Abstract
Aims
This cohort study examined the association of total and individual dairy products with the risk of incident MetS and its components in children and adolescents.
Methods
We prospectively assessed 531 participants aged 6–18 years without the MetS at baseline during an average 6.6-year follow-up period. Dairy consumption was estimated with a valid and reliable food frequency questionnaire. The MetS was defined according to the Cook criteria. The multivariable regression model was used to calculate the odds ratio (OR) for incident MetS associated with the consumption of dairy products.
Results
The incidence of MetS was 9.8% after an average 6.6-year follow-up. After adjusting for potential confounders, OR (95% confidence interval) for incident MetS was 0.48 (0.23–1.00) for total dairy, 0.44 (0.21–0.92) for low-fat dairy, 0.46 (0.22–0.98) for low-fat milk, and 0.45 (0.21–0.97) for low-fat yogurt when comparing participants in the highest versus lowest tertile. A moderate intake of regular cheese was associated with decreased risk of MetS (OR = 0.43, 95% CI: 0.19–0.97). Replacing one serving/day of total dairy with nuts was associated with a lower (OR: 0.63, 95% CI: 0.42–0.95), whereas replacement by red and processed meat was associated with higher (OR: 1.55, 95%CI: 1.21–1.97) MetS risk. No significant association was found between high-fat dairy and MetS risk.
Conclusions
Higher consumption of dairy products, particularly low-fat milk and yogurt, was associated with reduced risk of incident MetS, suggesting the capability of low-fat dairy products in the primary prevention of MetS in children and adolescents.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The metabolic syndrome (MetS), described by the clustering of cardiometabolic risk factors that include anthropometric and physiological parameters, appears to be caused through pathophysiological processes related to insulin resistance [1]. The prevalence and incidence of MetS has increased rapidly around the world [2, 3]. The prevalence of MetS in Iranian adolescents increased from 4.1% in 1990 to 7.6% in 2018 [4, 5]. In addition to higher presence of mental health disorders, non-alcoholic fatty liver, and obstructive sleep apnea in later childhood or adolescence [6], the evidence indicates that MetS and obesity in children and adolescents are strong predictors of future type 2 diabetes, insulin resistance, and cardiovascular diseases in adulthood [7, 8]. Although the etiology of the disease is not well known, dietary and lifestyle pattern are recognized as the leading modifiable risk factors that appear to have a substantial effect on its development [9]. From a nutritional point of view, various healthy dietary patterns and particularly some food groups, such as dairy products, may have a beneficial impact on MetS prevention in adults [10], but the evidence in children and adolescents is limited.
Dairy products have a desirable ratio of nutrient density to energy density [11] and supply protein and some micronutrients, especially calcium, in a regular diet. However, inclusion of dairy as part of a healthy diet and the effects of milk, yogurt, and cheese on health and longevity is controversial [12]. There are major differences in the components of individual dairy products that may alter their role in metabolic health. Diverse profiles of fatty acids and total content in each dairy food may influence the risk of MetS progression as well. Although dietary guidelines recommend to eat low or reduced fat dairy products, recent evidence casts doubt on the detrimental effect of dairy fat on metabolic health, indicating there is neutral or inverse association between full-fat dairy consumption and cardiometabolic health outcomes [13, 14]. The presence of lactic acid bacteria in fermented products such as yogurt and cheese, in addition to the content of specific saturated fatty acids (SFA), has distinct effects on cardiometabolic disease and insulin resistance [15, 16].
Contrary to the results of a cross-sectional study illustrating an inverse relationship between dairy consumption and abdominal obesity [17], Matthews et al. demonstrated that dairy consumption doubles the risk of obesity among children and adolescents [18]. A recent meta-analysis of prospective cohort studies reported a 23% reduction in the incidence of MetS with dairy product intakes in adults [19]. However, to our knowledge, no previous longitudinal study has examined the association of dairy products with the incidence of MetS in children and adolescents. Therefore, we aimed to investigate the association of total and individual dairy food consumption with the incidence of MetS among children and adolescents.
Materials and methods
Study population
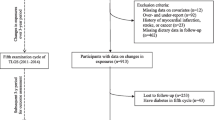
We conducted the current study within the framework of the Tehran Lipid and Glucose Study (TLGS), which is an ongoing community-based prospective investigation aiming to evaluate the risk factors for non-communicable diseases. The baseline survey was a cross-sectional study (1999–2001), and participants are monitored every 3 years [20].
In the fourth examination survey of the TLGS (2009–2012) of 12,523 participants who had complete medical history and physical examination data, a representative sample of 7956 participants was randomly selected for dietary assessment. There were 829 children and adolescents aged 6–18 years with complete nutritional data.
Participants who had missing data on MetS components (n = 10) were excluded. Furthermore, participants with over- or underreporting energy intake, i.e., those in the top and bottom 1% of the energy intake to estimated energy requirement (EER) [21] ratio (n = 9), were also excluded. Depending on outcome variables under analysis, participants were also excluded if they presented that outcome at baseline: (MetS, n = 91; hypertension, n = 72; high triglycerides (TGs), n = 144; low high-density lipoprotein cholesterol (HDL-C), n = 84; high fasting plasma glucose (FPG), n = 89; or abdominal obesity, n = 189). Some participants fell into more than one category of exclusion; the final sample size varied by the following outcomes: MetS (n = 531), hypertension (n = 517), high TGs (n = 449), low HDL-C (n = 509), high FPG (n = 504), and abdominal obesity (n = 412). Data from the sixth survey of the TLGS (response rate: 82%) were used to evaluate the incidence of MetS and its components after a mean 6.6 years of follow-up.
Protocols of this study were approved by the institutional ethics committee of the Research Institute for Endocrine Sciences, affiliated to the Shahid Beheshti University of Medical Sciences, and written informed consent was obtained from participants' parents.
Measurements
Dietary measurements
A valid and reliable semi-quantitative food frequency questionnaire (FFQ) was applied to assess the habitual dietary intake of children and adolescents at the baseline [22,23,24]. The relative validity of the FFQ for measuring dairy intake has been previously reported, and it shows a reasonable correlation with estimates from dietary records. Trained dietitians asked participants during a face-to-face interview to determine their consumption frequency for each food item consumed during the previous year on a daily, weekly, or monthly basis. Mothers were asked about the type and quantity of meals and snacks when children had difficulties or were unable to recall. Portion sizes of consumed foods that were reported in household measures were then converted to grams. To assist with determining portion size, interviewers provided photographs of household portions of each food item.
The questionnaire items for dairy products included low-fat milk, whole milk, low-fat yogurt, high-fat yogurt, regular cheese, cream cheese, and ice cream. The standard serving sizes were 240 mL for low-fat milk and whole milk; 120 mL for yogurt and ice cream; 220 mL for low- or high-fat yogurt, and 30 mL for cream cheese and regular cheese. Furthermore, we summed low-fat milk (< 2% of total fat content) with low-fat yogurt (< 2% of total fat content) and labeled as low-fat dairy, and high-fat yogurt (≥ 3% of total fat content), high-fat milk (≥ 3% of total fat content), ice cream, regular cheese, and cream cheese labeled as high-fat dairy.
Measurements of covariates
Physical activity (PA) was estimated by the Modifiable Activity Questionnaire (MAQ), and metabolic equivalent task (MET) minutes per week was calculated. The Persian-translated MAQ in adolescents showed high reliability (97%) and moderate validity (49%) [25]. A low level of PA was considered as MET < 600 min/week.
Weight was recorded in light clothing to the nearest 0.1 kg on a SECA digital weighing scale (Seca 707; Seca Corporation, Hanover, Maryland; range 0.1–150 kg), and height was measured without shoes to the nearest 0.1 cm. Waist circumference (WC) was measured to the nearest 0.1 cm using a non-elastic tape over light clothing, midway between the iliac crest and the lowest rib in a standing position. Arterial blood pressure (BP) was measured with a suitable cuff size for each participant, after a 15-min rest in a supine position. Systolic- (SBP) and diastolic blood pressure (DBP) were measured, and the average of two measurements was recorded as the participants' BP.
A blood sample was drawn after 12–14-h overnight fast. Fasting plasma glucose was measured on the day of blood collection by the enzymatic colorimetric method, using glucose oxidase. Serum TGs concentration was measured by commercially available enzymatic reagents (Pars Azmoon, Tehran, Iran), adapted to a Selectra auto analyzer. HDL-C was measured after precipitation of the apolipoprotein B-containing lipoproteins with phosphotungstic acid and evaluated using Pars Azmoon kits. Inter- and intra-assay coefficients of variations at baseline were 2.2% for FPG, 2.0, and 0.5% for HDL-C and 1.6 and 0.6% for TGs, respectively.
Definitions
In children and adolescents, MetS was defined according to the definition proposed by Cook et al. [26] as three or more of the following: WC ≥ 90th percentile for age and sex, according to national reference curves [27]; SBP and DBP ≥ 90th percentile for sex, age, and height (the National Heart, Lung, and Blood Institute's recommended cut-points) [28], and FPG ≥ 100 mg/dl (American Diabetes Association) [29]. Fasting TGs ≥ 110 mg/dl; HDL-C < 40 mg/dl; In subjects aged > 18 years after follow-up, MetS was defined according to the joint interim statement [30] as the presence of any 3 of 5 risk factors following: (i) Enlarged WC (≥ 95 cm according to the newly introduced cut-off points for Iranian adults for both genders) [31]; (ii) FPG ≥ 100 mg/dl or use of hyperglycemic medication; (iii) Fasting TGs ≥ 150 mg/dl or use of antihypertriglyceridemia medication; (iv) Fasting HDL-C < 50 mg/dl for women and < 40 mg/dl for men or drug treatment; and (v) High BP was defined as SBP ≥ 130 mm Hg, DBP ≥ 85 mm Hg, or antihypertensive drug treatment.
Statistical analysis
All dietary exposures were adjusted for total energy using the residual method [32]. The Kolmogorov–Smirnov test and histogram were applied to determine the normality of the quantitative variables. Continuous variables were presented as the mean (standard deviation (SD)) or median (inter-quartile 25th and 75th), whereas categorical data were presented as percentages. Energy-adjusted dairy intake and its subgroups were categorized into tertiles. Linear regression and chi-square were used to test the trend of continuous and categorical variables, respectively, across the tertile of total dairy intakes.
We applied multivariable regression models to estimate the odds ratio (OR) and 95% confidence interval (CIs) across categories of total dairy consumption with the risk of incident MetS, abdominal obesity, high FPG, high TGs, low HDL-C, and hypertension. For all outcomes, the basic model was adjusted for age in years. Consequently, we further adjusted for family history of diabetes, PA, total energy intake, and sex. We additionally adjusted for some dietary covariates, including percent of energy from protein and carbohydrate along with whole-grain consumption. We modeled dairy exposures as categorical variables and tested for a trend by modeling the median value within each category as a continuous variable. The interaction terms between age (grouped as from 6 to 12 years and from 13 to 18 years), sex (girl, boy), and PA (low, moderate, and vigorous), and total dairy consumption on the incidence of MetS were examined. No interaction was found between considered covariates and total dairy intake on the outcome in the final models.
We estimated the effect on MetS risk of increasing the intake of one serving per day of nuts, legumes, whole grain, poultry and fish, egg, or red and processed meat while concomitantly decreasing the dairy consumption, using substitution modeling. Thus, the difference in regression coefficients was used to estimate ORs for the substitution and covariance to estimate the 95% CIs. Statistical significance was set at P < 0.05. All statistical analyses were performed using the SPSS software (version 15.0; SPSS Inc, Chicago, IL).
Results
The average age of the population was 12.3 (3.1) years; 49.2% were girls, and 81% had low physical activity. The average dairy consumption was 2.8 serving/day. Among the 531 participants without MetS at baseline, there were 52 (9.8%) cases of incident MetS over an average 6.6-year follow-up. Baseline clinical and dietary characteristics of participants are presented across tertile categories of energy-adjusted dairy consumption in Table 1. In trend analyses from the lowest to the highest tertile category of dairy consumption, and those in the highest category were more likely to be younger. With increasing total dairy intake, intake of whole grains and percent of energy from carbohydrate tended to be lower. In contrast, intake of percent of energy from fatty acids, SFA, protein as well as consumption of low- and high-fat dairy tended to be higher.
Risk of incident MetS in those with normal status at baseline, according to total dairy consumption, is presented in Table 2. In the basic model, adjusted for age, increasing total dairy consumption was associated with a 55% lower risk of incident MetS (lowest (reference) compared with the highest intake, OR: 0.45, 95% CI: 0.22–0.94; P-trend = 0.029). This association remained after adjusting for sex, total energy intake, physical activity, and family history of diabetes (OR: 0.48, 95% CI = 0.23–1.00, P-trend = 0.042), and even after additionally adjusting for dietary factors (OR: 0.41, 95% CI = 0.19–0.88, P-trend = 0.018). In the final model, a 1 serving/day increase in dairy consumption was associated with lower MetS risk (OR: 0.68, 95% CI = 0.51 –0.92). In the fully adjusted model, low-fat dairy intake was inversely associated with incident MetS (highest compared with the lowest tertile, OR: 0.44, 95% CI: 0.21–0.92; P-trend = 0.023). Furthermore, one serving/day increase in low-fat dairy consumption in a diet was marginally associated with decreased MetS risk (OR: 0.74, 95% CI = 0.54–1.02).
We further evaluated associations of types of dairy products with risk of incident MetS (Table 3). After controlling for considered confounding factors, higher low-fat milk consumption was associated with a 54% lower risk of incident MetS (highest compared with lowest consumption, OR: 0.46, 95% CI: 0.22–0.98; P-trend = 0.050); and the higher low-fat yogurt intake was associated with a 65% lower risk of incident MetS (OR: 0.45, 95% CI: 0.21–0.97; P-trend = 0.031). Moderate but not the highest intake of regular cheese consumption had a decreased risk of MetS by 67% (second compared with first tertile consumption, OR: 0.43, 95% CI: 0.19–0.97). High-fat milk, high-fat yogurt, and ice cream intake were not associated with the risk of incident MetS.
After adjusting for confounding factors, higher dairy consumption was associated with 51%, 63%, and 57% lower risk of incident abdominal obesity (OR: 0.49, 95% CI = 0.26–0.94, P-trend = 0.032), high BP (OR: 0.37, 95% CI = 0.17–0.81, P-trend = 0.008), high TGs (OR: 0.43, 95% CI = 0.20–0.96, P-trend = 0.039) as presented in Supplementary Table 1.
We estimated that substituting one serving/day of total dairy for nuts was associated with a 47% lower risk of MetS incidence (OR: 0.63, 95% CI: 0. 42–0. 95; Fig. 1). However, a substitution for red and processed meat was associated with a 54% higher risk of MetS (OR: 1.55, 95% CI: 1.21–1.97) (Fig. 1).
Odds ratios and 95% confidence intervals from substitution models for incidence of metabolic syndrome associated with increasing consumption of a food group and concomitantly decreasing consumption of total dairy by one serving per day. The model was adjusted for baseline age, sex, total energy intake, physical activity, family history of diabetes, dietary intakes of protein, carbohydrate, and whole grain.
Discussion
In this community-based prospective study of children and adolescents with a 6.6-year follow-up period, we observed that consumption of total dairy products was related to lower incident MetS and some of its components, including abdominal obesity, high BP, and high TGs after adjustment for several diet and lifestyle covariates. Furthermore, specific dairy products, including low-fat dairy, low-fat milk, low-fat yogurt, and regular cheese, were associated with a lower risk of incident MetS. Decreasing intake of total dairy and simultaneously increasing nut intake by one serving/day was associated with a 47% lower risk of MetS, whereas substituting one serving/d of red and processed meat for total dairy consumption was associated with a 57% higher risk of MetS.
To our knowledge, the present study is the first to examine the association between total and individual dairy consumption and MetS risk in a longitudinal analysis in children and adolescents. We showed that a higher intake of total dairy decreased the odds of incident MetS in children and adolescents, whereas a cross-sectional study of Iranian adolescents found no association of dairy consumption with the risk of MetS and its component [33]. It should be noted that our results are in agreement with those of other prospective and cross-sectional studies indicating an inverse association of dairy consumption with the MetS risk in adults [14, 34, 35]. A meta-analysis among studies of adults reported a 15% (RR: 0.85; 95% CI: 0.73, 0.98) lower risk of MetS incidence for the highest versus lowest category of total dairy consumption [36]. We observed that by increasing total dairy consumption in a regular diet both low- and high-fat dairy consumption was concurrently increased. Accordingly, the percentage of energy from total fatty acids and SFA was also elevated. Notably, it remains controversial whether the intake of dairy fat is harmful to cardiometabolic health, and whether the intake of low-fat dairy provides advantages with regard to MetS risk relative high-fat dairy [37, 38]. Furthermore, we observed that participants who consumed more dairy had lower intakes of whole grain and percent of energy from carbohydrate, compared to those who consumed less. Thus, the association between dairy consumption and metabolic syndrome could be the relative reduction in carbohydrates rather than (or in combination with) the relative increase in dairy consumption; however, the association still remains significant when whole grain and percent of energy from carbohydrate were adjusted in the final model to attenuate its confounding effect.
In the present study, we determined that low-fat dairy consumption was inversely associated with the risk of the MetS, unlike high-fat dairy. This finding was further supported by the lower risk of MetS among those with dairy products consumption with low-fat content, like yogurt and milk [39, 40]. Although the beneficial effect of yogurt consumption on metabolic parameters has been reported in some studies, this finding is inconsistent [33, 41, 42]. One explanation could be the inability of studies to separate the low-fat yogurt from high-fat ones. In the current study, we observed that a higher intake of low-fat yogurt was associated with a 45% decreased risk of MetS; however, high-fat yogurt consumption had no significant association. The favorable association of low-fat dairy and MetS may be attributed to the higher amounts of protective constituents, such as calcium and whey protein instead of SFA content, in low-fat dairy [43].
Nevertheless, data on the risk–benefit of cheese intake is highly controversial. A positive association between cheese consumption and MetS prevalence was illustrated in one study [44], while higher consumption of cheese had an inverse association with the incidence of MetS in another [45]. In our study, moderate consumption of cheese was associated with a lower risk of MetS compared to lower consumption. Moderate intake of regular cheese may provide some advantages of dairy products family; however, high content of fat and salt in cheese may confound its beneficial effect on the metabolism [46]. Moreover, we did not have enough detail about the types of cheeses consumed in this cohort to assay MetS risk in relation to various cheese types. More research is needed to determine the underlying mechanism that may mediate the relation between cheese consumption and MetS risk. With regard to ice cream consumption, we did not observe any association with MetS risk among children and adolescents. These findings may be explained by the fact that ice cream contains high amounts of fat and sugar, which can eliminate the favorable effects of milk component [47]. Overall, our findings suggested that low-fat dairy products, like yogurt and milk, are associated with a lower risk of MetS. It should also be noted that consuming high-fat dairy did not raise risk of MetS.
Data from previous studies revealed the desirable association of dairy consumption with cardiometabolic risk factors such as overweight/obesity, high BP, and abdominal obesity among children and adolescents [17, 48, 49] and in adults [35]. In our longitudinal study, we found that participants in the highest tertiles of dairy consumption had a lower risk for the incidence of abdominal obesity, high BP, and high TGs. In a prospective study conducted in Australian children, as compared with those who were in the lowest category of dairy consumption, children in the highest dairy consumption had lower incident SBP and DBP risk [49]. Abreu et al. found that higher consumption of dairy was associated with a 78% reduction in abdominal obesity in boys after adjustment for confounders [17]. Likewise, a recent meta-analysis of prospective cohort comprising 46,011 children and adolescents showed that those in the highest category of dairy consumption had a 38% lower risk of overweight/obesity compared with subjects in the lowest consumption during a median follow-up of 3-years [48].
Several potential mechanisms for the beneficial effect of dairy consumption on MetS have been proposed. The nutrients found in dairy products, particularly calcium and protein, may exert a synergistic and protective role against metabolic dysregulation. Calcium inhibits fat absorption and increases fecal excretion in the intestine through binding to bile acids and fatty acids, especially SFA [50]. As a result, calcium intake can improve the HDL-C to LDL-C ratio by converting cholesterol to bile acids in the liver and decrease TGs levels [51, 52]. Moreover, dietary calcium, as shown in a meta-analysis of intervention studies [53], decreases BP through repression of 1,25-dihydroxy vitamin D3, resulting in normalization of intercellular calcium in vascular smooth muscle cells [54]. Peptide fractions of dairy protein may play a major role in preventing the MetS through regulating insulinemia, dyslipidemia, BP, and fat accumulation [55]. Dairy is a substantial source of two major forms of protein, casein and whey. These proteins inhibit the function of angiotensin-converting enzyme (ACE), which is known to increase BP and adipocyte lipogenesis by upregulated of fatty acid synthesis expression. Hence, the inhibition of ACE and consequently angiotensin II production by dairy-derived proteins can decrease BP and endogenous fat deposition [56, 57].
We performed substitution analyses to predict the impacts of replacing dairy products with other foods on the risk of MetS. We found that replacing dairy with red and processed meat was associated with a higher risk of MetS. Red and processed meat consumption may lead to an elevated risk of developing MetS by several pathways such as increased insulin resistance and the loss of β-cell function, obesity, hyperinsulinemia and hyperglycemia. These comorbidities are stemmed from the high content of fat and SFA, especially long-chain SFA compared to short- and medium-chain SFA in dairy, high levels of inflammatory mediators or through elevated free fatty acid and nitrosamines production [58,59,60]. A meta-analysis indicated that total, red, and processed meat consumption was associated with higher MetS risk [61]. In contrast to red meat, replacing total dairy with nuts was associated with a lower risk of MetS. These health effects may be explained by improving oxidative stress, inflammation, and endothelial function, which can reduce the risk of insulin resistance and diabetes, BP, abdominal obesity, and dyslipidemia as components of MetS [62]. These results suggest that replacing nuts for dairy products in a regular diet may provide further advantages to metabolic health than dairy consumption.
Our study has several strengths, including a longitudinal follow-up design and a high follow-up rate, which helps to evaluate the incidence of the MetS. We have also used a validated FFQ and national cutoff points for defining abdominal obesity, WC, and BP in children and adolescents.
The study also has limitations. Firstly, the information on the puberty status of our participants was not recorded, and we were not able to control the puberty stage in our analysis. Secondly, our cohort was made up of Tehranian urban participants; thus, our findings cannot be generalized to other populations. Thirdly, although habitual dietary intake of participants was assessed by a validated FFQ, potential measurement errors are unavoidable.
Conclusion
In conclusion, the results of the present prospective study suggest that higher consumption of dairy, particularly low-fat milk and yogurt, is related to a lower risk of incident MetS among children and adolescents. Furthermore, replacing total dairy with red and processed meat is associated with a higher MetS risk, whereas replacement by nuts is associated with a lower risk. Thus, it is imperative to invest in a strategy to improve the dietary pattern and consume healthy food groups, particularly low-fat dairy, to prevent cardio-metabolic risk factors in children and adolescents.
Availability of data and materials
The data set is the property of the Research Institute for Endocrine Sciences (RIES) and is made available upon approval of the research proposal by the research council and the ethics committee. The RIES ethics committee must issue an approval in case of a request for access to the de-identified dataset. The data request may be sent to the head of the RIES Ethics Committee, Dr. Shahram Alamdari, at email: alamdari@endocrine.ac.ir.
References
Gurka MJ, Golden SH, Musani SK et al (2017) Independent associations between a metabolic syndrome severity score and future diabetes by sex and race: the Atherosclerosis Risk In Communities Study and Jackson Heart Study. Diabetologia 60(7):1261–1270. https://doi.org/10.1007/s00125-017-4267-6
Dunkley A, Charles K, Gray L, Camosso-Stefinovic J, Davies M, Khunti K (2012) Effectiveness of interventions for reducing diabetes and cardiovascular disease risk in people with metabolic syndrome: systematic review and mixed treatment comparison meta-analysis. Diabetes Obes Metab 14(7):616–625
Grundy SM (2008) Metabolic syndrome pandemic. Arterioscler Thromb Vasc Biol 28(4):629–636. https://doi.org/10.1161/ATVBAHA.107.151092
Ahmadi N, Sadr SM, Mohammadi MR et al (2020) Prevalence of abdominal obesity and metabolic syndrome in children and adolescents: a community based cross-sectional study. Iran J Public Health 49(2):360–368
Kelishadi R, Hovsepian S, Djalalinia S, Jamshidi F, Qorbani M (2016) A systematic review on the prevalence of metabolic syndrome in Iranian children and adolescents. J Res Med Sci Official J Isfahan Univ Med Sci 21:90. https://doi.org/10.4103/1735-1995.192506
Magge SN, Goodman E, Armstrong SC (2017) The metabolic syndrome in children and adolescents: shifting the focus to cardiometabolic risk factor clustering. Pediatrics 140(2):e20171603. https://doi.org/10.1542/peds.2017-1603
Glueck CJ, Woo JG, Khoury PR, Morrison JA, Daniels SR, Wang P (2015) Adolescent oligomenorrhea (age 14–19) tracks into the third decade of life (age 20–28) and predicts increased cardiovascular risk factors and metabolic syndrome. Metab Clin Exp 64(4):539–553. https://doi.org/10.1016/j.metabol.2015.01.005
Llewellyn A, Simmonds M, Owen CG, Woolacott N (2016) Childhood obesity as a predictor of morbidity in adulthood: a systematic review and meta-analysis. Obes Rev Official J Int Assoc Study Obes 17(1):56–67. https://doi.org/10.1111/obr.12316
Wannamethee SG, Shaper AG, Whincup PH (2006) Modifiable lifestyle factors and the metabolic syndrome in older men: Effects of lifestyle changes. J Am Geriatr Soc 54(12):1909–1914. https://doi.org/10.1111/j.1532-5415.2006.00974.x
Perez-Martinez P, Mikhailidis DP, Athyros VG et al (2017) Lifestyle recommendations for the prevention and management of metabolic syndrome: an international panel recommendation. Nutr Rev 75(5):307–326. https://doi.org/10.1093/nutrit/nux014
Nicklas TA, O’Neil CE, Fulgoni VL 3rd (2009) The role of dairy in meeting the recommendations for shortfall nutrients in the American diet. J Am Coll Nutr 28(Suppl 1):73S-81S. https://doi.org/10.1080/07315724.2009.10719807
Mozaffarian D (2016) Dietary and policy priorities for cardiovascular disease, diabetes, and obesity: a comprehensive review. Circulation 133(2):187–225. https://doi.org/10.1161/CIRCULATIONAHA.115.018585
Hirahatake KM, Astrup A, Hill JO, Slavin JL, Allison DB, Maki KC (2020) Potential cardiometabolic health benefits of full-fat dairy: the evidence base. Adv Nutr 11(3):533–547. https://doi.org/10.1093/advances/nmz132
Astrup A, Geiker NRW, Magkos F (2019) Effects of full-fat and fermented dairy products on cardiometabolic disease: food is more than the sum of its parts. Adv Nutr 10(5):924s–930s. https://doi.org/10.1093/advances/nmz069
Fernandez MA, Marette A (2018) Novel perspectives on fermented milks and cardiometabolic health with a focus on type 2 diabetes. Nutr Rev 76(Suppl 1):16–28. https://doi.org/10.1093/nutrit/nuy060
Mozaffarian D, Wu JHY (2018) Flavonoids, dairy foods, and cardiovascular and metabolic health: a review of emerging biologic pathways. Circ Res 122(2):369–384. https://doi.org/10.1161/CIRCRESAHA.117.309008
Abreu S, Santos R, Moreira C et al (2012) Association between dairy product intake and abdominal obesity in Azorean adolescents. Eur J Clin Nutr 66(7):830–835. https://doi.org/10.1038/ejcn.2012.32
Matthews VL, Wien M, Sabate J (2011) The risk of child and adolescent overweight is related to types of food consumed. Nutr J 10:71. https://doi.org/10.1186/1475-2891-10-71
Mena-Sánchez G, Becerra-Tomás N, Babio N, Salas-Salvadó J (2019) Dairy product consumption in the prevention of metabolic syndrome: a systematic review and meta-analysis of prospective cohort studies. Adv Nutr 10 (suppl_2):S144–S153
Azizi F, Ghanbarian A, Momenan AA et al (2009) Prevention of non-communicable disease in a population in nutrition transition: Tehran Lipid and Glucose Study phase II. Trials 10:5. https://doi.org/10.1186/1745-6215-10-5
Micronutrients IoMPo (2005) Dietary reference intakes for energy, carbohydrate, fiber, fat, fatty acids, cholesterol, protein and amino acids. Panel on Macronutrients Panel on the Definition of Dietary Fiber, Subcommittee on Upper Reference Levels of Nutrients, Subcommittee on Interpretation and Uses of Dietary Reference Intakes, and the Standing Committee on the Scientific Evaluation of Dietary Reference Intakes, Food and Nutrition Board. National Academies Press,
Hosseini Esfahani F, Asghari G, Mirmiran P, Azizi F (2010) Reproducibility and relative validity of food group intake in a food frequency questionnaire developed for the Tehran Lipid and Glucose Study. J Epidemiol (0):1002100131
Asghari G, Rezazadeh A, Hosseini-Esfahani F, Mehrabi Y, Mirmiran P, Azizi F (2012) Reliability, comparative validity and stability of dietary patterns derived from an FFQ in the Tehran Lipid and Glucose Study. The British journal of nutrition 108(6):1109–1117. https://doi.org/10.1017/S0007114511006313
Esfahani FH, Asghari G, Mirmiran P, Azizi F (2010) Reproducibility and relative validity of food group intake in a food frequency questionnaire developed for the Tehran Lipid and Glucose Study. J Epidemiol Japan Epidemiol Assoc 20(2):150–158. https://doi.org/10.2188/jea.je20090083
Delshad M, Ghanbarian A, Ghaleh NR, Amirshekari G, Askari S, Azizi F (2015) Reliability and validity of the modifiable activity questionnaire for an Iranian urban adolescent population. Int J Preventive Med 6:3. https://doi.org/10.4103/2008-7802.151433
Cook S, Weitzman M, Auinger P, Nguyen M, Dietz WH (2003) Prevalence of a metabolic syndrome phenotype in adolescents: findings from the third National Health and Nutrition Examination Survey, 1988–1994. Arch Pediatr Adolesc Med 157(8):821–827. https://doi.org/10.1001/archpedi.157.8.821
Kelishadi R, Gouya MM, Ardalan G et al (2007) First reference curves of waist and hip circumferences in an Asian population of youths: CASPIAN study. J Trop Pediatr 53(3):158–164. https://doi.org/10.1093/tropej/fml090
National High Blood Pressure Education Program Working Group on High Blood Pressure in C, Adolescents (2004) The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics 114 (2 Suppl 4th Report):555–576. 114/2/S2/555 [pii]
Genuth S, Alberti KG, Bennett P , Buse J, Defronzo R, Kahn R, Kitzmiller J, Knowler WC, Lebovitz H, Lernmark A, Nathan D, Palmer J, Rizza R, Saudek C, Shaw J, Steffes M, Stern M, Tuomilehto J, Zimmet P, Expert Committee on the D, Classification of Diabetes M (2003) Follow-up report on the diagnosis of diabetes mellitus. Diabetes Care 26(11):3160–3167. https://doi.org/10.2337/diacare.26.11.3160
Alberti KG, Eckel RH, Grundy SM, Zimmet PZ, Cleeman JI, Donato KA, Fruchart JC, James WP, Loria CM, Smith SC, Jr., International Diabetes Federation Task Force on E, Prevention, Hational Heart L, Blood I, American Heart A, World Heart F, International Atherosclerosis S, International Association for the Study of O (2009) Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 120(16):1640–1645. https://doi.org/10.1161/CIRCULATIONAHA.109.192644
Azizi F, Hadaegh F, Khalili D et al (2010) Appropriate definition of metabolic syndrome among Iranian adults: report of the Iranian National Committee of Obesity. Arch Iranian Med 13(5):426–428
Willett WC, Howe GR, Kushi LH (1997) Adjustment for total energy intake in epidemiologic studies. Am J Clin Nutr 65 (4 Suppl):1220S––1228S; discussion 1229S–1231S. https://doi.org/10.1093/ajcn/65.4.1220S
Ghotboddin Mohammadi S, Mirmiran P, Bahadoran Z, Mehrabi Y, Azizi F (2015) The Association of Dairy Intake With Metabolic Syndrome and Its Components in Adolescents: Tehran Lipid and Glucose Study. Int J Endocrinol Metabolism 13(3):e25201. https://doi.org/10.5812/ijem.25201v2
Azadbakht L, Mirmiran P, Esmaillzadeh A, Azizi F (2005) Dairy consumption is inversely associated with the prevalence of the metabolic syndrome in Tehranian adults. Am J Clin Nutr 82(3):523–530. https://doi.org/10.1093/ajcn.82.3.523
Trichia E, Luben R, Khaw KT, Wareham NJ, Imamura F, Forouhi NG (2020) The associations of longitudinal changes in consumption of total and types of dairy products and markers of metabolic risk and adiposity: findings from the European Investigation into Cancer and Nutrition (EPIC)-Norfolk study, United Kingdom. Am J Clin Nutr 111(5):1018–1026. https://doi.org/10.1093/ajcn/nqz335
Kim Y, Je Y (2016) Dairy consumption and risk of metabolic syndrome: a meta-analysis. Diabet Med 33(4):428–440
Chowdhury R, Warnakula S, Kunutsor S et al (2014) Association of dietary, circulating, and supplement fatty acids with coronary risk: a systematic review and meta-analysis. Ann Intern Med 160(6):398–406. https://doi.org/10.7326/M13-1788
Drouin-Chartier JP, Cote JA, Labonte ME et al (2016) Comprehensive review of the impact of dairy foods and dairy fat on cardiometabolic risk. Adv Nutr 7(6):1041–1051. https://doi.org/10.3945/an.115.011619
Alvarez-Bueno C, Cavero-Redondo I, Martinez-Vizcaino V, Sotos-Prieto M, Ruiz JR, Gil A (2019) Effects of milk and dairy product consumption on Type 2 Diabetes: overview of systematic reviews and meta-analyses. Adv Nutr 10 (suppl_2):S154-S163. https://doi.org/10.1093/advances/nmy107
Rancourt-Bouchard M, Gigleux I, Guay V et al (2020) Effects of regular-fat and low-fat dairy consumption on daytime ambulatory blood pressure and other cardiometabolic risk factors: a randomized controlled feeding trial. Am J Clin Nutr 111(1):42–51. https://doi.org/10.1093/ajcn/nqz251
Sayón-Orea C, Bes-Rastrollo M, Martí A, Pimenta AM, Martín-Calvo N, Martínez-González MA (2015) Association between yogurt consumption and the risk of metabolic syndrome over 6 years in the SUN study. BMC Public Health 15:170. https://doi.org/10.1186/s12889-015-1518-7
Moslehi N, Shab-Bidar S, Mirmiran P, Sadeghi M, Azizi F (2015) Associations between dairy products consumption and risk of type 2 diabetes: Tehran lipid and glucose study. Int J Food Sci Nutr 66(6):692–699. https://doi.org/10.3109/09637486.2015.1034249
Barr SI (2003) Increased dairy product or calcium intake: is body weight or composition affected in humans? J Nutr 133(1):245S-248S. https://doi.org/10.1093/jn/133.1.245S
Beydoun MA, Gary TL, Caballero BH, Lawrence RS, Cheskin LJ, Wang Y (2008) Ethnic differences in dairy and related nutrient consumption among US adults and their association with obesity, central obesity, and the metabolic syndrome. Am J Clin Nutr 87(6):1914–1925. https://doi.org/10.1093/ajcn/87.6.1914
Fumeron F, Lamri A, Abi Khalil C, Jaziri R, Porchay-Balderelli I, Lantieri O, Vol S, Balkau B, Marre M, Data from the Epidemiological Study on the Insulin Resistance Syndrome Study G (2011) Dairy consumption and the incidence of hyperglycemia and the metabolic syndrome: results from a french prospective study, Data from the Epidemiological Study on the Insulin Resistance Syndrome (DESIR). Diabetes Care 34(4):813–817. https://doi.org/10.2337/dc10-1772
Hanning AR, Wang X, Hashemi Z et al (2019) Both low- and regular-fat cheeses mediate improved insulin sensitivity and modulate serum phospholipid profiles in insulin-resistant rats. J Nutr Biochem 64:144–151. https://doi.org/10.1016/j.jnutbio.2018.10.018
Yeon SJ, Kim JH, Hong GE et al (2017) Physical and sensory properties of ice cream containing fermented pepper powder. Kor J Food Sci Anim Resour 37(1):38–43. https://doi.org/10.5851/kosfa.2017.37.1.38
Lu L, Xun P, Wan Y, He K, Cai W (2016) Long-term association between dairy consumption and risk of childhood obesity: a systematic review and meta-analysis of prospective cohort studies. Eur J Clin Nutr 70(4):414–423. https://doi.org/10.1038/ejcn.2015.226
Rangan AM, Flood VL, Denyer G et al (2012) The effect of dairy consumption on blood pressure in mid-childhood: CAPS cohort study. Eur J Clin Nutr 66(6):652–657. https://doi.org/10.1038/ejcn.2011.218
Boon N, Hul GB, Stegen JH et al (2007) An intervention study of the effects of calcium intake on faecal fat excretion, energy metabolism and adipose tissue mRNA expression of lipid-metabolism related proteins. Int J Obesity 31(11):1704–1712. https://doi.org/10.1038/sj.ijo.0803660
Reid IR, Mason B, Horne A et al (2002) Effects of calcium supplementation on serum lipid concentrations in normal older women: a randomized controlled trial. Am J Med 112(5):343–347. https://doi.org/10.1016/s0002-9343(01)01138-x
Lorenzen JK, Nielsen S, Holst JJ, Tetens I, Rehfeld JF, Astrup A (2007) Effect of dairy calcium or supplementary calcium intake on postprandial fat metabolism, appetite, and subsequent energy intake. Am J Clin Nutr 85(3):678–687. https://doi.org/10.1093/ajcn/85.3.678
Bucher HC, Cook RJ, Guyatt GH et al (1996) Effects of dietary calcium supplementation on blood pressure. A meta-analysis of randomized controlled trials. Jama 275(13):1016–1022. https://doi.org/10.1001/jama.1996.03530370054031
Zemel MB (2001) Calcium modulation of hypertension and obesity: mechanisms and implications. J Am College Nutr 20 (5 Suppl):428S–435S; discussion 440S–442S. https://doi.org/10.1080/07315724.2001.10719180
Ricci-Cabello I, Herrera MO, Artacho R (2012) Possible role of milk-derived bioactive peptides in the treatment and prevention of metabolic syndrome. Nutr Rev 70(4):241–255. https://doi.org/10.1111/j.1753-4887.2011.00448.x
Dugan CE, Fernandez ML (2014) Effects of dairy on metabolic syndrome parameters: a review. Yale J Biol Med 87(2):135–147
Nilsson M, Holst JJ, Bjorck IM (2007) Metabolic effects of amino acid mixtures and whey protein in healthy subjects: studies using glucose-equivalent drinks. Am J Clin Nutr 85(4):996–1004. https://doi.org/10.1093/ajcn/85.4.996
Phillips CM, Kesse-Guyot E, McManus R et al (2012) High dietary saturated fat intake accentuates obesity risk associated with the fat mass and obesity-associated gene in adults. J Nutr 142(5):824–831. https://doi.org/10.3945/jn.111.153460
Risch HA (2012) Pancreatic cancer: Helicobacter pylori colonization, N-nitrosamine exposures, and ABO blood group. Mol Carcinog 51(1):109–118. https://doi.org/10.1002/mc.20826
Ley SH, Sun Q, Willett WC et al (2014) Associations between red meat intake and biomarkers of inflammation and glucose metabolism in women. Am J Clin Nutr 99(2):352–360. https://doi.org/10.3945/ajcn.113.075663
Kim Y, Je Y (2018) Meat consumption and risk of metabolic syndrome: results from the Korean Population and a Meta-Analysis of Observational Studies. Nutrients 10(4):390. https://doi.org/10.3390/nu10040390
Ros E (2010) Health benefits of nut consumption. Nutrients 2(7):652–682. https://doi.org/10.3390/nu2070683
Acknowledgment
We express our appreciation to the participants of the study for their enthusiastic support and to the staff of the involved TLGS unit for their valuable help.
Funding
This work was funded by a Research Institute for Endocrine Sciences, Shahid Beheshti University of Medical Sciences, Tehran, Iran. The funders had no role in study design, data collection, and analysis, decision to publish, or preparation of the manuscript. There was no additional external funding received for this study.
Author information
Authors and Affiliations
Contributions
E.Y. conceptualized and designed the study, analyzed and interpreted the data, prepared the manuscript, and approved the final manuscript as submitted. G.A. and M.N designed the study and drafted the initial manuscript and approved the final manuscript as submitted. C.C. interpreted the data and critically revised the manuscript and approved the final manuscript as submitted. P.M. supervised the project, drafted the initial manuscript, and approved the final manuscript as submitted. F.A. supervised the project, critically revised the manuscript, and approved the final manuscript as submitted.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Ethics approval
This study was approved by the Ethics Committee of the Institute of Endocrinology and Metabolism of Shahid Beheshti University of Medical Sciences.
Additional information
Managed by Antonio Secchi.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yuzbashian, E., Nosrati-Oskouie, M., Asghari, G. et al. Associations of dairy intake with risk of incident metabolic syndrome in children and adolescents: Tehran Lipid and Glucose Study. Acta Diabetol 58, 447–457 (2021). https://doi.org/10.1007/s00592-020-01651-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-020-01651-0