Abstract
Purpose
Anterior (ALIF) and transforaminal (TLIF) lumbar interbody fusion have shown similar clinical outcomes at short- and medium-term follow-ups. Possible advantages of ALIF in the long run could be better disc height and lumbar lordosis and reduced risk of adjacent segment disease. We aimed to study if ALIF could be associated with superior clinical outcomes than TLIF at long-term follow-up.
Methods
We analysed 535 patients treated with ALIF or TLIF of the L5-S1 spinal segment between 2007 and 2017 who completed long-term follow-up in a national spine registry database (NORspine). We defined treatment success after surgery as at least 30% improvement in Oswestry Disability Index (ODI) at long-term follow-up. Patients treated with ALIF and TLIF and who responded at long term were balanced by propensity score matching. The proportions of successfully treated patients within each group were compared by numbers and percentages with corresponding relative risk.
Results
The mean (95%CI) age of the total study population was 50 (49–51) years, and 264 (49%) were females. The mean (95%CI) preoperative ODI score was 40 (39–42), and 174 (33%) had previous spine surgery. Propensity score matching left 120 patients in each treatment group. At a median (95%CI) of 92 (88–97) months after surgery, we found no difference in proportions successfully treated patients with ALIF versus TLIF (68 (58%) versus 77 (65%), RR (95%CI) = 0.88 (0.72 to1.08); p = 0.237).
Conclusions
This propensity score-matched national spine register study of patients treated with ALIF versus TLIF of the lumbosacral junction found no differences in proportions of successfully treated patients at long-term follow-up.
Level of Evidence I
Diagnostic: individual cross-sectional studies with the consistently applied reference standard and blinding.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Disabling chronic low back pain, with or without leg pain, associated with degenerative disc disease (DDD) is the leading cause of global disability, and the lumbosacral junction is commonly affected [1, 2]. Spinal fusion is a widely used surgical treatment option for patients with DDD that do not respond to non-operative care. The surgery aims to fuse the supposedly painful segment of the spine by a bony union between the involved vertebras.
Two principally different surgical approaches exist—anterior and transforaminal lumbar interbody fusion (ALIF and TLIF, respectively) [3]. ALIF is done through the retroperitoneal space (sometimes transabdominally), while TLIF involves a traditional dorsal approach. ALIF is commonly recommended to avoid surgical trauma to the back muscles and for restoration of the physiological lumbosacral height and lordosis and is reported to lead to less adjacent segment disease [4,5,6,7,8,9,10,11]. However, recent reviews based on cohort studies have shown similar clinical outcomes after ALIF and TLIF, and one systematic review calls explicitly for prospectively multicenter register studies to assess any differences between the two surgical fusion procedures [4, 7, 9, 10].
Planning this observational study from the Norwegian Registry for Spine Surgery (NORSpine), we hypothesized that, in a real-world daily clinical practice, the clinical effectiveness of ALIF could be superior to TLIF at long-term follow-up.
Methods
We hypothesized that long-term clinical effectiveness of ALIF was superior to TLIF.
Currently, 40 Norwegian hospitals report to NORspine—a mandatory national spine register. The coverage and the one-year response rate approximate 70% and 73%, respectively [12]. A NORspine dataset consists of informed consent, patient-reported outcome measures (PROMs) and socioeconomic variables, and surgeon-reported diagnostics and surgical details. Patients report clinical status at baseline during hospital admission and clinical outcome at 3 and 12 months after surgery directly to NORspine, including any postoperative complications (at 3 months). The quality of NORspine data has been assessed and found acceptable for most variables [13]. NORspine has evolved, and questionnaires have been amended somewhat during the study period. Finally, NORspine does not include radiological images.
We screened NORspine patients who received a surgical fusion of the lumbosacral junction during eleven years (1st January 2007–31st December 2017) and included those treated by ALIF or TLIF; we only included patients with complete datasets in the final analysis of clinical outcomes. NORspine defines ALIF as fusion surgery performed by an anterior approach, with or without additional posterior fixation. We defined TLIF as transforaminal approach to the disc and posterior midline or paramedian (Wiltze’s) incisions. We did not include patients operated on with posterior lumbar interbody fusions (PLIF) and posterolateral fusions (PLFs). Different types of autologous or allogenic bone transplants, and in a few cases commercially available grafts, were used for both ALIF and TLIF.
To obtain long-term PROMs, we reached out to patients by mail and asked them to respond to the same questionnaires that they did at one-year-follow-up form. The following PROMs were collected: (1) Oswestry Disability Index (ODI), a continuous variable from 0 (no disability) to 100 (bed bound) [14, 15]. (2) Numeric Rating Scale (NRS) for back pain and leg pain; a continuous variable from 0 (no pain) to 10 for worst imaginable pain [16]. (3) Quality of life assessed by EuroQol 5 Dimensions (EQ-5D) index, ranging from − 0.6 (“worse than dead”) to 1.00 (“full health”) [17]. (4) Patient-perceived effect of treatment as assessed by Global Perceived Effect (GPE), a categorical transitional health scale from 1 to 7 (1 = “completely recovered”, 2 = “much improved”, 3 = ”somewhat improved”, 4 = ”unchanged”, 5 = “somewhat worse”, 6 = ”much worse”, 7 = ”worse than ever”) [18].
Outcomes
The primary outcome was a clinically relevant treatment effect, as defined by minimum 30% improvement in ODI at long-time follow-up [19].
Secondary outcomes were treatment success defined by GPE categories “much improved” or “completely recovered”, mean differences in ODI, and NRS back and leg pain scores at long-term follow-up. We also analysed any between-group differences in working status; complications (perioperative (surgeon-recorded at hospital stay) and postoperative (patient-recorded at 3 months); and re-operations (patient-recorded at long-term follow-up); mean length of hospital stay; and mean surgical time of the index surgery.
Statistics
We described patient characteristics for those who responded to our long-term follow-up survey versus those who did not (respondents vs. non-respondents). We evaluated differences between patients that received ALIF versus those that underwent TLIF. We performed an unadjusted comparative analysis of the aforementioned primary and secondary outcomes for the two treatment arms.
In order to account for any imbalances between the groups at baseline, we matched the groups by propensity score. The propensity score, derived from a logistic regression model, was defined as a patient’s baseline probability for receiving ALIF, conditioned on pre-specified plausible confounders (age, gender, smoking, BMI, working status, civil status, higher education, ASA classification, and previous spine surgery; preoperative symptoms as assessed by ODI, NRS back pain, NRS leg pain, EQ-5D, duration of back pain > 12 months, and radiological characteristics) [20]. We used the technique of 1:1 matching without replacement. ALIF patients were matched with TLIF patients if the difference in propensity scores was less than 0.2 of the logit of the standard deviation [21, 22].
Finally, we compared the matched groups regarding primary and secondary outcomes and performed post-hoc power analyses using the new population size after propensity score matching. The null hypothesis was that ALIF could be superior TLIF in the long term. We set the superiority margin at a 15 per cent higher proportion of patients reaching the primary outcome (at least 30% improvement in ODI). With the assumption of missing not at random, we only included patients who completed the long-term cross-sectional survey [23, 24].
Continuous variables were displayed as means and 95% CIs and categorical variables as numbers and proportions (%). We compared means using the Student t test and proportions using Z-statistics. We used SPSS version 26 (IBM Corp., Armonk, NY, USA) and Med calc (MedCalc Statistical Software version 19.2.6 (MedCalc Software bv, Ostend, Belgium; https://www.medcalc.org; 2020). We used ClinCalc.com to perform the power analysis (clincalc.org/Stats/power.aspx). The Norwegian Regional Committee for Medical and Health Research Ethics approved the study (identifier 75294). The study was conducted and presented according to the STROBE consortium [25].
Results
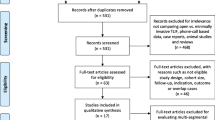
We identified 945 patients at baseline: 43 were dead, 29 had no valid postal address, and eight were duplicates, leaving 865 patients eligible for the study (Fig. 1). A total of 535 (62%) patients responded to the cross-sectional long-term questionnaire at a median of 8 years after index surgery. Among long-term respondents, 159 (30%) received ALIF and 376 (70%) TLIF. After propensity score matching, 120 patients remained in each treatment group. The mean follow-up time (95%CI) was 92.3 (87.6–97.0) months in the matched cohorts.
Table 1 displays baseline characteristics and indications for surgery for the patients who responded to the long-term follow-up and the non-respondents. The responders of this study had a mean age (95%CI) of 50 (49–51) years, 264 (49%) were females, and the mean (95%CI) BMI was 27.1 (26.8–27.5). At baseline, the mean (95%CI) ODI score was 40.4 (39.1–41.5), and 174 (33%) had previous spine surgery. Fewer respondents lived alone, smoked and reported any previous spine surgery than non-responders. Respondents were more often highly educated.
Unmatched cohorts
Baseline data and indications for surgery for patients that received ALIF compared to TLIF are displayed in Table 2. ALIF patients were more often living alone (RR = 0.66 (0.43–1.00); p = 0.050) and had a lower BMI (MD = 1.1 (0.3–1.8); p = 0.003) compared to TLIF patients. ALIF patients reported a lower preoperative ODI (MD = 4.2 (1.7–6.8); p < 0.001), NRS leg pain (MD = 1.15 (0.67–1.63); p < 0.001), and NRS back pain (MD = 0.59 (0.24–0.93); p < 0.001) than TLIF patients. Furthermore, fewer ALIF patients were operated due to disc herniation, lateral spinal stenosis, and degenerative spondylolisthesis (RRs 0.08–0.38) than those who received TLIF.
Table 3 shows the clinical outcomes for the unmatched cohort. We found no differences in the primary outcome or any other secondary clinical outcomes. Compared to TLIF, ALIF patients had a shorter duration of surgery (MD = 47 min (37–56); p < 0.001) and shorter hospital stays (MD = 0.9 days (0.3–1.4); p < 0.001). There were no differences in rates of perioperative (surgeon-recorded) or postoperative (patient-reported at 3 months) complications or re-operations (patient-reported at long-term follow-up).
Propensity score–matched cohort
Table 2 and Fig. 2 demonstrate that propensity score matching created similar groups concerning the observed baseline parameter distribution.
Results for matched cohorts are displayed in Table 4. The proportions of patients with an improvement in the ODI of at least 30% were 68/120 (57.6%) in the ALIF group versus 77/120 (64.7%) in the TLIF group; the difference was not statistically significant (RR (95%CI) = 0.88 (0.72 to 1.08); p = 0.237). Similarly, we found no statistically significant differences in the secondary clinical outcomes between the two treatments. However, ALIF patients had shorter operation time (109 (102–116) min vs. 150 (141–158) min; MD = 40 min (29–51); p < 0.001). The between-group difference in length of stay was no longer significant after matching (MD = 0.6 days ( − 0.2–1.4); p = 0.077).
We found no significant differences in the numbers of complications, neither perioperative (surgeon-recorded) nor postoperative (patient-recorded at 3 months) nor re-operation rates (patient-reported at long-term follow-up).
Most (117 of 120 (97.5%)) ALIFs were stand-alone variants, i.e. without additional posterior fixation. We performed an exploratory analysis of 84 patients with isthmic spondylolisthesis who received stand-alone ALIF (n = 38) versus those who underwent TLIF (n = 46) and found no significant between-group difference in number of patients who reported at least 30% ODI improvement (Isthmic stand-alone ALIF 24 of 37(65%) vs. Isthmic TLIF 36 of 46 (78%), RR (95% CI) = 0.90 (0.60–1.33); p = 0.588).
We performed a post hoc power analysis and found that our study achieved a statistical power of 89% (unmatched cohorts) and 65% (propensity score-matched cohorts) to detect a 15 percentage points difference in proportions of patients reaching at least 30% ODI improvement after fusion surgery.
Discussion
This national register-based study compared long-term results after ALIF versus TLIF of the lumbosacral junction. We used prospectively collected register data, supplemented by a long-term cross-sectional survey, and matched groups at baseline by propensity score. At an extended follow-up of 8 years after surgery, we found no differences in the proportions of patients with a clinically relevant improvement assessed by the ODI (primary outcome). Except for shorter operation time, secondary outcomes did not indicate support that ALIF could be superior to TLIF.
To our knowledge, no other studies have compared ALIF versus TLIF with a comparable long-term follow-up. Previously published meta-analyses included studies with short- or middle-term follow-ups (10–71 months) and found no differences in clinical outcomes comparing ALIF versus TLIF, supporting our findings [4, 9].
Loss of sagittal balance and lumbar lordosis have been associated with back pain and disability and adjacent segment disease (ASD) [26,27,28,29,30,31]. One study hypothesized that ALIF could be superior to posterior fusion in restoring a more physiological lumbar lordosis which in turn could yield better patient-reported outcomes [11]. Additionally, superior fusion rates after ALIF versus TLIF have been suggested because the anterior approach provides a wider access to debride the disc space and allows larger implants and bone transplant volumes than posterior techniques [4,5,6,7,8]. However, a recent systematic review found similar fusion rates comparing ALIF versus TLIF [32]. Although surgeons report preoperative radiological findings, NORspine does not store any radiological images. More importantly, fusion rates and adjacent segment disease should optimally be assessed in the long term, whereas the standard follow-up time in NORspine is one year after surgery. Most comparisons of ALIF versus TLIF are short-term studies and might not detect clinically relevant long-term differences in fusion rates or ASD [8, 10, 33,34,35,36]. Finally, results from our study suggest that previously published differences in radiological lumbar lordosis after ALIF versus TLIF may not be relevant for long-term patient-reported outcomes.
In line with previously published data, we found a shorter surgical time for ALIF than for TLIF [37]. However, one study reported longer surgical time for ALIF in patients who required additional pedicle screws (360-degree fusion procedure). Our finding of shorter surgical time for ALIF versus TLIF could rely on the finding that almost all ALIFs were stand-alone procedures without additional pedicle screws. Still, surgical time is relevant because the cumulative surgical trauma, risk of hypothermia, infection, and postoperative complications partly rely on the duration of surgery. Additionally, the surgical time has an economic aspect.
Different indications for surgery could also influence patient-reported outcomes, irrespective of the fusion procedure. A recent NORspine study showed superior results reported by patients who received a fusion due to isthmic spondylolisthesis [38]. In the present study, we found no difference in the proportions of patients with isthmic spondylolisthesis that reported a successful effect of lumbosacral fusion by ALIF stand-alone versus TLIF. However, this subgroup sample was too small to be statistically robust.
Finally, in the unadjusted results, the length of stay was about one day shorter for ALIF patients. However, the difference was not statistically significant (p = 0.077) after matching. Previously published data do not support this finding: Phan et al. found longer lengths of stay for ALIF patients in the meta-analysis [4]. Our data are based on operations done between 2007 and 2017; less invasive surgery could have developed during the study period, i.e. unilateral pedicle screws in TLIF procedures have become frequent [39]. Length of stay has an impact on patients' return to everyday living and on health economics.
Although a trend against less perioperative and postoperative complications in ALIF patients, the difference was not statistically significant. Re-operation rates were almost similar. These findings align well with previously published data: Teng et al. reported no differences in complications between the two treatments [9]. The complication profiles are, however, reported to be different between the two techniques [4, 39,40,41,42,43]. Also, the recording of perioperative complications in spine registers may be underestimated [13, 43, 44]. Re-operations were reported by patients themselves at the long-term follow-up and could be inaccurately recorded and subject to recall bias.
Limitations
Most importantly, this study was based on register data and had an observational design. There was no randomization between the two treatments; however, we did balance the groups by propensity score matching. Still, unobserved variables not included in the propensity score could introduce some allocation bias. In addition to the differences between the comparative treatments, the two cohorts might differ regarding additional procedures and indication. ALIF is a pure fusion procedure, while TLIF necessitates nerve root decompression in addition to the fusion procedure.
The loss to follow-up at 8-year follow-up was 38% and could have introduced attrition bias; however, the proportion of patients lost was within the acceptable limit of 20–40% for registers and comparable to other spine register studies [23, 24, 45,46,47]. This study used register data to include a large patient pool; still, post-hoc power analysis showed only 65% power to detect a relevant between-group difference for the primary outcome in the propensity score-matched cohorts. Additionally, we believe this cohort is too small to assess rare events such as complications and re-operation. Increasing the study population would only be possible by decreasing the follow-up time significantly, as we would have to include more recently operated patients.
NORspine does not include postoperative radiological parameters, and neither fusion rates nor lordosis could be assessed radiologically. Finally, and crucial to recognize, the results of our study apply to a heterogeneous population of patients with a variety of degenerative lumbosacral spine conditions who underwent variants of ALIF and TLIF of the L5-S1 segment.
Conclusions
In this propensity score-matched prospective national spine register study, the type of procedure—anterior (ALIF) or transforaminal (TLIF) lumbar interbody fusion of the lumbosacral junction—was not associated with the long-term outcomes reported by the patients. The ALIF procedure was associated with somewhat shorter surgical time than TLIF.
References
Muraki S, Oka H, Akune T, Mabuchi A, En-Yo Y, Yoshida M, Saika A, Suzuki T, Yoshida H, Ishibashi H, Yamamoto S, Nakamura K, Kawaguchi H, Yoshimura N (2009) Prevalence of radiographic lumbar spondylosis and its association with low back pain in elderly subjects of population-based cohorts: the ROAD study. Ann Rheum Dis 68(9):1401–1406. https://doi.org/10.1136/ard.2007.087296
Clark S, Horton R (2018) Low back pain: a major global challenge. The Lancet 391(10137):2302–2302
Mobbs RJ, Phan K, Malham G, Seex K, Rao PJ (2015) Lumbar interbody fusion: techniques, indications and comparison of interbody fusion options including PLIF, TLIF, MI-TLIF, OLIF/ATP LLIF and ALIF. J Spine Surg 1(1):2–18. https://doi.org/10.3978/j.issn.2414-469X.2015.10.05
Phan K, Thayaparan GK, Mobbs RJ (2015) Anterior lumbar interbody fusion versus transforaminal lumbar interbody fusion–systematic review and meta-analysis. Br J Neurosurg 29(5):705–711. https://doi.org/10.3109/02688697.2015.1036838
Rao PJ, Maharaj MM, Phan K, Abeygunasekara ML, Mobbs RJ (2015) Indirect foraminal decompression after anteriorlumbar interbody fusion: a prospective radiographic study using a new pedicle-to-pedicle technique. Spine J 15(5):817–824
Hsieh PC, Koski TR, O’Shaughnessy BA, Sugrue P, Salehi S, Ondra S, Liu JC (2007) Anterior lumbar interbody fusion in comparison with transforaminal lumbar interbody fusion: implications for the restoration of foraminal height, local disc angle, lumbar lordosis, and sagittal balance. J Neurosurg Spine SPI 7(4):379–386. https://doi.org/10.3171/SPI-07/10/379
Divi SN, Schroeder GD, Goyal DK, Radcliff KE, Galetta MS, Hilibrand AS, Anderson DG, Kurd MF, Rihn JA, Kaye ID, Woods BR, Vaccaro AR, Kepler CK (2019) Fusion technique does not affect short-term patient-reported outcomes for lumbar degenerative disease. Spine J 19(12):1960–1968
Kim JS, Kang BU, Lee SH, Jung B, Choi YG, Jeon SH, Lee HY (2009) Mini-transforaminal lumbar interbody fusion versus anterior lumbar interbody fusion augmented by percutaneous pedicle screw fixation: a comparison of surgical outcomes in adult low-grade isthmic spondylolisthesis. J Spinal Disord Techn 22(2):114–121. https://doi.org/10.1097/BSD.0b013e318169bff5
Teng I, Han J, Phan K, Mobbs R (2017) A meta-analysis comparing ALIF PLIF, TLIF and LLIF. J Clin Neurosci 44:11–17. https://doi.org/10.1016/j.jocn.2017.06.013
Thompsom P, Adogwa O, Knott KT, Toche U, Huang K, Cheng JS, Isaacs RE (2013) Outcomes after ALIF versus TLIF for treatment of symptomatic L5–S1 Spondylolisthesis: a prospective, multi-institutional comparative effectiveness study. Spine J 13:s159. https://doi.org/10.1016/j.spinee.2013.07.402
Min J, Jang J, Lee S (2007) Comparison of anterior- and posterior-approach instrumented lumbar interbody fusion for spondylolisthesis. J Neurosurg Spine SPI 2007 7(1):21–26. https://doi.org/10.3171/SPI-07/07/021
Norwegian registry for Spine Surgery (NORspine) Annual report 2017 Tore K Solberg, Lena Ringstad olsen Universtitetssykehuset I Nord Norge (UNN) 2SKDE (2018) https://www.kvalitetsregistre.no/sites/default/files/30_arsrapport_2017_ryggkirurgi_1.pdf, Accessed 17 July 2022
Alhaug OK, Kaur S, Dolatowski F, Småstuen MC, Solberg TK, Lønne G (2022) Accuracy and agreement of national spine register data for 474 patients compared to corresponding electronic patient records. Eur Spine J 31(3):801–811. https://doi.org/10.1007/s00586-021-07093-8
Fairbank JC, Pynsent PB (2000) The oswestry disability index. Spine. 2940–2952
Grotle M, Brox JI, Vøllestad NK (2003) Cross-cultural adaption of the Norwegian versions of the roland-morris disability questionnaire and the oswestry disability index. J Rehabil Med 35:241–247
McCaffery M (1989) Pain, clinical manual for nursing practice. Mosby
EuroQol Group (1990) EuroQol–a new facility for the measurement of health-related quality of life. Health Policy 16(3):199–208. https://doi.org/10.1016/0168-8510(90)90421-9
Kamper SJ, Ostelo RWJG, Knol DL, Maher CG, de Vet HCW, Hancock MJ (2010) Global Perceived Effect scales provided reliable assessments of health transition in people with musculoskeletal disorders, but ratings are strongly influenced by current status. J Clin Epidemiol 63(7):760–766. https://doi.org/10.1016/j.jclinepi.2009.09.009
Ostelo RW, Deyo RA, Stratford P, Waddell G, Croft P, Von Kroff M et al (2008) Interpreting change scores for pain and functional status in low back pain: towards international consensus regarding minimal important change. Spine (Phila Pa 1976) 33:90–94
Aalto TJ, Malmivaara A, Kovacs F, Herno A, Alen M, Salmi L, Kröger H, Andrade J, Jiménez R, Tapaninaho A, Turunen V, Savolainen S, Airaksinen O (2006) Preoperative predictors for postoperative clinical outcome in lumbar spinal stenosis: systematic review. Spine (Phila Pa 1976) 31(18):E648–E663. https://doi.org/10.1097/01.brs.0000231727.88477.da
Austin PC (2011) An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivar Behav Res 46(3):399–424. https://doi.org/10.1080/00273171.2011.568786
Wang Y, Cai H, Li C, Jiang Z, Wang L, Song J, Xia J (2013) Optimal caliper width for propensity score matching of three treatment groups: a Monte Carlo study. PLoS ONE 8(12):e81045. https://doi.org/10.1371/journal.pone.0081045
Solberg TK, Sørlie A, Sjaavik K, Nygaard ØP, Ingebrigtsen T (2011) Would loss to follow-up bias the outcome evaluation of patients operated for degenerative disorders of the lumbar spine? Acta Orthop 82(1):56–63. https://doi.org/10.3109/17453674.2010.548024
Kaur S, Alhaug OK, Dolatowski FC et al (2023) Characteristics and outcomes of patients who did not respond to a national spine surgery registry. BMC Musculoskelet Disord 24:164. https://doi.org/10.1186/s12891-023-06267-3
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP (2007) The strengthening the reporting of observational studies in epidemiology (STROBE) Statement: guidelines for reporting observational studies. Ann Intern Med 147(8):573–577 (PMID: 17938396)
Kim MK, Lee SH, Kim ES et al (2011) The impact of sagittal balance on clinical results after posterior interbody fusion for patients with degenerative spondylolisthesis: a pilot study. BMC Musculoskelet Disord 12:69. https://doi.org/10.1186/1471-2474-12-69
Yilgor C, Sogunmez N, Yavuz Y, Abul K, Boissiére L, Haddad S, Obeid I, Kleinstück F, Sánchez Pérez-Grueso FJ, Acaroğlu E, Mannion AF, Pellise F, Alanay A, European Spine Study Group (2017) Relative lumbar lordosis and lordosis distribution index: individualized pelvic incidence-based proportional parameters that quantify lumbar lordosis more precisely than the concept of pelvic incidence minus lumbar lordosis. Neurosurg Focus 43(6):E5. https://doi.org/10.3171/2017.8.FOCUS17498
Bari TJ, Heegaard M, Bech-Azeddine R, Dahl B, Gehrchen M (2021) Lordosis distribution index in short-segment lumbar spine fusion: Can ideal lordosis reduce revision surgery and iatrogenic deformity? Neurospine. 18(3):543–553. https://doi.org/10.14245/ns.2040744.372
Kumar MN, Baklanov A, Chopin D (2001) Correlation between sagittal plane changes and adjacent segment degeneration following lumbar spine fusion. Eur Spine J 10(4):314–319. https://doi.org/10.1007/s005860000239
Charles Malveaux WMS, Sharan AD (2011) Adjacent segment disease after lumbar spinal fusion: a systematic review of the current literature. Sem Spine Surg 23(4):266–274. https://doi.org/10.1053/j.semss.2011.05.008
Zheng G, Wang C, Wang T, Hu W, Ji Q, Hu F, Li J, Chaudhary SK, Song K, Song D, Zhang Z, Hao Y, Wang Y, Li J, Zheng Q, Zhang X, Wang Y (2020) Relationship between postoperative lordosis distribution index and adjacent segment disease following L4–S1 posterior lumbar interbody fusion. J Orthop Surg Res 15(1):129. https://doi.org/10.1186/s13018-020-01630-9
Lenz M, Mohamud K, Bredow J, Oikonomidis S, Eysel P, Scheyerer MJ (2022) Comparison of different approaches in lumbosacral spinal fusion surgery: a systematic review and meta-analysis. Asian Spine J. 16(1):141–149. https://doi.org/10.31616/asj.2020.0405
Crandall DG, Revella J (2009) Transforaminal lumbar interbody fusion versus anterior lumbar interbody fusion as an adjunct to posterior instrumented correction of degenerative lumbar scoliosis: three year clinical and radiographic outcomes. Spine (Phila Pa 1976) 34(20):2126–2133. https://doi.org/10.1097/BRS.0b013e3181b612db
Faundez AA, Schwender JD, Safriel Y, Gilbert TJ, Mehbod AA, Denis F, Transfeldt EE, Wroblewski JM (2009) Clinical and radiological outcome of anterior-posterior fusion versus transforaminal lumbar interbody fusion for symptomatic disc degeneration: a retrospective comparative study of 133 patients. Eur Spine J 8(2):203–211. https://doi.org/10.1007/s00586-008-0845-0
Kim JS, Lee KY, Lee SH, Lee HY (2010) Which lumbar interbody fusion technique is better in terms of level for the treatment of unstable isthmic spondylolisthesis? J Neurosurg Spine 12(2):171–177. https://doi.org/10.3171/2009.9.SPINE09272
Dorward IG, Lenke LG, Bridwell KH, O’Leary PT, Stoker GE, Pahys JM, Kang MM, Sides BA, Koester LA (2013) Transforaminal versus anterior lumbar interbody fusion in long deformity constructs: a matched cohort analysis. Spine (Phila 1976). 38(12):E755–E762. https://doi.org/10.1097/BRS.0b013e31828d6ca3
Upadhyayula PS, Curtis EI, Yue JK, Sidhu N, Ciacci JD (2018) Anterior versus transforaminal lumbar interbody fusion: perioperative risk factors and 30-day outcomes. Int J Spine Surg. 12(5):533–542. https://doi.org/10.14444/5065
Rossvoll I, Hammer S, Gulati S, Solberg T, Nygaard OP (2022) Outcome of surgery for isthmic spondylolisthesis from the Norwegian Registry for Spine Surgery (NORspine). Brain Spine 1(2):100960
Ren C, Qin R, Sun P, Wang P (2017) Effectiveness and safety of unilateral pedicle screw fixation in transforaminal lumbar interbody fusion (TLIF): a systematic review and meta-analysis. Arch Orthop Trauma Surg 137(4):441–450. https://doi.org/10.1007/s00402-017-2641-y
Sasso RC, Burkus JK, LeHuec JC (2003) Retrograde ejaculation after anterior lumbar interbody fusion: transperitoneal versus retroperitoneal exposure. Spine 28(10):1023–1026. https://doi.org/10.1097/01.BRS.0000062965.47779.EB
Rajaraman V, Vingan R, Roth P, Heary RF, Conklin L, Jacobs GB (1999) Visceral and vascular complications resulting from anterior lumbar interbody fusion. J Neurosurg Spine 91(1):60–64. https://doi.org/10.3171/spi.1999.91.1.0060
Phan K, Xu J, Scherman DB, Rao PJ, Mobbs RJ (2017) Anterior lumbar interbody fusion with and without an “access surgeon”: a systematic review and meta-analysis. Spine 42(10):E592–E601. https://doi.org/10.1097/BRS.0000000000001905
Villavicencio AT, Burneikiene S, Bulsara KR, Thramann JJ (2006) Perioperative complications in transforaminal lumbar interbody fusion versus anterior-posterior reconstruction for lumbar disc degeneration and instability. J Spinal Disord Tech 19(2):92–97. https://doi.org/10.1097/01.bsd.0000185277.14484.4e
Meyer B, Shiban E, Albers LE et al (2020) Completeness and accuracy of data in spine registries: an independent audit-based study. Eur Spine J 29:1453–1461. https://doi.org/10.1007/s00586-020-06342-6
Hooff MLV, Jacobs WC, Willems PC, Wouters MW, Kleuver MD, Peul WC, Ostelo RW, Fritzell P (2015) Evidence and practice in spine registries. Acta Orthop 86(5):534–544. https://doi.org/10.3109/17453674.2015.1043174
Endler P, Ekman P, Hellström F et al (2020) Minor effect of loss to follow-up on outcome interpretation in the Swedish spine register. Eur Spine J 29:213–220. https://doi.org/10.1007/s00586-019-06181-0
Højmark K, Støttrup C, Carreon L et al (2016) Patient-reported outcome measures unbiased by loss of follow-up. Single-center study based on DaneSpine, the Danish spine surgery registry. Eur Spine J 25:282–286. https://doi.org/10.1007/s00586-015-4127-3
Funding
Open access funding provided by Akershus University Hospital (AHUS).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors did not receive support from any organization for the submitted work. The authors have no relevant financial or non-financial interests to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The authors dr Alhaug and Dr Dolatowski would like to share fisrt authorship.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Alhaug, O.K., Dolatowski, F.C., Thyrhaug, A.M. et al. Long-term comparison of anterior (ALIF) versus transforaminal (TLIF) lumbar interbody fusion: a propensity score-matched register-based study. Eur Spine J 33, 1109–1119 (2024). https://doi.org/10.1007/s00586-023-08060-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-023-08060-1