Abstract
Background
Although measurement of intracranial pressure (ICP) has occasionally been utilized in children with craniosynostosis (CSS), data on parameters of pulsatile ICP in CSS are still lacking, and the role of pulsatile ICP measurements in the management of CSS is not well established.
Methods
From our department’s database, we retrieved the data from children in whom the measurement of static and pulsatile ICP was a part of the diagnostic work-up in different clinical situations related to CSS. Both clinical and ICP data were retrospectively reviewed and analyzed.
Results
We identified 49 children with CSS, median age 4.4 years (range 0.2–18.9), in whom a total of 67 diagnostic ICP measurements were undertaken between 2002 and 2014. The CSS was syndromal in 23 cases. The rationale for ICP measurement was a question of indication for cranial vault expansion surgery (CVES) in 12 patients (Group 1), of its timing in 10 patients (Group 2), of suspected abnormally elevated ICP or hydrocephalus in 11 patients (Group 3), of indication for repeated CVES in 13 patients (Group 4), or shunt dysfunction in three patients (Group 5). The average mean ICP for the whole cohort was 15.1 ± 5.5 mmHg and mean wave amplitude (MWA) 5.3 ± 2.2 mmHg. There was no significant difference in ICP parameters when compared between Groups 1–5. Fundoscopy revealed papilledema in five out of 32 children (15.6%). There were significantly higher parameters of pulsatile ICP (MWA) in patients with papilledema, but no statistically significant difference in parameters of static ICP.
Conclusions
In this cohort of pediatric patients with CSS presenting with various diagnostic challenges, we found the diagnostic measurement of static and pulsatile ICP useful in selecting the optimal treatment modality and timing of surgery. Papilledema was associated with elevated pulsatile ICP, a parameter that in previous studies has been shown to correlate with impaired intracranial compliance.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Craniosynostosis (CSS), a condition first described by Sömmerring in 1791 [1], results from premature closure of calvarial sutures and presents clinically most often in newborns and small infants. The most common forms of CSS are the sagittal (scaphocephaly), coronal (plagiocephaly), and metopic (trigonocephaly) single-suture synostoses, while the complex cases of multiple-suture CSS are less common and often associated with specific craniofacial conditions such as Apert, Crouzon, and Pfeiffer syndromes [2].
CSS leads to the limitation of normal growth of the skull and hence the restriction of the intracranial volume (ICV). In healthy children, ICV doubles from birth by 9 months of age and triples by 6 years of age [3], thus enabling normal development of the brain. In untreated CSS and as ICV becomes reduced, the brain growth is restricted and its development is threatened by an increase of the intracranial pressure (ICP). This may result in delayed psychomotor development, visual impairment, and ultimately severe brain damage. The combination of CSS and obstructive sleep apnea or hydrocephalus [4], which also may contribute to pathologically increased ICP, may be even more critical in preventing the brain from normal development.
Cranial vault expansion surgery (CVES) is necessary if normal brain growth is at risk. The goal of CVES is then to expand the ICV in order to prevent damage to the growing brain. The understanding of the association between ICV and ICP in children with CSS is therefore of utmost clinical importance.
In a recent study, we found no association between ICV and ICP values prior to CVES, nor any association between change in ICV and ICP values after CVES [5]. These results, as well as the observations made by others [6, 7], suggest that the ICV is no reliable predictor of the ICP, meaning that intracranial hypertension in CSS may not be caused by reduced ICV alone but rather by a distorted relationship between ICV and the volume of intracranial contents (brain tissue, cerebrospinal fluid, and blood in intracranial vessels). Alterations of static ICP in CSS have been described in several studies [6,7,8,9,10,11,12,13], as well as their effect on cerebral perfusion pressure [14]. However, several lines of evidence suggest that intracranial compliance (ICC) is better predicted by the pulsatile ICP [15,16,17], which alterations in the setting of CSS have been explored in only a few studies so far [5, 18].
While the surgical treatment of simple single-suture, non-syndromic CSS in small infants is technically straightforward and the results are generally favorable [19], some children may develop a complex CSS condition requiring a more intricate treatment strategy. In clinical practice, this latter subgroup of patients has proven to be particularly challenging. Besides CVES, other surgical interventions may be necessary: repeated CVES, decompressive subtemporal craniectomy, and also diversional procedures of the cerebrospinal fluid (CSF) such as ventriculo-peritoneal (or atrial) shunt, endoscopic third ventriculostomy, or fenestration of lamina terminalis .
At our institution, we have for many years performed diagnostic overnight measurement of static and pulsatile ICP in selected and more complex cases of children with CSS prior to determining the indication for corrective surgery with CVES or sometimes for repeated CVES. Furthermore, we have utilized diagnostic ICP measurement in patients suffering from a combination of CSS and underlying hydrocephalus and/or suspected intracranial hypertension. The ultimate goal of utilizing overnight ICP measurement in patients with CSS is to either strengthen the indication for surgical treatment in doubtful cases or—equally importantly—to prevent patients from being exposed to unnecessary surgical procedures.
In this retrospective descriptive study, we conducted an analysis of ICP measurements performed in patients with CSS in these different clinical situations. The goal of the study was to systematically review our experience and evaluate the value of measuring both static and pulsatile ICP in challenging cases of CSS.
Methods
Approvals and ethical considerations
The study was approved by the Oslo University Hospital as a quality control study (approval no. 2015/15993).
Patient sample
From the database of diagnostic ICP measurements performed at the Department of Neurosurgery, Oslo University Hospital–Rikshospitalet in the period from January 2002 to November 2014, we retrospectively identified all children with CSS who had undergone diagnostic ICP measurement. Criteria for inclusion in the present study were as follows: all children under 19 years of age with clinically and radiologically verified CSS who underwent diagnostic ICP measurement and whose raw-data from ICP measurements were found digitally stored in the department’s database. We also included children who underwent surgery related to CSS prior to the first ICP measurement. In all patients, the ICP scores were analyzed and patient charts reviewed in order to obtain clinical details.
The children had undergone ICP measurement as a part of diagnostic workup when the indication for treatment was considered. The patients could be divided into five groups according to the indication for ICP measurement:
-
1)
Those with evident CSS but an uncertain indication for CVES and in whom abnormal ICP could potentially strengthen the indication;
-
2)
Those with CSS and relatively clear indication for surgical correction, but with the question of optimal timing of CVES;
-
3)
Those with clinical signs indicative of abnormal ICP and in whom some form of CSS also was present;
-
4)
Those with previously performed CVES but still persistent or progressive clinical signs indicative of abnormal ICP and hence the question of the need for repeated CVES;
-
5)
Those with previously performed CSF diversion and present CSS condition, and in whom shunt dysfunction could be clinically suspected.
Measurements of pulsatile and static ICP
ICP was measured invasively. Usually in general anesthesia, a cranial burr hole was placed in front of the coronal suture, most often on the right side. The ICP sensor (MicroSensor; Codman, Johnson & Johnson) was calibrated in a standard manner and placed 1–2 cm into the brain parenchyma. The monitoring of ICP then continued overnight, lasting usually for 16–24 h. After finishing the recording, the ICP sensor was withdrawn.
Analysis of pressure measurements
The continuous ICP waveforms were sampled at 100–200 Hz and stored on a hospital server. The automatic algorithm for ICP analysis incorporated in the dedicated software (Sensometrics Software; dPCom) was used. This software identifies physiological waves in the continuous ICP waveform and ignores the artificial pressure waves caused by noise in the signal. Each ICP wave accepted for further analysis is characterized by the amplitude (pulse amplitude dP [pressure difference between diastolic minimum pressure and systolic maximum pressure]), rise time (dT [time interval from beginning diastolic pressure to systolic pressure]), and rise time coefficient (dP/dT). For ICP waves with multiple subpeaks, the software selects the largest peak. For each consecutive 6-s time window, the following ICP parameters are then determined: the mean ICP (averaged ICP over the 6-s time window, representing the static ICP), the mean ICP wave amplitude (MWA; averaged ICP pulse amplitude [dP] over the 6-s time window, representing the pulsatile ICP), the mean ICP wave rise time (averaged pulse rise time (dT) over the 6-s time window), and the mean ICP wave rise time coefficient (averaged pulse rise time coefficient [dP/dT] over the 6-s time window). For comparison of pressure values among individuals, we selected a standardized recording time from 11 PM to 7 AM. During this time, the children were normally asleep.
Threshold levels for pulsatile and static ICP scores
As ICP measurement requires an invasive procedure, data from ICP measurements in healthy individuals cannot be obtained. Based on our own experience with monitoring pulsatile and static ICP in populations of patients of different age and various conditions affecting the central nervous system [16,17,18, 20], we therefore use the following thresholds when considering averaged overnight recordings: MWA < 4 mmHg (below threshold), 4–5 mmHg (borderline threshold), and > 5 mmHg (above threshold); mean ICP < 10 mmHg (below threshold), 10–15 mmHg (borderline threshold), and > 15 mmHg (above threshold).
Statistical analysis
Statistical analyses were performed using the SPSS software (version 20.0 for Windows; IBM Corp.). Differences between continuous data were determined using independent-sample t tests, or by ANOVA with Bonferroni corrected post hoc tests when multiple comparisons were performed. Statistical significance was accepted at the 0.05 level (two-tailed).
Results
Forty-nine patients met the inclusion criteria. There were 34 boys and 15 girls (ratio 2.3:1), and the median age at the time of the first ICP measurement was 4.4 years (range 0.2–18.9 years). Head circumference at the time of the first ICP measurement was median 49.4 cm (range 39–58.5 cm), i.e., median 10-percentile (range from 4.5 cm below 2.5-percentile to 3.5 cm over 97.5-percentile).
Craniosynostosis was non-syndromal in 26 cases (53%), involving simple (n = 19: sagittal in 14, metopic in three, and coronar in two cases, respectively) or multiple sutures (n = 7). In 23 cases (47%), the craniosynostosis was syndromal in association with genetically verified Apert (n = 5), Crouzon (n = 6, one of which combined with Marfan), Pfeiffer (n = 3), and CHARGE (n = 1) syndromes, while syndrome could not be specified in another eight cases.
Reliable data on clinical signs of symptoms were available from 42 out of 49 patients and are listed in Table 1. The most frequent symptoms at the time of the first ICP measurement were delayed psychomotor development in 26 (62%), headache in 21 (50%), and sleep disturbance in 19 (45%) patients, respectively.
According to CT or MRI, the median value of Evans’ index (images available for measuring in 45 out of 49 patients) was 0.28 (range 0.18–0.50).
Twenty-six (53%) of patients had already undergone at least one surgery before the first ICP measurement: CVES in 25 patients and CSF diversion (VP-shunt) in four patients, respectively.
The rationale for diagnostic ICP measurement was as follows: the question of indication for CVES in 12 patients (25%; Group 1 in Table 2), of its timing in 10 patients (20%, Group 2), of suspected abnormally elevated ICP or hydrocephalus in 11 patients (22%, Group 3), of indication for repeated CVES in 13 patients (n = 27%, Group 4), or shunt dysfunction in three patients (6%, Group 5).
As ICP data from four patients were not digitally stored, recording data from only 45 patients were analyzed. The findings from ICP measurements are listed in Table 2. The average mean ICP for the whole cohort was 15.1 ± 5.5 mmHg and MWA 5.3 ± 2.2 mmHg. There was no significant difference in ICP parameters when compared between Groups 1–5.
Based on finding from the first ICP measurement, a conservative strategy was chosen in 24 patients (49%) while an indication for CVES was found in another 19 patients (39%). Three patients (6%) underwent CSF diversion: ventriculoperitoneal (VP) shunt in two and endoscopic third ventriculostomy (ETV) in one patient. Two patients (4%) were treated with CVES along with CSF diversion (VP shunt), and one patient (2%) with pre-existing VP shunt underwent shunt revision (Fig. 1).
A flow-chart schematically illustrating the rationale for diagnostic ICP measurement (red squares) as well as its clinical consequences (blue squares) in the present cohort of patients. The thickness of arrows corresponds graphically with the number of patients in each subgroup. One patient had a total of six ICP measurements (only four of them are presented in this figure)
Eleven patients (22.5%) underwent second ICP measurements after initial treatment or expectancy. As a result of repeated ICP measurement, another two patients received VP shunt and three patients were operated on with CVES (which was a repeated procedure in two of these cases). The third ICP measurement was undertaken in five patients (10%), the fourth in two (4%), while one patient (2%) underwent six ICP measurements, respectively. The rationale for and clinical impact of these measurements is schematically showed in Fig. 1 for illustration.
Thus, a total of 67 diagnostic ICP measurements were undertaken. We experienced complications related to invasive ICP measurement in two (4%) patients (i.e., in 3% of all procedures): a CSF leakage in one and meningitis with encephalitis and sepsis due to multiresistant Streptococcus pneumoniae in another patient. In the latter case, the infection occurred 15 days after the procedure.
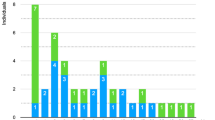
Fundoscopy prior to the first ICP measurement was undertaken by an ophthalmologist in 32 patients (65%), and papilledema was found in five of them (16%). In 31 of these patients, in whom ICP data were available, there were significantly higher parameters of pulsatile ICP (average MWA or percentage of MWA above 5 mmHg) in patients with papilledema, but no statistically significant difference in parameters of static ICP (Fig. 2).
For individuals without (No, n = 31) or with (Yes, n = 5) papilledema, the differences in a average of MWA, b percentage of MWA > 5 mmHg, c average of mean ICP, and d percentage of mean ICP > 15 mmHg are presented. Error bars with 95% confidence intervals are presented, including significance levels as determined by independent samples t test
Discussion
The main finding from this study was that in this cohort of CSS patients with various diagnostic challenges, the measurement of static and pulsatile ICP added diagnostic information that was useful in the clinical decision making. Notably, papilledema was not associated with increased static ICP, but with increased pulsatile ICP.
It is important to stress that the present patient cohort does not represent a CSS population in general, but only a minor part—below 20%—of approximately three hundred patients treated at our department for CSS in the same period. The majority of children with CSS would never require ICP measurement as the indication for CVES may be based on the clinical assessment alone. The present patient cohort is therefore a selected group of patients, in whom the indication for CVES (or repeated CVES) was doubtful, and/or in whom a component of underlying hydrocephalus and/or abnormal ICP might be suspected.
The main rationale for evaluation of ICP in these cases was to obtain an optimal basis in the decision making regarding indication and timing for surgery, i.e., to ensure that the child does not need CVES or CSF diversion or— equally importantly—that the patient would not be exposed to the risk of unnecessary surgical procedures when the indication was uncertain. This rationale also justified a risk of complications potentially related to invasive ICP measurement, which was quite low in our series (3% of all procedures).
As shown in Fig. 1, the children undergoing the first ICP measurement with the question of “need for CVES” (Group 1 in Table 2) ended up with conservative strategy equally often as with surgery (6:6), while in those with the question of the need for “repeated CVES” (Group 4), we tended to choose more often the conservative strategy (8:5). On the other hand, there was a clear majority of patients with the question of “timing of CVES” (Group 2) who proceeded to surgical treatment (9:1) after their diagnostic ICP measurement. These numbers point at valuable diagnostic information retrieved from ICP measurements, including analysis of the pulsatile ICP.
The main goal of the treatment of CSS is to prevent damage to the developing brain potentially caused by suspended skull growth and compromised ICV. Since ICV may not reliably predict the ICP [5,6,7], the measurement of ICP may be beneficial in selected cases. In contrary to the measurement of static ICP (i.e., the mean ICP) [6,7,8,9,10,11,12,13,14], the analysis of pulsatile ICP (i.e., the mean wave amplitude, MWA) has proven to better indicate the intracranial compliance (ICC) [15,16,17] in different neurosurgical conditions. Alterations of pulsatile ICP in the setting of CSS, however, have previously been explored in only a few studies [5, 18]. As illustrated in Table 3, the parameters of pulsatile ICP are abnormally elevated despite normal static ICP in most of the other non-acute neurosurgical conditions. However, in patients with CSS as well as in those with idiopathic intracranial hypertension (IIH), also static ICP usually tends to be higher, illustrating a more dramatic impact of reduced ICV (in CSS) or engorged brain (in IIH).
Radiologically definable hydrocephalus is only rarely observed in patients with non-syndromic CSS, in whom it usually is a coincidental condition, while it is more common in syndromic CSS [4]. Furthermore, it has been shown that parameters of neither static nor pulsatile ICP correlate with linear measures of ventricular size [18]. Fundoscopy with the assessment of papilledema has therefore traditionally been used in order to indirectly confirm or rule out abnormally elevated ICP. It was performed in only two thirds (65%) of patients in our series, and papilledema suggesting elevated ICP was found in 16% of them, similar to the finding of Tuite et al. [13] (12%). Important to note from the latter study, however, is that although papilledema was a highly specific indicator of elevated ICP, its sensitivity was age-dependent: while it was 100% sensitive in children older than 8 years, it indicated elevated (static) ICP in only 22% of younger children. Thus, the presence of papilledema reliably indicates elevated ICP but its absence does not rule out elevated ICP. Therefore, diagnostic ICP measurement has a role in demonstrating elevated ICP when it is clinically suspected, but not confirmed by fundoscopy. Furthermore and as observed in patients with idiopathic intracranial hypertension (IIH) [16] as well as in our previous study on pediatric patients [18], all patients with papilledema had abnormally elevated MWA, while some of them could have normal mean ICP. This is again confirmed in the present study, as there was no significant difference in parameters of static ICP between those with and without papilledema, but significantly higher parameters of pulsatile MWA (Fig. 2) in patients with papilledema.
Some limitations of the present study should be noted. The study design is retrospective with its inherent limitations. Moreover, the use of diagnostic ICP measurement in the work-up of children with CSS as presented here relied on individual considerations in each patient. Furthermore, the role of disturbed CSF circulation and its relation to CSS may be discussed. Four patients from the present cohort had a pre-existing CSF diversion (i.e., a ventriculoperitoneal shunt) at the time of the first ICP measurement, which was undertaken because of suspected shunt dysfunction in three and to assess the need for CVES in one patient, respectively. The inclusion of these patients in the whole cohort may appear controversial as CSF diversion could potentially have an impact on ICP values measured.
Finally, the present age distribution of CSS patients is not representative of today’s surgical management of CSS. During the study period (2002–2014), most of the typical CSS cases were operated around 7–12 months of age at the earliest, as CVES still was considered a demanding procedure not very well suitable for the youngest children due to expected blood loss and other specific risks. Hence, the median age of the present cohort was 4.4 years. Since 2013, we have utilized a technique of minimal invasive CVES with subsequent treatment with head orthosis [19, 24], which has allowed for CVES being performed in children just a few weeks or months old, before the issue of abnormally elevated ICP becomes clinically relevant. For this reason, the need for diagnostic ICP measurements in non-syndromic CSS has fallen significantly over the last few years and is now used only in specific, mostly syndromic cases.
Conclusion
In this cohort of pediatric patients with CSS presenting various diagnostic challenges, we found the diagnostic measurement of pulsatile and static ICP useful in selecting the optimal treatment modality and timing of surgery. Papilledema was associated with elevated pulsatile ICP, a parameter that in previous studies has been shown to correlate with impaired intracranial compliance.
References
Sömmerring ST (1839) Vom Bau des menschlichen Körpers. Bischoff, Henle u. a, Lepizig
Tuite GF, Chong WK, Evanson J, Narita A, Taylor D, Harkness WF, Jones BM, Hayward RD (1996) The effectiveness of papilledema as an indicator of raised intracranial pressure in children with craniosynostosis. Neurosurgery 38:272–278
Kamdar MR, Gomez RA, Ascherman JA (2009) Intracranial volumes in a large series of healthy children. Plast Reconstr Surg 124:2072–2075
Collmann H, Sorensen N, Krauss J (2005) Hydrocephalus in craniosynostosis: a review. Childs Nerv Syst 21:902–912
Langvatn EA, Frič R, Due-Tønnessen BJ, Eide PK (2019) Intracranial volume versus static and pulsatile intracranial pressure values in children with craniosynostosis. J Neurosurg Pediatrics 24:66–74
Fok H, Jones BM, Gault DG, Andar U, Hayward R (1992) Relationship between intracranial pressure and intracranial volume in craniosynostosis. Br J Plast Surg 45:394–397
Gault DT, Renier D, Marchac D, Jones BM (1992) Intracranial pressure and intracranial volume in children with craniosynostosis. Plast Reconstr Surg 90:377–381
Eide PK, Helseth E, Due-Tonnessen B, Lundar T (2002) Assessment of continuous intracranial pressure recordings in childhood craniosynostosis. Pediatr Neurosurg 37:310–320
Judy BF, Swanson JW, Yang W, Storm PB, Bartlett SP, Taylor JA, Heuer GG, Lang SS (2018) Intraoperative intracranial pressure monitoring in the pediatric craniosynostosis population. J Neurosurg Pediatrics 22:475–480
Renier D, Sainte-Rose C, Marchac D, Hirsch JF (1982) Intracranial pressure in craniostenosis. J Neurosurg 57:370–377
Thompson DN, Harkness W, Jones B, Gonsalez S, Andar U, Hayward R (1995) Subdural intracranial pressure monitoring in craniosynostosis: its role in surgical management. Childs Nerv Syst 11:269–275
Thompson DN, Malcolm GP, Jones BM, Harkness WJ, Hayward RD (1995) Intracranial pressure in single-suture craniosynostosis. Pediatr Neurosurg 22:235–240
Tønne E, Due-Tønnessen BJ, Wiig U, Stadheim BF, Meling TR, Helseth E, Heimdal KR (2020) Epidemiology of craniosynostosis in Norway. J Neurosurg Pediatr:1–8. https://doi.org/10.3171/2020.1.PEDS2051
Hayward R, Gonsalez S (2005) How low can you go? Intracranial pressure, cerebral perfusion pressure, and respiratory obstruction in children with complex craniosynostosis. J Neurosurg 102:16–22
Eide PK (2016) The correlation between pulsatile intracranial pressure and indices of intracranial pressure-volume reserve capacity: results from ventricular infusion testing. J Neurosurg 125:1493–1503
Eide PK, Kerty E (2011) Static and pulsatile intracranial pressure in idiopathic intracranial hypertension. Clin Neurol Neurosurg 113:123–128
Eide PK, Sorteberg W (2010) Diagnostic intracranial pressure monitoring and surgical management in idiopathic normal pressure hydrocephalus: a 6-year review of 214 patients. Neurosurgery 66:80–91
Eide PK, Egge A, Due-Tønnessen BJ, Helseth E (2007) Is intracranial pressure waveform analysis useful in the management of pediatric neurosurgical patients? Pediatr Neurosurg 43:472–481
Proctor MR, Meara JG (2019) A review of the management of single-suture craniosynostosis, past, present, and future. J Neurosurg Pediatrics 24:622–631
Frič R, Eide PK (2017) Comparative observational study on the clinical presentation, intracranial volume measurements, and intracranial pressure scores in patients with either Chiari malformation type I or idiopathic intracranial hypertension. J Neurosurg 126:1312–1322
Eide PK (2018) The pathophysiology of chronic noncommunicating hydrocephalus: lessons from continuous intracranial pressure monitoring and ventricular infusion testing. J Neurosurg 129:220–233
Frič R, Eide PK (2015) Comparison of pulsatile and static pressures within the intracranial and lumbar compartments in patients with Chiari malformation type 1: a prospective observational study. Acta Neurochir 157:1411–1423
Saehle T, Eide PK (2015) Characteristics of intracranial pressure (ICP) waves and ICP in children with treatment-responsive hydrocephalus. Acta Neurochir 157:1003–1014
Mathijssen IM (2015) Guideline for care of patients with the diagnoses of craniosynostosis: working group on craniosynostosis. J Craniofac Surg 26:1735–1807
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Per Kristian Eide has an ownership stake in the software company (dPCom AS), that manufactures the software (Sensometrics) used for the analysis of the ICP recordings. All other authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (name of institute/committee) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required. The study was approved by the Oslo University Hospital as a quality control study (approval no. 2015/15993).
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Pediatric Neurosurgery
Rights and permissions
About this article
Cite this article
Frič, R., Langvatn, E.A., Due-Tønnessen, B.J. et al. The role of pulsatile and static intracranial pressure measurements in the management of children with craniosynostosis–an institutional experience from 49 patients. Acta Neurochir 163, 2015–2023 (2021). https://doi.org/10.1007/s00701-020-04680-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-020-04680-4