Abstract
Background
Microsurgical circumferential stripping of intracerebral metastases is often insufficient in achieving local tumor control. Supramarginal resection may improve local tumor control.
Methods
A retrospective analysis was performed for patients who underwent supramarginal resection of a cerebral metastasis by awake surgery with intraoperative cortical and subcortical stimulation, MEPs, and SSEPs. Supramarginal resection was achieved by circumferential stripping of the metastasis and additional removal of approximately 3 mm of the surrounding tissue. Pre- and postsurgical neurological status was assessed by the NIH Stroke Scale. Permanent deficits were defined by persistence after 3-month observation time.
Results
Supramarginal resection of cerebral metastases in eloquent brain areas was performed in 34 patients with a mean age of 60 years (range, 33–83 years). Five out of 34 patients (14.7 %) had a new transient postoperative neurological deficit, which improved within a few days due to supplementary motor area (SMA) syndrome. Five out of 34 patients (14.7 %) developed a local in-brain progression and nine patients (26.4 %) a distant in-brain progression.
Conclusions
Supramarginal resection of cerebral metastases in eloquent locations is feasible and safe. Safety might be increased by intraoperative neuromonitoring. The better outcome in the present series may be entirely based on other predictors than extend of surgical resection and not necessarily on the surgical technique applied. However, supramarginal resection was safe and apparently did not lead to worse results than regular surgical techniques. Prospective, controlled, and randomized studies are mandatory to determine the possible benefit of supramarginal resection on local tumor control and overall outcome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Neurosurgical standard therapy of singular metastases is microsurgical circumferential stripping. Standard metastasis resection without an adjuvant whole-brain radiation therapy frequently results in local in-brain progression in the range of 70 % in older and 50–60 % in more recent studies [10, 13, 17]. This considerably high local in-brain progression rate is lowered by an adjuvant whole-brain radiation therapy to about 27 % [10]. Therefore, microsurgical standard therapy alone is often insufficient in achieving long-lasting tumor control. Several reasons for the high in-brain progression rate were assumed: Cerebral metastases are widely believed to be sharply delimitated from the surrounding brain tissue and thus could easily be stripped from the surrounding brain tissue. In fact, less than 40 % of cerebral metastases are sharply delimitated from the brain parenchyma, whereas over 60 % display an irregular tumor–brain interface with tongue-like extensions into the surrounding brain or even in infiltrative growth pattern [3, 4, 9]. Besides, unexpected incomplete surgical resection of cerebral metastases might be another explanation of the high incidence of local-brain progression. Addressing these concerns, supramarginal resection with an extension to a depth of about 5 mm after complete microsurgical stripping of non-eloquently situated metastases reduced the risk of a local in-brain progression in a multivariate analysis and improve 2-year survival rates [17]. The concept of supramarginal resection was initially analyzed only for non-eloquently localized metastases but it is also eligible for eloquently situated metastases: Here, tools as awake surgery and intraoperative electrophysiological monitoring may minimize postoperative neurological deficits and morbidity and maximize the extent of surgical resection [8]. However, additional studies analyzing the oncological impact and local control rates after supramarginal resection of eloquently situated metastases are lacking.
The aim of the present retrospective single-center study was to analyze 1-year outcome data after supramarginal resection of eloquent situated cerebral metastases and if these data are superior to those reported from prospective randomized trails.
Materials and methods
The single-center analysis was approved by the local Research Ethics Committee and institutional review board (internal study number: 4851). The charts of all patients who underwent resection of cerebral metastasis between 01/2009 and 11/2013 at our tertiary care center were reviewed. All patients included in the present analysis fulfilled the following criteria: (1) surgical resection of one to maximal three eloquent situated cerebral metastases; (2) intraoperative supramarginal extension to a depth of about 5 mm after complete microsurgical resection (as described by surgeon’s notes); (3) complete set of pre-operative standard imaging and early postoperative MRI within 72 h after surgery; (4) postoperative residual tumor was excluded by the postoperative MRI with 72 h after surgery; (5) histopathological confirmation of a cerebral carcinoma or malignant melanoma metastasis; (6) subsequent adjuvant therapy and follow-up of the cerebral metastasis was performed in our department; and (7) follow-up of at least 1 year except for those patients who died of systemic tumor progression. Patients with tumors other than cerebral carcinoma metastasis (e.g., patients suffering from glioma, cerebral lymphoma or e.g., sarcoma metastases), patients who underwent resection without additional supramarginal tissue removal to a depth of about 5 mm, and patients who refused follow-up in our department were excluded.
Surgery
All analyzed surgeries were performed with intra-operative monitoring (IOM) recording motor-evoked (MEPs) and somato-sensory evoked potentials (SSEPs). Furthermore, surgery was performed as awake surgery in an asleep-awake-asleep protocol as described before [8]. An eloquent brain region was defined according to the literature as a cortical or subcortical brain area at which we expect intraoperative stimulation to elicit changes in neurologic conditions (particularly regarding speech, movement, and tactile sensation) or to elicit a response in electrophysiological recordings in corresponding areas [8]. Cerebral metastases were resected by white-light-assisted microsurgical circumferential stripping from the surrounding brain parenchyma. Resection was extended to a depth of about 5 mm after complete microsurgical resection provided that cortical and subcortical stimulation (either by 60-Hz or monopolar stimulation) excluded the risk of induction of a neurological deficit.
Control of extent of surgical resection within 72 h after surgery
Pre- as well as postoperative standard imaging within 72 h after resection was performed by an contrast-enhanced 1.5 T-MRI (Avanto, Siemens, Erlangen, Germany) within 72 h after surgery including the following sequences: T1 (repetition time (TR)/echo time (TE) of 1900/3.37 ms), T2 gradient (TR/TE of 5500/102 ms), T2* images (TR/TE of 2300/29 ms), T2 Flair (TR/TE of 9000/118 ms), DWI, ADC repetition time (TR/TE of 3600/89 ms; image resolution: 192 × 192 mm3), T1 with gadolinium (TR/TE 1900/3.37 ms; concentration 0.5 mmol/kg, Guerbet, Sulzbach, Germany). Residual contrast-enhancing parts in the T1 sequences as well as T2 and diffusion sequences were considered as confirmation of residual tumor. Exact pre- and postoperative tumor volumes were estimated by lining tumor margins as well as autosegmentation using three-dimensional volume rendering as described before (OsiriX Version v.5.7; Pixmeo SARL, Bernex, Switzerland) [6]. The iplan Net 3.0.0 software (BrainLAB AG, Germany) was used for figure illustration.
Data collection, definition of progression, and follow-up
Epidemiological data (age, gender), data regarding tumor location, and primary tumor (first diagnosis, other metastasis) and pre- and postoperative images were collected as being available. Patients were followed from the time of surgery until death every 3 months including a standard neurologic assessment and evaluation according to the National Institutes of Health Stroke Scale (NIHSS; minimal score 0 to maximum possible score 42). A contrast-enhanced MRI was scheduled in parallel. Mean follow-up time was 15.8 ± 1.5 months.
Local in-brain progression was defined as a tumor recurrence or progression within or at the boarder of the resection cavity. Distant in-brain progression was defined as occurrence of new metastasis distant to the site of the resected metastasis. Leptomeningeal carcinosis was diagnosed by cranial MRI by a diffuse nodular tumor progression of meninges or by lumbar puncture and confirmation of malignant tumor cells in the cerebrospinal fluid (CSF). Time to (local/distant) in-brain progression is defined as time between surgery and diagnosis of the in-brain tumor progression.
Statistical analysis
Descriptive statistics including minimum, maximum, mean, and standard error of mean (SEM) were calculated for all continuous variables. Frequencies and ratios were calculated for binary variables. Statistical analysis was performed using IBM SPSS Statistics Build 19.0.0 (IBM). The Kaplan–Meier curve was constructed with GraphPad Prism 5 (GraphPad Software, Inc., La Jolla, CA, USA). Data are presented as the mean ± standard error of mean (SEM).
Results
Patients
In total, 34 patients were identified who underwent supramarginal resection of their cerebral metastases in eloquent brain regions. Twenty-four patients were female and ten were male. Therefore, the female-to-male ratio was 2.4:1. The mean age was 60 years (range, 33–83 years). Twenty-five patients suffered from an adenocarcinoma, two from small cell cancer, one from clear cell cancer, one from squamous cell cancer, and five from malignant melanoma. A non-small cell lung cancer (NSCLC) could be identified as primary tumor in 17 patients, a small cell lung cancer (SCLC) in two patients, a malignant melanoma in five patients, a mamma carcinoma in five patients, a carcinoma of the gastro-intestinal tract in four patients, a carcinoma arising from the uro-genital tract in one patient. All supramarginally resected cerebral metastases had a supratentorial localization and were situated in an eloquent brain region. Clinical data are summarized in Table 1. For an additional six patients not included in this series, supramarginal resection was intended but not possible, as electrical stimulation revealed that further resection might cause a severe neurologic deficit.
Neurological outcome after supramarginal resection
Patients were evaluated for neurologic deficits at admission, discharge, and at every 3 months in a standardized setting as outpatients: Preoperatively, six patients had no neurological deficits and cerebral metastasis was diagnosed by staging. Seven patients suffered from dys- or aphasia, five patients from paresis, each four patients from disturbances of gait or coordination, three patients from memory disorders, while seven patients displayed seizures as initial symptom of their cerebral metastases. Mean preoperative NIHSS score was 1.2 ± 0.4 (range, 0–12). After surgery, five patients (14.7 %) displayed postoperatively new or worsened temporary neurologic deficits (hemiparesis due to supplementary motor area (SMA) syndrome and disturbances of fine motor skills). Mean NIHSS score was 1.2 ± 0.4 (range, 0–9) at the time of discharge. All new or worsened neurologic deficits due to surgery improved completely in the following weeks. Accordingly, mean NIHSS score was 0.7 ± 0.3 (range, 0–9) at 3 months and 1.2 ± 0.4 (range, 0–9) after 1 year.
Adjuvant radiation therapy
After surgery, the vast majority of patients (28/34; 82.3 %) received an adjuvant whole-brain radiation therapy (WBRT). One patient underwent local radiation therapy of the resection cavity with the adjacent brain parenchyma. However, five patients (14.7 %) received no form of additional radiotherapy after resection, as they refused adjuvant radiation therapy.
In-brain progression
Mean follow-up after surgery was 16 ± 1.5 months (range, 4–41 months) and 5/34 patients died within 1 year after surgery due to systemic progression of the malignancy but without an in-brain progression. In-brain progression was observed in 12/34 patients (35.3 %): a local in-brain progression was observed for five patients (5/34, 14.7 %). Mean time to local in-brain progression was 12.2 ± 1.7 months (range, 8–18 months). Two of the five patients suffered from distant in-brain progression in parallel to their local recurrence. In total, nine patients (9/34, 26.4 %) developed a distant in-brain progression (Fig. 1). A Kaplan–Meier estimation of the incidence of distant and local in-brain progression for all analyzed patients is given in Fig. 2.
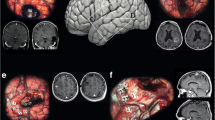
Complete resection. Figure 1 shows the pre- (a–c), postoperative (d–f), and 3-year follow-up MRI scans (g–l) of a 55-year-old patient who suffered from a central situated cerebral metastasis arising from a non-small lung cancer. The patient underwent supramarginal resection. Adjuvant whole-brain radiation therapy with 5 × 3 Gy per week to a total dose of 30 Gy was started 1 month after surgery. Three years after surgery, MRI showed no local in-brain progression but a new small cerebellar metastasis. k, l (preop: T1 ± gadolinium, T2; a–c; postop: T1 ± gadolinium, T2; d–f, MRI control 13 months after surgery: T1 ± gadolinium, T2: g–l)
Discussion
The main finding of this retrospective analysis in patients subject to experiencing supramarginal resection of cerebral metastases is that (1) about 15 % of patients displayed temporary new or worsened deficits, (2) no patients had new permanent deficits, and (3) that nearly 15 % suffered from local in-brain progression despite supramarginal resection.
Standard neurosurgical treatment of cerebral metastases with white-light assisted microsurgical circumferential stripping from the surrounding brain tissue is often insufficient to achieve local control. In the recently published prospective randomized and controlled EORTC 22952–26001 study, 27 % of patients with adjuvant whole-brain radiation therapy and 58 % of patients without adjuvant radiation therapy suffered from local in-brain progression [10]. The reasons for this high local recurrence rate have yet not been analyzed. However, evidence suggests that some metastases display an infiltrative growth pattern or an irregular tumor–brain interface with tongue-like extensions into the surrounding brain. Here, residual tumor parts or infiltrating tumor cells may not be visible and accessible during standard circumferential stripping and result in a local in-brain progression. The concept of supramarginal resection with extension to a depth of about 5 mm after complete microsurgical stripping of non-eloquently situated metastases reduced the risk of a local in-brain progression in the uni- and multivariate analysis [17]. In this series, 1- and 2-year local in-brain progression rates were 29.1 % after supramarginal resection without adjuvant radiotherapy. However, local control after supramarginal resection has as of yet only been analyzed in this single-center retrospective study for non-eloquent cerebral metastases [17]. In contrast, the present series analyzes neurologic deficits and local-control of eloquent situated metastases: Despite supramarginal resection, we observed no permanent neurologic deficits. However, supramarginal resection of eloquent situated cerebral metastases seems only to be applicable with intra-operative neurophysiological monitoring (IOM) and/or as awake surgery. Here, an early identification and prevention of eloquent brain regions is necessary to prevent permanent neurologic deficits. Furthermore, supramarginal resection might only be applicable for subpopulation as IOM revealed for some patients that an extension of resection to a supramarginal resection was not possible without risking permanent neurologic deficits. Furthermore, IOM and awake surgery may not prevent temporary deficit such as supplementary motor area syndrome (SMA syndrome). Persevered motor evoked potentials (MEPs) are indicative of SMA syndrome and help to guide within the postoperative course. Surgeries of glioma in the SMA frequently lead to SMA syndrome, which usually has a good prognosis [2, 7, 14, 16, 18].
In the present series, the local in-brain progression rate was 14.7 % (five/34 patients). It should be noted that 85 % of our present patients were subjected to adjuvant radiation therapy. Therefore, the local in-brain progression rate might be comparable to the patient cohort treated by surgery and adjuvant whole-brain radiation therapy in the more recently published prospective randomized and controlled EORTC 22952–26001 study with an incidence of local in-brain progression of 27 % [10]. These data suggest that the incidence rate may be halved by supramarginal resection and are in line with the study by Yoo and coworkers [17]: The authors demonstrated a 1-year local in-brain progression rate of 29.1 % after supramarginal resection as compared to 58.6 % after conventional resection. In this study, ten of 43 patients underwent supramarginal and 19 of 51 patients underwent conventional metastases resection were subjected to an adjuvant postoperative radiation therapy.
Although supramarginal resection might lower the local recurrence rate, it was not able to achieve local control in all patients in the present as well as in the Yoo series. The reasons therefore remain unclear. A residual microscopic tumor infiltration of the adjacent brain tissue may be one explanation. Resection margins were not histopathologically analyzed for residual microscopic tumor infiltration in the present series. However, unexpected contrast-enhancing tumor rest could be excluded as all patients of the present study were subjected to an early postoperative within 72 h after surgery. Furthermore, resection margins were pathologically confirmed to be tumor cell free by intraoperative fresh-frozen sectioning in the Yoo study. Other explanations for local in-brain progression despite supramarginal resection are spreading of single tumor cells during surgery (e.g., by piece-meal resection) or dural contact and infiltration of brain parenchyma metastases.
Besides surgery, radio-surgical treatment of cerebral metastases is a promising approach, especially as quality of life matters in patients with cerebral metastases [11, 15]. The 1-year local tumor control rate was 67 and 72.5 % in two prospective randomized and controlled phase III trials for patients with cerebral metastases treated with stereotactic radiosurgery alone, respectively [1, 5]. The final analysis of a prematurely stopped randomized controlled multicenter phase III trial showed no statistically significant differences in the local tumor control rates of the cohorts surgery plus whole-brain radiation therapy vs. radiosurgery [12]. In the more recent EORTC 22952–26001 study, the local recurrence rate after 2 years was 19 % for patients treated with radiosurgery and adjuvant WBRT, 31 % for patients after radiosurgery alone compared to 27 and 59 % for patients underwent surgery without or with additional WBRT, respectively [10]. A comparison of surgical vs. radio-surgical treatment was not performed [10]. However, it would be interesting to analyze if the radio-surgical tumor margin dose correlates with the local in-brain progression rate in a larger patient collective or in a meta-analysis as it was described in some studies [15].
We acknowledge several limitations of our present study: (1) the results of our analysis are limited by the retrospective study design. Furthermore, these results arise from a single-center study (2) the present patient population is heterogeneous with patients suffering different primary tumors, different tumor stages, and different adjuvant therapy concepts. It would be interesting to perform a controlled study with a more homogeneous patient population and a clearly defined adjuvant therapy concept after surgical resection of the metastasis. (3) Other predictors such as the preoperative tumor volume, tumor infiltration of the dural skin, and piecemeal vs. en bloc resection are known to influence local in-brain progression rate but were not analyzed in the present series. However, the limited number of patients included in the present analysis does not allow a detailed statistical analysis of these subgroups. Other predictors of local in-brain progression should be analyzed in a larger patient collective suffering from cerebral metastases. (4) In the present study, degree of resection was classified as “supramarginal resection” by the surgeon. A “surgeon-independent” confirmation of a supramarginal resection is hardly possible: A postoperative MRI as performed in the present series easily confirms a total metastasis resection defined as complete resection of the contrast-enhancing tumor. However, quantification of supramarginal resection beyond tumor resection is hardly possible for several reasons: Cerebral masses such as brain metastases cause not only a brain shift but also a compression of the surrounding brain parenchyma, which resolves during surgery. Therefore, differences of pre- and postoperative manual or automatic MRI-based segmentation and volumetry of the brain parenchyma does not represent the degree of supramarginal resection. Also, quantification of resection cavity volume does not reliably correlate this resection degree for the same reason and as the resection cavities are known to collapse frequently after surgery.
Further prospective and randomized studies are mandatory to evaluate the impact of extent of metastasis resection in achieving a better local tumor control.
Conclusions
Supramarginal resection of cerebral metastases in eloquent locations is feasible and safe. Safety might be increased by intraoperative neuromonitoring. The better outcome in the present series may be entirely based on other predictors than the extent of surgical resection and not necessarily on the surgical technique applied. However, supramarginal resection was safe and apparently did not lead to worse results than regular surgical techniques. Prospective, controlled, and randomized studies are mandatory to determine the possible benefit of supramarginal resection on local tumor control and overall outcome.
References
Aoyama H, Shirato H, Tago M, Nakagawa K, Toyoda T, Hatano K, Kenjyo M, Oya N, Hirota S, Shioura H, Kunieda E, Inomata T, Hayakawa K, Katoh N, Kobashi G (2006) Stereotactic radiosurgery plus whole-brain radiation therapy vs stereotactic radiosurgery alone for treatment of brain metastases: a randomized controlled trial. JAMA 295:2483–2491
Bannur U, Rajshekhar V (2000) Post operative supplementary motor area syndrome: clinical features and outcome. Br J Neurosurg 14:204–210
Baumert BG, Rutten I, Dehing-Oberije C, Twijnstra A, Dirx MJ, Debougnoux-Huppertz RM, Lambin P, Kubat B (2006) A pathology-based substrate for target definition in radiosurgery of brain metastases. Int J Radiat Oncol Biol Phys 66:187–194
Berghoff AS, Rajky O, Winkler F, Bartsch R, Furtner J, Hainfellner JA, Goodman SL, Weller M, Schittenhelm J, Preusser M (2013) Invasion patterns in brain metastases of solid cancers. Neuro-Oncology 15:1664–1672
Chang EL, Wefel JS, Hess KR, Allen PK, Lang FF, Kornguth DG, Arbuckle RB, Swint JM, Shiu AS, Maor MH, Meyers CA (2009) Neurocognition in patients with brain metastases treated with radiosurgery or radiosurgery plus whole-brain irradiation: a randomised controlled trial. Lancet Oncol 10:1037–1044
Etminan N, Beseoglu K, Heiroth HJ, Turowski B, Steiger HJ, Hanggi D (2013) Early perfusion computerized tomography imaging as a radiographic surrogate for delayed cerebral ischemia and functional outcome after subarachnoid hemorrhage. Stroke 44:1260–1266
Fontaine D, Capelle L, Duffau H (2002) Somatotopy of the supplementary motor area: evidence from correlation of the extent of surgical resection with the clinical patterns of deficit. Neurosurgery 50:297–303
Kamp MA, Dibue M, Niemann L, Reichelt DC, Felsberg J, Steiger HJ, Szelenyi A, Rapp M, Sabel M (2012) Proof of principle: supramarginal resection of cerebral metastases in eloquent brain areas. Acta Neurochir (Wien) 154:1981–1986
Kamp MA, Grosser P, Felsberg J, Slotty PJ, Steiger HJ, Reifenberger G, Sabel M (2012) 5-aminolevulinic acid (5-ALA)-induced fluorescence in intracerebral metastases: a retrospective study. Acta Neurochir (Wien) 154:223–228
Kocher M, Soffietti R, Abacioglu U, Villa S, Fauchon F, Baumert BG, Fariselli L, Tzuk-Shina T, Kortmann RD, Carrie C, Ben Hassel M, Kouri M, Valeinis E, van den Berge D, Collette S, Collette L, Mueller RP (2011) Adjuvant whole-brain radiotherapy versus observation after radiosurgery or surgical resection of one to three cerebral metastases: results of the EORTC 22952–26001 study. J Clin Oncol 29:134–141
Lavine SD, Petrovich Z, Cohen-Gadol AA, Masri LS, Morton DL, O’Day SJ, Essner R, Zelman V, Yu C, Luxton G, Apuzzo ML (1999) Gamma knife radiosurgery for metastatic melanoma: an analysis of survival, outcome, and complications. Neurosurgery 44:59–64
Muacevic A, Wowra B, Siefert A, Tonn JC, Steiger HJ, Kreth FW (2008) Microsurgery plus whole-brain irradiation versus Gamma Knife surgery alone for treatment of single metastases to the brain: a randomized controlled multicentre phase III trial. J Neuro-Oncol 87:299–307
Patchell RA, Tibbs PA, Regine WF, Dempsey RJ, Mohiuddin M, Kryscio RJ, Markesbery WR, Foon KA, Young B (1998) Postoperative radiotherapy in the treatment of single metastases to the brain: a randomized trial. JAMA 280:1485–1489
Russell SM, Kelly PJ (2003) Incidence and clinical evolution of postoperative deficits after volumetric stereotactic resection of glial neoplasms involving the supplementary motor area. Neurosurgery 52:506–516
Sheehan JP, Sun MH, Kondziolka D, Flickinger J, Lunsford LD (2003) Radiosurgery in patients with renal cell carcinoma metastasis to the brain: long-term outcomes and prognostic factors influencing survival and local tumor control. J Neurosurg 98:342–349
Ulu MO, Tanriover N, Ozlen F, Sanus GZ, Tanriverdi T, Ozkara C, Uzan M (2008) Surgical treatment of lesions involving the supplementary motor area: clinical results of 12 patients. Turk Neurosurg 18:286–293
Yoo H, Kim YZ, Nam BH, Shin SH, Yang HS, Lee JS, Zo JI, Lee SH (2009) Reduced local recurrence of a single brain metastasis through microscopic total resection. J Neurosurg 110:730–736
Zentner J, Hufnagel A, Pechstein U, Wolf HK, Schramm J (1996) Functional results after resective procedures involving the supplementary motor area. J Neurosurg 85:542–549
Conflict of interest
None.
Funding
The present study was not funded.
Author information
Authors and Affiliations
Corresponding author
Additional information
Comment
This is a single-center, retrospective study of 34 patients who underwent MRI-verified supramarginal resection of a single cerebral metastasis in an eloquent area by awake surgery with intraoperative monitoring. The study demonstrates that supramarginal resection was feasible as no permanent postoperative neurological deficits were observed. Furthermore, supramarginal resection achieved better tumor control.
The strengths of this study, albeit a retrospective one, are the clear definitions of eloquence, inclusion criteria and exclusion criteria, the very careful follow-up with outpatient clinic and MRI every 3 months, and that the limitations of the study are identified and clearly stated. Furthermore, the analysis is careful and the discussion is precise and pertinent. The results are not “over-sold”, although the supramarginal resection resulted in a very low local in-brain progression.
There are some weaknesses, such as the retrospective design and that proof of concept has been previously demonstrated for metastases in non-eloquent areas. The study group is selected. Six patients (15 %) were excluded from the study as the intraoperative monitoring suggested further resection would result in neurological deficits. Furthermore, due to the strict inclusion and exclusion criteria, a number of patients with metastases in eloquent regions must have been excluded. We are not given information about them. Lastly, although the neurological outcome was scored using standardized methods, it was not scored by an independent operator.
There are also some unanswered questions. One inclusion criterion is a complete set of pre- and postoperative MRI. How many were excluded due to lack of postoperative MRI? There is clearly a selection bias unless every single patient has a postoperative MRI and not at the surgeon’s discretion. Surgeon-independent confirmation of a supramarginal resection was not performed, nor quantification of supramarginal resection beyond tumor resection. What substantiates the notion that supramarginal resection is different from your standard MRI-verified complete extirpation?
Torstein Meling
Oslo, Norway
Marcel A. Kamp and Marion Rapp contributed equally to this work.
Rights and permissions
About this article
Cite this article
Kamp, M.A., Rapp, M., Slotty, P.J. et al. Incidence of local in-brain progression after supramarginal resection of cerebral metastases. Acta Neurochir 157, 905–911 (2015). https://doi.org/10.1007/s00701-015-2405-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-015-2405-9