2, 11,12,13, 25]. The offending vessel responsible for the neurovascular compression (NVC) is typically situated at the root exit zone (REZ) of the facial nerve [19]. Secure nerve decompression can relieve symptoms in most patients. The effectiveness of MVD at postoperative 1-year has been reported to exceed 90% following the initial surgical intervention [7]. However, even though repeat MVD after failure or recurrence has demonstrated efficacy, the underlying causes of failure can often be traced back to technical issues during the initial surgery [3, 6, 7, 15, 16, 27, 30]. These factors include missing the true offending vessel or improper use of Teflon. It is crucial to prevent these technical issues to avoid subjecting patients to potentially unnecessary re-explorations. Identifying and addressing these issues could potentially enhance the outcomes of the initial MVD procedures. This study aims to analyze radiological and surgical findings from repeat MVD procedures with long-term follow-up and provide recommendations for enhancing outcomes in initial MVD cases.
Methods
Patient cohort
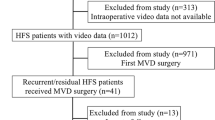
Among 1224 consecutive MVD procedures for HFS conducted at our institutes between April 2005 and March 2023, 62 patients (5.1%) underwent surgery due to failure or recurrence following prior MVD interventions. Of these, 52 patients (4.2%) with a one-year or longer follow-up period were included in this study (Fig. 1). Patient characteristics and information were gathered from their medical records. Recurrence was defined as the reappearance of facial spasms occurring more than 1 year after the initial MVD, while failure was defined as the absence of any improvement in facial spasms following the previous decompression procedure.
Imaging analyses
The assessment of preoperative computed tomography (CT) scans with bone window images aimed to ascertain the lateral and inferior extent of the previous craniotomy performed during the initial MVD. The primary focus was on the drilling procedure targeting the “inferior retrosigmoid point” [28]. This point is crucial as it marks the transition of the sigmoid sinus from its vertical to horizontal alignment at the lowest part of the sinus. A craniotomy reaching this inferior retrosigmoid point ensures secure exposure to both the lateral and inferior aspects of the posterior fossa. The previous craniotomies were characterized by two classifications: “Proper Craniotomy”, where the craniotomy reached the inferior retrosigmoid point, and the “Improper Craniotomy”, in which the inferior retrosigmoid point was not exposed (Fig. 2). Magnetic resonance imaging (MRI) with fast imaging employing steady-state acquisition (FIESTA) was used to measure the “Flocculus Height” (FH) value [8]. A line connecting the NVC to the medial edge of the sigmoid sinus is drawn to demonstrate the approach direction. The perpendicular length from the most distant point on the flocculus to the line is measured as the FH value, as described in our previous publication, enabling a simple estimation of the medial location of the NVC (Fig. 3).
Proper and improper craniotomy. a A three-dimensional computed tomography (CT) image of an “Improper Craniotomy” performed during the initial procedure. The craniotomy did not extend to the inferior retrosigmoid point (red point). The yellow dashed line represents the appropriate extent of the “Proper Craniotomy”. b An axial CT slice shows the location of the inferior retrosigmoid point, which corresponds to the medial and inferior border of the sigmoid sinus. c An intraoperative photograph showing the posterior border of the sigmoid sinus (blue dashed line) and the location of the inferior retrosigmoid point (red point), which is characterized by a triangular bony projection situated at the base of the posterior fossa. d A clear visualization of the neurovascular compression (asterisk) achieved with minimal cerebellar retraction via the proper craniotomy
Measurement of flocculus height (FH). The flocculus height (FH) is measured as the perpendicular length from the most distant point on the flocculus to the line (yellow) connecting the neurovascular compression (white arrowhead) and the medial edge of the sigmoid sinus (red arrowhead) (cited from reference [8])
Operative technique and grouping
All patients underwent surgery in the lateral position, with continuous monitoring of auditory brainstem evoked response (ABR) and the lateral spread response (LSR). ABR was employed to prevent hearing impairment during surgery, while LSR aimed to verify complete facial nerve decompression [14]. Stimulation electrodes were placed along the zygomatic branch of the facial nerve, with recording electrodes inserted in the orbicularis oris, the oculi, and the mentalis muscles. The facial nerve stimulator (NIMTM3.0, Medtronic, MN, USA) was utilized to trace the location of the facial nerve, particularly when dissecting excess Teflon. It was also used to confirm the extent of the REZ if it had been thoroughly decompressed. The operative scar from the previous surgery was incised to expose the existing craniotomy. In cases where the initial craniotomy was considered insufficient, the skin incision was extended to allow for additional drilling until reaching the inferior retrosigmoid point. The dura mater was incised to provide accesses both to the base and the lateral aspect of the posterior fossa. The spinal root of the accessory nerve was first identified, followed by extending the dissection rostrally to the exit of the lower cranial nerves. After exposing the lower cranial nerves, the choroid plexus was elevated to reach the REZ of the facial nerve beyond the glossopharyngeal nerve. If any offenders were found at the REZ, they were transposed using a Teflon sling or bridge, as described in our previous report [9]. In cases where a Teflon felt was placed on the REZ during the previous MVD, the Teflon mass was meticulously dissected and removed with a careful search for the location of the facial nerve using a nerve stimulator. The REZ was thoroughly identified with the stimulator to ensure it was free from any compressing object. The Teflon removal was not further pursued after the LSR disappeared, indicating successful decompression accomplished. The distal portion of the facial nerve was inspected to look for any vascular compression if the LSR persisted. The dura mater was reconstructed using a collagen matrix (DuraGen®, Integra Lifesciences, Plainsboro, NJ, USA) [10]. Calcium phosphate paste (BIOPEX-R®, HOYA Technosurgical, Tokyo, Japan) was used for cranioplasty in most patients.
The reasons for the repeated MVD were divided into two groups based on the findings of the REZ in the latest surgery (Fig. 1): (1) “Missing Compression” group with persistent arterial compression on the REZ, which constituted 29 patients; and (2) “Teflon Contact” group with either overpacking of Teflon, Teflon-induced adhesion, or a Teflon granuloma, which comprised 23 patients. Patients with improperly placed Teflon were categorized into the “Missing Compression” group because of the presence of a residual offender on the REZ.
Assessment of outcomes of repeat MVD
Postoperative neurological status after the repeat MVD procedures was assessed through evaluation at our clinics, telephone interviews, and mailed questionnaires for remote patients. Any neurological complications, including delayed facial palsy, postoperative facial weakness, hearing impairment, hoarseness, dysphagia, cerebrospinal fluid leakage, and infectious complications, were recorded throughout the follow-up period.
In describing demographic characteristics, p values were obtained by linear regression for continuous variables and the Mantel-Haenszel test for categorical variables. Each statistical test was set to be significant at p < 0.05 (two-sided p value). SAS software (version 9.4; SAS Institute, Inc., Cary, NC) was used for all statistical analyses.
Results
Patients’ characteristics and radiological measurement
The mean age of the patients undergoing a repeat MVD procedure was 55 years, varying from 28 to 76 years. Female and the right side of the face were predominantly affected. Failure cases following the initial MVDs were more common in the missing compression group (86%) compared to the Teflon contact group (70%). The mean duration of the reappearance of HFS after the initial MVD was 4 months, ranging from 0 to 24 months (surgery failure was recorded as 0 months). The median interval between the previous and the repeat procedures was 31 months, ranging from 0 to 277 months.
Radiological assessment of the previous craniotomy indicated that patients with improper craniotomies were more prevalent in the missing compression group (66%) than in the Teflon contact group (48%). The mean FH values were relatively high in both groups, with an average measurement of 9.7mm in the missing compression group and 9.9mm in the Teflon contact group. In comparison to the FH value distribution in our previous report, this suggests that NVC was located medially in both groups. The two groups had no statistically significant differences in patient characteristics and radiological measurements (Table 1).
Operative findings of repeat MVD
The most frequently missed artery was the anterior inferior cerebellar artery (AICA), followed by the posterior inferior cerebellar artery (PICA), the common trunk of AICA and PICA (APC), and the vertebral artery (VA). Three patients with missed compression exhibited distal NVC involving an AICA in the mid-cisternal portion, with no offending vessels identified at the REZ. VA and APC were more frequently observed in the missing compression group with improper craniotomies, occurring in 38% and 34% of the patients, respectively. In contrast, PICA was more frequently implicated in the Teflon contact group with excessive Teflon inserted in between. No statistically significant differences were observed in the operative findings.
In the missing compression group, Teflon sling retraction was the preferred technique used in 79% of the patients, while a Teflon bridge was employed in 21% of cases. In contrast, among the patients in the Teflon contact group, 65% underwent Teflon removal alone, while the remaining 35% required additional use of a Teflon sling or a bridge following the removal of the Teflon in contact. Statistical analyses indicated significant differences between the two groups with Teflon removal alone and necessity of another Teflon sling (p<0.0001).
A notable decrease in the amplitude of ABR to less than 50% of the control was more frequently observed in the Teflon contact group (30%) compared to the missing compression group (10%). This discrepancy can be attributed to the longer manipulation time required near the REZ during Teflon dissection (Table 2).
Surgical outcomes and complications
The follow-up period ranged from 12 to 175 months, with a median of 54 months. Immediate spasm relief was achieved in 42 patients (81%), with 23 (79%) in the missing compression group and 19 (83%) in the Teflon contact group. In contrast, delayed relief was observed in 6 patients (12%) overall, with a higher incidence in the missing compression group (14%) compared to the Teflon contact group (9%). The spasm-free rate at the final follow-up visit was 96%, with a rate of 97% in the missing compression group and 96% in the Teflon contact group.
Delayed facial palsy was identified in 3 patients (6%). All the three experienced complete resolution within 3 months. Immediate facial weakness occurred in 5 patients (10%), with four from the Teflon contact group, for whom an extensive dissection of the Teflon mass from the facial nerve was necessary. One patient in this group experienced permanent facial weakness. Additionally, two patients (4%) experienced CSF leakage and one (2%) developed meningitis. There were two cases of wound infection (4%) in total. No statistically significant differences were detected (Table 3).
Discussion
Considering re-exploration for persistent or recurrent facial spasms can be an unpleasant prospect for patients who have undergone an MVD surgery. While most patients benefit from the initial MVD, preventing failure and minimizing recurrence remain issues in this field. The literature shows an approximate 10% risk of failure or recurrence following an initial MVD [2, 19, 25]. This study revealed that the primary reason for requiring repeat surgery was the failure to identify the culprit vessel or improper Teflon placement, leading to further nerve irritation. Addressing these technical problems may improve the outcomes of the first surgery. While we did not observe any statistical differences, except for the decompression techniques, likely due to the relatively small number of cases, we can still draw some essential considerations from this study.
Our study demonstrated that a missing compression might frequently occur when the craniotomy is insufficient, and the NVC is located medially. These may cause a narrow operative field and imperfect observation around the REZ, resulting in missing the actual offenders [3, 6, 15, 16, 30]. Even in cases with improper craniotomy, a more aggressive cerebellar retraction may allow exposing the REZ [17]. Nevertheless, comprehensive observation can be accompanied by a decrease in amplitude and prolonged latency in ABR, which may be associated with the risk of hearing loss [20, 26]. The infrafloccular approach can be employed instead to achieve ample exposure of the REZ beyond the glossopharyngeal nerve, to minimize the retraction on the vestibulocochlear nerve [1, 4]. To enhance the benefits of this approach, it is crucial to position the craniotomy as low as possible towards the occipital condyle and ensure the exposure of the posterior border of the sigmoid sinus. This often requires drilling of the inferior retrosigmoid point [22, 28]. Inadequate exposure of the REZ due to a restricted operative field can lead to the oversight of the truly offending artery. Our previous study emphasized the significance of preoperative evaluation in expanding craniotomy laterally in cases with medially located NVC [8]. Measuring the flocculus height (FH) proves valuable predictor for assessing the challenges of maneuvering around the REZ in cases of medially located NVC within a limited operative field. In such situations, lateral mobilization of the sigmoid sinus can expand the operative field [21]. Alternatively, an endoscopic inspection can offer a clear visualization of the blind corner without retraction, thereby reducing the risks associated with brain retraction [5]. Using an endoscope provides an advantage over a microscope in visualizing structures and identifying neurovascular conflicts.
The insertion of a Teflon pledget between the offending vessels and the nerve root is a commonly employed decompression method known as an interposition technique. However, it is essential to note that excessive use of Teflon and the associated development of granulomas have been reported as potential causes of recurrent HFS in the long term [3, 6, 15, 16]. All cases involving Teflon revisions in our series utilized the interposition technique in the initial surgery. This suggests that any object in contact with the REZ could lead to treatment failure or recurrence. Determining the optimal quantity of Teflon that ensures sufficient nerve decompression without causing long-term harm is challenging. Hence, it is advisable to consider non-compressive techniques, such as sling retraction and the bridge technique, to minimize the risk of failure or recurrence [9, 18, 23, 24]. The surgical removal of previously inserted Teflon differs from the original MVD procedures. Shredded Teflon often cannot be extracted as a single mass due to its potential adherence to both the nerve root and the offending vessel. This situation carries the risk of causing newly developed neurological deficits. In our series, we frequently observed decreased ABR amplitude in patients requiring Teflon removal. This suggests a higher risk of hearing loss in those cases [20, 26].
The REZ is widely recognized as the most common site responsible for HFS [11,12,13, 25]. However, it is essential to acknowledge that there are reported instances of vascular contact at the distal root as a rare cause of HFS [29]. In our series, we identified three patients with distal NVC, even without any offending factors at the REZ. The contact between the AICA branch and the distal portion of the facial nerve is a common finding in normal anatomy. While performing decompression on the distal portion during every initial case is impractical due to the associated risk of hearing disturbance, exploration should be considered, particularly when the LSR persists despite achieving adequate decompression at the REZ.
This study has several limitations. Firstly, its retrospective design with a relatively small number of patients could impact the robustness of the findings. Secondly, the reliability of the grouping of the patients based on the findings in the surgery may need to be questioned. A study involving a larger patient cohort and a more extended follow-up period is warranted to solidify and validate our conclusions.
Conclusion
A repeat MVD surgery is effective for patients with persistent facial spasms, but the causes could often have been prevented in the initial surgery. Improper craniotomy may cause overlooking the offender due to insufficient exposure of the facial nerve REZ. To prevent treatment failure or recurrence, it is essential to avoid any contact between Teflon and the REZ. Additionally, the compression of the distal nerve root should be carefully considered as a potential cause of treatment failure.
Data availability
Data is available upon reasonable request.
Code availability
Not applicable.
References
Amagasaki K, Nishimura S, Uchida T, Tatebayashi K, Nakaguchi H (2021) Infrafloccular approach effectively prevents hearing complication in microvascular decompression surgery for hemifacial spasm. Br J Neurosurg 8:1–4
Barker FG 2nd, Jannetta PJ, Bissonette DJ, Shields PT, Larkins MV, Jho HD (1995) Microvascular decompression for hemifacial spasm. J Neurosurg 82:201–210
Bigder MG, Kaufmann AM (2016) Failed microvascular decompression surgery for hemifacial spasm due to persistent neurovascular compression: an analysis of reoperations. J Neurosurg 124:90–95
Bozkurt B, Kalani MYS, Yağmurlu K, Belykh E, Preul MC, Nakaji P, Spetzler RF (2018) Low retrosigmoid infratonsillar approach to lateral medullary lesions. World Neurosurg 111:311–316
El Refaee E, Fleck S, Matthes M, Marx S, Baldauf J, Schroeder HWS (2021) Outcome of endoscope-assisted microvascular decompression in patients with hemifacial spasm caused by severe indentation of the brain stem at the pontomedullary sulcus by the posterior inferior cerebellar artery. Oper Neurosurg (Hagerstown) 20:399–405
Engh JA, Horowitz M, Burkhart L, Chang YF, Kassam A (2005) Repeat microvascular decompression for hemifacial spasm. J Neurol Neurosurg Psychiatry 76:1574–1580
Holste K, Sahyouni R, Teton Z, Chan AY, Englot DJ, Rolston JD (2020) Spasm freedom following microvascular decompression for hemifacial spasm: systematic review and meta-analysis. World Neurosurg 139:e383–e390
Inoue T, Goto Y, Shitara S, Keswani R, Prasetya M, Arham A, Kikuta K, Radcliffe L, Friedman AH, Fukushima T (2022) Indication for a skull base approach in microvascular decompression for hemifacial spasm. Acta Neurochir 164:3235–3246
Inoue T, Shitara S, Goto Y, Arham A, Prasetya M, Radcliffe L, Fukushima T (2021) Bridge technique for hemifacial spasm with vertebral artery involvement. Acta Neurochir 163:3311–3320
Inoue T, Shitara S, Shima A, Goto Y, Fukushima T (2021) Double collagen matrix grafting for dural closure in microvascular decompression: an alternative use of autologous fascial grafting. Acta Neurochir 163:2395–2401
Jannetta PJ (1975) The cause of hemifacial spasm: definitive microsurgical treatment at the brainstem in 31 patients. Trans Sect Otolaryngol Am Acad Ophthalmol Otolaryngol 80:319–322
Jannetta PJ (1983) Hemifacial spasm: treatment by posterior fossa surgery. J Neurol Neurosurg Psychiatry 46:465–466
Jannetta PJ, Abbasy M, Maroon JC, Ramos FM, Albin MS (1977) Etiology and definitive microsurgical treatment of hemifacial spasm. Operative techniques and results in 47 patients. J Neurosurg 47:321–328
Jeon C, Jung NY, Kim M, Park K (2023) Intraoperative monitoring of the facial nerve during microvascular decompression for hemifacial spasm. Life (Basel) 13:1616
Jiang C, Xu W, Dai Y, Lu T, Jin W, Liang W (2017) Failed microvascular decompression surgery for hemifacial spasm: a retrospective clinical study of reoperations. Acta Neurochir 159(2):259–263
Lee S, Park SK, Lee JA, Joo BE, Park K (2019) Missed culprits in failed microvascular decompression surgery for hemifacial spasm and clinical outcomes of redo surgery. World Neurosurg 129:e627–e633
Liebelt BD, Huang M, Britz GW (2018) A comparison of cerebellar retraction pressures in posterior fossa surgery: extended retrosigmoid versus traditional retrosigmoid approach. World Neurosurg 113:e88–e92
Matsushima T, Yamaguchi T, Inoue TK, Matsukado K, Fukui M (2000) Recurrent trigeminal neuralgia after microvascular decompression using an interposing technique. Teflon felt adhesion and the sling retraction technique. Acta Neurochir 142:557–561
Mercier P, Sindou M (2018) The conflicting vessels in hemifacial spasm: Literature review and anatomical-surgical implications. Neurochirurgie 64:94–100
Polo G, Fischer C, Sindou MP, Marneffe V (2004) Brainstem auditory evoked potential monitoring during microvascular decompression for hemifacial spasm: intraoperative brainstem auditory evoked potential changes and warning values to prevent hearing loss--prospective study in a consecutive series of 84 patients. Neurosurgery 54:97–104
Quiñones-Hinojosa A, Chang EF, Lawton MT (2006) The extended retrosigmoid approach: an alternative to radical cranial base approaches for posterior fossa lesions. Neurosurgery 58:208–214
Raso JL, Gusmão SN (2011) A new landmark for finding the sigmoid sinus in suboccipital craniotomies. Neurosurgery 68:1–6
Rawlinson JN, Coakham HB (1988) The treatment of hemifacial spasm by sling retraction. Br J Neurosurg 2:173–178
Sindou M, Leston JM, Decullier E, Chapuis F (2008) Microvascular decompression for trigeminal neuralgia: the importance of a noncompressive technique--Kaplan-Meier analysis in a consecutive series of 330 patients. Neurosurgery 63:341–350
Sindou M, Mercier P (2018) Microvascular decompression for hemifacial spasm: outcome on spasm and complications. A review. Neurochirurgie 64:106–116
Sindou M, Mercier P (2018) Microvascular decompression for hemifacial spasm: Surgical techniques and intraoperative monitoring. Neurochirurgie 64:133–143
Wang X, Thirumala PD, Shah A, Gardner P, Habeych M, Crammond D, Balzer J, Burkhart L, Horowitz M (2013) Microvascular decompression for hemifacial spasm: focus on late reoperation. Neurosurg Rev 36:637–643
Wanibuchi M, Fukushima T, Zenga F, Friedman AH (2009) Simple identification of the third segment of the extracranial vertebral artery by extreme lateral inferior transcondylar-transtubercular exposure (ELITE). Acta Neurochir 151:1499–1503
Xu X, Su S, Cai L, Wang L, Fan H, Wu C, Chen Y (2022) Hemifacial spasm caused by distal neurovascular compression confirmed by lateral spread response monitoring. J Craniofac Surg 3:2555–2559
Zhong J, Li ST, Zhu J, Guan HX, Zhou QM, Jiao W, Ying TT, Yang XS, Zhan WC, Hua XM (2012) A clinical analysis on microvascular decompression surgery in a series of 3000 cases. Clin Neurol Neurosurg 114:846–851
Author information
Authors and Affiliations
Contributions
Study concept and design: Takuro Inoue. Data collection and analysis: Takuro Inoue, Yukihiro Goto, Yasuaki Inoue, Mustaqim Prasetya, and Takanori Fukushima. Data interpretation: Peter Adidharma. Writing the manuscript: Takuro Inoue. Reviewing and editing: Yukihiro Goto, Yasuaki Inoue, and Mustaqim Prasetya. Supervision: Takanori Fukushima. All authors approved the final version of the submitted manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Inoue, T., Goto, Y., Inoue, Y. et al. Potential reasons for failure and recurrence in microvascular decompression for hemifacial spasm. Acta Neurochir 165, 3845–3852 (2023). https://doi.org/10.1007/s00701-023-05861-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-023-05861-7