Abstract
Positron emission tomography/computed tomography (PET/CT) is a standard procedure for imaging cancer commonly used in the clinical practice for several diseases, in particular for cancer staging, restaging, treatment monitoring and radiation therapy planning. Despite the availability of many radiotracers, 18F-fluoro-2-deoxy-2-d-glucose ([18F]FDG) is the most used. International PET/CT guidelines propose protocols for patients’ correct preparation before [18F]FDG injection, in particular with the regard of diabetic patients and therapy management. Hyperglycemic conditions and oral or insulin medication showed advantages and disadvantages on PET/CT scan accuracy: A correct knowledge of effects of these conditions on glucose metabolism assumes a fundamental role on patients management before [18F]FDG PET/CT scan.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction: mechanism of [18F]FDG uptake
Positron emission tomography/computed tomography (PET/CT) is a standard procedure commonly used in clinical practice for staging, restaging, treatment monitoring and radiation therapy planning in several types of cancer [1] and also has a recognized role in inflammatory infection disease, in neurologic disease, mostly for differential diagnosis for dementia, and in vitality cardiac studies.
Despite many radiotracers are available, according to different applications, 18F-fluoro-2-deoxy-2-d-glucose ([18F]FDG) is the most widespread, especially in oncology. The chemical structure of [18F]FDG is similar to that of natural glucose, differing in a carbon-2 atom labeled with [18F] [2], a radioisotope with an half-life of 109.8 min and a positron emission decay. [18F]FDG use in oncological imaging is based on the phenomenon of aerobic glycolysis named “Warburg effect” [3]. In the 1920s, Otto Warburg first described that cancer tissue presents accelerated glucose metabolism even in the presence of oxygen with an associated increase in lactate production [4]. Warburg hypothesized that the cause of increased lactate was a mitochondrial dysfunction; further research supported only in part this hypothesis: aerobic glycolysis, despite generally lead to a lower production of ATP molecules compared to mitochondrial oxidative phosphorylation; on the other hand, it is much faster, so, in the same amount of time, produces a higher number of ATP molecules [5].
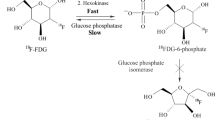
Glucose is generally transported by membrane-specific glucose transporters (GLUT) into the cytosol, where it is phosphorylated by the enzyme hexokinase to glucose 6-phosphate and subsequently metabolized to carbon dioxide and water. In the same way, [18F]FDG enters into the cell by means of GLUT and is phosphorylated by hexokinase to [18F]FDG 6-phosphate which, however, is not a good substrate for further enzyme action through the glycolytic chain and it is trapped into the cell [6]. [18F]FDG transport across the cell membrane is mediated by five different glucose transporters: GLUT-1 is ubiquitously expressed in the cell membrane of various tissues and it is up-regulated by several growth-factors [7]; GLUT-2 expression is regulated by glucose concentration and is mainly expressed by intestine, kidney, liver, pancreatic islets and brain [8]; GLUT-3 is expressed in neurons and ensures a glucose supply in the brain even in hypoglycemic conditions [9]; GLUT-4 is stimulated by insulin and it is expressed in skeletal and cardiac muscle and in brown and yellow adipose tissue [10]; GLUT-5 is expressed in the small intestine and is responsible for fructose transport [11].
The increased rate of glycolysis in cancer cells is associated both with increased GLUT and intracellular hexokinase expression [12], with the subsequently increased concentrations of [18F]FDG uptake into cancers cells that can vary substantially depending on the levels of glycolysis at one site.
Moreover, in tumoral cells, [18F]FDG 6-phosphate can be broken down by glucose 6-phosphatase, but this enzyme is markedly downregulated in cancer cells [6].
Furthermore, in 1992, Kubota et al. [13] firstly observed that FDG uptake in tumor tissue was related not only to metabolic activity of tumoral cells but also to peritumoral inflammatory cells. In 2001, Mochizuki et al. [14] investigated and compared the expression of GLUT subtypes between inflammatory lesions and malignant tumors in relation to [18F]FDG accumulation in rat models and showed that also inflammatory lesions presented a high GLUT-1 expression.
An interesting pictorial review published in 2015 by Vaidyanathan et al. [15] described the various mechanism of FDG uptake in inflammatory process according to three different phases of inflammation. The first phase, named “early vascular,” includes tissue hyperemia, enhanced vascular permeability and inflammatory mediator release that lead to higher tissue perfusion and subsequent higher [18F]FDG uptake; the second phase, named “acute cellular,” is characterized by a release of cytokines, with an up-regulation of GLUT-1 and GLUT-3 transporters and increase in both hexokinase activity and affinity of GLUT for its substrates. The last phase or “late cellular/healing” is characterized by the transition from acute to chronic inflammation cells with the persistence of shift toward cell glycolysis and escape the anabolic pathways.
For the above-mentioned mechanism, [18F]FDG PET/CT has found a wide field of application in inflammation and infection studies and it is now considered as a reliable tool in diagnosis, in particular in disease extent mapping, and follow-up of several inflammatory lesions such as fever of unknown origin, sarcoidosis, several musculoskeletal infections and prosthesis-related infections [16].
Physiologic biodistribution of [18F]FDG in human reflects GLUTs expression (Fig. 1): In euglycemic status, several organs and systems present a physiological [18F]FDG uptake, very intense in brain, moderate in liver and weak in skeletal muscle; [18F]FDG distribution is variable in the cardiac muscle because myocardial cells primarily use the beta-oxidation pathway of fatty acid, but following a glucose load may present a metabolic shift toward glucose metabolism; moreover, myocardial cells protect themselves from hypoxic state by means of the “glucose-fatty acid cycle” [17]. Finally, the uptake of [18F]FDG is high in the urinary system because of [18F]FDG physiological urinary excretion. In normal situation and euglycemic status, there is not a condition of glycosuria but in renal tubular cells, in addition to GLUTs, there is also the sodium-dependent glucose transporter (SGLT) that transport glucose against its concentration gradient and it is responsible for reuptake of glucose from the filtrate in the proximal tubules [18]. Due to the replacement of a hydroxy group in d-glucose with a [18F] atom [19], SGLT presents a lower affinity for [18F]-FDG compared to glucose and [18F]-FDG cannot be reabsorbed in the proximal tubules of the kidney, and so it is cumulated in the urine [20].
Physiologic biodistribution of [18F]FDG. In the fasting and euglycemic conditions, [18F]FDG uptake reflects GLUTs expression: [18F]FDG uptake is intense in brain, because of GLUT-3 expression that ensures correct glucose levels even in hypoglycemic conditions, weak in skeletal muscle for GLUT-4 expression stimulated by insulin, variable in cardiac muscle that primarily uses free fatty acids, but may present a metabolic shift to glucose metabolism after glucose load; intense in the urinary system because of [18F]FDG physiological urinary excretion
In neoplastic disease, [18F]FDG uptake is generally very high in more aggressive and proliferating tumor that present a high expression of GLUT and hexokinases but FDG avidity can differ among different histological subtype: For example, hematologic malignancies may present both high [18F]FDG uptake, such as Hodgkin Lymphoma, Diffuse Large B Cell Non-Hodgkin Lymphoma (DLBCL) or Follicular Lymphoma and low [18F]FDG uptake, such as Marginal Zone Lymphoma (ML) (Fig. 2). Also, lung tumors may have different [18F]FDG avidity: Typically, Non-Small Cell Lung Carcinoma (NSCLC) presents high [18F]FDG uptake, on the other hand tumors with neuroendocrine differentiation present low/absent [18F]FDG uptake (Fig. 3). Lastly, some well-differentiated tumors such as Hepatocellular Carcinoma (HCC) or Prostatic Carcinoma present a very low [18F]FDG avidity, using different metabolic pathway, such as membrane phospholipids turnover in cell membrane instead of glycolysis and should be studied specifically with other PET radiotracers.
[18F]FDG PET/CT scan in two patients with recent diagnosis of pulmonary nodule prior to histological diagnosis: left apical node of patient A presents very high [18F]FDG uptake compared to medium lobe node of patient B. Subsequent histological diagnosis revealed in case A a NSCLC instead in case B a typical carcinoid
Apart from tumoral histological characteristics [18F]FDG uptake may be influenced by, the presence of necrosis, that is responsible for low and heterogeneous uptake (Fig. 4), cells dedifferentiation (undifferentiated neuroendocrine tumors, for example, may present high [18F]FDG uptake), but also by the effect in the tissue of different therapies: If cytotoxic therapies such as chemotherapy or external radiotherapy may reduce [18F]FDG uptake in case of responder tumor, they may also cause flogistic reaction with increased [18F]FDG uptake at that site. For these reasons, especially in hematologic malignancies, an interval of at least 3 weeks from the last cycle of chemotherapy and 3 months from the end of radiotherapy is often required before performing [18F]FDG PET/CT scan to avoid false-positive findings.
Effects of hyperglycemia on [18F]FDG distribution and PET/CT scan interpretation
Several clinical conditions may affect [18F]FDG uptake, but the most important is hyperglycemia because it leads to a direct competition between plasmatic glucose and [18F]FDG uptake, both in normal and in cancer cells [21].
Several published studies described the effects of hyperglycemia and hyperinsulinemia on [18F]FDG biodistribution: Hyperglycemia may reduce the binding site of [18F]FDG for direct competition with plasmatic glucose [22]; on the other hand, the subsequent hyperinsulinemia causes up-regulation of GLUT-4, resulting in a higher skeletal and myocardial muscle [18F]FDG uptake [23]: This leads to lower uptake in cancer cells compared to physiological tissue leading to the risk of false-negative findings. Blood glucose levels may affect also the standardized uptake value (SUV), a semiquantitative parameter expressing FDG concentration in tissues [24]. SUV is the a-dimensional semiquantitative expression of the tracer uptake in a region of interest (ROI), for example, tumoral lesion, normalized by total amount of activity administered and body weight (body-weighted SUV − SUVbw) or body surface area (SUVbsa); it is used as SUVmax or mean or peak to compare [18F]FDG uptake on pre- and post-therapy scan and to define response to therapy [25]. Furthermore, SUVs of certain tissues, such as liver and mediastinal blood pool, are used as references to define disease and assess response to therapy in most lymphoproliferative disease [26] and also in inflammatory disease. Finally, SUV itself can be considered in certain diseases as a prognostic factor of tumor aggressiveness and worst prognosis.
In 2018, Sprinz et al. [27] investigated the effects of glycemia on [18F]FDG uptake in normal organs of interest (liver, brain and lung) in 5623 patients that underwent PET/CT scan for oncological indications and stratified them into four groups by serum glucose levels. All organs showed significant differences in mean SUVmax among different groups (p < 0.001); multivariate analysis adjusted for sex, age and BMI, confirmed significant differences only for the brain and the liver not for the lung.
A previous study published in 2013 by Büsing et al. [28] assessed the impact of chronically elevated blood glucose levels (BGL) on [18F]FDG tumor uptake and biodistribution in healthy organs in 90 patients with BGL ranging from 50 to 372 mg/dl [28] and found significant associations between BGL and both cerebral uptake reduction (p < 0.001) and muscle uptake increase (p < 0.001) and weak associations between BGL and liver uptake (p = 0.06), tumoral SUVmax (p = 0.133), fat, lung and spleen (p = 0.136–0.157).
A recent meta-analysis published in 2019 [29] assessed the effect of BGL in 20,807 individual on SUVmax and SUV mean measurements of 8380 patients. Patients were divided into five groups by BGL and SUVmax and SUVmean values of tumor, brain, muscle, liver and blood pool were recorded: Significant inverse correlations were found between BGL and SUVmax and SUVmean in brain (p < 0.001, p < 0.001) and muscle (p < 0.001, p < 0.001), while positive correlations were found between BGL and SUVmax and SUVmean in liver (p = 0.001, p = 0004) and blood pool (p = 0.008, p < 0.001). No significant correlation was found between BGL and SUVmax or SUVmean in tumors.
In multivariate analysis, SUVmax or SUVmean were considered dependent variables, and pre-scan BGL, sex, age, BMI, diagnosis of diabetes mellitus, injected dose of FDG, time interval between FDG injection and imaging were considered independent variables: All hyperglycemic groups compared with the euglycemic group presented brain and muscle SUVs significantly lower (p < 0.001 for both), while increased SUVmax and SUVmean in liver (p = 0.001, p = 0004) and blood pool (p = 0.008, p < 0.001) were recorded. Tumors presented significantly lower SUVmax only for BGL > 200 mg/dl: One explanation could be that tumoral cells overexpress GLUT in order to respond to the hypoxic condition subsequent to induced angiogenesis [29, 30] previously demonstrated in various cancer types [31, 32]; another could be the tumoral tissues loss of the control over glucose transportation and metabolism [31]: For these reasons, in tumoral tissue GLUT could be not saturated even in case of high BGL.
The reduction in brain [18F]FDG uptake in case of hyperglycemia is an issue in case of [18F]FDG PET/CT imaging of the brain routinely performed for dementing disorders (early and differential diagnosis), cerebral space-occupying lesions, epilepsy and differential diagnosis of movement disorders. EANM procedure guidelines [33] required BGL < 160 mg/dl in order to avoid stochastic noise increase and to preserve contrast between white and gray matter to maintain intact high diagnostic accuracy of the PET/CT scan.
Management of hyperglycemia and diabetic patients
Worldwide international societies and groups like the European Association of Nuclear Medicine (EANM) [34], the Society of Nuclear Medicine and Molecular Imaging (SNMMI) [35], the American College of Radiology (ACR) [36] and the National Cancer Institute (NCI) [37], proposed protocol guidelines to manage correct patient preparation for a [18F]FDG PET/CT scan, in order to avoid hyperglycemic status.
Non-diabetic patients should fast, and parenteral nutrition and intravenous fluids containing glucose should be discontinued for at least 4–6 h before [18F]FDG injection to prevent high insulinemia; BGL must be measured by a glucometer prior [18F]FDG administration [34,35,36,37]: For clinical studies, the upper BGL threshold accepted is 200 mg/dl, for research if ranges between 126 and 150 mg/dl [34]. In case of hyperglycemia, it is possible to lower BGL in patients by asking them to hydrate while ambulating until an acceptable level has been achieved [34].
Guidelines provide recommendations for patients with diabetic mellitus: In particular, European ones [34] are more specific for diabetes treated by oral medication and insulin.
For diabetic patients treated with oral medication such as metformin, they should fast for at least 4–6 h before [18F]-FDG injection, continuing to take oral medication to control blood sugar level and providing adequate hydration; PET/CT scan should preferably be performed in the late morning. Metformin decreases BGL by lowering gluconeogenesis, increasing insulin sensitivity, and enhancing glucose consumption by enterocytes [38]. Its known action on the bowel assumes a critical role in [18F]FDG physiological distribution: Metformin significantly increases [18F]FDG accumulation in the bowel, in particular in the colon [39] (Fig. 5a, b).
a [18F]FDG PET/CT scan performed for characterization of a hepatic lesion in a 41-year-old diabetic patient treated with metformin. High intestinal uptake is visible in particular in descending colon. b [18F]FDG PET/CT scan performed for suspected occult lesion in a 51-year-old diabetic patient treated with metformin and hepatic lesions suspected for metastases. High intestinal uptake is visible in particular in transverse and sigmoid colon
Massolo et al. [40] in 2013 verified this phenomenon in fifty-three mice submitted to dynamic acquisitions of [18F]FDG kinetics under fasting conditions over a 4-month study period and subdivided into four groups: untreated mice (group 1), mice exposed to metformin treatment for 48 h before each PET scan (group 2), mice treated for the whole study period (group 3) and mice in which treatment was interrupted 48 h before PET scan (group 4). They found that prolonged drug administration significantly increased bowel [18F]FDG uptake after a relatively long period of treatment and persisted after drug washout.
This increased [18F]FDG uptake has been also demonstrated in clinical practice.
A prospective study published by Gontier et al. [41] demonstrated an intense, diffuse and continuous pattern distribution of [18F]FDG along the bowel, strongly predominant in the colon, in patients treated with metformin. They enrolled fifty-five patients under oral medication for diabetes mellitus divided into two groups on the basis of anti-diabetic treatment (group 1a treated with metformin and group 1b in anti-diabetic treatment excluding metformin) and compared to control group (group 2, patients without diabetes mellitus). Patients treated with oral medication presented significantly increased [18F]FDG bowel uptake compared to controls (p < 0.001); [18F]FDG bowel uptake was significantly higher in group 1a compared to group 1b (p < 0.01).
The effect of metformin on [18F]FDG bowel uptake must kept on mind in case of PET/CT scan performed for abdominal evaluation, in particular in case of suspected peritoneal carcinomatosis, colic or gynecological neoplasm in which increased [18F]FDG bowel uptake could induce false-negative results [41]. No guidelines consensus is available on the oral anti-diabetic treatment management in case of abdominal evaluation, but several published studies on the timing of metformin discontinuation are available.
A retrospective study published in 2016 [42] aimed to assess the impact of metformin discontinuation up to 72 h in [18F]FDG bowel uptake. Two hundred and forty diabetic patients were divided into four groups on the basis of metformin discontinuation: < 24 h (group A), 24–48 h (group B), 48–72 h (group C) and no metformin (control group). Compared with the control group, [18F]FDG uptake increased significantly from the ileum to the rectosigmoid colon in group A (p < 0.001), from the transverse to the rectosigmoid colon in group B (p < 0.001) and from the descending colon to the rectosigmoid colon in group C (p < 0.001), highlighting a suboptimal metformin discontinuation for < 72 h for image interpretation, in particular for distal segments of the colon.
Conversely, a prospective study published in 2010 [43] concluded that discontinuation of metformin for 2 days is feasible to reduce high [18F]FDG bowel uptake. One hundred thirty-eight diabetic patients were divided into two groups: group A treated with metformin and group B in which the regimen did not include metformin and were compared to patients without diabetes mellitus (control group). Group A was divided into two subgroups on the basis of metformin discontinuation (group A1 continued metformin, group A2 stopped metformin treatment 2 days before PET/CT scan). Ten diabetic patients underwent two consecutive PET/CT scans before and after the discontinuation of metformin. Group A1 compared to group A2 and group B presented a significantly higher (p < 0.001) [18F]FDG bowel uptake. In 10 patients who underwent serial PET/CT scans, [18F]FDG bowel uptake decreased by 64% and hidden colorectal malignancies were revealed in two patients after the discontinuation of medication.
Despite no consensus is available on the timing metformin discontinuation, in case of [18F]FDG PET/CT scan for abdominal malignancies, a careful evaluation of patient’s drug treatment must be conducted, to ensure the optimal patient preparation in order to avoid false-negative result preventing the rise of BGL.
In case of treatment with insulin, European guidelines [34] suggest different options for scheduling [18F]FDG PET/CT scan on the basis of the treatment protocol. Commonly, insulin-dependent patients can be scheduled for PET/CT scan in late morning or midday, [18F]FDG should be injected no sooner than 4 h or 6 h after subcutaneous injection of rapid-acting and short-acting insulin, respectively, after having had breakfast in the early morning and subsequent fasting. For patients treated with intermediate-acting and/or long-acting insulin, [18F]FDG injection is not recommended on the same day of insulin administration and PET/CT scan should be scheduled in the early morning after insulin injection the evening before and night fasting. Particular attention needs long-acting insulin management that could have an interference with PET/CT scan, thus intermediate-acting replacement is mostly recommended. In case of continuous insulin infusion patients should be scheduled in the early morning and the insulin pump stopped for at least 4 h prior [18F]FDG injection.
The basis of this careful attention on insulin administration is the insulin’s affinity for GLUT-4, expressed in the skeletal and cardiac muscle, and in brown and yellow adipose cells [10]: Insulin causes the shift of GLUT-4 from intracellular location to the plasma membrane [44, 45] promoting both normal glucose and [18F]FDG intracellular uptake and resulting in altered radiotracer biodistribution and suboptimal image quality [39, 46]. The aforementioned study by Büsing et al. [28] also found out that diabetic and insulin patients compared to non-diabetics and non-insulin patients presented significant lower mean cerebral SUVmax (p < 0.001) and higher mean muscular SUVmax (p < 0.001).
An increase up to 50% was also observed in average fat tissue SUVmax and myocardial uptake in diabetic patients and insulin patients, respectively.
Despite this known effect of both endogenous and exogenous insulin on [18F]FDG biodistribution, several studies investigated the impact on image quality of insulin administration before [18F]FDG injection to correct hyperglycemia (Fig. 6).
In 2006, Turcotte et al. [47] assessed the impact of intravenous insulin 60 min before [18F]FDG injection on muscular, liver or lung [18F]FDG uptake. They compared 53 diabetic patients with BGL > 7.0 mmol/l versus 53 euglycemic non-diabetic patients and found no significant difference for the SUV calculated on the lung, liver, heart and skeletal muscles.
A study published in 2009 [48] aimed to assess the clinical impact of intravenous administration of short-acting insulin in 63 patients with glycemia greater than 10 mmol/L according to standardized protocol: two units for glycemia of 10.0–12.0 mmol/L, three units for glycemia of 12.1–14.0 mmol/L, and four–six units for glycemia of 14.1 mmol/L and above to reach a glycemia lower than 10.0 mmol/L. If glycemia after 30 min was still above 10.0 mmol/L, a second insulin dose was given. [18F]FDG was administered at least 60 min after the last insulin administration. After PET/CT scan patients were divided into two groups on the basis of [18F]FDG visual distribution: group A with adequate biodistribution (normal biodistribution, mild muscular uptake or muscular uptake involving more than one muscle group) and group B with altered biodistribution (diffuse muscular uptake of moderate intensity or diffuse, intense muscular uptake resulting in a non-diagnostic examination). [18F]FDG distribution was also semiquantitatively assessed by SUV mean of liver, gluteal muscles and myocardium. Group A vs group B presented a significantly longer time delay between insulin and [18F]FDG injections (p < 0.01), higher glycemia reduction after insulin injection (p < 0.01), higher hepatic mean SUV (p < 0.01) and lower gluteal muscular mean SUV (p < 0.01). This study concluded that an interval of at least 90 min between insulin and [18F]FDG administration should be considered and that hepatic and muscular SUVs could be useful tools to define adequate biodistribution of [18F]FDG.
In 2013, Caobelli et al. [49] evaluated the usefulness and impact on muscular [18F]FDG uptake of a protocol of intravenous insulin administration before [18F]FDG PET/CT scan in 130 diabetic patients. In 20 patients with BGL > 180 mg/dl endovenous insulin was administered 30 min earlier than 18F-FDG injection (group 1); in 10 patients with BGL > 160 mg/dl and < 200 mg/dl, no insulin was injected (group 2); 100 patients euglycemic were used as control group. Biodistribution was adequate in group 2 and control group and in 95% of patients in group 1. No significant differences were found between groups in gluteal muscle SUVmax (p = 0.20) and no false-negative result was recorded at 6-months follow-up evaluation.
In 2013, Song et al. [50] assessed the impact of intravenous ultra-short insulin administration 60 min before [18F]FDG injection in 105 diabetic patients: 52 patients with BGL > 190 mg/dl received 3–5 IU of insulin were compared to the remaining 53 with BGL < 190 mg/dl who did not receive insulin and no significant differences in image quality (p = 0.47), hepatic SUVmean (p = 0.13), gluteal muscle and brain uptake (p = 0.71 and p = 0.16, respectively) were found.
A study conducted by Garcia et al. [51] concluded that the quality of [18F]FDG PET/CT scan is not affected by subcutaneous administration of rapid-acting insulin if radiotracer is injected at least 4 h later. They enrolled 120 patients divided in four groups on the basis of insulin administration and delay to [18F]FDG injection: 30 diabetic patients with BGL < 160 mg/dl without further insulin administration (group 1), 30 diabetic patients with BGL ranging from 168 to 260 mg/dl in which subcutaneous rapid-acting insulin was administered and [18F]FDG was injected after a delay of 30–115 min waiting for BGL dropped below 160 mg/dl (group 2), 30 diabetic patients with BGL ranging from 192 to 324 mg/dl in which 18F-FDG was injected 4 h after subcutaneous rapid-acting insulin (group 3) and 30 non-diabetic patients with normal BGL (72–104 mg/dl) (control group). For each patient SUVmax of rectus femoris muscle was calculated: In group 2, SUVmax deviated without relation between BGL and [18F]FDG muscle uptake and the quality of PET-CT scan was suboptimal in 60% of patients in group 2, in 13% of patients in group 1, while was optimal in all patients of group 3.
Despite several study investigated protocols of intravenous administration of insulin before [18F]FDG administration in order to reduce BGLs, none has not yet been validated. EANM and SNMMI guidelines [34, 35] recommend, in case of necessity of insulin to correct hyperglycemia, an appropriate delay between [18F]FDG and insulin administration depending on the type and route of administration of insulin. Furthermore, EANM guidelines [34] both suggesting to avoid insulin administration unless this interval is less than 4 h and to prefer rapid-acting insulin subcutaneous injection (effective life 2–4 h), while short-acting, intermediate-acting or long-acting insulin are not recommended for their longer effective life (3–6 h, 12–18 h and 24 h, respectively).
Conclusions
The management of hyperglycemia in both diabetic and non-diabetic patients represents an issue in PET/CT practice, extensively described by the studies mentioned above. Both hyperglycemia and oral and insulin medications showed advantages and disadvantages on PET/CT scan accuracy: The respect of simple rules guarantees a good quality [18F]FDG PET/CT scan that sometimes assumes a pivotal role on patient clinical management.
References
Gallamini A, Zwarthoed C, Borra A (2014) Positron emission tomography in oncology. Cancers (Basel) 6:1821–1889
Ido T, Wan C, Casella J et al (1978) Labeled 2-deoxy-d-glucose analogs: 18F labeled 2-deoxy-2-fluoro-d-glucose, 2-deoxy-2-fluoro-d-mannose and 14C-2-deoxy-2-fluoro-d-glucose. J Label Compd Radiopharm 14:175–183
Warburg OP, Negelein E (1924) Uber den stoffwechsel der carcinomzelle. Biochem Z 152:309–335
Potter M, Newport E, Morten KJ (2016) The Warburg effect: 80 years on. Biochem Soc Trans 44:1499–1505
Chen Z, Liu M, Li L, Chen L (2017) Involvement of the Warburg effect in non-tumor diseases processes. J Cell Physiol 2017:2839–2849
Larson SM (2006) 18 F-FDG imaging: molecular or functional? J Nucl Med 47:31N–32N
Pauwels E, Sturm E, Bombardieri E, Cleton F, Stokkel M (2000) Positron-emission tomography with [18F]fluoro-deoxy glucose, part 1. Biochemical uptake mechanism and its implication for clinical studies. J Cancer Res Clin Oncol 126:549–559
Theorens B, Sarkar H, Kaback H, Lodish H (1988) Cloning and functional expression in bacteria of a novel glucose transporter present in liver, intestine, kidney, and beta-pancreatic islet cells. Cell 55:281–290
Maher F (1995) Immunolocalization of GLUT1 and GLUT3 glucose transporters inprimary cultured neurons and glia. J Neurosci Res 42:459–469
Rea S, James D (1997) Moving GLUT4: the biogenesis and trafficking of GLUT4 storage vesicles. Diabetes 46:1667–1677
Kayano T, Burant C, Fukumoto H, Gould G, Fan Y, Eddy R et al (1990) Human facilitative glucose transporters. Isolation, functional characterization, and gene localization of cDNAs encoding an isoform (GLUT5) expressed in small, and adipose tissue and an unusual glucose transporter pseudogene-like sequence (GLUT6). J Biol Chem 265:13276–13282
Brown R, Wahl R (1993) Over expression of glut-1 glucose transporter in human breast cancer: an immunohistochemical study. Cancer 72:2979–2985
Kubota R, Yamada S, Kubota K, Ishiwata K, Tamahashi N, Ido T (1992) Fluorodeoxyglucose in vivo: high accumulation in macrophages and granulation tissues studied by microautoradiography. J Nucl Med 33(11):1972–1980
Mochizuki T, Tsukamoto E, Kuge Y, Kanegae K, Zhao S, Hikosaka K et al (2001) FDG uptake and glucose transporter subtype expressions in experimental tumor and inflammation models. J Nucl Med 42(10):1551–1555
Vaidyanathan S, Patel CN, Scarsbrook AF, Chowdhury FU (2015) FDG PET/CT in infection and inflammation—current and emerging clinical applications. Clin Radiol 70(7):787–800. https://doi.org/10.1016/j.crad.2015.03.010
Jamar F, Buscombe J, Chiti A, Christian P, Delbeke D, Donohoe K et al (2013) EANM/SNMMI guideline for 18F-FDG use in inflammation and infection. J Nucl Med 54(4):647–658
Randle P, Garland P, Hales C, Newsholme E (1963) The glucose fatty-acid cycle. Its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. Lancet 1(7285):785–789
Szabo Z, Xia J, Mathews W, Brown P (2006) Future direction of renal PET. Semin Nucl Med 36(1):36–50
Moran JK, Lee HB, Blaufox MD (1999) Optimization of urinary FDG excretion during PET imaging. J Nucl Med 40(8):1352–1357
Qiao H, Bai J, Chen Y, Tian J (2007) Kidney modelling for FDG excretion with PET. Int J Biomed Imaging 2007:1–4
Rosica D, Cheng SC, Hudson M, Sakellis C, Van Den Abbeele AD, Kim CK et al (2018) Effects of hyperglycemia on fluorine-18-fluorodeoxyglucose biodistribution in a large oncology clinical practice. Nucl Med Commun 39(5):417–422
Wahl R, Henry C, Ethier S (1992) Serum glucose: effects on tumor and normal tissue accumulation of 2-[F-18]-fluoro-2-deoxy-d-glucose in rodents with mammary carcinoma. Radiology 183:643–647
Diederichs C, Staib L, Glatting G, Beger H, Reske S (1998) FDG PET: elevated plasma glucose reduces both uptake and detection rate of pancreatic malignancies. J Nucl Med 39:1030–1033
Juweid ME, Cheson BD (2006) Positron-emission tomography and assessment of cancer therapy. N Engl J Med 354:496–507
Wahl RL, Jacene H, Kasamon Y, Lodge MA (2009) From RECIST to PERCIST: evolving considerations for PET response criteria in solid tumors. J Nucl Med 50(Suppl_1):122S–150S
Barrington SF, Kluge R (2017) FDG PET for therapy monitoring in Hodgkin and non-Hodgkin lymphomas. Eur J Nucl Med Mol Imaging 44(Suppl 1):S97–S110
Sprinz C, Zanon M, Altmayer S, Watte G, Irion K, Marchiori E et al (2018) Effects of blood glucose level on 18F fluorodeoxyglucose (18F-FDG) uptake for PET/CT in normal organs: an analysis on 5623 patients. Sci Rep 8(1):6–11
Büsing KA, Schönberg SO, Brade J, Wasser K (2013) Impact of blood glucose, diabetes, insulin, and obesity on standardized uptake values in tumors and healthy organs on 18F-FDG PET/CT. Nucl Med Biol 40(2):206–213. https://doi.org/10.1016/j.nucmedbio.2012.10.014
Eskian M, Alavi A, Khorasanizadeh M, Viglianti BL, Jacobsson H (2019) Effect of blood glucose level on standardized uptake value (SUV) in 18 F-FDG PET-scan: a systematic review and meta-analysis of 20, 807 individual SUV measurements. Eur J Nucl Med Mol Imaging 46:224–237
Yang J, Wen J, Tian T, Lu Z, Wang Y, Wang Z et al (2017) GLUT-1 overexpression as an unfavorable prognostic biomarker in patients with colorectal cancer. Oncotarget 8(7):11788–11796
Macheda ML, Rogers S, Best JD (2005) Molecular and cellular regulation of glucose transporter (GLUT) proteins in cancer. J Cell Physiol 202(3):654–662
Carvalho K, Cunha I, Rocha R, Ayala F, Cajaiba M, Begnami M et al (2011) GLUT1 expression in malignant tumors and its use as an immunodiagnostic marker. Clinics (Sao Paulo) 66:965–972
Varrone A, Asenbaum S, Vander Borght T, Booij J, Nobili F, Någren K et al (2009) EANM procedure guidelines for PET brain imaging using [18F]FDG, version 2. Eur J Nucl Med Mol Imaging 36(12):2103–2110
Boellaard R, Delgado-Bolton R, Oyen WJG, Giammarile F, Tatsch K, Eschner W et al (2014) FDG PET/CT: EANM procedure guidelines for tumour imaging: version 2.0. Eur J Nucl Med Mol Imaging 42(2):328–354
Delbeke D, Coleman RE, Guiberteau MJ, Brown ML, Royal HD, Siegel BA et al (2006) Procedure guideline for tumor imaging with 18F-FDG PET/CT 1.0. J Nucl Med 47(5):885–895
American College of Radiology (2016) ACR–SPR practice guideline for performing FDG-PET/CT in oncology. https://www.acr.org/-/media/ACR/Files/Practice-Parameters/FDG-PET-CT.pdf
Shankar L, Hoffman J, Bacharach S et al (2006) Consensus recommendations for the use of 18F-FDG PET as an indicator of therapeutic response in patients in National Cancer Institute Trials. J Nucl Med 47:1059–1066
Rena G, Hardie DG, Pearson ER (2017) The mechanisms of action of metformin. Diabetologia 60(9):1577–1585
Martin J, Saleem N (2014) 18F-FDG PET-CT scanning and diabetic patients: what to do? Nucl Med Commun 35(12):1197–1203
Massollo M, Marini C, Brignone M, Emionite L, Salani B, Riondato M et al (2013) Metformin temporal and localized effects on gut glucose metabolism assessed using 18F-FDG PET in mice. J Nucl Med 54(2):259–266. https://doi.org/10.2967/jnumed.112.106666
Gontier E, Fourme E, Wartski M, Blondet C, Bonardel G, Le Stanc E et al (2008) High and typical 18F-FDG bowel uptake in patients treated with metformin. Eur J Nucl Med Mol Imaging 35(1):95–99
Lee SH, Jin S, Lee HS, Ryu JS, Lee JJ (2016) Metformin discontinuation less than 72 h is suboptimal for F-18 FDG PET/CT interpretation of the bowel. Ann Nucl Med 30(9):629–636
Oh JR, Song HC, Chong A, Ha JM, Jeong SY, Min JJ et al (2010) Impact of medication discontinuation on increased intestinal FDG accumulation in diabetic patients treated with metformin. Am J Roentgenol 195(6):1404–1410
Huang S, Czech MP (2007) The GLUT4 glucose transporter. Cell Metab 5(4):237–252
Bryant NJ, Govers R, James DE (2002) Regulated transport of the glucose transporter GLUT4. Nat Rev Mol Cell Biol 3(4):267–277
Surasi DS, Bhambhvani P, Baldwin JA, Almodovar SE, O’Malley JP (2014) 18F-FDG PET and PET/CT patient preparation: a review of the literature. J Nucl Med Technol 42(1):5–13. https://doi.org/10.2967/jnmt.113.132621
Turcotte E, Leblanc M, Carpentier A, Bénard F (2006) Optimization of whole-body positron emission tomography imaging by using delayed 2-deoxy-2-[F-18]fluoro-d-glucose injection following I. V. insulin in diabetic patients. Mol Imaging Biol 8(6):348–354
Roy F-N, Beaulieu S, Boucher L, Bourdeau I, Cohade C (2009) Impact of intravenous insulin on 18F-FDG PET in diabetic cancer patients. J Nucl Med 50(2):178–183. https://doi.org/10.2967/jnumed.108.056283
Caobelli F, Pizzocaro C, Paghera B, Guerra UP (2013) Proposal for an optimized protocol for intravenous administration of insulin in diabetic patients undergoing 18F-FDG PET/CT. Nucl Med Commun 34(3):271–275
Song HS, Yoon JK, Lee SJ, Yoon SH, Jo KS, An YS (2013) Ultrashort-acting insulin may improve on18F-FDG PET/CT image quality in patients with uncontrolled diabetic mellitus. Nucl Med Commun 34(6):527–532
Garcia JR, Sanchis A, Juan J, Tomas J, Domenech A, Soler M et al (2014) Influence of subcutaneous administration of rapid-acting insulin in the quality of 18F-FDG PET/CT studies. Nucl Med Commun 35(5):459–465
Acknowledgements
We thank Dr. Francesca Paola Giunta Md for assistance with the English language and for comments that greatly improved the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement of human and animal rights
This article does not contain research involving human or animal subjects.
Informed consent
For this type of study formal consent is not required.
Additional information
Managed by Massimo Federici.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Finessi, M., Bisi, G. & Deandreis, D. Hyperglycemia and 18F-FDG PET/CT, issues and problem solving: a literature review. Acta Diabetol 57, 253–262 (2020). https://doi.org/10.1007/s00592-019-01385-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00592-019-01385-8