Abstract
The present study was conducted to analyze the phylogenetic status of the genus Onobrychis and to evaluate the monophyly of its subgenera and sections and relationship among them. We sequenced the nuclear ribosomal DNA internal transcribed spacer (nrDNA ITS) and three chloroplast regions trnL-F, rpl32/rpl32-trnL(UAG) and ndhF-rlp32 for phylogenetic reconstruction of 51 species of Onobrychis. In all of our analyses, Eversmannia subspinosa, Corethrodendron scoparium, Greuteria membranacea and G. argyrea were chosen as outgroups. Phylogenetic analyses were performed by maximum parsimony, maximum likelihood and Bayesian methods. Our molecular data indicate that Onobrychis is monophyletic and composed of two main clades, each corresponding to the redefined subgenus Onobrychis (including sections Onobrychis and Hemicyclobrychis) and subgenus Sisyrosema (including sections Afghanicae, Laxiflorae, Heliobrychis, Hymenobrychis, Insignes, Lipskyanae and Litvinovianae), respectively. Sections Lipskyanae and Litvinovianae are newly established and described, representing distinct lineages within the genus. Onobrychis splendida, a species hitherto without a sectional position, along with some members of sect. Anthyllium were retrieved representatives of section Lipskyanae. Sections Afghanicae, Insignes, Heliobrychis and Hymenobrychis (with the inclusion of two species of section Anthyllium) are monophyletic. Sections Dendrobrychis and Lophobrychis are reduced to synonymy of section Onobrychis and Anthyllium to synonymy of section Hymenobrychis. A taxonomic treatment for the genus is presented.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Onobrychis Mill. with more than 130 species is ranked as the second largest genus of the tribe Hedysareae after Hedysarum (Mabberley 2008; Lock 2005; Amirahmadi et al. 2014a). The genus is distributed throughout temperate and subtropical regions of Eurasia, N and NE Africa (Townsend 1974). Most species of Onobrychis are distributed in the Flora Iranica area (77 species), the former USSR (62 species), Turkey (46 species) and Europe (23 species), respectively (Hedge 1970; Grossheim 1972; Ball 1978; Rechinger 1984). Recently, several new species of Onobrychis were described from Iran (e.g., Ranjbar et al. 2004, 2009; Amirabadizadeh et al. 2007, 2009; Amirahmadi et al. 2014b).
Širjaev (1925) based on floral characteristics divided the genus into two subgenera, Onobrychis (as Euonobrychis (Bunge ex Boiss.) Širj.) and Sisyrosema (Bunge ex Boiss.) Širj. each with four sections. Rechinger (1984) reclassified the two subgenera into four and five sections, namely Onobrychis, Dendrobrychis DC., Lophobrychis Hand.-Mazz., Laxiflorae (Širj.) Rech.f., Anthyllium Nábĕlek, Afghanicae Širj., Heliobrychis Bunge ex Boiss., Hymenobrychis DC. and Insignes (Širj.) Rech.f., respectively. However, the sectional positions of O. splendida Rech.f. & Podlech and O. freitagii Rech.f. (this species was not included in the present study) have remained hitherto uncertain. The infra-generic classification systems of Onobrychis are summarized in Table 1.
Several biosystematics studies ranging from non-molecular data to DNA sequences have been carried out on Onobrychis and allies (e.g., Yildiz et al. 1999; Pavlova and Manova 2000; Abou-El-Enain 2002; Ahangarian et al. 2007; Irfan et al. 2007; Hesamzadeh Hejazi and Ziaei Nasab 2010; Ranjbar et al. 2010, 2012; Arslan et al. 2012; Hayot Carbonero et al. 2012; Karamian et al. 2012; Avci et al. 2013, Lewke Bandara et al. 2013; Amirahmadi et al. 2014a; Safaei Chaei Kar et al. 2012, 2014; Duan et al. 2015; Zarrabian and Majidi 2015). But still, detailed phylogenetic analysis using multiple DNA sequence data and adequate taxon sampling of all recognized sections on the genus is lacking.
In this study, the nuclear ribosomal DNA internal transcribed spacer (nrDNA ITS) and three chloroplast regions, trnL(UAA) intron and trnL(UAA)-trnF(GAA) intergenic spacer (hereafter trnL-F), rpl32 gene and rpl32-trnL(UAG) intergenic spacer (hereafter rpl32/rpl32-trnL(UAG)) and ndhF-rlp32 intergenic spacer, were sequenced for phylogenetic reconstructions. The internal transcribed spacer (ITS) contains the signals needed to process the rRNA transcript (Baldwin et al. 1995) and has often been used for inferring phylogeny at intra- and intergeneric levels (e.g., Wojciechowski et al. 1999; Kazempour Osaloo et al. 2005; Amirahmadi et al. 2014a). The trnL-F and both ndhF-rpl32 and rpl32/rpl32-trnL(UAG) are located in the large single copy and the small single copy regions of the chloroplast genome, respectively. The latest is the best non-coding region for low-level molecular studies (Shaw et al. 2007; Dong et al. 2012).
Our objectives in the present study are to use molecular sequence data, to generate hypotheses on the phylogenetic status of Onobrychis and relationships among its analyzed taxa herein (subgenera, sections and O. splendida) so to evaluate past classifications of the genus, to identify morphological features that characterize the main clades detected by the molecular analyses and to present a new phylogenetic classification of the genus.
Materials and methods
Taxon sampling
A total of 199 DNA sequences were generated from specimens deposited at the following herbaria: FUMH, GAZI, MSB, TARI, TUH and Tarbiat Modares Univ. Herb. or in several cases (especially species distributed in Iran) from silica-dried leaves. Two species of Greuteria (G. membranacea (Coss. & Bal.) Amirahm. & Kaz.Osaloo and G. argyrea (Greuter & Burdet) Amirahm.& Kaz.Osaloo), Corethrodendron scoparium (Fisch & Meyer) Fisch & Basiner and Eversmannia subspinosa (Fisch) B.Fedtsch. were selected as outgroups following our previous study (Amirahmadi et al. 2014a). The sampling strategy was to include representatives of all nine recognized sections of Onobrychis (sensu Rechinger 1984) including sects. Heliobrychis (9 species), Hymenobrychis (8 species), Onobrychis (9 species), Lophobrychis (5 species), Dendrobrychis (5 species), Anthyllium (5 species), Afghanicae (4 species), Insignes (3 species) and Laxiflorae (one species with two accessions), plus O. splendida (a species of unassigned section) which are listed in Table 2.
DNA extraction, amplification, and sequencing
Total genomic DNA was isolated from fresh or dried leaf material using modified CTAB method of Doyle and Doyle (1987). The nrDNA ITS region was amplified using the primers ITS5 m (Sang et al. 1995) and ITS4 (White et al. 1990) or AB101F and AB102R (Douzery et al. 1999). The trnL-F region was amplified using the universal “c” and “f” primers (Taberlet et al. 1991). The rpl32/rpl32–trnL(UAG) and ndhF-rpl32 regions were amplified using two primer pairs: rpl32-F/trnL(UAG) (Shaw et al. 2007) and ndhFm/rpl32RR (both were designed in this study: 5′-AAGATTGATGTGTATATTC-3′ and 5′-TACGTTTTTTTGGAACTG-3′), respectively. The PCR amplification was carried out in the volume of 20 μl, containing 8 μl deionized water, 10 μl of the 2× Taq DNA polymerase master mix Red (Amplicon, Cat. No. 180301, 150 mM Tris–HCl pH = 8.5, 40 mM (NH4)2SO4, 3.0 mM MgCl2, 0.4 mM dNTPs, 0.05 units μl−1 Amplicon Taq DNA polymerase, inert red dye and a stabilizer) 0.5 μl of each primer (10 pmol/μl), and 1 μl of template DNA (20 ng/μl). PCR cycles consisted of pre-denaturation at 94 °C for 3 min followed by 28–35 cycles: denaturation at 94 °C for 1 min, annealing at a temperature depending on the region (55 °C for nrDNA and 58 °C for cpDNA) for 1 min and elongation at 72 °C for 1 min. A final elongation step of 7 min at 72 °C was performed. The quality of the PCR products were checked by electrophoresis on a 1 % (w/v) agarose gel (using 1× TBE as the gel buffer) stained with ethidium bromide and then was photographed with a UV gel documentation system (UVItec, Cambridge, UK). Each region was sequenced using the Big dye terminator cycle sequencing ready reaction kit (Applied Biosystems, USA) with the appropriate primers in an ABI Prism 3730XL DNA sequencer (Applied Biosystems, USA).
Alignment and phylogenetic analyses
Each of the single dataset was aligned using the web-based version of MUSCLE (Edgar 2004; at http://www.ebi.ac.uk/Tools/msa/muscle/) under default parameters followed by manual adjustment. Sequences of all datasets showed length variation (because of noncoding region), and it was necessary to introduce insertions/deletions in the alignment. Positions of indels were treated as missing data for all datasets. Phylogenetic analyses were performed based on the maximum parsimony (MP) and maximum likelihood (ML) methods as well as Bayesian inference (BI). Parsimony analyses were conducted using PAUP* version 4.0b10 (Swofford 2002). The heuristic search option was employed for each dataset, using tree bisection-reconnection (TBR) branch swapping, with 100 replications of random addition sequence and an automatic increase in the maximum number of trees. Branch support values were calculated using a full heuristic search with 1000 bootstrap replicates (Felsenstein 1985) each with simple addition sequence. In Bayesian method, models of sequence evolution were selected using the program MrModeltest version 2.3 (Nylander 2004) based on the Akaike information criterion (AIC) (Posada and Buckley 2004). This program indicated GTR + G for nrDNA ITS and plastid sequence datasets (trnL–F, rpl32/rpl32-trnL(UAG) and ndhF-rpl32) and GTR + G+I for the combined dataset, as the best model for nucleotide substitution. The program MrBayes version 3.2.4 (Ronquist et al. 2012) was used for the Bayesian phylogenetic analyses. Posteriors on the model parameters were estimated from the data, using the default priors. The analysis was carried out with 6 million generations, using the Markov chain Monte Carlo (MCMC) search. MrBayes performed two simultaneous analyses starting from different random trees (Nruns = 2) each with four Markov chains and trees sampled at every 100 generations. The first 25 % trees were discarded as the burn-in. The remaining trees were then used to build a 50 % majority rule consensus tree accompanied with posterior probability (PP) values. Tree visualization was carried out using TreeView version 1.6.6 (Page 2001). Also ML analyses were performed for the datasets in the program GARLI (Zwickl 2006) and raxmlGUI (Silvestro and Michalak 2012). The model of evolution employed for each dataset is the same as that of Bayesian analyses. Parametric bootstrap values for ML were calculated in GARLI and raxmlGUI based on 1000 replicates with one search replicate per bootstrap replicate.
Overall mean p-distance for each dataset was computed using MEGA6 (Tamura et al. 2013).
ILD test
Combinability of these datasets was assessed by use of the partition homogeneity test [the incongruence length difference test (ILD) of Farris et al. (1995)] as implemented in PAUP* (Swofford 2002). The test was conducted with the exclusion of invariant characters (Cunningham 1997) using the heuristic search option involving simple addition sequence and TBR branch swapping with 1000 homogeneity replicates.
Results
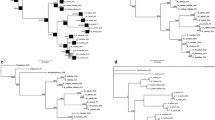
Detailed information about alignment characteristics and statistics from the analyses is presented in Table 3. The Parsimony, Likelihood and Bayesian analyses of nrDNA ITS and plastid datasets, trnL–F, rpl32/rpl32-trnL(UAG) and ndhF-rpl32 (as Online resources 1, 2, 3, 4) as well as the combined nuclear-plastid dataset (Fig. 1), produced congruent trees without any major difference. The ILD test did not reveal significant incongruence (p < 0.07) between the individual datasets. Therefore, only the results of the Bayesian inference of the combined dataset are discussed below (Fig. 1). The monophyly of Onobrychis was well supported (PP = 1.0, ML/BS = 100/96), and two main lineages could be identified within the genus, clades “A” and “B” (Fig. 1). The clade “A” is composed of members of sects. Onobrychis, Lophobrychis and some members of sect. Dendrobrychis; the clade “B” comprised members of sects. Anthyllium, Insignes, Laxiflorae, Afghanicae, Heliobrychis, Hymenobrychis and some members of sect. Dendrobrychis plus O. splendida. Sections Dendrobrychis, Lophobrychis, Onobrychis, Anthyllium, Laxiflorae and Hymenobrychis in all analyses are non-monophyletic. Conversely, sections Afghanicae, Insignes (except in nrDNA ITS) and Heliobrychis (except in rpl32/rpl32-trnL(UAG), ndhF-rpl32) are monophyletic. The aligned data matrix used in this study is presented as Online Resource 5.
Fifty percent majority rule consensus tree resulting from Bayesian inference of the combined nrDNA ITS-plastid (trnL-F, rpl32/rpl32-trnLUAG and ndhF-rpl32) dataset. Numbers above branches are posterior probability and likelihood as well as parsimony bootstrap values, respectively. Values <50 % were not shown. Subgen. Onobrychis and subgen. Sisyrosema, corresponding to the clades “A” and “B”, respectively, were redefined at the present study. Characters distinguishing the clades “A” and “B” were mapped as boxed above them. The sectional classification for the genus Onobrychis based on Rechinger (1984) and the present study was given at the right side of the tree
Discussion
Phylogenetic status and composition of Onobrychis
Recent phylogenetic studies (Amirahmadi et al. 2014a; Safaei Chaei Kar et al. 2012, 2014; Duan et al. 2015; Zarrabian and Majidi 2015) retrieved all Onobrychis species in a well-supported monophyletic group with Eversmannia subspinosa and allied genera (Corethrodendron, Greuteria; Amirahmadi et al. 2014a) showing close relationship. Nonetheless, based on only nrDNA ITS data, few studies (Hayot Carbonero et al. 2012; Lewke Bandara et al. 2013) suggested that Onobrychis, due to the inclusion of Eversmannia within it, is not monophyletic. The present results confirm, however, the monophyly of Onobrychis as a well-supported clade (PP = 1.00, ML/BS = 100/96). Our analyses are in agreement with recent findings (Carbonero et al. 2012; Lewke Bandara et al. 2013; Amirahmadi et al. 2014a; Safaei Chaei Kar et al. 2014; Duan et al. 2015) which reveal that Onobrychis is divided into two strongly supported clades. As shown in Fig. 1, the clade “A” includes the great majority of subgen. Onobrychis sensu Rechinger (1984), comprising sects. Lophobrychis and Onobrychis as well as some members of sect. Dendrobrychis (O. cornuta (L.) Desv. and O. elymaitica Boiss. & Hausskn. ex Boiss.). The clade “B” includes the remaining members of subgen. Onobrychis comprising other members of sect. Dendrobrychis (O. arnacantha Bunge ex Boiss., O. afghanica Širj. & Rech f. and O. echidna Lipsky) and sect. Laxiflorae along with all members of subgen. Sisyrosema sensu Rechinger (1984) comprising sects. Anthyllium, Afghanicae, Heliobrychis, Hymenobrychis and Insignes as well as O. splendida.
New subgeneric concept in Onobrychis
As noted above, our tree topologies (Fig. 1, Online Resources 1, 2, 3, 4) indicate that some members of subgen. Onobrychis are nested within subgen. Sisyrosema, and thus they are not monophyletic at the current status. This conclusion was already reached by several studies (Yildiz et al. 1999; Hayot Carbonero et al. 2012; Amirahmadi et al. 2014a; Safaei Chaei Kar et al. 2014; Duan et al. 2015; Zarrabian and Majidi 2015). Molecular results presented by us and recent authors (Hayot Carbonero et al. 2012; Lewke Bandara et al. 2013; Amirahmadi et al. 2014a; Safaei Chaei Kar et al. 2014) revealed inconsistencies with the two traditionally recognized subgenera (Širjaev 1925, 1926; Rechinger 1984) based on morphological characters. For example, sect. Laxiflorae with a semi-curved suture pod was treated by Širjaev (1926) as subgen. Sisyrosema sect. Hymenobrychis subsect. Laxiflorae, while Rechinger (1984) transferred this subsection to subgen. Onobrychis and treated it as sect. Laxiflorae without any clear explanation. According to this study, the clade “A” includes all members of subgen. Onobrychis (sensu Rechinger 1984) with the exception of members of sect. Laxiflorae and some members of sect. Dendrobrychis (O. arnacantha, O. afghanica and O. echidna). We concluded that the clade “A” based on our phylogenetic hypothesis is presented as subgen. Onobrychis in emend us and recognized by the pod with straight ventral suture, the deciduous corolla, the presence of crystal in the calyx epidermal cells and relatively small flowers and pods. The clade “B” includes all members of subgen. Sisyrosema (sensu Rechinger 1984) along with sect. Laxiflorae, other members of sect. Dendrobrychis (O. arnacantha, O.afghanica and O.echidna) and O. splendida. The synapomorphies for members of the clade “B” are the pod with semi-curved to curved, rarely straight ventral suture, the persistent corolla, the absence of crystal in the calyx epidermal and relatively large flowers and pods. The clade “B” is presented here as subgen. Sisyrosema in emend us (Table 1 and “Taxonomic treatment”).
Monophyly and relationships of sections
Members of sects. Onobrychis, Lophobrychis and some members of sect. Dendrobrychis (O. elymaitica and O. cornuta) analyzed herein are intermixed with each other (Fig. 1; PP = 1.0 ML/BS = 100/100). They are gathered in two subclades of the clade “A” (subgen. Onobrychis). In the first subclade, members of sect. Onobrychis from O. viciifolia Scop. (type species of sect. Onobrychis) through O. supine (Vill.) DC. form a monophyletic group. In addition, O. aequidentata d’Urv. and O.caput-galli (L.) Lam. (type species of sect. Lophobrychis) are successive sisters to this group. In the second subclade, O. elymaitica and O. cornuta (type species of Dendrobrychis) are sister taxa for which O. verae Širj. and O. sosnovskyi Grossh. from sect. Onobrychis, and O. crista-galli (L.) Lam. from sect. Lophobrychis are successive grades, respectively. Finally, this newly formed group is sister to a branch including O. micrantha Schrenk and O. pulchella Schrenk from sect. Lophobrychis. These findings are congruent with previous studies (Hayot Carbonero et al. 2012; Lewke Bandara et al. 2013; Safaei Chaei Kar et al. 2014) which showed that sects. Onobrychis, Lophobrychis and Dendrobrychis are closely related, but none are monophyletic. Limited numbers of characters have been used for delimitation of the sections in the literature. Section Onobrychis (sensu Širjaev 1925; Rechinger 1984) comprises perennial herbs, rarely woody at the base, with many flowered, long or short wings and 1-seeded pod as opposed to annual herbs, with few flowered, long wings and 1 or 2-seeded pods. However, there are some morphological similarities between sects. Onobrychis and Lophobrychis, particularly in shape of pods and the teeth of the crest and having long-wing petals. It seems that annual or perennial habit at least in the studied genus should not be diagnostic features to separate species as two different sections, namely Lophobrychis and Onobrychis. Consequently, based on the present molecular study, sect. Lophobrychis is considered as a synonym of sect. Onobrychis (see “Taxonomic treatment”). Section Dendrobrychis only includes the spiny cushion-forming species. Širjaev (1925) subdivided the section into the two series, Dielsianae Širj. and Litvinovianae Širj., and distinguished them with the spiny peduncle and petiole, respectively. Phylogenetic analysis showed that O. cornuta and O. elymaitica from series Dielsianae (with the spiny peduncle, semi-orbicular legume, and crystal in the calyx epidermal cells) are nested within the clade “A”. Onobrychis afghanica, O. arnacantha and O. echidna from series Litvinovianae are nested within the clade “B” and comprise a distinct lineage in Onobrychis. On the basis of our phylogenetic results and morphological similarities (e.g., having long-wing petals and pod shape), sect. Dendrobrychis is also treated as a synonym of sect. Onobrychis (see “Taxonomic treatment”).
It should be noted that although O. hemicycla C.I. Blanche ex Boiss., a sole member of sect. Hemicyclobrychis, restricted to Syria, was not analyzed here, we followed Širjaev’s treatment (1925) to retain the species in its own section as a member of subgen. Onobrychis (see Table 1 and “Taxonomic treatment”).
Sections Anthyllium, Insignes, Laxiflorae, Afghanicae, Heliobrychis and Hymenobrychis along with the rest of sect. Dendrobrychis and O. splendid are members of the clade “B” (subgen. Sisyrosema). The present phylogenetic study revealed that sect. Anthyllium with 8 species, represented herein by 5 species, is not monophyletic, indicating that the delimitation of Anthyllium on the basis of morphological features is artificial and the diagnostic features of it overlap with other sections/species. Its representatives were placed in three distinct subclades. One of which, O. merxmuelleri Podlech was well united with O. splendida (a species of unassigned section) and, in turn, both formed the basal lineage in the clade “B”. This relationship has never been mentioned in earlier studies (e.g., Rechinger 1984). Podlech (1967) established O. merxmuelleri as a new species and designated it as the closest relative of O. grandis Lipsky of Anthyllium subsect. Lipskyanae and placed it in this subsection. This relationship was also retrieved in cpDNA tree of Duan et al. (2015). These two species and O. splendida do share several morphological characteristics such as taller perennial habit, leaves with remote leaflets, lax inflorescence, legume with straight ventral suture and 1-seeded (Rechinger 1984). Hence, based on the molecular data, we excluded the two species from sect. Anthyllium and along with O. splendida placed them in the newly erected section Lipskyanae (see “Taxonomic treatment”). Two other species of sect. Anthyllium, O. dealbata Stocks and O. schugnanica B.Fedtsch. with the inclusion of two taxa of O. laxiflora Baker of sect. Laxiflorae (typified by O. laxiflora) formed a monophyletic group. The placement of O. dealbata with O. laxiflora corroborates Lewke Bandara et al. (2013), although they analyzed O. dasycephala Baker (a synonym of O. dealbata) and did not mention such close species relationship. Hence, these two species are considered to be members of sect. Laxiflorae. Two another species of sect. Anthyllium (O. acaulis Bornm. and O. Susiana Nábĕlek) were nested within sect. Hymenobrychis and united with O. ptolemaica (Del.) DC. This is consistent with findings of previous studies (Ahangarian et al. 2007; Hayot Carbonero et al. 2012; Lewke Bandara et al. 2013; Safaei Chaei Kar et al. 2014), which found O. acualis within sect. Hymenobrychis. The characteristics of the two species are well suited with that of members (in particular O. ptolemaica) of sect. Hymenobrychis. Due to nomenclatural priority, we merged sect. Anthyllium, typified here by O. susiana, in the sect. Hymenobrychis, (see “Taxonomic treatment”). Onobrychis arnacantha, O. afghanica and O. echidna, which are here recognized as members of new section Litvinovianae (type species: O. arnacantha), form the second lineage within Clade “B”. They are distinct in having cushion-forming habit, spiny petiole, semi-lunar pod and without crystal in the calyx epidermal cells. The placement of O. arnacantha in subgen. Sisyrosema (Clade “B”) is congruent with the analyses of Hayot Carbonero et al. (2012), Lewke Bandara et al. (2013) and Safaei Chaei Kar et al. (2014). Section Insignes, represented with three species, is monophyletic and along with Anthyllium + Laxiflorae formed the third diverging subclade. Both sections Afghanicae and Heliobrychis comprise independent lineages and are monophyletic. The monophyly of sect. Heliobrychis was supported by Ahangarian et al. (2007), Hayot Carbonero et al. (2012) and Lewke Bandara et al. (2013), whereas the monophyly of both sections was questioned by nrDNA ITS data of Safaei Chaei Kar et al. (2014). This discrepancy might be because of PCR contamination of O. iranenesis Amirab. & Ghanavati with O. aucheri Boiss. in the study of Safaei Chaei Kar et al. It merits to note that in this and previous works, O. teheranica Bornm. (= O. aucheri ssp. teheranica (Bornm.) Rech. f.), an annual species, is sister to the remaining of Heliobrychis. This indicates that annual habit in the section might be an ancestral state. Analyses of molecular data showed that sect. Hymenobrychis with the inclusion of two members of Anthyllium (O. acualis and O. susiana) formed a distinct lineage and, in turn, well united with sect. Heliobrychis.
Conclusions
The results of the present analysis based on multiple DNA regions provide more convincing evidence as to the phylogenetic relationships among Onobrychis taxa examined. The present molecular study illustrated that the current infrageneric classification of the genus is no longer tenable. A new taxonomic classification of the genus at both subgeneric and sectional levels along with the typification is hereby presented. An inclusive phylogenetic study dealing with species rich sections of Onobrychis such as Onobrychis (sensu us), Heliobrychis and Hymenobrychis using several DNA markers and comprehensive taxon sampling is especially needed to clarify their evolutionary history as we have already progressed in this regard.
Taxonomic treatment
Onobrychis Mill., Gard. Dict. Abr. Ed. 4. 1754
= Onobruchus Medik., Vorles. Churpfälz. Phys.-Öcon. Ges. 2:372. 1787
= Eriocarpaea Bertol., Nov. Comm. Acad. Bonon. 6:234. 1843.—TYPE (designated by Hanelt 2001): Onobrychis viciifolia Scop.
Subg. Onobrychis emend Amirahm. & Kaz. Osaloo ≡ Sect. Euonobrychis Bunge ex Boiss., Fl. Or. 2:526. 1872 ≡ Subgen. Euonobrychis (Bunge ex Boiss.) Širj., Spisy Přír. Fak. Masarykovy Univ. 56:18. 1925.—TYPE: Onobrychis viciifolia Scop.
Sect. Onobrychis emend Amirahm. & Kaz. Osaloo ≡ Sect. Eubrychis DC. Prodr. 2: 344. 1825.—TYPE: Onobrychis viciifolia Scop.
= Sect. Dendrobrychis DC., Prodr. 2:347. 1825. syn. nov.—TYPE (designated here): O. cornuta (L.) Desv.
= Sect. Dendrobrychis DC. ser. Dielsianae Širj., Spisy Přír. Fak. Masarykovy Univ. 56:22. 1925.—TYPE (designated here): O. cornuta (L.) Desv.
= Sect. Lophobrychis Hand.-Mazz., Osterr. Bot. Zeitschr. 59:373. 1909. syn. nov.—TYPE (designated here): O. caput-galli (L.) Lam.
= Sect. Lophobrychis Hand.-Mazz. subsect. Occidentales Širj., Spisy Přír. Fak. Masarykovy Univ. 56:34. 1925. syn. nov.—TYPE (designated here): O. caput-galli (L.) Lam.
= Sect. Lophobrychis Hand.-Mazz. subsect. Orientales Širj., Spisy Přír. Fak. Masarykovy Univ. 56:51. 1925. syn. nov.—TYPE (designated here): O. micrantha Schrenk.
Note The original publication is sometimes cited as Flore Française 4: 511. 1805, but this page deals with Ononis L. Onobrychis can be found on page 611, but Dendrobrychis is not mentioned here (Lamarck and De Candolle 1805).
Description: Annual or perennial herbs, sometimes cushion-forming shrublets. Peduncle rarely spiny. Flowers relatively small. Epidermal cells of calyx with crystal. Corolla deciduous, glabrous, wings longer or shorter than keel. Legume relatively small, semi-orbicular, with 1, 2 seeds, sessile or rarely stipitate, with straight ventral (seminiferous) suture, with or without crest, disk and crest smooth, spiny or dentate.
Sect. Hemicyclobrychis Širj., Spisy Přír. Fak. Masarykovy Univ. 56:56. 1925.—TYPE (designated here): O. hemicycla C.I.Blanche ex Boiss.
Description: Perennial herbs. Wings shorter than keel. Legume semi-orbicular, 1-seeded, sessile, with straight ventral suture, crest broad, dentate.
Subg. Sisyrosema (Bunge ex Boiss.) Širj., Spisy Přír. Fak. Masarykovy Univ. 76:5. 1926. emend. Amirahm. & Kaz. Osaloo ≡ Sect. Sisyrosema Bunge ex Boiss., F1. Orient. 2:526. 1872.—TYPE (designated here): O. radiata (Desf.) M.Bieb.
Description: Perennial, rarely annual herbs, sometimes cushion-forming shrublets. Flowers relatively large. Epidermal cells of calyx without crystal. Corolla persistent, more or less pubescent. Legume relatively large, with semi-curved to fully curved, rarely straight ventral (seminiferous) suture, with or without crest, disk and crest spineless or spiny or dentate.
Sect. Afghanicae Širj., Spisy Přír. Fak. Masarykovy Univ. 76:18. 1926.—TYPE (designated here): O. tavernieraefolia Stocks ex Boiss.
Description: Annual herbs. Leaves with 1-2 pairs of leaflets. Legume 2 (-1)-locular, 2(-1)-seeded, coiled inwards from the tip, flattened out, bristled or sometimes cottony-woven together at margins; disk with distinct pits, rarely spiny.
Sect. Laxiflorae (Širj.) Rech.f., Fl. Iranica 157:415. 1984. emend. Amirahm. & Kaz. Osaloo ≡ Sect. Hymenobrychis DC. subsect. Laxiflorae Širj., Spisy Přír. Fak. Masarykovy Univ. 76:106. 1926.—TYPE (designated by Rechinger 1984): O. laxiflora Baker.
= Sect. Anthyllium Nábělek subsect. Fedcenkoanae Širj., Spisy Přír. Fak. Masarykovy Univ. 76:8. 1926. syn. nov.—TYPE (designated here): O. schugnanica B.Fedtsch.
= Sect. Anthyllium Nábělek subsect. Mirae Širj., Spisy Přír. Fak. Masarykovy Univ. 76:13. 1926. syn. nov.—TYPE (designated here): O. dealbata Stocks.
Description: Perennial herbs, caulescent or acaulescent. Stipules mostly free rarely connate. Wings shorter, sometimes longer than keel. Legume semiglobular, straight or slightly curved at ventral suture, with narrow to rather broad crest and dentate.
Sect. Lipskyanae (Širj.) Amirah. & Kaz. Osaloo, stat. nov. ≡ Sect. Anthyllium Nábělek subsect. Lipskyanae Širj., Spisy Přír. Fak. Masarykovy Univ. 76:11. 1926.—TYPE (designated here): O. grandis Lipsky.
Description: Perennial herb, caulescent. Leaves with remote leaflets. Inflorescence lax, peduncle curved after flowering; Legume semiglobular, 1-seeded, with straight ventral suture and dentate crest.
Sect. Heliobrychis (Bunge ex Boiss.) Sirj., Spisy Přír. Fak. Masarykovy Univ. 76:19. 1926 ≡ Sect. Sisyrosema Bunge ex Boiss. subsect. Heliobrychideae Bunge ex Boiss., Fl. Orient. 2:527. 1872.—TYPE (designated here): O. heterophylla C.A.Mey.
Description: Perennial rarely annual herbs, caulescent or almost acaulescent. Wings shorter than keel. Legume sub-orbicular, covered with pinnate bristles, stipitate, with curved ventral suture and without crest.
Sect. Hymenobrychis DC., Prodr. 2:346. 1825. emend. Amirahm. & Kaz. Osaloo.—TYPE (designated here): O. radiata (Desf.) M. Bieb.
= Sect. Anthyllium Nábělek, Spisy Přír. Fak. Masarykovy Univ. 35:96. 1923. syn. nov.—TYPE (designated here): O. susiana Nábělek
= Sect. Anthyllium Nábělek subsect. Nabélekianae Širj., Spisy Přír. Fak. Masarykovy Univ. 76:14. 1926. syn. nov.—TYPE (designated here): O. susiana Nábělek.
Description: Perennial rarely biennial herbs, caulescent rarely acaulescent. Wings short. Legume stipitate, with semi-curved to curved ventral suture; crest well-developed, more or less dentate, disk hairy, bristly or rarely glabrous.
Sect. Insignes (Širj.) Rech.f., Fl. Iranica 157: 460. 1984 ≡ Sect. Hymenobrychis DC. subsect. Insignes Širj., Spisy Přír. Fak. Masarykovy Univ. 76:58. 1926.—TYPE (designated by Rechinger 1984): O. eubrychidea Boiss.
Description: Perennial or annual herb, more or less caulescent, Wings shorter than keel. Legume orbicular-reniform with curved ventral suture; crest membranous, irregularly dentate.
Sect. Litvinovianae (Širj.) Amirahm. & Kaz. Osaloo, stat. nov. ≡ Sect. Dendrobrychis DC. ser. Litvinovianae Širj., Spisy Přír. Fak. Masarykovy Univ. 56: 29. 1925.—TYPE (designated here): O. arnacantha Bunge ex Boiss.
Description: Cushion-forming, strongly branching shrublets. Petioles spinescent. Legume semi-lunar.
References
Abou-El-Enain M (2002) Chromosomal criteria and their phylogenetic implications in the genus Onobrychis Mill. sect. Lophobrychis (Leguminosae), with special reference to Egyptian species. Bot J Linn Soc 139:409–414. doi:10.1046/j.1095-8339.2002.00075.x
Ahangarian S, Kazempour Osaloo S, Maassoumi AA (2007) Molecular phylogeny of the tribe Hedysareae with special reference to Onobrychis (Fabaceae) as inferred from nrDNA ITS sequences. Iranian J Bot 13:64–74
Amirabadizadeh H, Abbassi M, Ranjbar M (2007) A new species of Onobrychis Sect. Heliobrychis (tribe Hedysarae) from Iran. Iranian J Bot 13:53–56
Amirabadizadeh H, Ghanavati F, Abbassi M, Ranjbar M (2009) A new species of Onobrychis sect. Afghanicae (Fabaceae) from Iran. Iranian J Bot 15:45–50
Amirahmadi A, Kazempour Osaloo S, Moein F, Kaveh A, Maassoumi AA (2014a) Molecular systematics of the tribe Hedysareae (Fabaceae) based on nrDNA ITS and plastid trnL-F and matK sequences. Pl Syst Evol 300:729–747. doi:10.1007/s00606-013-0916-5
Amirahmadi A, Kazempour Osaloo S, Khoshsokhan-Mozaffar M, Charkhchian MM (2014b) A new species of Onobrychis sect. Onobrychis (Fabaceae) from Iran. Turkish J Bot 38:658–664. doi:10.3906/bot-1309-54
Arslan E, Ertuğrul K, Tugay O, Dural H (2012) Karyological studies of the genus Onobrychis Mill. and the related genera Hedysarum L. and Sartoria Boiss. & Heldr. (Fabaceae, Hedysareae) from Turkey. Caryologia 65:11–17. doi:10.1080/00087114.2012.678079
Avci S, Sancak C, Can A, Acar A, Pınar NM (2013) Pollen morphology of the genus Onobrychis (Fabaceae) in Turkey. Turkish J Bot 37:669–681. doi:10.3906/bot-1207-52
Baldwin BJ, Sanderson MJ, Porter MJ, Wojciechowski MF, Campbell C, Donoghue JM (1995) The ITS region of nuclear ribosomal DNA: a valuable source of evidence of Angiosperm phylogeny. Ann Missouri Bot Gard 82:247–277. doi:10.2307/2399880
Ball PW (1978) Onobrychis. In: Tutin TG, Heywood VH, Burges NA, Moore DM, Valentine DH, Walters SM, Webb DA (eds) Flora Europaea, vol 2. Cambridge University Press, Cambridge, pp 187–191
Cunningham CW (1997) Can three incongruence tests predict when data should be combined? Molec Biol Evol 14:733–740
Dong W, Liu J, Yu J, Wang L, Zhou S (2012) Highly variable chloroplast markers for evaluating plant phylogeny at low taxonomic levels and for DNA barcoding. PLoS ONE 7:e35071. doi:10.1371/journal.pone.0035071
Douzery E, Pridgeon A, Kores P, Linder HP, Kurzweil H, Chase M (1999) Molecular phylogenetics of Diseae (Orchidaceae): a contribution from nuclear ribosomal ITS sequences. Amer J Bot 86:887–899
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Duan L, Wen J, Yang X, Liu PL, Arslan E, Ertuğrul K, Chang ZY (2015) Phylogeny of Hedysarum and tribe Hedysareae (Leguminosae: Papilionoideae) inferred from sequence data of ITS, matK, trnL-F and psbA-trnH. Taxon 64:49–64. doi:10.12705/641.26
Edgar RC (2004) Muscle: multiple sequence alignment with high accuracy and high throughput. Nucl Acids Res 32:1792–1797. doi:10.1093/nar/gkh340
Farris JS, Kallersjo M, Kluge AG, Bult C (1995) Testing significance of incongruence. Cladistics 10:315–319. doi:10.1111/j.1096-0031.1994.tb00181.x
Felsenstein J (1985) Confidence limits on phylogenies: an approach using the bootstrap. Evolution 38:783–791. doi:10.2307/2408678
Grossheim AA (1972) Onobrychis Adans. (Leguminosae). In: Komarov VL, Shishkin BK, Bobrov EG (eds) Flora of the USSR, vol 13. Israel Program for Scientific Translations, Jerusalem, pp 244–281
Hayot Carbonero C, Carbonero F, Smith LMJ, Brown TA (2012) Phylogenetic characterization of Onobrychis species with special focus on the forage crop Onobrychis viciifolia Scop. Genet Resources Crop Evol 59:1777–1788. doi:10.1007/s10722-012-9800-3
Hedge IC (1970) Onobrychis (Leguminosae-Hedysareae). In: Davis PH, Chamberlain DF, Matthews VA (eds) Flora of Turkey and the East Aegean Islands, vol 3. Edinburgh University Press, Edinburgh, pp 560–590
Hesamzadeh Hejazi SM, Ziaei Nasab M (2010) Cytotaxonomy of some Onobrychis (Fabaceae) species and populations in Iran. Caryologia 63:18–31. doi:10.1080/00087114.2010.589705
Irfan E, Turgut-Balik D, Sahin A, Kursat M (2007) Total electrophoretic band patterns of some Onobrychis species growing in Turkey. Amer Eurasian J Agric Environ Sci 2:123–126
Karamian R, Moradi Behjou A, Ranjbar M (2012) Anatomical findings of Onobrychis sect. Heliobrychis (Fabaceae) in Iran and their taxonomic implications. Turkish J Bot 36:27–31. doi:10.3906/bot-1010-2
Kazempour Osaloo S, Maassoumi AA, Murakami N (2005) Molecular systematics of the Old World Astragalus (Fabaceae) as inferred from nrDNA ITS sequence data. Brittonia 57:367–381. doi:10.1663/0007-196X(2005)057[0367:MSOTOW]2.0.CO;2
Lamarck MM, De Candolle AP (1805) Flore Française. vol. 4, part 2, Paris, pp 401–946
Lewke Bandara N, Papini A, Mosti S, Brown T, Smith LMJ (2013) A phylogenetic analysis of genus Onobrychis and its relationships within the tribe Hedysareae (Fabaceae). Turkish J Bot 37:981–992. doi:10.3906/bot-1210-32
Lock JM (2005) Tribe Hedysarae. In: Lewis G, Schrire B, Mackinder B, Lock M (eds) Legumes of the world. Royal Botanical Gardens, Kew, pp 489–495
Mabberley DJ (2008) The plant-book. A portable dictionary of the higher plants, 3rd edn. Cambridge University Press, Cambridge
Nylander JAA (2004) MrModeltest v2. Program distributed by the author. Evolutionary Biology Centre, Uppsala University, Uppsala
Page DM (2001) Treeview (Win32) version 1.6.6. Available at: http://taxonomy.zoology.gla.ac.uk/rod/treeview.html
Pavlova DK, Manova VI (2000) Pollen morphology of the genera Onobrychis and Hedysarum (Hedysareae, Fabaceae) in Bulgaria. Ann Bot Fenn 37:207–217
Podlech D (1967) Neue und bemerkenswerte Fabaceae aus Nordost-Afghanistan (beiträge zur Flora von Afghanistan II). Mitt Bot Staatssamml München 6:547–591
Posada D, Buckley TR (2004) Model selection and model averaging in phylogenetics: advantages of akaike information criterion and Bayesian approaches over likelihood ratio tests. Syst Biol 53:793–808. doi:10.1080/10635150490522304
Ranjbar M, Amirabadizadeh H, Karamian R, Ghahremani MA (2004) Notes on Onobrychis sect. Heliobrychis (Fabaceae) in Iran. Willdenowia 34:187–190. doi:10.3372/wi.34.34116
Ranjbar M, Karamian R, Hajmoradi F (2009) Taxonomic notes on Onobrychis sect. Hymenobrychis (Fabaceae, Hedysareae) in Iran. Novon 19:215–218. doi:10.3417/2007119
Ranjbar M, Karamian R, Hadadi A (2010) Cytosystematics of three Onobrychis species (Fabaceae) in Iran. Caryologia 63:237–249. doi:10.1080/00087114.2010.10589733
Ranjbar M, Hajmoradi F, Karamian R (2012) An overview on cytogenetics of the genus Onobrychis (Fabaceae) with special reference to O. sect. Hymenobrychis from Iran. Caryologia 65:187–198. doi:10.1080/00087114.2012.735887
Rechinger KH (1984) Hedysareae. In: Rechinger KH (ed) Flora Iranica, vol 157. Akademische Druck, Graz, pp 365–475
Ronquist F, Teslenko M, van der Mark P, Ayres DL, Darling A, Höhna S, Larget B, Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61:539–542. doi:10.1093/sysbio/sys029
Safaei Chaei Kar S, Ghanavati F, Mozafari J, Naghavi MR, Amirabadizadeh H, Darvish F (2012) Phylogenetic relationships of Onobrychis Mill. (Fabaceae: Papilionoideae) based on ITS sequences of nuclear ribosomal DNA and morphological traits. Crop Breed J 2:91–99
Safaei Chaei Kar S, Ghanavati F, Naghavi MR, Amirabadi-zade H, Rabiee R (2014) Molecular phylogenetics of the Onobrychis genus (Fabaceae: Papilionoideae) using ITS and trnL–trnF DNA sequence data. Austral J Bot 62:235–250. doi:10.1071/BT13279
Sang T, Crawford DJ, Stuessy T (1995) Documentation of reticulate evolution in peonies (Paeonia) using internal transcribed spacer sequences of nuclear ribosomal DNA: implication for biogeography and concerted evolution. Proc Natl Acad Sci USA 92:6813–6817
Shaw J, Lickey EB, Schilling EE, Small RL (2007) Comparison of whole chloroplast genome sequences to choose noncoding regions for phylogenetic studies in Angiosperms: the tortoise and the hare III. Amer J Bot 94:275–288. doi:10.3732/ajb.94.3.275
Silvestro D, Michalak I (2012) raxmlGUI: a graphical front-end for RAxML. Organisms Divers Evol 12:335–337. doi:10.1007/s13127-011-0056-0
Širjaev G (1925) Onobrychis generis revisio critica, pars prima. Spisy Přír Fak Masarykovy Univ 56:1–197
Širjaev G (1926) Onobrychis generis revisio critica, partes secunda et tertia. Spisy Přír Fak Masarykovy Univ 76:1–165
Swofford DL (2002) PAUP*: Phylogenetic analysis using parsimony (*and other methods), Version 4.0b10. Sinauer Associates, Sunderland
Taberlet P, Gielly L, Pautou G, Bouvet J (1991) Universal Primers for amplification of three non-coding regions of chloroplast DNA. Pl Molec Biol 17:1105–1109. doi:10.1007/BF00037152
Tamura K, Stecher G, Peterson D, Filipski A, Kumar S (2013) MEGA6: molecular evolutionary genetics analysis version 6.0. Molec Biol Evol 30:2725–2729. doi:10.1093/molbev/mst197
Townsend CC (1974) Papilionaceae. In: Townsend CC, Guest ER (eds) Flora of Iraq, vol 3. Ministry of Agriculture and Agrarian Reform of the Republic of Iraq, Baghdad, pp 54-601
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis DH (ed) PCR protocols: a guide to methods and applications. Academic Press, San Diego, pp 315–322
Wojciechowski MF, Sanderson MJ, Hu JM (1999) Evidence on the monophyly of Astragalus (Fabaceae) and its major subgroups based on nuclear ribosomal DNA ITS and chloroplast DNA trnL intron data. Syst Bot 24:409–437. doi:10.2307/2419698
Yildiz B, Ciplak B, Aktoklu E (1999) Fruit morphology of sections of the genus Onobrychis Miller (Fabaceae) and its phylogenetic implications. Israel J Pl Sci 47:269–282. doi:10.1080/07929978.1999.10676784
Zarrabian M, Majidi MM (2015) Genetic diversity and relationships within and among Onobrychis species using molecular markers. Turkish J Bot 39:681–692. doi:10.3906/bot-1406-47
Zwickl D (2006) Genetic algorithm approaches for the phylogenetic analysis of large biological sequence datasets under the maximum likelihood criterion. PhD thesis, University of Texas, Austin
Acknowledgments
The present study was financially supported in part by Grant-in-Aids for Scientific Research, No. 89002433, to S.K. O (corresponding author) from INSF (Iran National Science Foundation). We would like to thank the staff of the herbaria of, FUMH, HKNRRC, HQNRRC, MSB, TARI and TUH to allow studying herbarium specimens and providing leaf materials. We also thank H. Zare for editing and improving linguistic of the text. The trees resulting from Parsimony, Likelihood and Bayesian analyses of nrDNA ITS and plastid datasets, trnL–F, rpl32/rpl32-trnL(UAG) and ndhF-rpl32 are presented as Electronic Supplementary Material in Online resource 1, 2, 3, 4. The aligned data matrix used in this study is presented as Online Resource 5.
Author information
Authors and Affiliations
Corresponding author
Additional information
Handling editor: Christoph Oberprieler.
Electronic supplementary material
Below is the link to the electronic supplementary material.
606_2016_1343_MOESM1_ESM.pdf
Online Resource 1 Fifty percent majority rule consensus tree resulting from Bayesian inference of the nrDNA ITS dataset. Numbers above branches are posterior probability and likelihood as well as parsimony bootstrap values, respectively. Values<50 % were not shown (PDF 132 kb)
606_2016_1343_MOESM2_ESM.pdf
Online Resource 2 Fifty percent majority rule consensus tree resulting from Bayesian inference of the plastid trnL-F dataset. Numbers above branches are posterior probability and likelihood as well as parsimony bootstrap values, respectively. Values<50 % were not shown (PDF 129 kb)
606_2016_1343_MOESM3_ESM.pdf
Online Resource 3 Fifty percent majority rule consensus tree resulting from Bayesian inference of the plastid rpl32/rpl32- trnLUAG dataset. Numbers above branches are posterior probability and likelihood as well as parsimony bootstrap values, respectively. Values<50 % were not shown (PDF 129 kb)
606_2016_1343_MOESM4_ESM.pdf
Online Resource 4 Fifty percent majority rule consensus tree resulting from Bayesian inference of the plastid ndhF-rpl32 dataset. Numbers above branches are posterior probability and likelihood as well as parsimony bootstrap values, respectively. Values<50 % were not shown (PDF 132 kb)
Information on Electronic Supplementary Material
Information on Electronic Supplementary Material
Online Resource 1. Fifty percent majority rule consensus tree resulting from Bayesian inference of the nrDNA ITS dataset.
Online Resource 2. Fifty percent majority rule consensus tree resulting from Bayesian inference of the plastid trnL-F dataset.
Online Resource 3. Fifty percent majority rule consensus tree resulting from Bayesian inference of the plastid rpl32/rpl32- trnLUAG dataset.
Online Resource 4. Fifty percent majority rule consensus tree resulting from Bayesian inference of the plastid ndhF-rpl32 dataset.
Online Resource 5. The aligned data matrix used in this study.
Rights and permissions
About this article
Cite this article
Amirahmadi, A., Kazempour-Osaloo, S., Kaveh, A. et al. The phylogeny and new classification of the genus Onobrychis (Fabaceae-Hedysareae): evidence from molecular data. Plant Syst Evol 302, 1445–1456 (2016). https://doi.org/10.1007/s00606-016-1343-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00606-016-1343-1