Abstract
Background
Intracranial hemorrhage (ICH) is often associated with cardiac events. Twelve-lead electrocardiography (ECG) and transthoracic echocardiography are essential diagnostic tools for preoperative risk assessment. B-type natriuretic peptide (BNP) is a well-known predictor of cardiac outcome in various clinical settings. This study examined whether BNP levels on admission are associated with in-hospital cardiac events among ICH patients.
Method
This prospective study enrolled 77 ICH patients who were admitted to this hospital for emergency neurosurgery. On admission, BNP levels, 12-lead ECG and transthoracic echocardiography were carried out for all patients. These patients were divided into two groups: Group I included 19 patients (10 men and 9 women) having 24 in-hospital cardiac events (mean age of 57 ± 15 years); Group II included 57 patients (29 men, mean age of 71 ± 11 years) without cardiac events.
Results
Admission BNP levels of Group I patients were significantly higher than those of Group II patients (683.8 ± 1,043.8 pg/ml vs 168.5 ± 173.5 pg/ml, p = 0.001). In multivariate analysis, BNP levels and T-wave inversion are independent predictors of in-hospital cardiac events. A cutoff value of BNP levels (156.6 pg/ml) predicted in-hospital cardiac events in ICH patients with 80 % sensitivity and 66 % specificity.
Conclusions
Serum BNP levels and electrocardiographic T-wave inversion on admission are independent predictors of in-hospital cardiac events in patients with ICH who undergo emergency neurosurgery.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
An association between intracranial hemorrhage and cardiac dysfunction was first reported in 1954. In the report, T-wave abnormality was observed in patients with subarachnoid hemorrhage (SAH) [1]. It is well known that SAH is often associated with cardiopulmonary complications including cardiac arrhythmias and conduction abnormalities [2].
In previous studies, SAH was associated with adverse cardiac events caused by rhythm disturbances or myocardial necrosis in up to 40 % of the patients [3–5]. Electrocardiography (ECG) and transthoracic echocardiography (TTE) are important diagnostic tools for preoperative risk assessment [6]. Nevertheless, these tests often cannot be performed due to time constraints during an emergency situation. B-type natriuretic peptide (BNP) has a diagnostic or prognostic value in various clinical settings [7,8]. Furthermore high BNP levels are associated with an increased mortality after stroke [9]. In previous studies, high BNP levels were associated with poor neurological outcome after SAH. BNP levels increased soon after development of SAH, but these BNP levels returned to the baseline by 2 weeks. It was related to myocardial dysfunction evidenced by sudden catecholamine surge after SAH [10,11]. However, there are limited data on a correlation between BNP levels and clinical outcomes in patients with intracranial hemorrhage (ICH) other than SAH. The aim of this study was to evaluate whether BNP levels could predict cardiac events, such as cardiac arrhythmias, myocardial infarction or cardiac arrest for in-patients with ICH.
Methods
Study population
This prospective study enrolled 77 patients (with a mean age of 66 ± 15 years; 39 men (50.6 %) and 38 women (49.4 %). They were admitted to this hospital with a diagnosis of ICH, and underwent emergency neurosurgery from January 2009 through May 2011.
ICH was confirmed by computed tomography or magnetic resonance imaging. ICH cases included 30 subdural hemorrhages (SDHs), 4 epidural hemorrhages (EDHs), 32 intracerebral hematomas (ICbHs), and 11 SAHs. All 30 patients with SDH were in the acute stage of hemorrhage. Concerning the ICbH, the location of the hemorrhages for ten patients was in the basal ganglia and for 15 patients was in the frontal lobe. As for the remaining seven patients, the location of the hemorrhages was in the parietal lobe. Eleven SAHs were due to spontaneously ruptured aneurysms. All of these patients underwent intracranial operation within 24 h after admission. This study excluded patients with any history of ischemic heart disease, cardiomyopathy, heart death, myocardial infarction, or chronic kidney disease. Patients were divided into two groups: Group I included 19 patients with in-hospital cardiac events, while Group II included 58 patients without a cardiac event. This study was approved by the institutional review board of the Kang Nam General Hospital.
Baseline clinical data
The patients’ past medical history was obtained from the patients themselves, family members and previous medical records. Age, gender, diabetes mellitus, hypertension, dyslipidemia, obesity (BMI > 23 kg/m2) and history of heart disease were checked. Heart rate, systolic and diastolic blood pressure were monitored periodically during hospitalization. Chest PA, 12-lead ECG and TTE were performed within 48 h of admission.
Laboratory test and measurement of BNP levels
Venous blood samples were collected in a tube containing lithium-heparin and transported to the laboratory at room temperature. Plasma BNP levels were measured using the fluorescent enzyme immunoassay (Abbot Diagnostics, Abbott Park, IL). The upper limit of normal BNP level is 100 pg/ml in our hospital. The level of cardiac troponin I (TnI) was also measured using the fluorescent enzyme immunoassay and the upper limit of normal TnI level was 0.032 ng/ml at this laboratory. The levels of hemoglobin, hematocrit, electrolyte, blood urea nitrogen, creatinine, liver enzymes, activated partial thromboplastin time, and prothrombin time were taken on admission.
ECG and echocardiography
ECG was carried out for all patients and rhythm strips were continuously monitored in the intensive care unit. Patients were put on a 24-h Holter monitoring whenever ECG irregularities were detected in the hospital. ECG abnormalities were analyzed with respect to pathologic Q wave, QT prolongation, U wave, ST elevation and depression, T-wave inversion, tall T-wave, giant T-wave inversions, and nonspecific ST-T-wave abnormalities (NSTTA).
Abnormal rhythmic disturbances were specified as sinus tachycardia (>100 beats/min), sinus bradycardia (<60 beats/min), supraventricular tachycardia (SVT), atrial fibrillation (AF), ventricular tachycardia (VT), or ventricular fibrillation (VF). Atrio-ventricular (AV) conduction disturbance was also examined (see Electronic supplementary material, Methods).
Transthoracic echocardiography (TTE) was performed using GE vivid 7 Dimension Ultrasound System (GE Healthcare, Milwaukee, WI, USA). For TTE examination, standard two-dimensional images, Doppler recordings of mitral inflow and pulmonary venous flow were obtained [12]. Left ventricular ejection fraction (LVEF), regional wall motion abnormality (RWMA) and diastolic dysfunction were also verified [13].
Clinical outcome
The clinical outcome included cardiac death, nonfatal myocardial infarction, as well as rhythm disturbances (ventricular tachycardia, ventricular fibrillation, paroxysmal supraventricular tachycardia, atrial tachycardia, atrial flutter, and atrial fibrillation). Cardiac arrest is defined as the abrupt loss of heart function. Myocardial infarction was define as ST-segment change with increased TnI levels above the normal range (TnI >0.032 ng/ml) (see Electronic supplementary material, Fig. 1).
Statistical analysis
Continuous data with normal distribution were expressed as the mean ± standard deviation and the independent t-test or Mann–Whitney U test was utilized for comparative analysis of continuous data. The Fischer exact test was used for the comparisons of categorical data.
The Spearman’s correlation was used to assess the relationship between BNP levels on admission and ECG parameters. The linear regression analysis was used to test a correlation between BNP levels and LVEF. Univariate and multivariate logistic regression analyses were used to identify the predictor of in-hospital cardiac events, reporting odds ratios (ORs) and 95 % confidence intervals (CIs). The receiver-operating characteristic (ROC) analysis was performed to determine sensitivity, specificity, and cutoff BNP levels in the prediction of cardiac events. All analyses were performed using the Statistical Package for the Social Sciences (SPSS) version 17.0 statistical software (SPSS, Chicago, IL, USA)
Results
Baseline characteristics
The baseline characteristics of subjects are shown in Table 1. The mean duration of hospitalization was 45 ± 47 days. The mean LVEF of patients in Group I was lower than that of patients in Group II (47.9 ± 14.7 vs 57.6 ± 7.5, p = 0.004). There were no significant differences in other baseline parameters between these two groups.
Biochemistry and ECG abnormalities
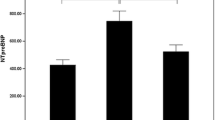
The mean BNP level was 295.6 ± 575.0 pg/ml. The BNP levels of patients in Group I were significantly higher than those of patients in Group II (683.8 ± 1,043.8 pg/ml vs 168.5 ± 173.5 pg/ml) (p = 0.001) (Fig. 1). The TnI levels of patients in Group I were also significantly higher than those of patients in Group II (1.2 ± 5.1 ng/ml vs 0.03 ± 0.05 ng/ml) (p = 0.002). There were no differences in other laboratory tests between these two groups (Table 2). The LVEF values were significantly correlated with admission BNP levels on (correlation coefficient 0.232, p < 0.001). Thirty one patients had at least one or more ECG abnormalities (40.3 %). The most common ECG abnormality was T-wave inversion (24 patients, 31.2 %). T-wave inversion, NSTTA and ST depression appeared more frequently among patients in Group I than patients in Group II (Table 2).
Predictors of in-hospital cardiac events in patients with ICH
The mean hospital duration of ICH patients was 45 ± 47 days (Group I, 47 ± 42 days; Group II, 40 ± 50 days). The mortality rate during hospitalization was 19.4 % (15/77 patients).
Nineteen patients experienced 24 cardiac events including 6 cardiac arrests (25 %), 2 myocardial infarctions (8 %), 2 VTs (8 %), 13 AFs (54 %), and 1 SVT (5 %) (see Electronic supplementary material, Fig. 1). The incidence of new-onset AF was significantly higher among patients with an elevated BNP level. Eleven (84.6 %) out of 13 patients with new-onset AF had an elevated BNP level, while two (15.4 %) patients had a normal BNP level (p < 0.001). There were no significant differences in other cardiac events between these two groups.
In multivariate logistic regression analyses, only higher BNP levels (OR, 1.003; 95 % CI, 1.001–1.005; p = 0.01) and T-wave inversions (OR, 4.218; 95 % CI, 1.256–14.166; p = 0.02) were independently associated with in-hospital cardiac events (Table 3). Using the cutoff value of BNP (156.6 pg/ml), the sensitivity and specificity of BNP levels predicting cardiac events were 68 and 66 %, respectively. The area under the ROC curve was 0.749 in Fig. 2 (95 % CI = 0.631–0.867, p = 0.001).
A receiver operating characteristics curve was constructed to determine the cutoff value of B-type natriuretic peptide for predicting cardiac events. A cutoff value of 156.6 pg/ml yielded the maximal combined efficacy with a sensitivity of 68 % and specificity of 66 %. Area under the curve = 0.749 (95 % CI = 0.631–0.867, p = 0.001)
High BNP levels are significantly associated with cardiac events in the subgroup of patients with spontaneous ICH (n = 43: 32 ICbH and 11 SAH). However, such an association has not been established in patients with traumatic ICH (n = 34: 30 SDH and 4 EDH) (see Electronic supplementary material, Table 1). We have also evaluated the prognostic implication of the admission BNP and ECG parameters in the subgroup of ICbH (n = 32) and SDH (n = 30). In the subgroup analysis for the patients with ICbH, the mean BNP level was significantly higher in patients with cardiac events than in patients without cardiac events. However, the BNP level was not associated with in-hospital cardiac events in patients with SDH (see Electronic supplementary material, Table 2).
Discussions
This prospective observational study demonstrated that BNP levels were independently associated with in-hospital cardiac events in patients with ICH undergoing neurosurgery, not only SAH but also the other types of ICH. In a multivariate logistic regression analysis model for predicting in-hospital cardiac events in patients with ICH after adjustment of age, sex, LVEF, underlying disease (hypertension, diabetes) and the type of ICH, the admission BNP level was the only independent predictor of the in-hospital cardiac events in ICH patients (OR, 1.004; 95 % CI, 1.000–1.008; p = 0.029) (see Electronic supplementary material, Table 3)
Elevated BNP levels in patients with SAH were associated with poor prognosis [10,11]. Excessive catecholamine release after SAH has been proposed as the most probable pathophysiologic mechanism of myocardial injury. After development of SAH, local release of norepinephrine from myocardial sympathetic nerve endings has been shown to induce cardiac-toxicity [14,15]. A transient relationship has been described between BNP levels and cerebral vasospasm [16]. BNPs and their receptors were detected from the human brain tissues, particularly in the brain stem, hypothalamus, and the circumventricular organs [17].
LV systolic dysfunction has been described in approximately 10 % of patients after SAH. An elevated TnI level was associated with RWMA in TTE [18,19]. In this study, however, BNP, TnI and LVEF were significant predictors of in-hospital cardiac events, as shown in the univariate analysis. Among the biochemical parameters, only BNP levels were independently associated with cardiac events in multivariate analysis.
Preoperative BNP concentration is one of the powerful independent predictors of perioperative cardiovascular complications. However, a recent meta-analysis has reported that an additional postoperative BNP measurement enhances risk stratification for the cardiac events at 30 days after non-cardiac surgery compared with a preoperative BNP measurement alone [20]. In a previous study by Spatenkova et al. [21], postoperative NT-proBNP level was significantly higher on postoperative day 2 than on day 1. However, there was no relationship between the post-operative BNP levels and the 1-year cardiac events in 27 patients with C-spine surgery. The previous study population was younger and was comprised of fewer female patients than the study population of the current study
This investigation also examined whether specific ECG abnormalities had been associated with in-hospital cardiac events in ICH patients. The most common and specific ECG change in ICH patients was QT prolongation [22–24]. The most common types of arrhythmia were sinus bradycardia and AF, while rhythm abnormalities were also detected in about 4 % of patients with SAH [25]. Coghlan et al. [26] showed that preoperative bradycardia, relative tachycardia (heart rate >80 beats/min) and nonspecific ST-T-wave abnormalities had been strongly associated with 3-month mortality after infliction of SAH. In this study, only T-wave inversion on admission was independently associated with clinical outcomes, as evidenced in the multivariate analysis. However, the sensitivity of T-inversion in ECG in the prediction of in-hospital cardiac event was only 45.8 %. The consideration is that the diagnostic attribute of ECG has been limited to the detection of myocardial dysfunction(s) in patients with ICH [4,27]. Thus, TTE has been regarded as the ‘gold standard’ in the detection of cardiac dysfunction(s). Nevertheless, in many instances, TTE may not be performed in an emergency circumstance due to the time constraint and poor patient condition. From this investigation, we suggest that patients with high BNP levels on admission necessitate careful monitoring of possible development of cardiac event(s) in the hospital. Thus, routine BNP levels taken on admission may prove to be diagnostically valid in the assessment of cardiac functions and identification of high risk ICH patients vulnerable to in-hospital cardiac events.
Study limitations
This study has several important limitations. First, the follow-up observations were only possible until the time of hospital discharge. Data on long-term follow-up observations remain to be scrutinized in a future study. Second, this study could not conclude that early detection of cardiac dysfunction(s) would improve clinical outcome in these patients. Third, this study did not demonstrate the extent of neurologic consequences after development of ICH, which correlated with neuro-cardiac injuries. Some studies reported that neuro-cardiac injuries were more adversely associated with infliction of poor neurological grade. The clinical importance of these observations remain to be defined [28]. Lastly, the heterogeneous ICH diagnoses pool together in the current study. In subgroup analysis, admission BNP was an independent risk factor for adverse cardiac event even after adjusting the type of ICH. However, our data should only be applied in patients undergoing brain surgery.
Conclusion
Elevated BNP levels and T-inversion on admission were independently associated with in-hospital cardiac events in patients with ICH. BNP levels on admission may prove to be a useful predictor of adverse cardiac events than other time-consuming diagnostic parameters in providing in-hospital cardiac prognosis in patients with ICH.
References
Burch GE, Meyers R, Abildskov JA (1954) A new electrocardiographic pattern observed in cerebrovascular accidents. Circulation 9:719–723
Macmillan CS, Grant IS, Andrews PJ (2002) Pulmonary and cardiac sequelae of subarachnoid haemorrhage: time for active management? Intensiv Care Med 28:1012–1023
Crago EA, Kerr ME, Kong Y, Baldisseri M, Horowitz M, Yonas H, Kassam A (2004) The impact of cardiac complications on outcome in the SAH population. Acta Neurol Scand 110:248–253
Mayer SA, Lin J, Homma S, Solomon RA, Lennihan L, Sherman D, Fink ME, Beckford A, Klebanoff LM (1999) Myocardial injury and left ventricular performance after subarachnoid hemorrhage. Stroke 30:780–786
Tung P, Kopelnik A, Banki N, Ong K, Ko N, Lawton MT, Gress D, Drew B, Foster E, Parmley W, Zaroff J (2004) Predictors of neurocardiogenic injury after subarachnoid hemorrhage. Stroke 35:548–551
Poldermans D, Bax JJ, Boersma E, De Hert S, Eeckhout E, Fowkes G, Gorenek B, Hennerici MG, Iung B, Kelm M, Kjeldsen KP, Kristensen SD, Lopez-Sendon J, Pelosi P, Philippe F, Pierard L, Ponikowski P, Schmid JP, Sellevold OF, Sicari R, Van den Berghe G, Vermassen F (2009) Guidelines for pre-operative cardiac risk assessment and perioperative cardiac management in non-cardiac surgery. Eur Heart J 30:2769–2812
de Lemos JA, Morrow DA, Bentley JH, Omland T, Sabatine MS, McCabe CH, Hall C, Cannon CP, Braunwald E (2001) The prognostic value of B-type natriuretic peptide in patients with acute coronary syndromes. N Engl J Med 345:1014–1021
Doust JA, Glasziou PP, Pietrzak E, Dobson AJ (2004) A systematic review of the diagnostic accuracy of natriuretic peptides for heart failure. Arch Intern Med 164:1978–1984
Makikallio AM, Makikallio TH, Korpelainen JT, Vuolteenaho O, Tapanainen JM, Ylitalo K, Sotaniemi KA, Huikuri HV, Myllyla VV (2005) Natriuretic peptides and mortality after stroke. Stroke 36:1016–1020
Tomida M, Muraki M, Uemura K, Yamasaki K (1998) Plasma concentrations of brain natriuretic peptide in patients with subarachnoid hemorrhage. Stroke 29:1584–1587
Tung PP, Olmsted E, Kopelnik A, Banki NM, Drew BJ, Ko N, Lawton MT, Smith W, Foster E, Young WL, Zaroff JG (2005) Plasma B-type natriuretic peptide levels are associated with early cardiac dysfunction after subarachnoid hemorrhage. Stroke 36:1567–1569
Schiller NB, Shah PM, Crawford M, DeMaria A, Devereux R, Feigenbaum H, Gutgesell H, Reichek N, Sahn D, Schnittger I et al (1989) Recommendations for quantitation of the left ventricle by two-dimensional echocardiography. American society of echocardiography committee on standards, subcommittee on quantitation of Two-dimensional echocardiograms. J Am Soc Echocardiogr 2:358–367
Khouri SJ, Maly GT, Suh DD, Walsh TE (2004) A practical approach to the echocardiographic evaluation of diastolic function. J Am Soc Echocardiogr 17:290–297
Banki NM, Kopelnik A, Dae MW, Miss J, Tung P, Lawton MT, Drew BJ, Foster E, Smith W, Parmley WW, Zaroff JG (2005) Acute neurocardiogenic injury after subarachnoid hemorrhage. Circulation 112:3314–3319
Mertes PM, Carteaux JP, Jaboin Y, Pinelli G, el Abassi K, Dopff C, Atkinson J, Villemot JP, Burlet C, Boulange M (1994) Estimation of myocardial interstitial norepinephrine release after brain death using cardiac microdialysis. Transplantation 57:371–377
McGirt MJ, Blessing R, Nimjee SM, Friedman AH, Alexander MJ, Laskowitz DT, Lynch JR (2004) Correlation of serum brain natriuretic peptide with hyponatremia and delayed ischemic neurological deficits after subarachnoid hemorrhage. Neurosurgery 54:1369–1373, discussion 1373-1364
Gerbes AL, Dagnino L, Nguyen T, Nemer M (1994) Transcription of brain natriuretic peptide and atrial natriuretic peptide genes in human tissues. J Clin Endocrinol Metab 78:1307–1311
Banki N, Kopelnik A, Tung P, Lawton MT, Gress D, Drew B, Dae M, Foster E, Parmley W, Zaroff J (2006) Prospective analysis of prevalence, distribution, and rate of recovery of left ventricular systolic dysfunction in patients with subarachnoid hemorrhage. J Neurosurg 105:15–20
Zaroff JG, Rordorf GA, Ogilvy CS, Picard MH (2000) Regional patterns of left ventricular systolic dysfunction after subarachnoid hemorrhage: evidence for neurally mediated cardiac injury. J Am Soc Echocardiogr 13:774–779
Rodseth RNBB, Le Manach Y, Sessler DI, Lurati Buse GA, Thabane L, Schutt RC, Bolliger D, Cagini L, Cardinale D, Chong CP, Chu R, Cnotliwy M, Di Somma S, Fahrner R, Lim WK, Mahla E, Manikandan R, Puma F, Pyun WB, Radović M, Rajagopalan S, Suttie S, Vanniyasingam T, van Gaal WJ, Waliszek M, Devereaux PJ (2014) The prognostic value of pre-operative and post-operative B-type natriuretic peptides in patients undergoing noncardiac surgery: B-type natriuretic peptide and N-terminal fragment of pro-B-type natriuretic peptide: a systematic review and individual patient data meta-analysis. J Am Coll Cardiol 63:170–180
Spatenkova VBO, Kazda A, Suchomel P (2011) N-terminal pro-B-Type natriuretic peptided with fractional excretion and clearance of sodium in relation to cardiovascular events after elective cervical spine surgery. Neuro Endocrinol Lett 32:874–878
Di Pasquale G, Pinelli G, Andreoli A, Manini G, Grazi P, Tognetti F (1987) Holter detection of cardiac arrhythmias in intracranial subarachnoid hemorrhage. Am J Cardiol 59:596–600
Jung JH, Min PK, Rim SJ, Ha JW, Chung N, Lee KC (2010) Are electrocardiographic changes in patients with acute subarachnoid hemorrhage associated with Takotsubo cardiomyopathy? Cardiology 115:98–106
Khechinashvili G, Asplund K (2002) Electrocardiographic changes in patients with acute stroke: a systematic review. Cerebrovasc Dis 14:67–76
Andreoli A, di Pasquale G, Pinelli G, Grazi P, Tognetti F, Testa C (1987) Subarachnoid hemorrhage: frequency and severity of cardiac arrhythmias. A survey of 70 cases studied in the acute phase. Stroke 18:558–564
Coghlan LA, Hindman BJ, Bayman EO, Banki NM, Gelb AW, Todd MM, Zaroff JG (2009) Independent associations between electrocardiographic abnormalities and outcomes in patients with aneurysmal subarachnoid hemorrhage: findings from the intraoperative hypothermia aneurysm surgery trial. Stroke 40:412–418
Kawahara E, Ikeda S, Miyahara Y, Kohno S (2003) Role of autonomic nervous dysfunction in electrocardio-graphic abnormalities and cardiac injury in patients with acute subarachnoid hemorrhage. Circ J 67:753–756
Jensen JK, Korsholm L, Hoilund-Carlsen PF, Atar D, Kristensen SR, Mickley H (2007) The relation between electrocardiographic ST-T changes and NT-proBNP in patients with acute ischemic stroke. Scand Cardiovasc J 41:294–298
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
J.H. Park and Y.R. Kim contributed equally to this article
Rights and permissions
About this article
Cite this article
Park, J.H., Kim, Y.R., Kim, S.H. et al. Admission B-type natriuretic peptide levels are associated with in-hospital cardiac events in patients with intracranial hemorrhage. Acta Neurochir 157, 21–27 (2015). https://doi.org/10.1007/s00701-014-2260-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-014-2260-0