Abstract
Backgroud
The role of radical resection for nongerminomatous pineal region tumors is still controversial. The purpose of this study was to present the surgical results in a large series and evaluate the feasibility of radical surgical strategy.
Methods
We retrospectively reviewed the records of 143 patients with nongerminomatous pineal region tumors surgically treated via an occipital transtentorial approach between 2000 and 2011. The tumor was small (<2 cm) in 14.7 % of patients, medium (2–4 cm) in 52.4 %, and large (>4 cm) in 32.9 %.
Results
Gross total tumor removal was achieved in 91.6 % of patients, subtotal in 7.0 %, and partial in 1.4 %. Histological diagnosis was nongerminomatous germ cell tumor in 41.3 %, pineal parenchymal tumor in 14.7 %, glial tumors in 28.7 %, and miscellaneous in 15.4 %. The overall complication and mortality rate was 18.2 % and 0.7 %, respectively. Permanent morbidity occurred in 5.6 % of patients, including hemianopsia in 3.5 % and Parinaud syndrome in 2.1 %. Hydrocephalus was resolved in 82.1 % without surgery for the CSF diversion. Sixty-eight patients with malignant tumors underwent radiotherapy; 35 also received adjuvant chemotherapy. One hundred thirty patients were successfully followed up with a mean duration of 43 months. Finally, 86.9 % of the patients achieved a favorable functional outcome (mRS ≤ 2), 3.1 % had an mRS score of 3, 1.5 % had an mRS score of 4, and 8.5 % had died (mRS = 6).
Conclusions
Radical surgery was recommended as the optimal treatment for nongerminomatous pineal region tumors. Favorable results could be achieved by experienced neurosurgeons. Hydrocephalus could be cured by radical tumor removal in the majority of cases. The occipital transtentorial approach was indicated for most pineal region tumors, but surgeon’s preference and experience should also be considered. New understanding of the arachnoid membranes of this region may be helpful for tumor resection.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Pineal region tumors present with a spectrum of histological heterogeneous neoplasms from benign to malignant and have a higher incidence in Far Eastern countries (2.1–3.2 %) than in Western countries (0.4–1 %) [1–4]. The diversity of pineal region tumors makes histology-specific therapy mandatory for improving outcome [5–7]. It has been well established that surgical resection plays a small role in the management of germinomas because of their sensitive response to radiotherapy or chemotherapy. However, the optimal treatment strategy for nongerminomatous pineal region tumors remains controversial, especially for malignant tumors or tumors containing mixed components.
With advances in microsurgical technique and postoperative care in recent years, encouraging surgical outcomes of pineal region tumors have been improved dramatically with the mortality reduced to a level under 4 % [5–10]. However, these tumors are still surgically challenging to the neurosurgeon because of the complicated adjacent anatomy and deep location. In most publications, special emphasis was given to the anatomical relationship between deep cerebral veins and the tumor [2, 6, 8, 9, 11–18]. The relationship between the tumor removal and arachnoid membranes distributing in this region has not been well described. Besides, the role of radical surgery in the recovery of the accompanying hydrocephalus was also seldom mentioned in previous studies. Therefore, we presented a large consecutive series of 143 patients with nongerminomatous pineal region tumors undergoing radical resection via the occipital transtentorial approach. The surgical results, outcome of hydrocephalus, and follow-up results were described in detail. Our purpose was to evaluate the feasibility of an aggressive surgical strategy for these tumors based on the arachnoid membranes and a specific approach.
Materials and methods
Neither approval of the institutional review board (IRB) nor patient informed consent were required by the ethics committee of our institution (IRB of Nanfang Hospital) for retrospective analyses of patients’ records and imaging data.
Patient characteristics
Between 2000 and 2011, 143 patients with nongerminomatous pineal region tumors underwent surgical resection using an occipital transtentorial approach at the Department of Neurosurgery, Nanfang Hospital, Guangzhou, China. Of these patients, there were 97 males and 46 females, and the mean age was 18.0 years (4–67 years). A total of 76 (53.1 %) tumors occurred in children, and 67 (46.9 %) occurred in adulthood. The most common presenting symptoms and signs were obstructive hydrocephalus and Parinaud’s syndrome, which were noted in 106 (74.1 %) and 57 (39.9 %) patients, respectively. Other major clinical manifestations included cerebellar syndrome in 23 (16.1 %), hemisyndromes in 12 (8.4 %), hearing impairment in 9 (6.3 %), cognitive/memory disturbances in 7 (4.9 %), and precocious puberty in 2 (1.4 %) cases.
Tumor markers
All patients underwent serum analysis for human β-human choriogonadotropin (β-HCG) and α-fetoprotein (AFP). The cerebrospinal fluid (CSF) was obtained in 118 patients for analysis of β-HCG and AFP through external ventricular drain (EVD), endoscopic third ventriculostomy (ETV), or lumbar puncture. Elevated levels of CSF tumor markers were observed in 18 (30.5 %) of 59 patients with nongerminomatous germ cell tumors (NGGCTs).
Neuroradiological studies
All patients underwent MRI routinely for evaluation of the tumor intensity, size and extension, and relationship between the tumor and deep cerebral veins. CT scans were also performed in all patients to detect calcification, tumor density, or intratumoral hemorrhage for the differential diagnosis. The tumor size was determined by the maximal diameter on MRI, and the tumors were thus classified into 3 groups: small (<2 cm) in 21 (14.7 %) patients, medium (2–4 cm) in 75 (52.4 %), and large (>4 cm) in 47 (32.9 %). The mean tumor size was 38.3 mm (13–67 mm).
Preoperative intervention
Permanent CSF diversion procedures, including shunt placement and endoscopic third ventriculostomy (ETV), were performed prior to the open surgery in 11 and 2 patients, respectively. Fifteen patients underwent external ventricular drainage (EVD) because of rapid deterioration. Twenty-four patients had received direct radiotherapy or chemotherapy 6 to 35 months before admission without histological diagnosis.
Surgical techniques
The patients were placed in the prone position or the three-quarters prone position. In the prone position, the head is slightly rotated with the approach side down to facilitate gravity-dependent retraction of the occipital lobe from the falcotentorial region. For most patients with no or mild hydrocephalus, brain relaxation could be easily achieved by the egress of CSF from the perimesencephalic cisterns. Otherwise, when brain relaxation was still unsatisfactory or the patient presented with severe hydrocephalus, an intraoperative puncture of the occipital horn was always necessary. The puncture point was usually located 6–7 cm above the transverse sinus and 3 cm lateral to the midline, and the catheter was then passed toward the mid-pupillary line. The procedure should be performed before the opening of the dura in order to avoid occipital contusion caused by high intracranial pressure. When an EVD was placed before surgery, it was clapped 8 h before surgery to allow ventricular dilation.
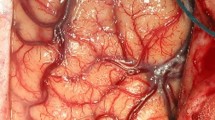
Because of the density of the important deep venous system in the pineal region, it is mandatory to remove tumors through the narrow space between these veins. Based on our anatomical studies on the arachnoid envelope over the pineal region (Fig. 1) [19–21], different surgical techniques were used to facilitate the procedure and reduce the risk of venous injury. For those tumors located within the arachnoid envelope, the envelope should be sequentially dissected prior to tumor removal to minimize the retraction or injury of the deep veins (Fig. 2 and Video 1). For tumors arising outside the envelope, however, the arachnoid envelope should be preserved as intact as possible to provide a protective boundary for deep veins during tumor removal (Fig. 3).
A case of immature teratoma. a Preoperative sagittal MRI; b-e intraoperative photographs. In b, the tumor was located within the arachnoid envelope and remained invisible before dissection of the envelope; in c, the tumor was exposed after sequential dissection of the envelope; in d, the tumor was removed en bloc after dissection; in e, the vein of Galen and its tributaries were well preserved without injury after tumor removal. f Immediate MRI after surgery demonstrating total tumor removal. AEPR, arachnoid envelope over the pineal region; BV, basal vein; CC, corpus callosum; IOV, internal occipital vein; PPCV, posterior pericallosal vein; T, tumor; TV; third ventricle; VG, vein of Galen
A case of meningioma. a Preoperative sagittal MRI; b–d intraoperative photographs. In b, the tumor was situated outside the arachnoid envelope; in c, the arachnoid envelope was preserved intact at the end of tumor removal, providing a protective boundary for deep veins; in d, a detailed observation of the surgical field under higher magnification showed the content of the arachnoid envelope. e Immediate MRI after surgery demonstrating total tumor resection and favorable preservation of the quadrigeminal plate (arrowhead). e A 2-year follow-up MRI showing no evidence of tumor recurrence. AEPR, arachnoid envelope; ICV, internal cerebral vein; QP, quadrigeminal plate; SPR, suprapineal recess; VG, vein of Galen
En-bloc resection was recommended in cases of small tumors. For large and tough tumors, however, intratumoral decompression or piecemeal resection was necessary to avoid venous retraction. The internal cerebral veins were frequently displaced superiorly by the tumor and hidden under the splenium of the corpus callosum. In such cases, a cotton sheet was inserted into the velum interpositum cistern to push the tumor downward to enter the surgical field. This technique was especially helpful when the anterior part of the tumor projected into the posterior third ventricle. Tumors slightly extending to infratentorial or contralateral space could be well exposed by retraction of the tentorial edge or falx. Otherwise, a tentorial or falx incision was necessary to gain additional exposure. At the end of tumor removal, the suprapineal recess should be opened if still kept intact, in an attempt to relieve the hydrocephalus without a second procedure.
Results
Extent of tumor resection
The extent of tumor resection was evaluated by both intraoperative findings and immediate MRI review within 48 h after surgery (Fig. 4). However, in one patient who died early postoperatively, the extent of tumor removal could only be estimated by the intraoperative findings. Accordingly, gross total resection (GTR) was defined as 100 % macroscopic removal of the tumor or no visible tumor remaining, subtotal resection (STR) was defined as >80 % but <100 % resection, and partial resection (PR) was defined as no more than 80 % resection. GTR was achieved in 131 patients (91.6 %); 10 patients (7.0 %) had STR, and 2 (1.4 %) underwent PR.
Postoperative immediate sagittal MRI showing total tumor removal compared with preoperative MRI in several cases with pineal region tumors. a and b Mixed germ cell tumor in a 13-year-old male; c and d pineoblastoma in a 22-year-old male; e and f oligodendroglioma in a 16-year-old female; g and h meningioma in a 56-year-old male
Pathohistology
All 143 tumors were histologically verified. The distribution of histological subtypes is noted in Table 1. Nearly half of the tumors (59 cases, 41.3 %) were NGGCTs, in which teratomas (29 cases, 20.3 %) and mixed GCTs (19 cases, 13.3 %) were most common subtypes. The histological diagnosis was a pineal parenchymal tumor in 21 patients (14.7 %), neuroepithelial tumors in 41 (28.7 %), and miscellaneous in 22 (15.4 %).
Outcome of hydrocephalus
Of the 106 patients presented with hydrocephalus preoperatively, permanent recovery was achieved in 87 (82.1 %) patients after radical tumor removal; 8 (7.5 %) patients remained unchanged, and 11 (10.4 %) recovered temporarily but had a recurrence within 1 to 3 months, possibly due to the reocclusion of the opening of the posterior third ventricle after surgery. Finally, 19 patients needed a second CSF diversion surgery, including ETV in 16 and shunting in 3. Of the 11 patients who had undergone shunt placement before tumor removal, 4 (36.4 %) required shunt revision or ETV due to shunt malfunction. (Fig. 5).
Adjuvant therapy
Sixty-eight patients with malignant tumors (including 37 NGGCTs, 7 pineal parenchymal tumors, 21 neuroepithelial tumors, and 3 miscellaneous tumors) underwent postoperative radiotherapy; 35 of these patients also received adjuvant chemotherapy according to standard protocols [22–24].
Complications
Overall postoperative complications occurred in 26 (18.2 %) of 143 patients. The most life-threatening complication was intracranial hemorrhage, which was noted in five patients (3.5 %). All these patients required an urgent reoperation to remove the hematoma. However, one patient died within 3 days because his parent refused the reoperation. Homonymous hemianopsia was the most common complication associated with surgery and occurred in 23 patients (16.1 %). However, the condition began to improve in the early postoperative period in most patients, and a permanent visual field defect was observed in only five patients (3.5 %). All five patients had hemianopsia immediately after surgery and MRI showed T2 hyperintense areas in the medial occipital lobe, suggesting damage to the visual cortex attributed to the excessive brain retraction. It should be mentioned that most of these permanent defects occurred in our early period. With increased experience, permanent hemianopsia only occurred in 1 of the last 81 cases (1.2 %, 2006–2011). New cases of Parinaud syndrome occurred in nine patients (6.3 %), three (2.1 %) of whom developed permanent disturbances. Other complications were mostly transient and reversible, including mild hemiparesis in three patients, bacterial meningitis in three, mental dysfunction in two, and subdural hygroma in two (Table 2). Overall permanent morbidity was noted in eight patients (5.6 %), most commonly associated with large and malignant tumors.
Follow-up and prognosis
One hundred thirty (90.9 %) patients were successfully followed up by way of outpatient visit, telephone, or e-mail. We were unable to get information on the remaining 13 (9.1 %) cases after discharge from the clinic. The mean duration of follow-up was 43 months (range, 6–125 months). The final mortality and functional outcomes were determined according to the modified Rankin Scale (mRS) score at the end of follow-up. Finally, 113 patients (86.9 %) achieved a favorable functional outcome (mRS ≤ 2), 4 (3.1 %) had an mRS score of 3, 2 (1.5 %) had an mRS score of 4, and 11 (8.5 %) had died (mRS = 6) (Table 3). The 11 patients included those with pineoblastoma (n = 2), NGGCT (n = 5), and astrocytoma (n = 4). All patients with benign or low-grade lesions are still alive.
Discussion
Role of radical surgery
The primary role of radical surgery is to obtain a sufficient volume of tumor tissue for accurate histological diagnosis of pineal region tumors, which has been regarded as an important step in guiding histology-specific therapy [5–7]. Some authors chose stereotactic or endoscopic biopsy as the initial surgical decision and mentioned minimal invasiveness and low risk associated with both procedures [25–30]. Endoscopic procedures have even demonstrated the potential to relieve hydrocephalus. However, the disadvantages of both methods lie mainly in the limited tissue sampling and insufficient ability to control bleeding. Kanamori et al. [31] performed endoscopic biopsy in 18 patients with pineal region tumors and found that endoscopic biopsy may be useful for distinguishing GCTs from other lesions, but not for determining the subtypes of GCTs in patients with normal and moderately elevated levels of tumor markers. In contrast, open surgery allows for obtaining a maximal tissue sample and provides wide exposure to lower the risk of bleeding, especially in large and highly vascular tumors. Furthermore, for those patients with nongerminomatous tumors, a second open surgery was usually inevitably required, and the accumulated risk associated with two surgical procedures was obviously higher than a single open surgery.
A second role of radical surgery is to provide maximal removal of tumor volume. It has been clearly defined that aggressive surgical resection is the optimal treatment for benign or low-grade pineal region tumors involving meningiomas, mature teratomas, pineocytomas, and epidermoids [8, 32–34]. For malignant tumors, however, no consensus has been reached on the extent of resection. With the exception of pure germinomas, malignant pineal region tumors frequently show poor response to either radiotherapy or chemotherapy. Under the circumstance, radical surgery can eliminate the tumor mass effect, reduce the tumor burden prior to adjuvant therapy, or remove the tumor components resistant to adjuvant therapy. Numerous reports have associated extensive resection of the malignant tumors with an improved response to adjuvant therapy and increased survival rate, although most studies are retrospective [35–38]. Another benefit of radical resection is the considerable recovery rate of hydrocephalus without a second procedure, which was noted in 82.1 % patients of this series. The risk of postoperative hemorrhage into the residual tumor bed may also be reduced by radical surgery. Most importantly, all the benefits should be established on the basis of acceptable mortality and morbidity. The radical surgical strategy was further justified by the low rates of mortality and permanent morbidity (0.7 % and 5.6 %, respectively) achieved in this series.
Selection of surgical approach
Various surgical approaches were adopted in the treatment of pineal region tumors [2, 6–9, 16, 17, 39, 40]. The supracerebellar infratentorial approach is the most widely used approach, followed by an occipital transtentorial and interhemispheric approach. The results of recent major surgical series using different approaches are listed in Table 4 [1, 2, 6–9, 15–17, 41–45]. The overall GTR and mortality rates of surgery were comparable between different approaches. However, neither the transient nor permanent morbidity could be compared between series because of the insufficiently provided data in nearly half of the series and the lack of unified standards of morbidity. Besides, the tumor size was not presented in most studies. Therefore, it was difficult to conclude which approach is the optimal one.
As we know, each approach has its advantages and disadvantages. The supracerebellar infratentorial approach, placing the patient in the sitting position, allows a relaxed natural surgical corridor to the pineal region without cerebellar retraction, reduces the risk of bleeding, and provides a clean surgical field because of the low venous pressure and outflow of fluid. Other benefits include facilitating dissection of the tumor from the deep venous system and avoiding cortical injury during the approach. The main limitation of the supracerebellar infratentorial approach is the risk of air embolism, although it rarely occurs. We have also used this approach in some cases. However, we found that the patient’s neck had to be extremely flexed in a sitting position, especially when a steep tentorial apex existed [14]. In these cases, the surgeon had to lean forward with the arms outstretched to overcome the long working distance, which may cause discomfort and rapid fatigue, as mentioned by Reid and Clark [40]. In comparison, the occipital transtentorial approach allows the surgeon more comfort and easy access to the pineal region. This is one of the main reasons why we preferred the occipital transtentorial approach in dealing with most pineal region tumors.
On the other hand, the supracerebellar approach is usually considered to be indicated for relatively small tumors without lateral expansion and primarily below the deep cerebral veins [9, 13, 40]. Besides, a very steep tentorium or extension of the tumor above the deep veins is a relative contraindication to the supracerebellar approach [13, 39]. In contrast, the occipital transtentorial approach is more appropriate for larger tumors because it can provide an excellent view of the tectum and tentorial notch [13]. It should also be noted that medium- and large-size tumors accounted for nearly 90 % of all cases in our series. The goal of radical tumor resection may not be achieved in some cases when using the supracerebellar approach. This is another reason why we choose the occipital transtentorial approach as the main method to remove pineal region tumors (in approximately 90 % of all cases). Based on our experience, this approach was indicated for most of the pineal region tumors.
The major limitation of the occipital transtentorial approach is the relatively difficult manipulation between complex deep cerebral veins during tumor removal, especially for large tumors. However, based on our increased experience and new understanding of arachnoid membranes in the pineal region, this is not such a big problem. Another drawback of the transtentorial approach is postoperative hemianopsia due to intervention of the medial occipital lobe. The hemianopsia, although mostly transient, developed into a permanent defect in 3.5 % of patients in this series. A possible reason for this complication was excessive retraction of the occipital lobe or vein injury when managing large tumors in our early period (2000–2005). With increasing experience, the permanent hemianopsia was reduced to a low level of 1.2 % in the last 81 cases (2006–2011). Based on our experience, the risk of permanent morbidity can be reduced by several methods: (1) do not use brain retractors as far as possible when managing small or medium tumors; (2) if a retractor must be used, place it superiorly to avoid direct contact with the visual cortex around the calcarine fissure; (3) use retractors more to hold the brain out of the way rather than aggressively retract.
Overall, there is not a specific surgical approach suitable for all pineal region tumors. The optimal approach for a given pineal region tumor depends on the tumor size, anatomical features of the tumor extension, the tumor’s relationship to the deep cerebral veins, the intended extent of tumor removal, and the surgeon’s experience and preference [5, 7, 39].
Arachnoidal consideration during tumor removal
Numerous anatomical and surgical studies of the pineal region have been reported in the past literature, but the arachnoid membranes of this region as well as their relationships with neighboring structures were seldom described. Based on our previous anatomical studies [19–21], the vein of Galen and its tributaries, as well as the pineal body and the suprapineal recess, were typically wrapped by a thickened arachnoid envelope over the pineal region. So far, no evidence has explained how the arachnoid membrane influences the growth pattern of pineal region tumors for their invasive entities. However, it is conceivable that the arachnoid membrane may play a role in benign or low-grade tumors and even in malignant tumors at their early phase. This presumptive theory was especially useful in our series due to the fact that benign or low-grade tumors account for nearly one half of all cases. As such, understanding of the relationship between the tumor and the arachnoid envelope may be helpful to protect deep veins during tumor removal. For example, it could be useful when deciding whether and how the membranes should be sequentially dissected or carefully reserved prior to tumor resection. We believe that surgical strategies based on arachnoid considerations may at least partially contribute to the favorable results achieved in this series.
Management of hydrocephalus
Treatment of obstructive hydrocephalus is one of the most important goals for surgical management of pineal region tumors. For a given patient with a nongerminomatous pineal region tumor, the treatment of the accompanying hydrocephalus should be carefully planned during the perioperative period based on several concerns: (1) shunt placement should be avoided whenever possible [5, 29,39]; (2) a single-stage surgical procedure is preferred to a two-stage one except for emergency conditions; (3) the intervention for the hydrocephalus should be designed with the consideration of facilitating the tumor removal and the specific approach selected.
The relationship between tumor removal and the beforehand relief of the hydrocephalus has been demonstrated by several authors. Konovalov et al. [9] found that total tumor removal could be achieved in 72 % of cases among shunt-free patients, whereas the rate was only 39 % in cases of previously shunted patients. They thereby concluded that collapsed ventricles may hamper radical tumor resection because of unsatisfactory exposure. Weshphal, in his comment on the study by Deshmukh et al. [46], also stressed the importance of controlling the ventricular size and advocated an ETV when the hydrocephalus needed to be relieved before the operation. As he stated, the ventricular system will remain dilated for a few days after this procedure and allows for easier manipulation of the tumor and protection of the posterior third ventricular wall. We agreed with this theory but preferred to place an EVD before surgery if needed. Compared with an ETV, an EVD would provide a more flexible ventricular volume by changing the drainage level or controlling the valve of EVD. Moreover, the hydrocephalus was resolved in the majority of cases (82.1 %) after radical tumor removal; thus, a second surgery for CSF diversion could be avoided.
Hydrocephalus is also an important factor hampering tumor exposure during the surgical procedure. For the supracerebellar infratentorial approach, rapid relaxation of the cerebellum can be achieved by opening the cisterna magna. In our experience, for most cases with moderate hydrocephalus when using the transtentorial approach, the occipital lobe can be relaxed by step-by-step dissection of the perimesencephalic cisterns and egression of the CSF. If it was still not sufficient or the patient presented with severe hydrocephalus, we advocated an intraoperative EVD to offer a size-controllable ventricle, which can definitely facilitate rapid brain relaxation during surgery and therefore minimize the risk of hemianopsia inherent to this approach.
Limitations of the study
This retrospective analysis has some inherent biases and drawbacks. Although this study is a large series, its retrospective character and the fact that the analyzed data derived from a single center limit the strength of the conclusions. Furthermore, because of the lack of statistical comparison among several surgical approaches, our selection of surgical approach was mainly established based on the surgeon’s preference, personal experience, and existing literature. This series also represents the continuous evolution of indications and treatment strategies for various tumor types and reflects the lack of consensus regarding their management. Besides, the follow-up was still insufficient for some cases. Further prospective randomized controlled trails based on the surgical approaches, treatment strategies, and long-term follow-up will be more helpful to guide clinicians managing these patients.
Conclusions
Radical surgery is recommended as the optimal treatment for nongerminomatous pineal region tumors. Favorable results can be expected with modern microsurgical techniques and experienced neurosurgeons. An occipital transtentorial approach was indicated for most pineal region tumors, but the surgeon’s preference and experience with each approach should also be considered in the selection of the surgical approach. Detailed knowledge about the arachnoid membranes spanning the pineal region may be helpful for total tumor removal and protection of the adjacent neurovascular structures. Hydrocephalus caused by tumors may be eliminated by radical tumor removal in the majority of the cases. In cases of unresolved hydrocephalus after surgery, an ETV procedure is preferred to the insertion of a shunt.
References
Cho BK, Wang KC, Nam DH, Kim DG, Jung HW, Kim HJ, Han DH, Choi KS (1998) Pineal tumors: experience with 48 cases over 10 years. Childs Nerv Syst 14:53–58
Jia W, Ma Z, Liu IY, Zhang Y, Jia G, Wan W (2011) Transcallosal interforniceal approach to pineal region tumors in 150 children. J Neurosurg Pediatr 7:98–103
Nomura K (2001) Epidemiology of germ cell tumors in Asia of pineal region tumor. J Neurooncol 54:211–217
Tomita T (1999) Pineal region tumors. In: Albright AL, Pollack IF, Adelson PD (eds) Priciples and practice of pediatric neurosurgery. Thieme New York, pp 571–589
Bruce JN, Ogden AT (2004) Surgical strategies for treating patients with pineal region tumors. J Neurooncol 69:221–236
Hernesniemi J, Romani R, Albayrak BS, Lehto H, Dashti R, Ramsey C 3rd, Karatas A, Cardia A, Navratil O, Piippo A, Fujiki M, Toninelli S, Niemela M (2008) Microsurgical management of pineal region lesions: personal experience with 119 patients. Surg Neurol 70:576–583
Pendl G (2002) Management of pineal region tumors. Neurosurg Quart:279–298
Bruce JN, Stein BM (1995) Surgical management of pineal region tumors. Acta Neurochir (Wien) 134:130–135
Konovalov AN, Pitskhelauri DI (2003) Principles of treatment of the pineal region tumors. Surg Neurol 59:250–268
Pettorini BL, Al-Mahfoud R, Jenkinson MD, Avula S, Pizer B, Mallucci C (2013) Surgical pathway and management of pineal region tumours in children. Childs Nerv Syst 29:433–439
Davidson L, McComb JG (2013) The safety of the intraoperative sacrifice of the deep cerebral veins. Childs Nerv Syst 29:199–207
Giordano M, Ludemann WO, Stieglitz L, Gerganov VM, Columbano L, Samii A, Samii M (2011) Identification of venous variants in the pineal region with three-dimensional preoperative magnetic resonance imaging navigation in patients harbouring tumors in this area: significance for surgical approach to the lesion. Clin Neurol Neurosurg 113:387–392
Hart MG, Santarius T, Kirollos RW (2013) How I do it—pineal surgery: supracerebellar infratentorial versus occipital transtentorial. Acta Neurochir (Wien) 155:463–467
Hasegawa M, Yamashita J, Yamashima T (1991) Anatomical variations of the straight sinus on magnetic resonance imaging in the infratentorial supracerebellar approach to pineal region tumors. Surg Neurol 36:354–359
Kodera T, Bozinov O, Surucu O, Ulrich NH, Burkhardt JK, Bertalanffy H (2011) Neurosurgical venous considerations for tumors of the pineal region resected using the infratentorial supracerebellar approach. J Clin Neurosci 18:1481–1485
Luo SQ, Li DZ, Zhang MZ, Wang ZC (1989) Occipital transtentorial approach for removal of pineal region tumors: report of 64 consecutive cases. Surg Neurol 32:36–39
Tanaka R, Washiyama K (2003) Occipital transtentorial approach to pineal region tumors. Oper Tech Neurosurg 6:215–221
Yamamoto I (2001) Pineal region tumor: surgical anatomy and approach. J Neurooncol 54:263–275
Qi ST, Fan J, Zhang XA, Pan J (2011) Reinvestigation of the ambient cistern and its related arachnoid membranes: an anatomical study. J Neurosurg 115:171–178
Qi ST, Zhang XA, Fan J, Huang GL, Pan J, Qiu BH (2011) Anatomical study of the arachnoid envelope over the pineal region. Neurosurgery 68:7–14, discussion 14–15
Zhang XA, Qi S, Fan J, Huang G, Peng J, Xu J (2012) The distribution of arachnoid membrane within the velum interpositum. Acta Neurochir (Wien) 154:1711–1715
Habrand JL, De Crevoisier R (2001) Radiation therapy in the management of childhood brain tumors. Childs Nerv Syst 17:121–133
Matsutani M, Sano K, Takakura K, Fujimaki T, Nakamura O (1998) Combined treatment with chemotherapy and radiation therapy for intracranial germ cell tumors. Childs Nerv Syst 14:59–62
Oi S (1998) Recent advances and racial differences in therapeutic strategy to the pineal region tumor. Childs Nerv Syst 14:33–35
Morgenstern PF, Osbun N, Schwartz TH, Greenfield JP, Tsiouris AJ, Souweidane MM (2011) Pineal region tumors: an optimal approach for simultaneous endoscopic third ventriculostomy and biopsy. Neurosurg Focus 30:E3
Morgenstern PF, Souweidane MM (2013) Pineal region tumors: simultaneous endoscopic third ventriculostomy and tumor biopsy. World Neurosurg 79(S18):e19–13
Pople IK, Athanasiou TC, Sandeman DR, Coakham HB (2001) The role of endoscopic biopsy and third ventriculostomy in the management of pineal region tumours. Br J Neurosurg 15:305–311
Regis J, Bouillot P, Rouby-Volot F, Figarella-Branger D, Dufour H, Peragut JC (1996) Pineal region tumors and the role of stereotactic biopsy: review of the mortality, morbidity, and diagnostic rates in 370 cases. Neurosurgery 39:907–912, discussion 912–904
Teo C, Young R 2nd (1999) Endoscopic management of hydrocephalus secondary to tumors of the posterior third ventricle. Neurosurg Focus 7:e2
Zacharia BE, Bruce JN (2011) Stereotactic biopsy considerations for pineal tumors. Neurosurg Clin N Am 22:359–366, viii
Kanamori M, Kumabe T, Tominaga T (2008) Is histological diagnosis necessary to start treatment for germ cell tumours in the pineal region? J Clin Neurosci 15:978–987
Barnett DW, Olson JJ, Thomas WG, Hunter SB (1995) Low-grade astrocytomas arising from the pineal gland. Surg Neurol 43:70–75, discussion 75–76
MacKay CI, Baeesa SS, Ventureyra EC (1999) Epidermoid cysts of the pineal region. Childs Nerv Syst 15:170–178
Vaquero J, Ramiro J, Martinez R, Coca S, Bravo G (1990) Clinicopathological experience with pineocytomas: report of five surgically treated cases. Neurosurgery 27:612–618, discussion 618–619
Lutterbach J, Fauchon F, Schild SE, Chang SM, Pagenstecher A, Volk B, Ostertag C, Momm F, Jouvet A (2002) Malignant pineal parenchymal tumors in adult patients: patterns of care and prognostic factors. Neurosurgery 51:44–55, discussion 55–46
Matsutani M, Sano K, Takakura K, Fujimaki T, Nakamura O, Funata N, Seto T (1997) Primary intracranial germ cell tumors: a clinical analysis of 153 histologically verified cases. J Neurosurg 86:446–455
Reddy AT, Janss AJ, Phillips PC, Weiss HL, Packer RJ (2000) Outcome for children with supratentorial primitive neuroectodermal tumors treated with surgery, radiation, and chemotherapy. Cancer 88:2189–2193
Weiner HL, Finlay JL (1999) Surgery in the management of primary intracranial germ cell tumors. Childs Nerv Syst 15:770–773
Little KM, Friedman AH, Fukushima T (2001) Surgical approaches to pineal region tumors. J Neurooncol 54:287–299
Reid WS, Clark WK (1978) Comparison of the infratentorial and transtentorial approaches to the pineal region. Neurosurgery 3:1–8
Mottolese C, Szathmari A, Ricci-Franchi AC, Beuriat PA, Grassiot B (2014) The sub-occipital transtentorial approach revisited base on our own experience. Neurochirurgie. doi:10.1016/j.neuchi.2013.12.005
Oliveira J, Cerejo A, Silva PS, Polonia P, Pereira J, Vaz R (2013) The infratentorial supracerebellar approach in surgery of lesions of the pineal region. Surg Neurol Int 4:154
Pluchino F, Broggi G, Fornari M, Franzini A, Solero CL, Allegranza A (1989) Surgical approach to pineal tumours. Acta Neurochir (Wien) 96:26–31
Sajko T, Kudelic N, Lupret V, Lupret V Jr, Nola IA (2009) Treatment of pineal region lesions: our experience in 39 patients. Coll Anthropol 33:1259–1263
Vaquero J, Ramiro J, Martinez R, Bravo G (1992) Neurosurgical experience with tumours of the pineal region at Clinica Puerta de Hierro. Acta Neurochir (Wien) 116:23–32
Deshmukh VR, Smith KA, Rekate HL, Coons S, Spetzler RF (2004) Diagnosis and management of pineocytomas. Neurosurgery 55:349–355, discussion 355–347
Acknowledgments
We are grateful to Xiaoyu Qiu and Yiping Mo (Medical Secretary, Department of Neurosurgery, Nanfang Hospital, Southern Medical University, Guangzhou, China) for their help with the data collection.
Disclosure
The authors have no personal financial or institutional interest in any of the drugs, materials, or devices described in this article.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Drs. Songtao Qi, Jun Fan, and Xi-an Zhang contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Surgical case illustration. An occipital transtentorial approach was performed in a patient with a mixed germ cell tumor in pineal region. As shown by the preoperative MRI, the tumor arose within the arachnoid envelope wrapping the vein of Galen and its tributaries. Therefore, the envelope was dissected carefully prior to the tumor resection. After the deep veins had been totally released, a satisfactory exposure of the tumor could be achieved, and the retraction of veins could be reduced to minimize the risk of injury. During the procedure, the pineal vein was identified, coagulated, and separated from the tumor. Once all tumor-brain interfaces had been dissected, the tumor was removed en bloc. The adjacent neurovascular structures remained intact after tumor resection. Early postoperative MRI showed total removal of the tumor. Histology-specific adjuvant therapy was then performed as planned. A 5-year follow-up postoperative MRI presented no tumor recurrence. (WMV 16,185 kb)
Rights and permissions
About this article
Cite this article
Qi, S., Fan, J., Zhang, Xa. et al. Radical resection of nongerminomatous pineal region tumors via the occipital transtentorial approach based on arachnoidal consideration: experience on a series of 143 patients. Acta Neurochir 156, 2253–2262 (2014). https://doi.org/10.1007/s00701-014-2224-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00701-014-2224-4