Abstract
Purpose
To investigate the incidence and characteristics of subsequent vertebral fracture after osteoporotic vertebral fractures (OVFs) and identify risk factors for subsequent vertebral fractures.
Methods
This post-hoc analysis from a prospective randomized multicenter trial included 225 patients with a 48-week follow-up period. Differences between the subsequent and non-subsequent fracture groups were analyzed.
Results
Of the 225 patients, 15 (6.7%) had a subsequent fracture during the 48-week follow-up. The annual incidence of subsequent vertebral fracture after fresh OVFs in women aged 65–85 years was 68.8 per 1000 person-years. Most patients (73.3%) experienced subsequent vertebral fractures within 6 months. At 48 weeks, European Quality of Life-5 Dimensions, the Japanese Orthopedic Association Back Pain Evaluation Questionnaire pain-related disorder, walking ability, social life function, and lumbar function scores were significantly lower, while the visual analog scale (VAS) for low back pain was higher in patients with subsequent fracture. Cox proportional hazards analysis showed that a VAS score ≥ 70 at 0 weeks was an independent predictor of subsequent vertebral fracture. After adjustment for history of previous fracture, there was a ~ 67% reduction in the risk of subsequent vertebral fracture at the rigid-brace treatment.
Conclusion
Women with a fresh OVF were at higher risk for subsequent vertebral fracture within the next year. Severe low back pain and use of soft braces were associated with higher risk of subsequent vertebral fractures. Therefore, when treating patients after OVFs with these risk factors, more attention may be needed for the occurrence of subsequent vertebral fractures.
Level of evidence
III
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The clinical characteristics of osteoporosis are recurrent fractures. Particularly, fracture history is associated with an increased risk of subsequent fracture [1, 2]. It is important to note that the risk of subsequent fracture is relatively high within 5 years following the initial fracture [1]. Approximately 10–23% of all subsequent fractures occurred within 1 year after the first fracture [1, 3]. Recurring fractures were associated with severe deterioration in the quality of life, greater impaired function, and mortality [4, 5].
A recent study in women aged 65–74 years reported that 14% of them experienced a clinical fracture within 1 year following an initial vertebral fracture [1]. However, previous reports on the incidence of subsequent fractures after osteoporotic fractures were mainly based on administrative claims databases and post-hoc analysis of a large prospective studies examining the effects of an osteoporosis drug [1, 2, 6, 7]. Moreover, using these data have certain limitations such as lower accuracy in the timing and identification of fractures and details of symptoms. Thus, current evidence is limited because few reports have closely investigated the incidence and characteristics of subsequent vertebral fracture after osteoporotic vertebral compression fractures (OVFs) using detailed radiographic assessments and patient-reported outcome measures (PROMs). Therefore, a more detailed analysis of patients with subsequent fracture after OVF is needed to implement preventive measures to decrease the incidence of subsequent vertebral fractures. Furthermore, to date, no study has examined the incidence or risk factors for subsequent fractures using data from a prospective study examining the effect of braces on OVFs.
This study aimed to investigate the incidence of subsequent vertebral fracture after OVFs, characterize patient population with subsequent fracture after acute OVFs with a focus on PROMs and radiographic assessments, and identify predictors for subsequent vertebral fractures.
Methods
This study was a secondary analysis of a previous prospective randomized study (UMIN000014876) that compared the effectiveness of rigid and soft braces for acute thoracolumbar OVFs [8]. Briefly, the original trial enrolled 284 patients aged between 65 and 85 years who had received a diagnosis of one acute (within four weeks before inclusion) OVF between T10 and L2; 141 of these patients were randomly assigned to wear rigid braces, and 143 were assigned to wear soft braces. Patients wore ready-made braces until a custom-made thoracolumbar sacral rigid or soft brace was applied. Patients in the rigid-brace group received a rigid thoracolumbosacral orthosis. Patients in the soft-brace group received a soft thoracolumbosacral orthosis. Patients in both the rigid and soft bracing groups were instructed to wear the braces at all times whenever possible. All participants were instructed to wear the braces for a total of 12 weeks. The previous study was approved by each hospital’s institutional review board, and all participants provided oral and written informed consent prior to enrollment of the randomized controlled trial. In terms of anti-osteoporosis treatment, patients were allowed to use only the same medication they were taking before the injury or a newly prescribed active vitamin D to exclude any negative or positive effects of osteoporosis medication on vertebral deformity. Previous osteoporosis treatment consisted of bisphosphonates in 31 patients and selective estrogen receptor modulators in 2 patients. Accordingly, active vitamin D was not included as a previous osteoporosis treatment in this post-hoc analysis. Detailed inclusion and exclusion criteria of the study have been described previously [8].
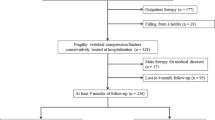
The current analysis included patients with 1-level acute thoracolumbar OVF who had received either rigid or soft-brace treatment and had undergone lateral radiography at 0, 12, and 48 weeks and magnetic resonance imaging at 48 weeks. In this study, patients who could not be followed up to week 48 and those with incomplete imaging studies were excluded in order to determine the exact number of vertebral fractures that had occurred by week 48. This study was approved by the institutional review board at the Tokyo Medical and Dental University and was compliant with the Declaration of Helsinki.
Patient-reported outcome measures
For the PROMs, scores on the European Quality of Life-5 Dimensions instrument (EQ-5D-3L; range, − 0.111 to 1, with higher scores indicating a better quality of life) [9], visual analog scale (VAS) scores for low back pain (range, 0–100; higher scores indicating more severe pain) [10], and the Japanese Orthopedic Association Back Pain Evaluation Questionnaire (JOABPEQ) for pain-related disorders, lumbar spine function, walking ability, social life function, and mental health (range, 0–100; higher scores indicating better function) [11] were used. JOABPEQ includes five categories (25 items) selected from the Roland Morris Disability Questionnaire and Short Form 36. These PROMs were provided when patients visited the hospital at 0, 12, and 48 weeks.
Radiographic assessment
In this study, a subsequent vertebral fracture was defined as a fracture occurring within 1 year of an incident fracture at a different site. In the radiographic analysis, the anterior vertebral body compression percentage (AVBCP) [12], defined as the ratio between the vertical height of the compressed anterior section of the injured vertebral body and the posterior vertebral body height at the same level, was measured independently by two radiologists as previously described in the original study. The mean values from the two evaluators were used. In addition to AVBCP measured in the original study, thoracic and lumbar vertebrae were graded on visual inspection of lateral radiography using the semiquantitative method of Genant et al. [13]. In this study, a new vertebral fracture was defined as a decrease of at least 20% in the height of any vertebral body during the 48-week follow-up to accurately assess a new vertebral fracture [14]. The Genant classification and the diagnosis of subsequent fractures were evaluated by a spine surgeon with 17 years of experience.
Data analysis
Differences between the subsequent fracture and non-subsequent fracture groups were analyzed. We analyzed continuous variables using an unpaired two-tailed t-test for normally distributed data and continuous data with skewed distribution using the Mann–Whitney U test after assessing the normality with the Shapiro–Wilk test. Fisher’s exact test was used for nominal variables. We then categorized the continuous variables using the cutoff values based on clinical experience for survival analysis. Because in previous studies, VAS 70 or greater has been defined as severe pain [15, 16], VAS 70 was used as the cutoff value in this study as well. For JOABPEQ, a clear criterion for the cutoff value is currently unclear, but since there is a report that each subdomain score on JOABPEQ in patients with lumbar spinal disease was around 50 [17], 50 was chosen as the cutoff value. Regarding AVBCP, because a reduction of more than 40% of the anterior vertebral body height is considered a severe fracture [13], a cutoff value of 60 was also used in this study. Regarding EQ-5D, as EQ-5D less than 0.3 has been defined as a severe condition [18], a cutoff value of 0.3 was used in this study as well. The log-rank test was used in univariate survival analysis to identify independent baseline variables (age, osteoporosis treatment, previous vertebral fracture, brace treatment, EQ-5D, VAS, JOABPEQ, AVBCP, and Genant classification) that could predict subsequent fractures. Using univariate and multivariate Cox’s proportional hazard models, we compared the baseline variables for the hazard of subsequent vertebral fractures during the 48-week follow-up. In multivariate Cox’s proportional hazard model, the model was adjusted for history of vertebral fracture. For all statistical analyses, JMP version 12 (SAS Institute, Cary, NC, USA), EZR (Saitama Medical Center, Jichi Medical University, Saitama, Japan), and a graphic user interface for R (The R Foundation for Statistical Computing, Vienna, Austria) [19, 20] were used. Regarding AVBCP, VAS, JOABPEQ, and EQ-5D, we imputed the missing data measurements with data obtained using the multiple imputation by chained equations package in R. We chose 20 iterations for multiple imputations. All tests were two-sided, and p values < 0.05 were considered significant.
Results
Demographics
In total, 225 patients with 48 weeks of follow-up were included in this study. The exclusion criteria are shown in Fig. 1. Of the 225 patients analyzed in the present study, 15 (6.7%) had a subsequent fracture during the 48-week follow-up. None of the 225 patients analyzed in this study had undergone vertebroplasty. The baseline characteristics of the patients are shown in Table 1. Interestingly, while there was a trend toward a higher proportion of patients wearing a rigid brace in the non-subsequent fracture group in the distribution of braces (p = 0.07), there were no significant differences between the two groups in any of the background variables (Table 1). The number of subsequent fractures for each patient ranged from 0 to 7. A total of 25 vertebral fractures were sustained during the follow-up period (Table 2). Thus, the annual incidence was 68.8 per 1000 person-years.
Details of the subsequent fracture group
Table 2 shows the detailed data of the subsequent fracture group. Of the 15 patients, 6 patients had subsequent fracture within 3 months after the original vertebral fracture, and 11 patients (73.3%) had subsequent fracture within 6 months. As a result, the incidence of subsequent vertebral fracture at 12 weeks was 106.7 per 1000 person-years. Interestingly, most of the subsequent vertebral fractures occurred in the lower vertebrae than in the original fracture (14/15 patients).
PROMs and radiographic assessment
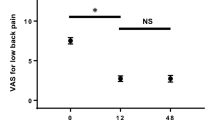
Table 3 shows differences in the PROMs and radiographic assessments between the subsequent and non-subsequent fracture groups. At enrollment, there were no differences in any scores between the two groups. At 12 weeks, JOABPEQ walking ability and lumbar function scores were significantly lower in the subsequent fracture group than in the non-subsequent fracture group (p = 0.02 and 0.03, respectively). At 48 weeks, EQ-5D and JOABPEQ pain-related disorder, walking ability, social life function, and lumbar function scores were significantly lower, and VAS for low back pain was higher in the subsequent fracture group than in the non-subsequent fracture group (p = 0.004, 0.03, 0.001, 0.01, 0.008, and 0.02, respectively). Regarding the radiographic assessment, there were no differences in AVBCP and Genant’s grades between the two groups.
Predictors for subsequent fracture
Then, we evaluated the risk of subsequent fracture using the log-rank test to identify variables that could predict a subsequent fracture. Clinical experience was used to categorize the continuous variables. The univariate analysis of the prognostic factors affecting subsequent fracture is shown in Table 4. The VAS score for low back pain showed a significant effect on subsequent fracture in the univariate analysis (p = 0.04). The brace treatment approached significance (p = 0.06), suggesting a difference in the occurrence of subsequent fracture (Table 4).
Univariate Cox proportional hazard models showed that a VAS score ≥ 70 alone was predictive of fracture-free survival rate (p = 0.02, hazard ratio (HR) = 6.31, 95% confidence interval [CI] 1.27 to 114.42) (Table 5). Furthermore, the univariate Cox proportional hazard model showed that the brace treatment alone was approaching as a significant predictor (p = 0.05, HR = 0.35, 95% CI 0.10 to 1.02). To assess whether subsequent vertebral fracture rates were significantly lower in the rigid-brace treatment compared to the soft-brace treatment after accounting for the difference in patient characteristics, we performed an additional analysis. Multivariate Cox proportional hazard model showed that there was a ~ 67% reduction in the risk of subsequent vertebral fracture with rigid-brace treatment (HR 0.33, 95% CI 0.09–0.99, p = 0.048) after adjustment for history of vertebral fracture (Table 5).
Discussion
This study investigated the incidence, characteristics, and predictors of subsequent fracture after acute OVFs. We found that the annual incidence of subsequent vertebral fracture after fresh OVFs in women aged 65–85 years was 68.8 per 1000 person-years. A population-based cohort study in Japan showed that the annual incidence of women aged 70–79 years without a vertebral fracture was 24.5 per 1000 person-years [14]. Thus, the incidence of vertebral fractures in the year following a fresh vertebral fracture is likely to be higher than in patients without a prior vertebral fracture.
We also found that most of the subsequent vertebral fractures occurred within 6 months of the original vertebral fracture. In this study, to exclude the effect of various anti-osteoporosis medications on vertebral fracture healing, patients were only allowed the same medication used before injury or newly prescribed active vitamin D. Given the relatively early occurrence of many subsequent fractures, the earlier the osteoporosis treatment is started, in theory, the better. Conversely, concerns have been raised about the possible negative impact of bisphosphonates on bone healing [21, 22]. Accordingly, there has been no consensus to date regarding the exact timing of starting anti-osteoporosis treatment after acute OVFs. Regarding the use of anti-resorptive agents, alendronate reduced the risk of clinical vertebral fracture by month 12 and of non-vertebral fracture by month 24 in the Fracture Intervention Trial [23]. Zoledronic acid reduced the risk of all clinical fractures by 12 months [24]. Denosumab reduced the risk of vertebral fractures after 1 year [25]. Regarding anabolic agents, anti-sclerostin antibody and teriparatide decreased vertebral fracture risk over 12 months and 21 months, respectively [26, 27]. Hence, these agents have been shown to be effective in inhibiting vertebral fractures, although there is still insufficient evidence regarding which treatment strategies can best reduce sequential fractures after fresh OVFs. Therefore, future prospective studies will be needed to determine which of these agents will most effectively reduce subsequent fractures after OVFs. In addition, further evidence on the timing of drug administration should be accumulated.
At 12 weeks after OVFs, while there were no significant differences in EQ-5D scores and VAS for low back pain between the groups, JOABPEQ walking ability and lumbar function scores were significantly lower in the subsequent fracture group than in the non-subsequent fracture group. We also found that EQ-5D and JOABPEQ pain-related disorder, walking ability, social life function, and lumbar function scores were significantly lower, and VAS for low back pain was significantly higher in patients with subsequent fracture than those without subsequent fracture at 48 weeks. Hence, subsequent fractures have a significantly negative impact on the quality of life, walking ability, and social life activities of patients.
Interestingly, most subsequent fractures occurred in the lower vertebrae than at the original fracture site. A biomechanical study found that while a significantly greater compression force was observed at one level below the fracture site compared with the non-fracture group, a compression force was not different one level above the fracture site [28]. Moreover, another biomechanical study found that vertebral deformities due to vertebral fractures are exacerbated by the anterior translation of the upper spinal column, which increases compressive loads on the lower vertebrae [29]. Thus, as biomechanical studies have shown, the kyphotic deformity caused by OVFs may have caused subsequent fractures in the vertebra below the fracture site due to an increased compression force applied to the lower vertebrae.
The Cox proportional hazard models showed that the predictor for subsequent vertebral fracture until 48 weeks following vertebral fracture was a VAS score ≥ 70 at 0 weeks. Thus, it is important to note that patients with severe low back pain immediately after an injury may be more prone to subsequent vertebral fractures. We speculate that this risk is related to the limitation on body movement due to pain because skeletal unloading causes regional loss of bone mineral density (BMD), primarily in the spine [30]. In the present study, although not significant at week 0, there was a clear trend toward severe pain and reduced walking ability in the subsequent fracture group. Therefore, it is inferred that daily movements were restricted by the intense pain experienced in the acute phase. The accompanying decrease in the loading stimuli on vertebrae may have reduced the bone strength and made them prone to subsequent fractures. In patients with fresh OVFs, a retrospective study reported that acute pain was negatively correlated with the functional independence measure (FIM) improvement ratio and that the FIM improvement ratio was an independent risk factor for subsequent vertebral fractures [31], which supports our speculation. Regarding the relationship between pain and bone fragility after acute OVFs, Lyritis et al. reported that fresh OVF patients with more severe vertebral deformity had more pain in the acute phase and significantly lower BMD than those with less severe vertebral deformity [32]. However, the association between acute pain and bone fragility in patients with acute OVFs is still unclear and should be investigated in future.
Finally, we found that the use of a soft brace was associated with an increased risk for subsequent vertebral fractures compared to the use of rigid braces in this study. In addition, the incidence of subsequent fracture up to 12 weeks was higher than the incidence of subsequent fracture up to 48 weeks. In theory, because a rigid brace is firmer than a soft brace, its use is expected to inhibit the anterior translation of the upper spine compared to the use of a soft brace. Regarding the biomechanical effects of the brace, the molded TLSO showed 94% restriction in lateral bending and 69% restriction of flexion–extension in the lumbar spine [33]. Restricting the overall movement of the upper body can reduce the load on the lumbar spine [34], so a molded rigid brace may have reduced the load on the lumbar spine leading to fewer subsequent fractures. Moreover, in the original study results, while there was no difference in the AVBCP between the two groups at 48 weeks, the group treated with rigid braces had significantly less vertebral deformity than the group treated with soft braces at week 12 [8]. Given that most subsequent fractures occur relatively early after injury, the importance placed on preventing vertebral deformity in the early post-injury period may have led to a reduction in subsequent vertebral fractures in the rigid brace group. Collectively, until new evidence is presented for brace treatment of fresh OVFs, rigid rather than soft braces may be a better choice in terms of preventing subsequent fractures. However, the inhibitory effect of rigid braces on subsequent vertebral fractures will need to be confirmed in future prospective comparative studies. In this case, it is preferable to consider the unbraced group as a comparison group.
There are some limitations to this study. First, we did not investigate the BMD of patients. While risk factors of subsequent fracture have been proposed as bone-related factors such as osteoporosis, fall-related factors, and the use of medications targeting the central nervous system [35], several previous studies showed that subsequent fracture risk among women with a previous fracture may be independent of BMD [1, 5, 36]. For example, a meta-analysis reported that low BMD explained only 8% of the subsequent fracture risk in patients with a previous fracture [36]. Thus, irrespective of having low BMD or not, postmenopausal women with a fresh OVF may need a rapid and strong treatment and fall prevention measures to reduce the risk of subsequent fracture. Second, numerous patients were excluded after enrollment. Accordingly, there may be a possibility that the rate of subsequent vertebral fracture was underestimated as patients may have been diagnosed with a subsequent vertebral fracture at other hospitals, which may reflect an underestimation of the actual number of subsequent fracture cases. Third, there is a lack of consensus on a precise definition of osteoporotic vertebral fractures by spine radiography [37]. For example, in one study, a new vertebral fracture was defined as a reduction in vertebral height of 20% or more [14], while in another study, it was defined as a reduction of 15% or more [38]. Unfortunately, the imaging definition of a subsequent vertebral fracture was not clearly defined in the original study. Thus, we clearly defined subsequent vertebral fractures on imaging for the current study and investigated the occurrence of subsequent vertebral fractures in patients who completed 48 weeks of follow-up. Because the original study included patients who dropped out during the study period, the numbers of patients involved differed between the original and current studies. As a result, the number of subsequent vertebral fractures and the conclusion drawn from the current study were slightly different from those in the original study. However, if the occurrence of subsequent vertebral fractures is diagnosed using the imaging definition used in this study, we believe that the use of rigid braces will be more preferable than soft braces to prevent the occurrence of subsequent vertebral fractures. With these limitations, further studies are required to address these issues and validate our findings.
In conclusion, the current study demonstrated that the annual incidence of subsequent vertebral fracture after fresh OVFs in women aged 65–85 years was 68.8 per 1000 person-years. Subsequent fractures result in deterioration in the quality of life and chronic low back pain. We also found that most of the subsequent vertebral fractures occurred within 6 months after the original fracture. Finally, severe low back pain and the use of soft braces were associated with an increased risk of subsequent vertebral fractures. Therefore, when treating patients after OVFs with these risk factors, greater attention is needed for the occurrence of subsequent vertebral fractures.
References
Balasubramanian A, Zhang J, Chen L, Wenkert D, Daigle SG, Grauer A, Curtis JR (2019) Risk of subsequent fracture after prior fracture among older women. Osteoporos Int 30:79–92. https://doi.org/10.1007/s00198-018-4732-1
Banefelt J, Akesson KE, Spangeus A, Ljunggren O, Karlsson L, Strom O, Ortsater G, Libanati C, Toth E (2019) Risk of imminent fracture following a previous fracture in a Swedish database study. Osteoporosis Int 30:601–609. https://doi.org/10.1007/s00198-019-04852-8
van Geel TA, van Helden S, Geusens PP, Winkens B, Dinant GJ (2009) Clinical subsequent fractures cluster in time after first fractures. Ann Rheum Dis 68:99–102. https://doi.org/10.1136/ard.2008.092775
Jinbayashi H, Aoyagi K, Ross PD, Ito M, Shindo H, Takemoto T (2002) Prevalence of vertebral deformity and its associations with physical impairment among Japanese women: the Hizen-Oshima Study. Osteoporosis Int 13:723–730. https://doi.org/10.1007/s001980200099
Bliuc D, Alarkawi D, Nguyen TV, Eisman JA, Center JR (2015) Risk of subsequent fractures and mortality in elderly women and men with fragility fractures with and without osteoporotic bone density: the Dubbo Osteoporosis epidemiology study. J Bone Miner Res 30:637–646. https://doi.org/10.1002/jbmr.2393
van Staa TP, Dennison EM, Leufkens HG, Cooper C (2001) Epidemiology of fractures in England and Wales. Bone 29:517–522. https://doi.org/10.1016/s8756-3282(01)00614-7
Lindsay R, Silverman SL, Cooper C, Hanley DA, Barton I, Broy SB, Licata A, Benhamou L, Geusens P, Flowers K, Stracke H, Seeman E (2001) Risk of new vertebral fracture in the year following a fracture. JAMA 285:320–323. https://doi.org/10.1001/jama.285.3.320
Kato T, Inose H, Ichimura S, Tokuhashi Y, Nakamura H, Hoshino M, Togawa D, Hirano T, Haro H, Ohba T, Tsuji T, Sato K, Sasao Y, Takahata M, Otani K, Momoshima S, Tateishi U, Tomita M, Takemasa R, Yuasa M, Hirai T, Yoshii T, Okawa A (2019) Comparison of rigid and soft-brace treatments for acute osteoporotic vertebral compression fracture: a prospective, randomized. Multicenter Study J Clin Med. https://doi.org/10.3390/jcm8020198
(1990) EuroQol--a new facility for the measurement of health-related quality of life. Health Policy 16:199–208. doi: https://doi.org/10.1016/0168-8510(90)90421-9
McCormack HM, Horne DJ, Sheather S (1988) Clinical applications of visual analogue scales: a critical review. Psychol Med 18:1007–1019. https://doi.org/10.1017/s0033291700009934
Fukui M, Chiba K, Kawakami M, Kikuchi S, Konno S, Miyamoto M, Seichi A, Shimamura T, Shirado O, Taguchi T, Takahashi K, Takeshita K, Tani T, Toyama Y, Yonenobu K, Wada E, Tanaka T, Hirota Y (2009) JOA back pain evaluation questionnaire (JOABPEQ)/JOA cervical myelopathy evaluation questionnaire (JOACMEQ). The report on the development of revised versions. April 16, 2007. The subcommittee of the clinical outcome committee of the Japanese orthopaedic association on low back pain and cervical myelopathy evaluation. J Orthop Sci 14:348–365. https://doi.org/10.1007/s00776-009-1337-8
Keynan O, Fisher CG, Vaccaro A, Fehlings MG, Oner FC, Dietz J, Kwon B, Rampersaud R, Bono C, France J, Dvorak M (2006) Radiographic measurement parameters in thoracolumbar fractures: a systematic review and consensus statement of the spine trauma study group. Spine 31:E156-165. https://doi.org/10.1097/01.brs.0000201261.94907.0d
Genant HK, Jergas M, Palermo L, Nevitt M, Valentin RS, Black D, Cummings SR (1996) Comparison of semiquantitative visual and quantitative morphometric assessment of prevalent and incident vertebral fractures in osteoporosis. The study of osteoporotic fractures research group. J Bone Miner Res 11:984–996. https://doi.org/10.1002/jbmr.5650110716
Fujiwara S, Kasagi F, Masunari N, Naito K, Suzuki G, Fukunaga M (2003) Fracture prediction from bone mineral density in Japanese men and women. J Bone Miner Res 18:1547–1553. https://doi.org/10.1359/jbmr.2003.18.8.1547
Aubrun F, Langeron O, Quesnel C, Coriat P, Riou B (2003) Relationships between measurement of pain using visual analog score and morphine requirements during postoperative intravenous morphine titration. Anesthesiology 98:1415–1421. https://doi.org/10.1097/00000542-200306000-00017
Kelly A (2001) The minimum clinically significant difference in visual analogue scale pain score does not differ with severity of pain. Emerg Med J EMJ 18:205–207. https://doi.org/10.1136/emj.18.3.205
Ohtori S, Ito T, Yamashita M, Murata Y, Morinaga T, Hirayama J, Kinoshita T, Ataka H, Koshi T, Sekikawa T, Miyagi M, Tanno T, Suzuki M, Aoki Y, Aihara T, Nakamura S, Yamaguchi K, Tauchi T, Hatakeyama K, Takata K, Sameda H, Ozawa T, Hanaoka E, Suzuki H, Akazawa T, Suseki K, Arai H, Kurokawa M, Eguchi Y, Suzuki M, Okamoto Y, Miyagi J, Yamagata M, Toyone T, Takahashi K (2010) Evaluation of low back pain using the Japanese Orthopaedic Association Back Pain Evaluation Questionnaire for lumbar spinal disease in a multicenter study: differences in scores based on age, sex, and type of disease. Journal of Orthop Sci 15:86–91. https://doi.org/10.1007/s00776-009-1426-8
M. Versteegh M, M. Vermeulen K, M. A. A. Evers S, de Wit GA, Prenger R, A. Stolk E, (2016) Dutch tariff for the five-level version of EQ-5D. Value Health 19:343–352. https://doi.org/10.1016/j.jval.2016.01.003
Kanda Y (2013) Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant 48:452–458. https://doi.org/10.1038/bmt.2012.244
Ihaka R, Gentleman R (1996) R: A language for data analysis and graphics. J Comput Gr Stat 5:299–314. https://doi.org/10.1080/10618600.1996.10474713
Molvik H, Khan W (2015) Bisphosphonates and their influence on fracture healing: a systematic review. Osteopor Int 26:1251–1260. https://doi.org/10.1007/s00198-014-3007-8
Tsuchie H, Miyakoshi N, Kasukawa Y, Nishi T, Abe H, Segawa T, Shimada Y (2016) The effect of teriparatide to alleviate pain and to prevent vertebral collapse after fresh osteoporotic vertebral fracture. J Bone Miner Metab 34:86–91. https://doi.org/10.1007/s00774-014-0646-y
Black DM, Thompson DE, Bauer DC, Ensrud K, Musliner T, Hochberg MC, Nevitt MC, Suryawanshi S, Cummings SR (2000) Fracture risk reduction with alendronate in women with osteoporosis: the Fracture Intervention Trial. FIT research group. J Clin Endocrinol Metab 85:4118–4124. https://doi.org/10.1210/jcem.85.11.6953
Black DM, Delmas PD, Eastell R, Reid IR, Boonen S, Cauley JA, Cosman F, Lakatos P, Leung PC, Man Z, Mautalen C, Mesenbrink P, Hu H, Caminis J, Tong K, Rosario-Jansen T, Krasnow J, Hue TF, Sellmeyer D, Eriksen EF, Cummings SR (2007) Once-yearly zoledronic acid for treatment of postmenopausal osteoporosis. New England J Med 356:1809–1822. https://doi.org/10.1056/NEJMoa067312
Cummings SR, San Martin J, McClung MR, Siris ES, Eastell R, Reid IR, Delmas P, Zoog HB, Austin M, Wang A, Kutilek S, Adami S, Zanchetta J, Libanati C, Siddhanti S, Christiansen C (2009) Denosumab for prevention of fractures in postmenopausal women with osteoporosis. New England J Med 361:756–765. https://doi.org/10.1056/NEJMoa0809493
Cosman F, Crittenden DB, Adachi JD, Binkley N, Czerwinski E, Ferrari S, Hofbauer LC, Lau E, Lewiecki EM, Miyauchi A, Zerbini CA, Milmont CE, Chen L, Maddox J, Meisner PD, Libanati C, Grauer A (2016) Romosozumab Treatment in postmenopausal women with osteoporosis. New England J Med 375:1532–1543. https://doi.org/10.1056/NEJMoa1607948
Neer RM, Arnaud CD, Zanchetta JR, Prince R, Gaich GA, Reginster JY, Hodsman AB, Eriksen EF, Ish-Shalom S, Genant HK, Wang O, Mitlak BH (2001) Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. New England J Med 344:1434–1441. https://doi.org/10.1056/nejm200105103441904
Briggs AM, Wrigley TV, van Dieën JH, Phillips B, Lo SK, Greig AM, Bennell KL (2006) The effect of osteoporotic vertebral fracture on predicted spinal loads in vivo. Eur Spine J 15:1785–1795. https://doi.org/10.1007/s00586-006-0158-0
Keller TS, Harrison DE, Colloca CJ, Harrison DD, Janik TJ (2003) Prediction of osteoporotic spinal deformity. Spine 28:455–462. https://doi.org/10.1097/01.Brs.0000048651.92777.30
Spector ER, Smith SM, Sibonga JD (2009) Skeletal effects of long-duration head-down bed rest. Aviat Space Environ Med 80:A23-28. https://doi.org/10.3357/asem.br02.2009
Yamauchi K, Adachi A, Kameyama M, Murakami M, Sato Y, Kato C, Kato T (2020) A risk factor associated with subsequent new vertebral compression fracture after conservative therapy for patients with vertebral compression fracture: a retrospective observational study. Arch Osteop 15:9. https://doi.org/10.1007/s11657-019-0679-x
Lyritis GP, Mayasis B, Tsakalakos N, Lambropoulos A, Gazi S, Karachalios T, Tsekoura M, Yiatzides A (1989) The natural history of the osteoporotic vertebral fracture. Clin Rheumatol 8(Suppl 2):66–69. https://doi.org/10.1007/bf02207237
Buchalter D, Kahanovitz N, Viola K, Dorsky S, Nordin M (1988) Three-dimensional spinal motion measurements. Part 2: A noninvasive assessment of lumbar brace immobilization of the spine. J Spinal Disord 1:284–286. https://doi.org/10.1097/00002517-198800140-00001
Lantz SA, Schultz AB (1986) Lumbar spine orthosis wearing. I Restriction of gross body motions. Spine 11:834–837. https://doi.org/10.1097/00007632-198610000-00019
Roux C, Briot K (2017) Imminent fracture risk. Osteopor Int 28:1765–1769. https://doi.org/10.1007/s00198-017-3976-5
Kanis JA, Johnell O, De Laet C, Johansson H, Oden A, Delmas P, Eisman J, Fujiwara S, Garnero P, Kroger H, McCloskey EV, Mellstrom D, Melton LJ, Pols H, Reeve J, Silman A, Tenenhouse A (2004) A meta-analysis of previous fracture and subsequent fracture risk. Bone 35:375–382. https://doi.org/10.1016/j.bone.2004.03.024
Santiago FR, Muñoz PT, Sánchez EM, Paniza MR, Martínez AM, Abela ALP (2016) Classifying thoracolumbar fractures: role of quantitative imaging. Quant Imaging Med Surg 6:772–784. https://doi.org/10.21037/qims.2016.12.04
Harris ST, Watts NB, Genant HK, McKeever CD, Hangartner T, Keller M, Chesnut CH 3rd, Brown J, Eriksen EF, Hoseyni MS, Axelrod DW, Miller PD (1999) Effects of risedronate treatment on vertebral and nonvertebral fractures in women with postmenopausal osteoporosis: a randomized controlled trial. Vertebral Efficacy with Risedronate Therapy (VERT) study group. JAMA 282:1344–1352. https://doi.org/10.1001/jama.282.14.1344
Acknowledgements
The authors thank Tomoko Nakajima for her help with data collection, Dr. Makoto Tomita for his help with the statistical analysis, and Dr. Ukihide Tateishi for his help with the radiological analysis.
Funding
None declared.
Author information
Authors and Affiliations
Contributions
Hiroyuki Inose, Tsuyoshi Kato, Shoichi Ichimura, Hiroaki Nakamura, Masatoshi Hoshino, Daisuke Togawa, Toru Hirano, Yasuaki Tokuhashi, Tetsuro Ohba, Hirotaka Haro, Takashi Tsuji, Kimiaki Sato, Yutaka Sasao, Masahiko Takahata, Koji Otani, Suketaka Momoshima, Kunihiko Takahashi, Masato Yuasa, Takashi Hirai, Toshitaka Yoshii, and Atsushi Okawa (1) provided substantial contributions to the conception or design of the study or the acquisition, analysis, or interpretation of data; (2) drafted the paper or revised it critically for important intellectual content, (3) provided final approval of the version to be published; and (4) agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Ethical approval
This study was approved by the institutional review board at the Tokyo Medical and Dental University and was compliant with the Declaration of Helsinki.
Availability of data and material
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Inose, H., Kato, T., Ichimura, S. et al. Risk factors for subsequent vertebral fracture after acute osteoporotic vertebral fractures. Eur Spine J 30, 2698–2707 (2021). https://doi.org/10.1007/s00586-021-06741-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-021-06741-3