Abstract
Crinum asiaticum is phenotypically specialised with white, scented, very long-tubed flowers, suggesting that only long-proboscid hawkmoths may be effective pollinators. However, this species has frequently escaped cultivation in many tropical and subtropical regions. We therefore investigated the reproductive biology of C. asiaticum to understand how it is able to naturalise, despite phenotypic specialisation. We examined the ability for autonomous selfing and reliance on sexual versus vegetative reproduction in C. asiaticum var. sinicum, using a group individuals growing and propagating naturally in a suburban botanical garden. We also analysed the floral syndrome, recorded floral visitors, determined the pollination effectiveness of floral visitors, and reviewed records of floral visitors at different observation sites. Sexual reproduction provides a much greater potential for dispersal than vegetative reproduction in C. asiaticum var. sinicum. This plant does not perform autonomous selfing and shows poor self-compatibility. The floral syndrome strongly points to long-proboscid hawkmoth pollination, whereas both hawkmoths and swallowtail butterflies with varied tongue lengths were observed as floral visitors because the nectar can accumulate to high levels and therefore fills the majority or all of the perianth tube. Both butterflies and hawkmoths effectively deposited pollen on stigmas. Thus, natural propagation should be attributed to sufficient cross pollination by local lepidopterans. Our findings suggest that autonomous selfing and vegetative reproduction may not be necessary for naturalisation and long-term persistence of plant populations in highly disturbed habitats or new ranges, even if the plants exhibit an extremely specialised floral syndrome.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Floral phenotypic specialisation characterises numerous lineages in the radiation of angiosperms (Fenster et al. 2004; van der Niet and Johnson 2012). It favours the use of a narrow group of pollinators (Armbruster 2006; Ollerton et al. 2007) and may lead to various ecological consequences. For instance, it constrains the spectrum of potential pollinators of a plant species, possibly leading to a specialised pollination system (Fenster et al. 2004; Ollerton et al. 2007). Plants exhibiting significant floral phenotypic specialisation may more often experience sexual reproduction failure in the face of human disturbance than will other plants (e.g. Johnson et al. 2004; Reiter et al. 2017; Walsha et al. 2019). Compared with phenotypic generalisation, it could in theory put plant populations at risk because of pollinator loss (Waser et al. 1996; Memmott et al. 2004). Moreover, if introduced to new habitats, these plants are less likely to be naturalised and become further invasive because they have a lower chance of encountering effective pollinators (Ollerton et al. 2012).
On the other hand, floral phenotypic specialisation does not necessarily lead to these ecological consequences. Some plants are capable of long-term persistence without frequent sexual reproduction mediated by pollination agents. These plants may exhibit highly specialised pollination systems but produce seeds by autonomous selfing or expand ranges by vegetative reproduction (Clark-Tapia and Molina-Freaner 2004; van Kleunen et al. 2008). Alternatively, floral phenotypic specialisation does not incur specialised pollinator use (Ollerton et al. 2007; Waser et al. 2011; Celep et al. 2014). In other words, flowers may attract and be pollinated by floral visitors which their highly specialised phenotypes would not imply. A third possibility is that phenotypic specialisation does result in exclusive adaptation to a narrow range of pollinators with specific morphology and behaviour, but these pollinators are themselves widespread and disturbance-adapted or may have co-invaded new ranges with the plants they pollinate (Gardner and Early 1996; Liu and Pemberton 2010; Johnson and Raguso 2016); consequently, a plant species will enjoy its own pollinator niche despite highly disturbed habitats or the introduction to new habitats. These scenarios are not mutually exclusive. For instance, Nicotiana glauca Graham is native to northern Argentina and has bird-adapted flowers. It has invaded many regions in both the New and Old World because it produces abundant seeds by autonomous selfing and pollination by birds in invaded ranges (Geerts and Pauw 2009; Ollerton et al. 2012).
Many plants have evolved pale sweet-scented flowers with long tubular structures, which provide copious nectar for hawkmoths and are pollinated by these long-proboscid insects (Martins and Johnson 2013; Johnson et al. 2017). One might expect that such specialised mutualisms are fragile because the deep floral tubes would exclude most flower-visiting animals. This is particularly true for hawkmoth flowers with extraordinarily long floral tubes because even most hawkmoths species cannot gain access to the deeply concealed nectar. For example, a guild of African plants with long-tubed (> 8 cm) flowers are exclusively pollinated by two local long-proboscid hawkmoth species (Johnson and Raguso 2016). The loss or rarity of these hawkmoths would probably lead to a dramatic decline in the sexual reproduction of such plants. To our knowledge, two endangered plant species showing floral syndromes suggestive of hawkmoth pollination have indeed been reported to suffer the loss of pollinators and consequent seedling recruitment failure (Johnson et al. 2004; Walsha et al. 2019). On the other hand, there are also hawkmoth-pollinated plants that become naturalised or invasive (Johnson and Raguso 2016), but few studies have investigated the mechanisms of the maintenance of their populations. One example is Lilium formosanum Wallace, which is native to Asia and attracts hawkmoths with white fragrant nectar-rich flowers in South Africa (Rodger et al. 2010). However, it has the capacity for autonomous selfing so it remains unknown whether long-tubed hawkmoth-adapted flowers form a barrier against plant sexual reproduction in invaded or disturbed habitats.
Crinum asiaticum L. displays a syndrome strongly pointing to the adaptation to long-proboscid hawkmoth pollination (i.e. with floral tubes of approximately 8 cm). We noticed that it could naturally set seeds and propagate in three urban and suburban botanical gardens in southern China (i.e. Guilin Botanical Garden, South China Botanical Garden, and Xishuangbanna Tropical Botanical Garden). There have been anecdotal reports of insects visiting C. asiaticum elsewhere (Knuth and Loew 1904–1905; Miyake et al. 1998; Matyot 2005), but the reproductive ecology has not been formally investigated. Here, we explored how C. asiaticum var. sinicum is propagated in an urban botanical garden. Our aims were to (1) test for the dependence of C. asiaticum on sexual versus vegetative reproduction for propagation, (2) test for self-compatibility and the capability of autonomous selfing, (3) analyse the floral syndrome, (4) determine the identity of floral visitors and their contributions to reproductive success, and (5) review documents of floral visitors at different observation sites worldwide.
Materials and methods
Study species and sites
Crinum asiaticum var. sinicum is a perennial herb that produces one or more umbels with white, long-tubed, salver-form fragrant flowers. C. asiaticum often grows near rivers and is naturally distributed in southeastern Asia and Oceania. It is widely cultivated as an ornamental in tropical and subtropical regions and has extensively escaped cultivation in both hemispheres (WCSP 2016). The variety C. asiaticum var. sinicum (Roxb. ex Herb.) Baker is extensively cultivated or naturally distributed in southern China. To our knowledge, it is usually difficult to tell whether individuals of this variety growing at a site are wild or cultivated in China, although it is reported to occur naturally in several southernmost provinces, such as Guangxi and Guangdong (Ji and Meerow 2000). It was introduced into a suburban garden, the Guilin Botanical Garden in 1976 and has propagated naturally from eight to more than 30 individuals. Guilin is within the natural historical range of C. asiaticum var. sinicum because it is located in one of the southernmost provinces of China. However, the Guilin Botanical Garden is surrounded by farmlands and hills without primitive vegetation so the habitat is highly disturbed. New plantlets emerge every year in large numbers, but most were removed by gardeners. Sometimes one or two well-grown plantlets were left, which is the reason why the number of adult plants has increased. This study was conducted in the Guilin Botanical Garden, which is located on Yanshan Street, Guilin City, Guangxi, China (25° 0454″ N, 110° 1815″ E, 124 m altitude).
Dispersal modes
To estimate the contribution of sexual and vegetative reproduction to natural propagation, we first compared their dispersal distances without assistance of agents. As fruits developed, the flowering stem was forced by the weight of the fruits to fall and contact the ground. The seeds in the fruits germinate in October, such that each infructescence produces several seedlings growing side by side. On 15 November 2019, using a plastic ruler, we measured the distance (e.g. the dispersal distance by sexual reproduction) between the stem and the central seedling among these seedlings (Fig. 1). To assess the dispersal distance by vegetative reproduction, we measured the distance between the main bulb and the largest shoot from the bulbils produced by the main bulb (Fig. 1). The measurement was conducted on the ground level.
Possibility of secondary dispersal was also tested. We examined fruits of C. asiaticum var. sinicum to determine whether they provided edible tissues for animals. To test whether fruits could be dispersed by water like the fruits of other Crinum (Bjorå et al. 2006), we tested whether fruits float and estimated the extent to which seeds tolerate soaking. We randomly chose three fruits from each of 30 individuals and divided them into 3 groups. Then each group contained 30 fruits from 30 individuals. Each group was put in a glass container nearly full of water. Groups 1, 2 and 3 soaked for 0, 6 and 12 days, respectively. Then we scattered them on a seed bed that was kept moist by watering every 3 days in a greenhouse in Guilin Botanical Garden. We counted the seeds that had germinated in December 30.
Floral visitors and proboscis lengths
We observed diurnal and nocturnal floral visitors from 2017 to 2020 during peak flowering in late July and early August. Nocturnal observations were conducted from 19:30 to 22:30 when nocturnal visitors were most active. Diurnal observations were conducted randomly from 7:00 to 19:00. The total time spent observing nocturnal or diurnal visitors each year is listed in Table 1. Four to eight umbels were observed during each observation session. Each umbel includes four or more open flowers. During each observation session, we recorded the number of visits to individual inflorescences by each of the three visitor groups.
The proboscis length of three floral-visiting hawkmoth species was measured. The individuals of hawkmoths for this measurement were captured over several years when they were visiting other flowers (including Quisqualis indica, Luffa acutangula, Gardenia jasminoides, and Hymenocallis littoralis) around Guilin Botanical Garden.
We summarised all reports on floral visitors to C. asiaticum and determined proboscis lengths of the visitors by searching Google Scholar for papers containing the terms “Crinum asiaticum” and “pollination”. The data on proboscis length of the visitors that we did not measure were retrieved from previous studies.
Floral traits
To test whether the anthesis of C. asiaticum var. sinicum tended to start around dusk, we randomly chose one floral bud at 18:00 h from each of 35 individuals to document when it started to open. We defined the time that the perianth lobes burst as when anthesis started.
Perianth tube length of 25 flowers from 25 individuals were measured to the nearest millimetre with a plastic ruler.
To examine the amount of nectar that a flower could provide for nocturnal visitors, we caged one flower of 24 individuals in a nylon net (aperture: 0.3 × 0.3 mm) from 19:00 h to 8:30 h the next day. Nectar volume was measured using glass capillary tubes (0.5 mm in diameter) and nectar concentration was measured using a hand-held sucrose refractometer (0%–50%, g solute per 100 g solution; Bellingham and Stanley, Ltd., London, UK).
We documented regular floral visitors with proboscises much shorter than the perianth tubes of C. asiaticum var. sinicum in all the four successive flowering seasons, and we investigated why this occurred. We that nectar could accumulate quickly and easily rose to a level near the tube entrance, such that short-proboscid insects could access some of the nectar. To examine the accessibility of nectar for short-proboscid visitors, we estimated nectar column length in flowers and the distance between the entrance of the perianth tube and the nectar (nectar depth) in the summer of 2020. We inserted a glass capillary tube (0.1 mm in diameter) into the perianth tube at 4 mm and removed the tube to test whether there was any nectar in the tube. If no nectar was in the tube, we inserted the tube for the second time at 8 mm and removed it. If there was no nectar, we continued to push the tube deeper and the length of the tube insertion increased for 4 mm (i.e. 4, 8, 12, 16, …) until there was a noticeable nectar column in the tube. Nectar depth was estimated as (a + b)/2, where a is the length of the inserted portion when the tube tip obtained an amount of nectar, whereas b is the length of the inserted portion immediately before insertion, during which the tube tip obtained an amount of nectar. Nectar column length was estimated as “average perianth tube length–nectar depth”. We measured nectar depth in four flowers from each of two inflorescences of a plant at 7:30 h when diurnal nectar foragers became active after a few minutes; nectar depth in another four flowers from a second inflorescence of the same plant was measured at 19:30 h when nocturnal nectar foragers became active. A total of 21 plants were used for nectar depth. The measurements were conducted over 7 days for three plants per day. We then determined the number of flowers that presented nectar that was accessible to short-proboscid floral visitors.
Mating system and pollination effectiveness
To test incompatibility, the capacity of autonomous selfing and the overall pollination effectiveness of floral visitors in C. asiaticum, we chose 16 flowers from each of the 12 individuals to conduct four pollination treatments. The 16 flowers were equally divided into four groups, and four flowers of each group were used for one pollination treatment. Flowers used in the autonomous selfing treatment were bagged and did not receive hand pollination. A second group of flowers was also bagged and hand-pollinated with pollen from newly opened flowers from the same plant for the self-pollination treatment. Flowers used in the cross-pollination treatment were bagged and hand-pollinated with outcrossing pollen from two different pollen donors. The bags (apertures: 0.3 × 0.3 mm) used in the above three treatments were removed when the flowers wilted. Another group of flowers remained open to all potential visitors throughout anthesis was the natural-pollination treatment. The four pollination treatments were conducted in early August 2019. We checked the number of the ovaries that were significantly expanded 60 d after the wilting of the treated flowers.
To compare the pollination effectiveness of diurnal and nocturnal floral visitors, we bagged a group of 62 floral buds before opening in the afternoon in a nylon net (apertures: 0.3 × 0.3 mm). A total of 31 individual plants were used, and each had two bagged floral buds. Once the flowers opened, we emasculated the flowers and bagged them again. One day later, they were in the female phase and we exposed half of them to nocturnal visitors for a night (from 19:00 to 7:00 h the next day) and the other half to diurnal visitors (from 7:00 to 18:00 h). We then counted the pollen grains on each stigma using the methods introduced by Kiepiel and Johnson (2014). One of the two flowers in each plant was exposed at night, whereas the other was exposed during the daytime. The experiment was conducted in mid-August.
Statistical analysis
To compare pollen grains deposited by diurnal and nocturnal floral visitors, a generalised linear model with a Poisson distribution and a logit link function was applied. To compare the fruit sets and seed germination rates of different pollination treatments, we applied a generalised linear model with a binomial distribution with a log link function. The difference between the dispersal distances by sexual versus vegetative reproduction was analysed using the Mann–Whitney U test. Statistical analyses were conducted using SPSS 22.0 (IBM, Armonk, NY, USA).
Results
Dispersal modes
The dispersal distance by sexual reproduction was significantly longer than that by vegetative reproduction (U = 0, P < 0.001); the former was approximately 7 times longer than the latter (Fig. 1).
Bulbils could not be spontaneously separated from the main bulb, such that they always grew next to the main bulb. In contrast, fruits were shed from infructescence when they ripened, providing an opportunity to move away from the parental plant.
Fruit coats were dry, membranaceous and translucent. Seed coats were dry and chartaceous, not fleshy. Fruits therefore did not seem to provide any edible tissues for animals and we have never found signs of being gnawed by any animals. Seeds floated and the seed germination rates of the seeds experiencing 0-day soaking, 4-day soaking and 12-day soaking were 46.7%, 50.0% and 43.3% respectively. The three soaking treatments did not significantly differed in germination rate (Wald χ2 = 0.187, df = 2, P = 0.911), suggesting that soaking for 12 days did not even significantly reduce seed vigour.
Floral visitors and proboscis lengths
Both hawkmoths and butterflies were common floral visitors to C. asiaticum var. sinicum in each of the four observation years, despite inter-annual fluctuation in abundance (Table 1). Four hawkmoth species visited flowers in the evenings (Table 1, Fig. 2a, Online Resource 1). Agrius convolvulii alighted or hovered around flowers when extracting nectar. The remaining species alighted and did not hover. All these hawkmoths contacted anthers and stigmas, carrying pollen on various parts of the body and wings (Fig. 2, Online Resource 1).
Flower-visiting butterflies belonged to Papilionidae (i.e. swallowtail butterflies). These butterflies fluttered their wings when visiting a flower, such that the wings contacted the anthers and stigma, carrying pollen on the ventral surfaces of the body and wings (Fig. 2b). Diurnal hawkmoths (Macroglossum sp.) and pollen-collecting bees (Lasioglossum sp.) were rare visitors (Table 1). The former alighted on the flowers but they might not contact the floral sexual organs as firmly as did the nocturnal hawkmoths because of their small stature; the latter never contacted stigmas because the stigma and the anthers were so widely spaced.
Hawkmoths were seen visiting C. asiaticum in all four localities where attempts to observe floral visitors were made, whereas swallowtail butterflies were seen in our study site and in Japan (Table 2). Proboscis length varied greatly among the visitor species (Table 2).
Floral traits
The floral syndrome of C. asiaticum coincides well with the reports on hawkmoth flowers in the literature (Table 3). The anthesis of C. asiaticum var. sinicum began at dusk. Flowers were fragrant. Nectar was abundant and dilute, and contained in a remarkably long perianth tube. The anthers immediately dehisced after the protandrous flower opened when the style curved outwards so the visitor could contact the newly exposed pollen but had little chance to contact the stigma; the style became erect the next day so that the visitor was more likely to contact the stigma. The stigma did not contact the anthers throughout the anthesis.
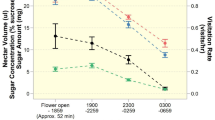
Among the 84 measured flowers from 21 plants, nectar in 48 flowers was accessible to the swallow butterfly Papilio helenus to some extent at 7:30 h, which was the dominant butterfly visitor at Guilin Botanical Garden (Fig. 3). Among the other group of 84 measured flowers at 19:30, nectar in 62 flowers could more or less be reached by the hawkmoth, Acosmeryx pseudomissa, which had the shortest proboscis among the nocturnal nectar foragers.
Nectar column length and nectar depth. The red bar indicates nectar column length (84 flowers were measured in total) at 7:30 (a) and 19:30 (b). The higher blue dotted line indicates the average height of the perianth tube (i.e. the entrance of the tube); the distance between the higher blue dotted line and a red bar represents nectar depth. The lower blue dotted line indicates the estimated level that the insect could reach within the perianth tube and nectar above this line could be accessed by the insect. The black vertical dotted line indicates the average length of fully uncoiled proboscises of the insect. The butterfly and hawkmoth are Papilio helenus and Acosmeryx pseudomissa, respectively. The former was active by day and was the most abundant diurnal nectar forager, whereas the latter was the shortest-proboscid nocturnal nectar forager for Crinum asiaticum var. sinicum at Guilin Botanical Garden
Mating system and pollination effectiveness
No ovaries expanded in the autonomous selfing treatments; therefore, this treatment was not included in the statistical analysis. Fruit set differed among the self-pollination, cross-pollination, and natural-pollination treatments (Wald χ2 = 21.105, df = 2, P < 0.001, Fig. 4). The fruit set in both the cross-pollination and natural-pollination treatments (both P < 0.001) were significantly higher than those from the self-pollination treatment, but the two treatments did not differ significantly from each other in fruit set (P = 0.552, Pollen Limitation Index = 0.08, 2019). Thus, the study population of C. asiaticum var. sinicum could perform sexual reproduction without pollinators and has a low level of self-compatibility (Self-Incompatibility Index = 0.69).
Mean proportion fruit set after three pollination treatments (± SE). n = 12 for each treatment. Fruit set with different letters differed significantly at the 5% level in a generalised linear model with a binomial distribution and a logit link function. The autonomous selfing treatment led to no fruit production and is not shown here
The flowers exposed to diurnal and nocturnal visitors did not differ significantly in pollen deposition (Wald χ2 = 1.219, df = 1, P = 0.270, Fig. 5).
Mean pollen grain deposition per stigma (± SD) on virgin flowers exposed to nocturnal (hawkmoths) and diurnal (mostly swallowtail butterflies) floral visitors. n = 31 for each treatment. A generalised linear model with a binomial distribution and a logit link function revealed no significant difference between diurnal and nocturnal deposition (Wald χ2 = 1.219, df = 1, P = 0.270)
Discussion
Seedlings exhibited even longer dispersal distances than plantlets produced by vegetative reproduction in C. asiaticum var. sinicum (Fig. 1). Seeds floated, tolerated soaking for at least 12 days and therefore could be potentially dispersed further. The floral syndrome implied a specialised pollination system involving hawkmoths with proboscises of approximately 8 cm in length or longer. Its flowers did attract hawkmoths with relatively long proboscises (Fig. 2; Table 2). However, swallowtail butterflies were also frequent visitors, depositing pollen grains as effectively as hawkmoths (Fig. 5). Flowers did not produce seeds without pollinators and were only poorly self-compatible (Self-Incompatibility Index = 0.69). No pollen limitation was found in 2019 (Pollen Limitation Index = 0.08) (Fig. 4).
The comparison of dispersal distances by sexual versus vegetative reproduction highlighted the advantage of sexual reproduction in dispersal (Fig. 1). Clonal reproduction is common in Amaryllidaceae and other families, enhancing the establishment and persistence of plant populations in intensely disturbed habitats or new locations (Klimes et al. 1997; Clark-Tapia and Molina-Freaner 2004; Liu et al. 2006). However, C. asiaticum var. sinicum does not have rhizomes. Furthermore, the bulbils were closely attached to the centre bulb and could not be autonomously separated and be moveable. On the other hand, the long flowering stem bent sideways to contact the ground with the heavy fruits, consequently positioning the seedlings distant from the main stem of the plant. In comparison to seedlings, the bulbils compete more intensely with the centre bulb because they are extremely close, suggesting that seedlings have a greater chance of surviving. This may further constrain the contribution of vegetative reproduction to the natural propagation of C. asiaticum var. sinicum at Guilin Botanical Garden and other localities. Thus, the individuals may have grown from seedlings because they were all widely spaced. Moreover, fruits and seeds displayed no signs of attracting animals but seeds retained vigour after soaking for a long period, which might promote secondary dispersal by flowing water. Given that C. asiaticum var. sinicum often occurs near rivers or streams, seed dispersal by water seems particularly probable. Adaptations of seeds to dispersal by water may be common in the genus Crinum (Bjorå et al. 2006).
Autonomous selfing is another trait that enables plants to regenerate populations in the absence of pollinators. For example, an investigation of invasive vs. non-invasive Iridaceae demonstrated that invasive species displayed significantly higher levels of fecundity by autonomous selfing (van Kleunen et al. 2008). Lilium formosanum, which invaded South Africa, is capable of autonomous selfing, although hawkmoths also contribute to reproductive success (Rodger et al. 2010). Crinum asiaticum var. sinicum produced no seeds with floral visitors excluded and hand self-pollination initiated much less fruit than did hand cross-pollination and natural pollination (Fig. 4), confirming that abundant seed production of this species resulted from cross-pollination by floral visitors in Guilin Botanical Garden. Insects were observed visiting this species at all four sites where observations were conducted (Table 2). Our findings are similar to those in some other plants (Herrera and Nassar 2009; Nienhuis and Stout 2009; Skogen et al. 2016), in which flowers show phenotypic specialisation and pollinators are necessary for seed production, but high levels of seed set in invaded or disturbed habitats were documented because of the abundant efficient pollinators. Our study demonstrates that plants with extremely long-tubed flowers such as C. asiaticum var. sinicum can also be pollinated enough and flourish in those habitats without autonomous selfing and breakdown of self-incompatibility (Fig. 4).
Crinum asiaticum exhibits floral phenotypic specialisation that points to hawkmoth pollination (Table 3). Although floral scent composition was not investigated here, an analysis of floral scent for this species in Japan revealed volatile organic compounds typically associated with hawkmoth pollination (Table 3). In particular, the extraordinarily long perianth tubes might imply adaptation to hawkmoths with long proboscises of similar length (ca. 8 cm). In the Old World, only a few hawkmoth species possess such proboscises (Miller 1997), suggesting that plants with similar floral tube lengths may have remarkably specialised pollination systems. In Africa, there is indeed a guild of plant species whose nectar is concealed in floral tubes > 8 cm, which is mainly pollinated by the long-proboscid Agrius convolvuli (Johnson and Raguso 2016). This hawkmoth is widespread in the Old World (Pittaway and Kitching 2020) and was recorded as one of the visitors to C. asiaticum in three widely spaced sites (Table 2; Online Resource 1). Unlike the long-tubed flowers in Africa studied by Johnson and Raguso (2016), long-proboscid hawkmoths other than A. convolvuli, i.e. Psilogramma discistriga and P. increta (Table 2), were also common floral visitors to the population we observed. Both Psilogramma species are widespread and common in Asia; several other hawkmoth species (e.g. Pergesa actea and Apocalypsis velox, with proboscises not shorter than 70 mm, unpublished data) with long proboscises not recorded in this study can also be easily found in tropical Asia (Pittaway and Kitching 2020). There might therefore be a long-proboscid hawkmoth pollinator niche composed of more abundant hawkmoths species for the Asian flora.
The hawkmoth visitors to C. asiaticum var. sinicum could not avoid contacting the highly exserted style and the neighbouring stamens and thereby transferring pollen. This was shown by the photos of these moths visiting flowers and pollen deposition data (Figs. 2, 5; Online Resource 1). Notably, the document of A. convolvuli as floral visitors to C. asiaticum in Seychelles, where C. asiaticum has been introduced (Matyot 2005), demonstrated that C. asiaticum could be pollinated by hawkmoths outside its native range.
A striking aspect of pollination ecology in C. asiaticum is that unlike many plants with long-tubed flowers (e.g. Paudel et al. 2016; Johnson and Raguso 2017; Balducci et al. 2019a, b), it is visited not only by long-proboscid insects but also by those with relatively short proboscises, i.e. swallowtail butterflies and the hawkmoth Acosmeryx pseudomissa (Table 2). The butterflies could deliver pollen (Fig. 5), whereas the short-proboscid hawkmoth probably also delivered pollen since it brushed against the high exserted floral sexual organs and picked up pollen (Online Resource 1). Both butterflies and short-proboscid hawkmoths were also recorded as floral visitors to a C. asiaticum population in Japan (Table 2). These short-proboscid lepidopterans were not expected to visit C. asiaticum according to the pollination syndrome concept, which hypothesises an association between flowers of a species with a functional group of floral visitors if the flowers display a specific floral syndrome (Fenster et al. 2004).
Although C. asiaticum largely conforms to the long-proboscid hawkmoth pollination syndrome, two characters are likely to lead to a relatively generalised pollination system. First, given that flowers of C. asiaticum remain open for several days and do not close at night, as we can see in some typical sphingophilous plants (e.g. Hirota et al. 2013, 2019), it is not surprising that they attract diurnal visitors, such as butterflies, by day. Lonicera japonica Thunb. provides another example, in which fragrant long-tubed flowers are visited by hawkmoths in the evening and do not close in the daytime and attract pollen-collecting bumblebees, leading to a bimodal pollination system (Miyake and Yahara 1998). Second, nectar can accumulate to such a high level in flowers of C. asiaticum that it can in part be accessed by short-proboscid lepidopterans (Fig. 3; see Manning and Goldblatt 2005 for a similar case). Thus, C. asiaticum may represent an example of a specialised floral syndrome with a relatively generalised pollination system (Ollerton et al. 2007).
Hawkmoths and swallowtail butterflies are common floral visitors in tropical and subtropical Asia (Miyake and Yahara 1998; Morinaga et al. 2009; Nakajima et al. 2018; Liu et al. 2019); therefore, it is likely that C. asiaticum is also pollinated by such insects at other sites in Asia (Table 2). We can further predict that it may benefit from similar pollinator niches where it is not native but has been naturalised because large hawkmoths and swallowtails also occur in other tropical zones (Johnson et al. 2004; Epps et al. 2015; Johnson et al. 2017). Moreover, C. asiaticum can become a very large plant and can persist for decades (personal observation), flowering frequently, so sexual reproduction may be assured in at least some years if pollinators show substantial annual fluctuations. Although floral specialisation does not seem to pose a barrier against naturalisation in new ranges, there are indeed examples that confirm the role of specialised floral syndromes in hindering naturalisation or invasion (Richardson 2000). Perhaps floral specialisation leads to poor or no sexual reproduction in new ranges only when no local floral visitors have pollinator niches similar to those in the native ranges of plants or plants presents floral rewards that are not sufficiently competitive in local communities.
Conclusion
Sexual reproduction provides a much greater potential for dispersal than vegetative reproduction in C. asiaticum var. sinicum. Despite poor self-compatibility and the absence of autonomous selfing, C. asiaticum var. sinicum, showing a highly specialised floral syndrome, could attract flower-visiting lepidopterans with a wide range of proboscis lengths that promoted the sexual reproduction and natural propagation of this plant in a suburban botanical garden. The naturalisation and long-term persistence of populations of C. asiaticum are likely attributed to pollination by various large local lepidopterans in many tropical and subtropical regions. Our results suggest that a highly specialised floral syndrome does not necessarily act as a barrier against the naturalisation and sexual reproduction of a plant species, even if there is no capacity for autonomous self-pollination or highly expansive vegetative reproduction (Figs. 1, 4).
References
Armbruster WS (2006) Evolutionary and ecological aspects of specialized pollination: Views from the Arctic to the Tropics. In: Waser NM, Ollerton J (eds) Plant–pollinator interactions: from specialization to generalization. University of Chicago Press, Chicago, pp 260–282
Balducci MG, Martins DJ, Johnson SD (2019a) Pollination of the long-spurred African terrestrial orchid Bonatea steudneri by long-tongued hawkmoths, notably Xanthopan morganii. Pl Syst Evol 305:765–775. https://doi.org/10.1007/s00606-019-01605-2
Balducci MG, Martins DJ, Johnson SD (2019b) Pollination of the long-spurred African terrestrial orchid Bonatea steudneri by long-proboscid hawkmoths, notably Xanthopan morganii. Pl Syst Evol 305:765–775. https://doi.org/10.1007/s00606-019-01605-2
Bjorå CS, Kwembeya EG, Nordal I (2006) Crinum jasoniia: a new endemic pan species of the Luangwa Valley in Zambia with notes on different seed structures in the genus. Kew Bull 61:569–577
Celep F, Atalay Z, Dikmen F, Doğan M, Classen-Bockhoff R (2014) Flies as pollinators of melittophilous Salvia species (Lamiaceae). Amer J Bot 101:2148–2159. https://doi.org/10.3732/ajb.1400422
Clark-Tapia R, Molina-Freaner F (2004) Reproductive ecology of the rare clonal cactus Stenocereus eruca in the Sonoran desert. Pl Syst Evol 247:155–164. https://doi.org/10.1007/s00606-003-0118-7
Dobson HEM (2006) Relationship between floral fragrance composition and type of pollinator. In: Dudareva N, Pichersky E (eds) Biology of floral scent. Taylor & Francis, Boca Raton, pp 147–198
Epps MJ, Allison SE, Wolfe LM (2015) Reproduction in Flame Azalea (Ericaceae): a rare case of insect wing pollination. Am Nat 186(2):294–301
Fenster CB, Armbruster WS, Wilson P, Dudash MR, Thomson JD (2004) Pollination syndromes and floral specialization. Annual Rev Ecol Syst 35:375–403. https://doi.org/10.1146/annurev.ecolsys.34.011802.132347
Gardner RO, Early JW (1996) The naturalization of Banyan figs (Ficus spp. Moraceae) and their pollinating wasps (Hymenoptera: Agaonidae) in New Zealand. New Zealand J Bot 34:103–110. https://doi.org/10.1080/0028825X.1996.10412697
Geerts S, Pauw A (2009) African sunbirds hover to pollinate an invasive hummingbird-pollinated plant. Oikos 118:573–579. https://doi.org/10.1111/j.1600-0706.2008.17167.x
Haber A, Frankie GW (1989) A tropical hawkmoth community: Costa Rican dry forest Sphingidae. Biotropica 21:155–172. https://doi.org/10.2307/2388706
Herrera I, Nassar JM (2009) Reproductive and recruitment traits as indicators of the invasive potential of Kalanchoe daigremontiana (Crassulaceae) and Stapelia gigantea (Apocynaceae) in a Neotropical arid zone. J Arid Environm 73:978–986. https://doi.org/10.1016/j.jaridenv.2009.05.004
Hirota SK, Nitta K, Suyama Y, Kawakubo N, Yasumoto AA, Yahara T (2013) Pollinator-mediated selection on flower color, flower scent and flower morphology of Hemerocallis: evidence from genotyping individual pollen grains on the stigma. PLoS ONE 8:e85601. https://doi.org/10.1371/journal.pone.0085601
Hirota SK, Miki N, Yasumoto AA, Yahara T (2019) UV bullseye contrast of Hemerocallis flowers attracts hawkmoths but not swallowtail butterflies. Ecol Evol 9:52–64. https://doi.org/10.1002/ece3.4604
Ji Z-H, Meerow A-W (2000) Amaryllidaceae Jaume Saint Hilaire. In: Wu ZY, Raven PH & Hong DY (Eds.), Flora of China, vol. 24. Science Press, Beijing & Missouri Botanical Garden Press, St. Louis
Johnson SD, Moré M, Amorim FW, Haber WA, Frankie GW, Stanley DA, Cocucci AA, Raguso RA (2017) The long and the short of it: a global analysis of hawkmoth pollination niches and interaction networks. Funct Ecol 31:101–115. https://doi.org/10.1111/1365-2435.12753
Johnson SD, Neal PR, Peter CI, Edwards TJ (2004) Fruiting failure and limited recruitment in remnant populations of the hawkmoth-pollinated tree Oxyanthus pyriformis subsp. pyriformis (Rubiaceae). Biol Conserv 120:31–39. https://doi.org/10.1016/j.biocon.2004.01.028
Johnson SD, Raguso RA (2016) The long-proboscid hawkmoth pollinator niche for native and invasive plants in Africa. Ann Bot (Oxford) 117:25–36. https://doi.org/10.1093/aob/mcv137
Kiepiel I, Johnson SD (2014) Shift from bird to butterfly pollination in Clivia (Amaryllidaceae). Am J Bot 101(1):190–200
van Kleunen M, Manning JC, Pasqualetto V, Johnson SD (2008) Phylogenetically independent associations between autonomous self-fertilization and plant invasiveness. Amer Naturalist 171:195–201. https://doi.org/10.1086/525057
Klimes L, Klimesová J, Hendriks R, van Groenendael J (1997) Clonal plant architecture: Clonal plant architecture: a comparative analysis of form and function. In: de Kroon H, van Groenendael J (eds) The Ecology and Evolution of Clonal Plants. Backhuys Publishers, Leiden, pp 1–30
Knuth P, Loew E (1904–1905) Handbuch der blütenbiologie, vol 3. Wilhem Engelmann, Leipzig
Liu J, Dong M, Miao S-L, Li ZY, Song MH, Wang RQ (2006) Invasive alien plants in China: role of clonality and geographical origin. Biol Invasions 8:1461–1470. https://doi.org/10.1007/s10530-005-5838-x
Liu C-Q, Gao Y-D, Niu Y, Xiong Y-Z, Sun H (2019) Floral adaptations of two lilies: implications for the evolution and pollination ecology of huge trumpet-shaped flowers. Amer J Bot 106:1–11. https://doi.org/10.1002/ajb2.1275
Liu H, Pemberton R (2010) Pollination of an invasive orchid, Cyrtopodium polyphyllum (Orchidaceae), by an invasive oil-collecting bee, Centris nitida, in southern Florida. Botany 88:290–295. https://doi.org/10.1139/B10-017
Manning JC, Goldblatt P (2005) Radiation of pollination systems in the Cape genus Tritoniopsis (Iridaceae: Crocoideae) and the development of bimodal pollination strategies. Int J Pl Sci 166:459–474. https://doi.org/10.1086/428703
Martins DJ, Johnson SD (2013) Interactions between hawkmoths and flowering plants in East Africa: polyphagy and evolutionary specialization in an ecological context. Biol J Linn Soc 110:199–213. https://doi.org/10.1111/bij.12107
Matyot P (2005) The hawkmoths (Lepidoptera: Sphingidae) of Seychelles: identification, historical background, distribution, food plants and ecological considerations. Phelsuma 13:55–80
Memmott J, Waser NM, Price MV (2004) Tolerance of pollination networks to species extinctions. Proc Roy Soc London B 271:2605–2611. https://doi.org/10.1098/rspb.2004.2909
Miller WE (1997) Diversity and evolution of tongue length in hawkmoths (Sphingidae). J Lepid Soc 51:9–31
Miyake T, Yamaoka R, Yahara T (1998) Roral scents of hawkmoth-pollinated flowers in Japan. J Pl Res 111:199–205. https://doi.org/10.1007/BF02512170
Miyake T, Yahara T (1998) Why does the flower of Lonicera japonica open at dusk? Canad J Bot 76:1806–1811. https://doi.org/10.1139/b98-119
Morinaga SI, Kumano Y, Ota A, Yamaoka R, Sakai S (2009) Day-night fluctuations in floral scent and their effects on reproductive success in Lilium auratum. Populat Ecol 51:187–195. https://doi.org/10.1007/s10144-008-0097-1
Nakajima R, Okamiya H, Shimokawa S, Yamamoto K, Kato H, Murakami N (2018) Comparison of floral traits and pollinator assemblages of insular and mainland varieties of Lilium auratum. Pl Spec Biol 33:276–288. https://doi.org/10.1111/1442-1984.12222
Nienhuis C, Stout JC (2009) Effectiveness of native bumblebees as pollinators of the alien invasive plant Impatiens glandulifera. J Pollinat Ecol 1:1–11. https://doi.org/10.26786/1920-7603(2009)1
Ollerton J, Watts S, Connerty S, Lock J, Parker L, Wilson I, Schueller SK, Nattero J, Cocucci AA, Izhaki I, Geerts S, Pauw A, Stout JC (2012) Pollination ecology of the invasive tree tobacco Nicotiana glauca: comparisons across native and non-native ranges. J Pollinat Ecol 9:85–95
Ollerton J, Killick A, Lamborn E, Watts S, Whiston M (2007) Multiple meanings and modes: on the many ways to be a generalist flower. Taxon 56:717–728. https://doi.org/10.2307/25065855
Paudel BR, Shrestha M, Burd M, Adhikari S, Sun Y-S, Li Q-J (2016) Coevolutionary elaboration of pollination-related traits in an alpine ginger (Roscoea purpurea) and a tabanid fly in the Nepalese Himalayas. New Phytol 211:1402–1411. https://doi.org/10.1111/nph.13974
Pittaway AR, Kitching IJ (2020) Sphingidae of the Eastern Palaearctic (including Siberia, the Russian Far East, Mongolia, China, Taiwan, the Korean Peninsula and Japan), The Natural History Museum, London. Available at: http://tpittaway.tripod.com/china/china.htm
Reiter N, Vlcek K, O’Brien N, Gibson M, Pitts D, Brown GR, Bower CC, Phillips RD (2017) Pollinator rarity limits reintroduction sites in an endangered sexually deceptive orchid (Caladenia hastata): implications for plants with specialized pollination systems. Biol J Linn Soc 184:122–136. https://doi.org/10.1093/botlinnean/box017
Richardson DM (2000) Plant invasions—the role of mutualisms. Biol Rev 75:65–93
Rodger JG, van Kleunen M, Johnson SD (2010) Does specialized pollination impede plant invasions? Int J Pl Sci 171:382–391. https://doi.org/10.1086/651226
Skogen KA, Jogesh T, Hilpman ET, Todd SL, Rhodes MK, Still SM, Fant JB (2016) Land-use change has no detectable effect on reproduction of a disturbance-adapted, hawkmoth-pollinated plant species. Amer J Bot 103:1950–1963. https://doi.org/10.3732/ajb.1600302
Van der Niet T, Johson SD (2012) Phylogenetic evidence for pollinator-driven diversification of angiosperms. Trends Ecol Evol 27:353–361. https://doi.org/10.1016/j.tree.2012.02.002
Walsha SK, Pender RJ, Junker RR, Daehlerb CC, Morden CW, Lorence DH (2019) Pollination biology reveals challenges to restoring populations of Brighamia insignis (Campanulaceae), a critically endangered plant species from Hawai‘i. Flora 259:151448. https://doi.org/10.1016/j.flora.2019.151448
Waser NM, Chittka L, Price MV, Williams NM, Ollerton J (1996) Generalization in pollination systems, and why it matters. Ecology 77:1043–1060. https://doi.org/10.2307/2265575
Waser NM, Ollerton J, Erhardt A (2011) Typology in pollination biology: lessons from an historical critique. J Pollinat Ecol 3:1–7. https://doi.org/10.26786/1920-7603(2011)2
WCSP (2016) World checklist of selected plant families, The Royal Botanic Gardens, Kew. Available at: http://apps.kew.org/wcsp/
Acknowledgements
We thank Ji-Bai He and Zhuo-Heng Jiang for the identification of lepidopterans and Zhuo-Heng Jiang for providing the photo of a Papilio helenus specimen used in Fig. 5. We thank Ying-Ze Xiong and Ze-Yu Tong for advice on statistical analysis. This study was supported by National Natural Science Foundation of China (Grant No. 31971563 to C.-Q. Liu and 31560068 to X.-H. Hu), the Science and Technology Basic Resources Investigation Program of China (Grant No. 2017FY100100) and Elevation of Talent Program of Guangxi Karst Ecological Construction and Sustainable Utilization of Plant Resources in 2018.
Author information
Authors and Affiliations
Contributions
Chang-Qiu Liu devised this study, revised the draft critically and took the photographs of the insects visiting flowers. The remaining authors were responsible for observing floral visitors and measuring nectar and flower and insect morphologies. Yang Huang and Lan-Ying Liu conducted the pollination treatments. Huang Yang wrote the first draft of the manuscript. Qing-Biao Lu helped Yang Huang conduct statistical analysis. Bo Cai helped Yang Huang prepare the figures. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights statement
The study did not involve research involving human participants and/or animals.
Ethical Statement
In the study “Diverse large Lepidoptera pollinators promote the naturalisation of Crinum asiaticum under cultivation despite apparent floral specialisation”, we observed the following rules: The manuscript should not be submitted to more than one journal for simultaneous consideration. The submitted work should be original and should not have been published elsewhere in any form or language (partially or in full), unless the new work concerns an expansion of previous work. (Please provide transparency on the re-use of material to avoid the concerns about text-recycling (‘self-plagiarism’). A single study should not be split up into several parts to increase the quantity of submissions and submitted to various journals or to one journal over time (i.e. ‘salami-slicing/publishing’). Concurrent or secondary publication is sometimes justifiable, provided certain conditions are met. Examples include: translations or a manuscript that is intended for a different group of readers. Results should be presented clearly, honestly, and without fabrication, falsification or inappropriate data manipulation (including image based manipulation). Authors should adhere to discipline-specific rules for acquiring, selecting and processing data. No data, text, or theories by others are presented as if they were the author’s own (‘plagiarism’). Proper acknowledgements to other works must be given (this includes material that is closely copied (near verbatim), summarized and/or paraphrased), quotation marks (to indicate words taken from another source) are used for verbatim copying of material, and permissions secured for material that is copyrighted.
Additional information
Handling editor: Ferhat Celep.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Information on Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Online Resource 1
. The three hawkmoth visitors not included in Fig. 2. a. Agrius convolvuli. b. Acosmeryx pseudomissa. c. Psilogramma increta. See the yellow pollen attached to these moths.
Information on Electronic Supplementary Material
Information on Electronic Supplementary Material
Online Resource 1. The three hawkmoth visitors not included in Fig. 2. a. Agrius convolvuli. b. Acosmeryx pseudomissa. c. Psilogramma increta. See the yellow pollen attached to these moths.
Rights and permissions
About this article
Cite this article
Huang, Y., Liu, LY., Liu, CQ. et al. Diverse large lepidopteran pollinators promote the naturalisation of Crinum asiaticum in invaded and disturbed habitats, despite apparent floral specialisation. Plant Syst Evol 307, 23 (2021). https://doi.org/10.1007/s00606-021-01748-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00606-021-01748-1