Abstract
The authors describe a colorimetric method for the determination of Hg(II) ion. It is based on the color change from red to colorless as displayed by gold nanoparticle (AuNP) modified with thymine - rich DNA. Signal amplification is accomplished by free strand displacement recycling. In this strategy, Hg(II) unfolds the arch-trigger duplex due to the high affinity between Hg(II) and the thymines to form T-Hg(II)-T structures, thereby causing the release of trigger. The liberated trigger unfolds the hairpin structure of H1, and unfolded H1 further unfolds with H2. As a result, the H2 hairpin displaces trigger, and the released trigger unfolds another H1. This results in strong and enzyme-free strand displacement recycling amplification. The aggregation of DNA-AuNPs occurs in the presence of the duplex formed by hairpins H2 and H1. This results in a color change from red to colorless that can be visually observed. Under optimal conditions, the assay has a detection range over 4 orders of magnitude and a 3.4 nM detection limit. The assay is selective, sensitive, rapid and cost-effective. In our perception, it represents a useful platform for determination of Hg(II).

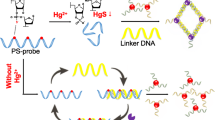
Schematic presentation of the simple, rapid, low cost colorimetric detection of mercury(II) based on enzyme-free strand displacement amplification along with DNA-labeled AuNP.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The presence of mercury in the environment originates from a variety of natural sources and human activities [1]. An annual release of 4400–7500 metric tons of mercury was estimated by the United Nations Environment Programme [2]. According to the World Health Organisation, mercury is one of the ten most dangerous elements to public health [3]. It is a highly toxic heavy metal that contaminates the water and is often accumulated throughout the food chain, thus posing a serious threat to the environment, animals, and humans by causing damage to the brain, heart, kidneys, lungs, and the immune system [4]. The maximum allowable level of Hg2+ in drinking water is 10 nM, a standard set by the U.S. Environmental Protection Agency [5]. Therefore, it is important to monitor Hg2+ levels with sensitivity and selectivity in aqueous systems. There is an urgent need for developing novel Hg2+ detection methods, which are facile, rapid, low-cost, highly sensitive and applicable to aqueous systems, to protect our environment and health.
Standard methods for Hg2+ detection include atomic absorption/emission spectroscopy [6, 7], selective cold vapor atomic fluorescence spectrometry [8], X-ray fluorescence spectrometry [9], inductively coupled plasma mass spectrometry [10] and anodic stripping voltammetry [11, 12]. These methods are very sensitive and selective, but they require complicated sample preparation and sophisticated instruments which limit their applications to routine Hg2+ detection [13]. Thus, it is still of great challenge to develop a simple method for aqueous Hg2+ detection. Nanotechnology based strategies have been reported for mercury detection. Among them, noble metal-based nanomaterials, such as Au, have been usually used for Hg2+ detection with electrochemical techniques and colorimetric measurements [14, 15]. However, synergistic metal nanoparticle-electrochemical strategies require multiple steps, including extensive pre-treatments of the electrode surface (polishing, surface contamination oxidation, etc.) [14], which cannot be easily automated and thus affect their applications as an ideal system. Colorimetric methods, which negate the above disadvantages, are extremely attractive and the detection results can be easily read by bare eyes [16, 17].
Gold nanoparticle (AuNP) has attracted much attention in colorimetric detection because of its high extinction coefficient in the visible region and color-tunable behavior that depends on the interparticle distance [18, 19]. Several groups reported colorimetric detection methods based on AuNP for the assay of heavy metal ions [20]. One such strategy is based on manipulating AuNP with different recognition units such as oligonucleotides, DNAzymes, peptides, proteins, and small thiolate ligands to induce aggregation [21]. However, the stability of this detection method is not sufficiently good for real-water applications because of that unmodified gold nanoparticle is unstable and it is easily influenced by the pH, temperature and other ions in reaction solution [22]. Subsequently, strategies using horseradish peroxidase [23], glucose oxidase [24], exonuclease I [25], exonuclease III [26], polymerase [27], and nicking endonuclease [28] coupled with DNA-AuNP have been reported with high sensitivities. Nevertheless, compared with other methods, enzymes make the detection relatively costly and complicated. Furthermore, the target, Hg2+, may denature and affect the activity of the enzymes. Therefore, it is desirable to develop a simple approach for Hg2+ detection that offers the advantages of enzyme-free, high sensitivity and good selectivity, particularly excellent stability performance in real-life sample analysis.
Enzyme-free strand displacement and branch migration have been studied since the 1970s [29], but have only been applied to DNA nanotechnology within the past decade. Strand displacement refers to the displacement of one or more pre-hybridized strands from partial or full complementary dsDNAs, which is initiated at the complementary single-stranded domains [30]. The strand displacement process is guided by the biophysics of DNA and occurs independently without the presence of enzymes. Due to the nucleic acid nature of this process, strand displacement has been demonstrated to be useful in designing new enzyme-free signal amplification approaches for detecting targets. As far as we know, enzyme-free strand displacement has not been used for Hg2+ detection. Herein, we describe a simple, rapid, excellent stability and enzyme-free detection approach for Hg2+ by using the color change of DNA-AuNP and strand displacement amplification (SDA) strategy. In this sensing system, thymine-Hg2+-thymine (T-Hg2+-T) coordination chemistry is used to specific recognize Hg2+. Besides, an arched probe is designed to contain a thymine-rich DNA probe (T) and a complementary DNA (T*), which as trigger can initiate strand displacement reaction with the aid of two hairpin probes (HAPs, H1 and H2). DNA-AuNP with stable property is used as a signal indicator. DNA-AuNP is extremely stable in the absence of Hg2+, but, when Hg2+ is added into the reaction mixture, owing to the affinity between Hg2+ and thymine in the DNA sequence, Hg2+ specifically combines to thymine and form “T-Hg2+-T mismatch” structure, leading to the dissociative T*. Immediately T* unfolds the hairpin structure of H1, thus the opened H1 further unfolds with H2, which results T* is away from H1 and free in buffer. Then T* hybridizes with another H1 again and initiates the cyclic strand displacement reaction, forming abundant of H1-H2 duplex. Because two same L* is included in the H1-H2 duplex, which induces the aggregation of L-labelled AuNPs through the hybridization of L with L*. Thus, the DNA-AuNP aggregation was achieved through reducing distance between DNA-AuNP. The output signals can be directly observed by the bare eye and also observed under UV light. In addition, SDA strategy is a key component in this highly sensitive assay to detect Hg2+. Therefore, this colorimetric sensing strategy can overcome some of the limitations of previously mentioned techniques.
Materials and methods
Reagents and materials
Oligonucleotides used in this work were HPLC-purified and synthesized by Sangon Biotechnology Co. Ltd. (Shanghai, China). The sequences of oligonucleotides are listed in Table 1. It involved the design and synthesis of five oligonucleotides (T, T*, H1, H2 and Linker). What’s more, an arched probe is designed to contain a thymine-rich DNA probe (T) and a complementary DNA (T*), which as trigger can initiate strand displacement reaction with the aid of two hairpin probes (HAPs, H1 and H2). The two specially designed HAPs contain the same DNA segments (L*) at their 5′ convex end, which is complementary to linker-DNA (L).
Posphine hydrochloride was obtained from Alfa-Aesar (Tianjing, China). Chloroauric acid (HAuCl4·4H2O) and trisodium citrate were obtained from Shanghai Reagent Company (Shanghai, China). Phosphate Buffer (PB, 100 mM, pH 7.4), phosphate-buffered saline (PBS, 5×, pH 7.4), and Tris acetate-EDTA (TAE) buffer (1×, pH 8.2) were purchased from Sigma-Aldrich (St. Louis, MO). All other reagents were of analytical reagent grade and were used without further purification. Ultrapure water obtained from a Millipore filtration system was used throughout. Sterile water was obtained via using the traditional high-temperature sterilization method.
Apparatus
Absorption spectra were recorded on a UV2600 UV-vis spectrophotometer (Shimadzu international trading Co. Ltd., Japan; http://www.shimadzu.com.cn/) at room temperature. Transmission electron microscopy (TEM) measurements were made on a JEM-3010 transmission electron microscope (http://www.jeol.co.jp/). The samples for TEM characterization were prepared by adding a drop of AuNP solution on a carbon-coated copper grid and drying at room temperature. Gel eletrophoresis was conducted using a DYCZ-24DN electrophoresis cell (LIUYI, Beijing, China; http://www.zg17w.cn/brand/beijingliuyi.html) and a Bio-Rad Gel imaging system (Bio-Rad, USA; http://www.bio-rad.com/).
Preparation of AuNP and DNA-conjugated with AuNP
AuNP with an average diameter of 20 nm were prepared as previously described [30]. Before the experiments, all glassware was cleaned in aqua regia (HCl/HNO3, 3:1), rinsed with ultrapure 66water, and then oven-dried. Next, an aqueous solution of HAuCl4 (0.04 g/ml, 500 μL) was added to 200 mL ultrapure water while stirring in a round-bottom flask fitted with a reflux condenser. It was heated with electric heating furnace. Then 3 mL of 1% (w/v) trisodium citrate solution was quickly added to the boiling solution, while it was being vigorously agitated, which resulted in a color change from pale yellow to deep red. Boiling was continued for 15 min. After the solution was cooled to room temperature, it was filtered and stored in a refrigerator at 4 °C before use. 1 mM HAuCl4 was brought to boil with vigorous stirring in a round-bottom flask fitted with a reflux condenser.
The DNA-AuNP were prepared as follows. First, AuNP was concentrated to 1 nM via centrifugation at 15000 rpm (21380 rcf) for 10 min followed by re-suspension of the sediment in ultrapure water. Second, DNA probe was then added into the mixture followed by standing at 4 °C for 24 h. Third, the mixture was added with 66 μL phosphate buffer (PB, 100 mM, pH 7.4), followed by the addition with 35 μL PBS (10 mM, pH 7.4). Fourth, after 48 h incubation at 4 °C, the mixture was slowly added with 81 μL PBS followed by incubation at 4 °C for another 24 h. Fifth, the unmodified oligonucleotides were removed via centrifugation at 15000 rpm (21,380 rcf) for 10 min followed by re-suspension of the sediments in 1 mL ultrapure water. Then this step was repeated three times to sufficiently remove all excessive oligonucleotides. Last, the AuNP decorated with DNA were re-dispersed in 300 μL ultrapure water and stored at 4 °C until use. The final concentration of DNA-conjugated with AuNP was ~1 nM, assuming that there was no significant loss of AuNPs during the preparation process.
Gel electrophoresis
The gel electrophoresis was performed at 145 V for 30 min using DNA samples (10 μL per well) on a 4% agarose gel with a fluorescence stain SYBR Gold (0.5 g/ mL) in 50 mM Tris-borate running buffer (pH 8.2) containing 2 mM EDTA. After electrophoresis, the gel was visualized via fluorescence detection using a gel imaging system.
Transmission electron microscopy
The samples for transmission electron microscopy characterization were prepared by pipetting 5 μL of the solution of AuNP on a carbon-coated copper grid. After solvent evaporation, the grid was dried overnight. All of the images were bright-field images.
Analytical protocol
All samples were prepared in PBS for the detection of Hg2+ and the detailed procedure was as follows: Firstly, T (3 μL, 10 μM), T* (3 μL, 10 μM), H1(3 μL, 10 μM), H2 (3 μL, 10 μM) and equal volumes of different concentrations (5 nM, 10 nM, 50 nM, 100 nM, 500 nM, 1 μM, 5 μM, 10 μM) of Hg2+ (or other heavy metal ions) were mixed at 37 °C for 30 min. Subsequently, the DNA-modified AuNP (30 μL, 3 nM) was added into the reaction mixture and incubated for 30 min at 37 °C, before the above mixture (60 μL) was detected by UV-visible spectrometry.
Results and discussion
Design principle of the colorimetric biosensor for highly sensitive detection of Hg2+
Scheme 1 shows the principle of enzyme-free SDA-based colorimetric assay for Hg2+. An arched probe is designed to contain a thymine-rich DNA probe (T) and a complementary DNA (T*), which as trigger can initiate SDA reaction with the aid of two hairpin probes (HAPs, H1 and H2). The two specially designed HAPs contain the same DNA segments (L*) at their 5′ convex end, which is complementary to linker-DNA (L). In the absence of Hg2+, the arched probe is a stable double-strands structure, thus T*can’t hybridize with H1 and trigger SDA reaction. However, when Hg2+ is added into the reaction mixture, owing to the affinity between Hg2+ and thymine in the DNA sequence, Hg2+ specifically combines to thymine and form “T-Hg2+-T mismatch” structure, leading to the dissociative T*. Immediately T* unfolds the hairpin structure of H1, thus the opened H1 further unfolds with H2, which results T* is away from H1 and free in buffer. Then T* hybridizes with another H1 again and initiates the cyclic strand displacement reaction, forming abundant of H1-H2 duplex. Because two same L* is included in the H1-H2 duplex, which induces the aggregation of L-labeled AuNP through the hybridization of L with L*. Therefore, the target Hg2+ detection can be visualized by color change and also UV-Vis absorption due to the change of the surface plasmon absorption.
UV-visible spectrometric characterization
Biosensing was performed in the absence of Trigger, H1, H2 or Hg2+, in the presence of Pb2+ and Hg2+. As shown in Fig. 1, in the absence of T*(curve a), H1 (curve b) or H2 (curve c) respectively, there are three strong peaks. This signifies that the enzyme-free SDA reaction is dependent on the combined action of T*, H1 and H2. Two powerful peaks were achieved for blank sample (curve d) and a sample of Hg2+ replaced by Pb2+ (curve e). In contrast, it is found a negligible peak in the presence of Hg2+ (curve f). This clearly reveals that the decreased signal is caused by the specific recognition of Hg2+ and Hg2+ that triggers the SDA reaction. On the basis of these results, it is reasonably concluded that our designed route should be feasible for Hg2+ assay.
Gel electrophoresis analysis
Direct proof of the biosensor mechanism was acquired through gel electrophoresis analysis, as shown in Fig. 2. Lane 1 and lane 2 respectively display a bright band of H1 and H2 on the image. At the same position the band becomes brighter (lane 3) after H1 incubated with H2. The result implies that H1 and H2 do not unfold each other independently. However, after T* incubation with H1 and H2, a new band with high molecular weight appeared. This indicates that the binding of trigger and H1 results in a structural switch and H2 associates with the opened H1 (Lane4). After incubation with T and T*, lane 5 displays a bright band of an arch probe. As seen in lane 6, it has a band of the same mobility with that of lane 4 and one new band is observed for positive sample. The top band demonstrates that the hybridized H1 with H2 is primed by T* in the presence of Hg2+, while the band at the bottom indicats that the repeated strand-displacement reaction produces T* and T. In contrast, the result in lane 7 was obtained using a blank sample shows two bands of the same mobility with that of lane 3 and lane 5. By incubating T, T*, H1 and H2 in Pb2+, instead of Hg2+, the result is shown in lane 8. Here, two bands of the similar mobility to those in lane 7 are observed. This further reveals the feasibility of the enzyme-free strand displacement mechanism.
Gel electrophoresis image obtained for different sample. Lane M, DNA marker; lane 1, H1; lane 2, H2; lane 3, H1 and H2; lane 4, T*, H1 and H2; lane 5, arch probe (T-T*); lane 6, T, T*, H1 and H2 incubated with Hg2+; lane 7, T, T*, H1 and H2 incubated without Hg2+; lane 8, T, T*, H1 and H2 incubated with Pb2+
Characterization of gold nanoparticle and DNA-AuNP
Transmission electron microscopy was utilized to characterize the prepared AuNP. As shown in Fig.3a, the spherical structure of AuNP shows good dispersion and homogeneous diameter. To verify the formation of DNA-AuNP, UV-vis spectroscopy experiments were further performed. As shown in Fig. 3b, a strong absorption peak was observed at 519 nm at bare AuNP. After conjugation these AuNP with DNA, the characteristic absorption peak of gold nanoparticle red shifted from 519 to 524 nm and the peak decreased due to the decoration of biomolecules on the Au nanoparticle. These results demonstrate the accomplishment of the DNA-AuNP nanoprobes.
Optimization of experimental conditions
In order to avoid sterically preventing the SDA process, we added the DNA–AuNP until SDA finished. To achieve optimal analytical performance of the biosensor, some key experimental parameters including the concentration of T*, H1 and H2 and the reaction time were investigated. Fig.4a depicts the effect of the concentration of T* on the UV-Vis peak absorbance of biosensor. As anticipated, the absorbance decreased with the increasing of the concentration of T*, and then reached equilibrium when the concentration of T* was 1 μM. Then, the influence of the concentrations of hairpins (H1 and H2) was investigated, since the hairpins not only constituted important components of SDA but also functioned in the process of DNA-AuNP aggregation. The results show that the optimized concentration of H1 and H2 was 1 μM (Fig.4b, c). Therefore, 1 μM was chosen for the optimal H1 (or H2) concentration in the following experiments. Generally, a longer reaction time yields a more stable absorbance. The absorbance was recorded along with the reaction time increasing (Fig.4d). It is found that the absorbance decreased with the reaction time increasing, then reached a minimum at 1 h, and kept almost a constant value until 1.5 h. To ensure the completeness of reaction, we choose 1 h as the optimum reaction time.
a Effect of the concentration of T* on the UV-Vis peak absorbance of biosensor. b Effect of the concentration of H1 on the UV-Vis peak absorbance of biosensor. c Effect of the concentration of H2 on the UV-Vis peak absorbance of biosensor.d Effect of the reaction time of AuNPs on the UV-Vis peak absorbance of biosensor
Calibration plot
According to the above standard procedures and under the optimized assay conditions, the constructed biosensor was adopted for the determination of a series of different concentrations of Hg2+. Fig. 5a depicts typical colorimetric responses of the biosensor to Hg2+ of varying concentration. It was observed that the absorbance decreased with the increasing concentration of Hg2+. The plot of the peak absorbance versus the logarithm value of Hg2+ concentration displayed a good linear relationship in the range from 5 nM to 10 μM (Fig. 5b), the linear regression equations is Absorbance =0.434–0.050logC Hg2+, with a correlation coefficient of −0.997 and the limit of detection (expressed as three times the standard deviation of a blank response) of 3.4 nM was estimated.
a Colorimetric responses of the biosensor to different concentrations of Hg2+. b The calibration curve of UV-Vis peak absorbance for different concentrations of Hg2+. Error bars are standard deviations across three repetitive experiments. Error bars are standard deviations across three repetitive experiments
Selectivity
To evaluate the selectivity of this protocol, a control experiment was conducted. The difference of absorbance for Hg2+ and other non-target heavy metal ions, including Cu2+, Mg2+, Fe2+, Ca2+, Pb2+, Zn2+, Mn2+, Cd2+ and Ni2+ detection, under optimum conditions was compared. As indicated in Fig. S1, in contrast to significant response (~0.24) as observed for Hg2+, negligible signal response (~0.41) was observed upon the addition of other non-target heavy metal ions. Hence, the results showed excellent selectivity toward Hg2+ over other heavy metal ions because of the specificity structure of T-Hg2+-T.
Comparison to previously reported methods
The analytical performance of our biosensor in the quantitative assay of Hg2+ was compared with that of some previously reported methods [31–38] . The results were shown in Table S1. It was found that the method presented here displays improved sensitivity and a wider dynamic range compared with these methods. Moreover, our method has the advantages of rapid, excellent stability and cost-effective. As a result, our biosensor might satisfy the requirements for the profiling low abundance of Hg2+ in water due to its very low detection limit.
Environmental water sample analysis
To test the practical application of our method, several environmental water samples spiked with Hg2+, with concentrations of 10 nM, 100 nM, 1000 nM, were tested using this work and atomic fluorescence spectroscopy. The samples used in this study were tap water. The samples spiked with different concentrations of Hg2+ were detected according to the general procedure with four replicates. The results are summarized in Table S2 and show good agreement with the found values determined by AFS. These data clearly demonstrate that our biosensor can be applied to complex samples.
Conclusion
In summary, we have developed an enzyme-free colorimetric assay for highly sensitive and selective detection of Hg(II) based on DNA conjugated to gold nanoparticle along with strand displacement amplification. This biosensor relies on T-Hg2+-T coordination chemistry to achieve specific recognition for Hg(II). Owing to enzyme-free strand displacement amplification strategy, the resulting colorimetric assay can detect Hg(II) with a wider detection range from 5 nM to 10 μM and a limit of detection of 3.4 nM. This biosensor also exhibits very high selectivity toward Hg(II) against other interfering metal ions. Moreover, our strategy offers the advantage of facilitated instrumentation, shortened analysis time, and simplified operations without the need of sample pretreatment and multiple washing steps. Besides, our biosensor can be used for the determination of Hg(II) in environmental water with high specificity and sensitivity. Additionally, this sensing strategy holds the potential of being extended for the detection of various aptamer binding molecules by the specific design of the Arched DNA probe. Therefore, this biosensor might create a useful and versatile platform in detecting Hg(II) and aptamer binding molecules.
References
Chen JH, Zhou SG, Wen JL (2014) Disposable strip biosensor for visual detection of Hg2+ based on Hg2+-triggered toehold binding and exonuclease III-assisted signal amplification. Anal Chem 86:3108–3114
Li HL, Zhai JF, Tian JQ, Luo YL, Sun XP (2011) Carbon nanoparticle for highly sensitive and selective fluorescent detection of mercury(II) ion in aqueous solution. Biosens Bioelectron 26:4656–4660
WHO (2007) Exposure to mercury: a major public health concern. WHO Preventing Disease Through Healthy Environments. Geneva, Switzerland, World Health Organization
Huang PJ, Wang F, Liu JW (2015) Cleavable molecular beacon for Hg2+ detection based on Phosphorothioate RNA modifications. Anal Chem 87:6890–6895
Office of Water (2001) Mercury update: impact on fish advisories, EPA fact sheet EPA-823-F-01−011. U.S. Environmental Protection Agency, Washington, D. C
Lo JM, Yu JC, Hutchison FI, Waei CM (1982) Solvent extraction of Dithiocarbamate complexes and back-extraction with mercury(II) for determination of trace metals in seawater by atomic absorption spectrometry. Anal Chem 54:2536–2539
Zenko Y, Masao T (1977) Indirect determination of Submicrogram amounts of sulfide by flameless atomic absorption spectrometry of mercury. Mikrochim Acta 67(5):459–468
Balint L, Vedrina-Dragojević I, Šebečić B, Momirović-Čuljat J, Horvatić M (1997) Spectrofluorometric method for determination of the Total mercury content in environmental samples-waste waters. Mikrochim Acta 127:61–65
Kim HN, Ren WX, Kim JS, Yoon JY (2012) Fluorescent and colorimetric sensors for detection of lead, cadmium, and mercury ions. Chem Soc Rev 41:3210–3244
Gomez-Ariza J, Lorenzo F, Garcia-Barrera T (2005) Comparative study of atomic fluorescence spectroscopy and inductively coupled plasma mass spectrometry for mercury and arsenic multispeciation. Anal Bioanal Chem 382:485–492
Gholivand MB, Parvin MH (2010) Differential pulse anodic stripping Voltammetric simultaneous determination of copper (II) and silver (I) with Bis(2-hydroxyacetophenone) butane-2,3-dihydrazone modified carbon paste electrodes. Electroanalysis 22:2291–2296
Deng W, Tan Y, Li Y, Wen Y, Su Z, Huang Z, Huang S, Meng Y, Xie Q, Luo Y, Yao S (2010) Square wave voltammetric determination of hg(II) using thiol functionalized chitosan-multiwalled carbon nanotubes nanocomposite film electrode. Microchim Acta 169:367–373
Leermakers M, Baeyens W, Quevauviller P, Horvat M (2005) Mercury in environmental samples: speciation, artifacts and validation. Trends Anal Chem 24:383–393
Ratner N, Mandler D (2015) Electrochemical detection of low concentrations of mercury in water using gold nanoparticles. Anal Chem 87:5148–5155
Xue XJ, Wang F, Liu XG (2008) One-step, room temperature, colorimetric detection of mercury (Hg2+) using DNA/nanoparticle conjugates. J Am Chem Soc 130:3244–3245
F Zarlaida, M. Adlim, (2016) Gold and silver nanoparticles and indicator dyes as active agents in colorimetric spot and strip tests for mercury(II) ions: a review; Microchim Acta 2016; on the web. DOI: 10.1007/s00604-016-1967-4
Wu S, Li Q, Duan N, Ma H, Wang Z (2016) DNA aptamer selection and aptamer-based fluorometric displacement assay for the hepatotoxin microcystin-RR. Microchim Acta 183(9):2555–2562
Zhang JF, Lim CS, Cho BR, Kim JS (2010) A two-photon excited luminescence of water-soluble rhodamine–platinum(II) complex: fluorescent probe specific for Hg2+ detection in live cell. Talanta 83:658–662
Xu Q, Du S, Jin GD, Li HB, Hu XY (2011) Determination of acetamiprid by a colorimetric method based on the aggregation of gold nanoparticles. Microchim Acta 173:323–329
Zhang J, Tang Y, Lv J, Fang SQ, Tang DP (2015) Glucometer-based quantitative determination of hg(II) using gold particle encapsulated invertase and strong thymine-hg(II)-thymine interaction for signal amplification. Microchim Acta 182:1153–1159
Shi QN, Shi YP, Pan Y, Yue ZF, Zhang H, Yi CQ (2015) Colorimetric and bare eye determination of urinary methylamphetamine based on the use of aptamers and the salt-induced aggregation of unmodified gold nanoparticles. Microchim Acta 182:505–511
Wang H, Wang YX, Jin JY, Yang RH (2008) Gold nanoparticle-based colorimetric and “turn-on” fluorescent probe for mercury(II) ions in aqueous solution. Anal Chem 80:9021–9028
Wang GF, Huang H, Zhang XJ, Wang L (2012) Electrically contacted enzyme based on dual hairpin DNA structure and its application for amplified detection of Hg2+. Biosens Bioelectron 35:108–114
Cao GX, Wu XM, Dong YM, Li ZJ, Wang GL (2016) Colorimetric determination of melamine based on the reversal of the mercury(II) induced inhibition of the light-triggered oxidase-like activity of gold nanoclusters. Microchim Acta 183:441–448
Yuan M, Zhu YG, Lou XH, Chen C, Wei G, Lan MB, Zhao JL (2012) Sensitive label-free oligonucleotide-based microfluidic detection of mercury(II)ion by using exonuclease I. Biosens Bioelectron 31:330–336
Wang GF, Xu G, Zhu YH, Zhang XJ (2014) A “turn-on” carbon nanotube–ag nanoclusters fluorescent sensor for sensitive and selective detection of Hg2+ with cyclic amplification of exonuclease III activity. Chem Commun 50:747–750
Zhu X, Zhou XM, Xing D (2011) Ultrasensitive and selective detection of mercury(II) in aqueous solution by polymerase assisted fluorescence amplification. Biosens Bioelectron 26:2666–2669
Zhu GC, Li Y, Zhang CY (2014) Simultaneous detection of mercury(II) and silver(I)ions with picomolar sensitivity. Chem Commun 50:572–574
Xu YY, Zhou WJ, Zhou M, Xiang Y, Yuan R, Chai YQ (2015) Toehold strand displacement-driven assembly of G-quadruplex DNA for enzyme-free and non-label sensitive fluorescent detection of thrombin. Biosens Bioelectron 64:306–310
Quan K, Huang J, Yang XH, Yang YJ, Ying L, Wang H, He Y, Wang KM (2015) An enzyme-free and amplified colorimetric detection strategy via target–aptamer binding triggered catalyzed hairpin assembly. Chem Commun 51:937–940
Yan FY, Kong DP, Luo YM, Ye QH, He JJ, Guo XF, Chen L (2016) Carbon dots serve as an effective probe for the quantitative determination and for intracellular imaging of mercury(II). Microchim Acta 183:1611–1618
Lu Y, Yu J, Ye WC, Yao X, Zhou PP, Zhang HX, Zhao SQ, Jia LP (2016) Spectrophotometric determination of mercury(II) ions based on their stimulation effect on the peroxidase-like activity of molybdenum disulfide nanosheets. Microchim Acta 183:2481–2489
Shi DC, Yan FY, Zhou XG, Zheng TC, Shi YY, Fu WG, Chen L (2016) Preconcentration and fluorometric detection of mercury ions using magnetic core-shell chitosan microspheres modified with a rhodamine spirolactam. Microchim Acta 183:319–327
Mojtaba S, Afsaneh S, Zahra M, Raheleh A (2016) Highly selective aggregation assay for visual detection of mercury ion based on competitive binding of sulfur-doped carbon nanodots to gold nanoparticles and mercury ions. Microchim Acta 183:2327–2335
Khosro ZK, Alagarsamy P, Subramaniam J, Ramasamy R, Hong NL, Boon HO, SDB C, Yeh YK, Huang NM (2016) Amalgamation based optical and colorimetric sensing of mercury(II) ions with silver@graphene oxide nanocomposite materials. Microchim Acta 183:369–377
Hu TY, Yan X, Na WD, Su XG (2016) Aptamer-based aggregation assay for mercury(II) using gold nanoparticles and fluorescent CdTe quantum dots. Microchim Acta 183:2131–2137
Tang WJ, Wang Y, Wang PP, Di JW, Yang JP, Wu Y (2016) Synthesis of strongly fluorescent carbon quantum dots modified with polyamidoamine and a triethoxysilane as quenchable fluorescent probes for mercury(II). Microchim Acta 183:2571–2578
An’amt MN, Perumal R, Huang NM, Wei LS (2016) Visual and spectrophotometric determination of mercury(II) using silver nanoparticles modified with graphene oxide. Microchim Acta 183:597–603
Acknowledgements
This work was supported by NSFC (21405060, 1471644), Shandong Province Natural Science Funds for Distinguished Young Scholars (JQ201410), and Shandong Province Natural Science Funds (ZR2015CM027).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The author(s) declare that they have no competing interests.
Electronic supplementary material
ESM 1
(DOC 680 kb)
Rights and permissions
About this article
Cite this article
Liu, S., Leng, X., Wang, X. et al. Enzyme-free colorimetric assay for mercury(II) using DNA conjugated to gold nanoparticles and strand displacement amplification. Microchim Acta 184, 1969–1976 (2017). https://doi.org/10.1007/s00604-017-2182-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00604-017-2182-7