Abstract
Objective
To investigate the changes of spinal cord angle between anterior controllable antedisplacement and fusion (ACAF) and posterior laminectomy in treating severe ossification of the posterior longitudinal ligament (OPLL).
Patients and methods
Seventy-one patients with cervical OPLL were enrolled. Patients in this study were divided into group A and group P. Japanese Orthopaedic Association (JOA) score was utilized to evaluate the neurological function. Radiological assessments included the spinal cord angle, Cobb angle, and area of the spinal cord. Surgery-related complications were also recorded.
Results
At the final follow-up, patients in group A had better recovery of local and whole cord angle, and the area of the cord than those in group P (all p < 0.05). A strong correlation between the change of local cord angle and the recovery of the spinal cord area was observed (r = − 0.867, p < 0.05). In addition, patients in group P had worse Cobb angle (9.15° ± 1.10°) than in group A (18.58° ± 0.73°) (p < 0.05). The final mean JOA score and its improvement rate were better in the group A than in group P (p < 0.05). During the follow-up, 15.15% patients in group P experienced surgery-related complications and 7.89% in group A.
Conclusion
This present study revealed that ACAF can achieve better recovery of the expansion of the spinal cord, spinal cord alignment, and Cobb angle, with better postoperative JOA score and less complications, compared with posterior laminectomy in treating severe cervical OPLL.
Graphical abstract
These slides can be retrieved under Electronic Supplementary Material.

Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Cervical ossification of the posterior longitudinal ligament (OPLL) has been one of the major contributors to cervical myelopathy, characterized by bone formation in the posterior ligament tissue. Patients with OPLL frequently exhibit sensory, motor, and even sphincteric dysfunction [1]. Surgical intervention has been proven to be the most effective treatment, especially for severe OPLL with an occupying rate of more than 50% [2,3,4]. The key idea of surgical treatment for cervical OPLL is to release the compression and reconstruct the physiological curvature of cervical spine. Anterior cervical corpectomy and fusion (ACCF) could decompress the spinal cord directly. However, this technique is highly technically demanding, and for those patients with a high occupying rate and/or multilevel segments, the surgery-related complications were inevitably encountered, including cerebrospinal fluid (CSF) leakage, spinal cord injury, and graft dislodgment [5, 6]. On the contrary, posterior approach (laminoplasty and laminectomy with or without fusion) is relatively safe with fewer complications. Good results have been reported after posterior surgery for OPLL in many studies [7, 8]. However, the benefits for patients with K-line (-) or larger ossified mass would be limited, and posterior axial pain, loss of cervical lordosis, and C5 nerve palsy have been frequently observed [9].
Many factors were reported to correlate with the surgical outcomes, including age, duration of symptoms, preoperative cervical alignment, and occupying rate of the spinal canal [10, 11]. However, it is still controversial to identify the best clinical indicator to predict postoperative prognosis among the aforementioned parameters. In fact, the main symptoms of cervical OPLL result from the chronic compression and damage of spinal cord. Posterior laminectomy can achieve decompression indirectly. However, many patients who underwent posterior decompression technique cannot acquire satisfactory recovery of spinal cord morphology, such as postoperative cord kyphosis [12, 13]. The posterior shift itself we believe could interrupt the normal spinal cord function, and the morphology of spinal cord, including the whole cord alignment and local compression, is theoretically optimum for neurological function.
We have previously proposed a novel technique, ACAF, to treat severe OPLL [14]. Instead of resecting the OPLL, ACAF moves the vertebrae with OPLL ventrally in a hoisting manner and patients can acquire direct decompression with good cord expansion and alignment [15]. Here, we have proposed a novel radiological parameter, spinal cord angle, to reflect the morphology of spinal cord and further compared it with posterior laminectomy.
Methods and materials
Patients’ population
We retrospectively reviewed the medical records of patients with symptoms of cervical myelopathy due to OPLL who had underwent surgical decompression from August 2016 to July 2017 in our institution.
The inclusion criteria were: (1) cervical myelopathy due to OPLL; (2) severe OPLL (occupying rate more than 60%); the occupying rate was used to reflect the condition of occupied spinal canal, which was calculated as follows: occupying rate = the thickness of ossified mass at the most compressed level/the anteroposterior diameter of the spinal canal at the same level × 100%, and the OPLL with the occupying rate of more than 60% was frequently considered as the severe OPLL; (3) treated by ACAF, or posterior laminectomy; and (4) without any dorsal compression to the spinal cord.
The exclusion criteria were: (1) cervical myelopathy caused by cervical disk herniation or spondylosis; (2) those with a history of cervical trauma, infection, and tumor; (3) those without a complete data of follow-up; (4) clinical symptoms resulting from thoracic or lumbar degenerative disease; (5) congenital spinal deformity; (6) combined with neurological disease such as Parkinson and Alzheimer dementia; (7) diabetes or other metabolic diseases without regular treatment and well control; and (8) a history of psychosis, or alcoholism or drug addiction.
Indications for surgery were neurological vulnerability due to myelopathy, radiculopathy, and/or intractable pain. Neurophysiologic monitoring including somatosensory-evoked potentials, spontaneous electromyogram, and/or motor-evoked potential intraoperatively was used in all patients during surgery. Patients were divided into two group A (ACAF) and group P (posterior laminectomy, including laminectomy with instrumented fusion and hemilaminectomy and unilateral fixation). Table 1 shows the demographic data of the included patients. All patients underwent X-ray films, computed tomography (CT), and MRI before and after surgery.
This study was approved by the institutional review board of our institution, and all patients signed the informed consent.
Selection of surgical procedure
The sample size in each group was determined by the number of eligible cases during the study period, and a total of 71 patients were finally enrolled in this study. All patients were informed of the purpose of this study before surgery, with well-explained manipulation differences of surgical technique (ACAF or posterior laminectomy). Before surgery, we would inform all patients of related advantages and potential disadvantages of different surgical methods. Simultaneously, we would take into consideration the characteristics of the cervical OPLL, including the extent, type, and position of the ossified mass, and patients’ physical conditions. In addition, for patients with severe kyphosis, ACAF would be recommended.
Finally, the selection of surgical decompression method (ACAF or posterior laminectomy) was confirmed by both patients’ acceptance, sagittal alignment, severity of OPLL, and doctors’ experience.
Surgical technique
The surgical procedures of posterior laminectomy including hemilaminectomy in this study have been well described in previous literature [16, 17]. For the surgery of ACAF, the process of ACAF has been reported previously and is illustrated in Fig. 1. Briefly, after general endotracheal anesthesia, the patient was placed in a supine position appropriately with the neck slightly extended. The vertebrae at both ends of the OPLL were defined as the upper- (UV) and lower-end vertebrae (LV), respectively, and the surgical exposure range of ACAF included the cephalic adjacent disk level to the UV and caudal adjacent disk level to the LV. The surgical level was confirmed via intraoperative radiography, and a standard right- or left-sided Smith–Robinson incision was conducted to expose the subcutaneous tissue and deep structures. After necessary discectomies, the resection of posterior longitudinal ligament at caudal and cranial ends of involved levels was performed to facilitate the further hoisting of the vertebrae–OPLL complex (VOC). The proper amount of anterior vertebral bodies of VOC was resected according to the thickness of the ossified mass and the anteroposterior diameter of the spinal canal. Following this, bilateral osteotomies for complete isolation of the VOC from the surrounding bony structures were conducted using high-speed drill, and this procedure was repeated at each affected level. Simultaneously, the intervertebral carbon fiber cages with autogenic bone were placed at corresponding levels. And the precurved cervical plate was fixed at caudal and cephalad vertebrae by screws which was inserted halfway for temporary fixation. Finally, the screws were gradually tightened in each vertebral body at the same pace to achieve anteriorly hoisting of the VOC and allogenic iliac bone was implanted into the groove to ensure fusion. All patients had neurophysiologic monitoring (somatosensory-evoked potentials), spontaneous electromyogram, and/or motor-evoked potential intraoperatively. All patients were operated by the spine surgeons who had at least ten-year experience of spine surgery from the same surgical team. After surgery, all patients wore a Philadelphia collar routinely postoperatively for at least 3 months.
Illustration of anterior controllable antedisplacement and fusion (ACAF) in sagittal (a–d) and axial (e–j) views. a Removal of the disk tissues and osteophytes of the affected segments. b Removal of the anterior portion of the involving vertebral bodies. c Prefixation of the cervical plate and screws and slotting on both sides of the affected vertebral bodies. d Controllable antedisplacement of the vertebrae–OPLL complex (VOC). e Ossification of the posterior longitudinal ligament (OPLL) at the operative level. f Removal of the anterior portion of the vertebral body. g Slotting at one side lateral to the margin of the OPLL. h Prefixation of cervical plate and screws. i Slotting at the other side lateral to the margin of the OPLL. j Controllable antedisplacement of the VOC. LV lower-end vertebrae, UV upper-end vertebrae
Clinical assessment
All patients were followed up at 6 months, and 12 months after surgery, respectively. Japanese Orthopaedic Association (JOA) score was utilized to evaluate the neurological function, and the improvement rate (IR) of JOA was calculated as IR = (final JOA score − preoperative JOA score/17 − preoperative JOA score) × 100%. Complications of CSF leakage, C5 nerve palsy, dysphagia, and hematoma were also recorded.
Radiological assessment
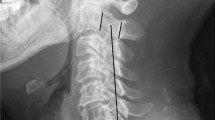
A diagnosis of OPLL was established for all the patients according to the results of a preoperative radiological examination. The measurement technique is presented in Fig. 1. Cervical Cobb angle was defined as the angle formed by two lines between the lower end plate of C2 and the lower end plate of C7 on lateral X-rays (angle α) (Fig. 2a) [18]. The spinal cord angle was divided into local cord angle and whole cord angle. The local cord angle (angle β) was defined as the angle formed by the two lines tangential to the bilateral rims of the compressed spinal cord area at the maximal compression level on mid-sagittal view of T2-weighted MRI, which was used to evaluate the local expansion of the spinal cord after decompression surgery (Fig. 2b, ossified mass indicated by red dotted circle). The whole cord angle (angle γ) was to evaluate the overall alignment of the spinal cord which has been reported to correlate with postoperative clinical outcomes previously [19]. When measuring the whole cord angle, two lines were drawn parallel to the lower end plate of C2 and C7 and intersected the posterior rim of the cord at point A and point C. The line connecting point A and point C was defined as the spine line (S line), which served as a baseline to evaluate the alignment of the spinal cord. The mid-perpendicular line of S line intersected the posterior rim of the cord at point B, and the line connecting point B and point C and S line formed the whole cord angle (angle γ) (Fig. 2c). The area of the spinal cord was measured to evaluate the expansion of the spinal cord (ESC) after decompression on T2-weighted MRI (Fig. 2d). Fusion was determined by CT, and the CT criteria for fusion include bridging bone inside or outside the graft and no lucencies extending > 50% of the graft–host interface.
Illustration of the measurement of radiological parameters. a Cobb angle was defined as the angle formed by two lines between the lower end plate of C2 and the lower end plate of C7 on lateral X-rays; b local spinal cord angle β was defined as the angle formed by the two lines tangential to the bilateral rims of the compressed spinal cord area at the maximal compression level on mid-sagittal view of T2-weighted MRI; c the method to measure the whole spinal cord angle; d the measurement of axial area of the spinal cord
All radiologic images were reviewed on a single day and again 2 weeks later by three independent spine surgeons who were blinded to this study. Anatomic measurements were performed using digital calipers on a uniform magnification of 200%. To obtain reliable data and reduce errors, the average of at least three measurements was taken for each result.
Statistical analysis
Statistical analyses were performed using SPSS (Version 20.0; IBM Corp., Armonk, New York, USA). The data were presented as the mean ± standard deviation. Comparisons between preoperative and postoperative parameters within the same groups were made using paired t test. Intergroup comparisons were made using the analysis of variance. The gender was analyzed by Chi-squared test. The relationship between the change of cord angle and the area of the cord was also analyzed using the Pearson correlation coefficient. The correlation values were graded as follows: strong (values from − 1.0 to − 0.5 or 1.0–0.5), moderate (− 0.5 to − 0.3 or o 0.3 to 0.5), weak (− 0.3 to − 0.1 or 0.1 to 0.3), and none (− 0.1 to 0.1). Values less than 0.05 (p < 0.05) were considered statistically significant.
Results
A total 71 patients were included in this study (38 in group A and 33 in group P). The mean age in group A was 58.18 ± 1.82 years old and 58.06 ± 2.33 years old in group P (p > 0.05). The duration of symptoms of patients in group A was 19 ± 5.8 months, and in group P, the value was 18 ± 4.9 (p > 0.05). No significant difference was observed between patients in group A and group P in terms of preoperative area of the spinal cord (59.72 ± 26.88 mm2 vs. 58.01 ± 28.33 mm2). At the final follow-up, patients in group A had better improvement of spinal cord area than in group P (p < 0.05). During the follow-up, only one patient in group A suffered postoperative dysphagia, one patient had CSF leakage, and one had C5 nerve palsy, while three months after ACAF, the related symptoms improved significantly. Patients in group P had higher occurrence of complications (22.7%), including one with CSF leakage, one with postoperative hematoma, and three with C5 nerve palsy. At the final follow-up, no patients have complaint of wound healing disorders in this study. In addition, all the patients in group A had solid bone fusion at bilateral bony grooves at the final follow-up, and patients who underwent posterior laminectomy had solid fixation at the final follow-up.
Table 2 shows the JOA score of patients. In general, neurologic status was both significantly improved in the two groups. Six months after the operation, patients in group A had higher JOA score (13.37 ± 0.34) than those in the group P (12.58 ± 0.25) (p < 0.05). At the final follow-up, patients in group A had the better improvement rate of JOA score (78.6% ± 3.4%) than the in group P (64.5% ± 3.0%) (p < 0.05).
The spinal cord angle and cervical Cobb angle of all patients are summarized in Table 3. No statistical difference was observed between the two groups before surgery in terms of Cobb angle, local cord angle, and whole cord angle before surgery (p > 0.05). At the final follow-up, the Cobb angle in group A significantly improved to 18.58° ± 0.73° (p < 0.05). However, patients in group P showed a decrease in Cobb angle, with the final Cobb angle of 9.15° ± 1.10° (p < 0.05). Patients in group A had smaller local cord angle (7.19° ± 1.24°) and higher change of local cord angle (19.95° ± 1.79°) than those in group P (14.06° ± 1.85° and 13.36° ± 1.51°, respectively) (p < 0.05). In terms of the whole cord angle, patients in group A had better whole spinal cord angle (14.36° ± 1.06°) than in group P at the final follow-up (p < 0.05). Inter-observer reliability was excellent for all cervical radiographic parameters in the two independent observers. At the final follow-up, a strong correlation between the change of local cord angle and the change of spinal cord area was found (r = − 0.867, p < 0.05).
Case presentation
Case 1
A 59-year-old male patient complained of pain at his upper extremities, associated with slight gait disturbance. Preoperative imaging confirmed the diagnosis of cervical OPLL from C5 to C7, with the most compression at C6/7 level (Fig. 3a). His JOA score was 10 before surgery. ACAF of C2-5 was given (Fig. 3b). His preoperative local cord angle was 65.9°, and the whole cord angle was 6.2° (Fig. 3c). After the operation, his local and whole cord angle was 3.6° and 12.7°, respectively (Fig. 3d). As shown in figure D-F, the most compression at C6/7 disappeared completely, which indicated a satisfactory recovery of local spinal cord expansion (Fig. 3d–f). In addition, his spinal cord was kept lordotic after decompression. His JOA score improved to 16 at the final follow-up, with the improvement rate of 85.7%. There was no CSF leakage or other complications during the follow-up.
Case 1 A 59-year-old male patient complained of pain at his upper extremities, associated with slight gait disturbance. Preoperative imaging confirmed the diagnosis of cervical OPLL from C5 to C7, with the most compression at C6/7 level (a). His JOA score was 10 before surgery. ACAF of C2-5 was given (b). His preoperative local cord angle was 65.9°, and the whole cord angle was 6.2° (c). After the operation, his local and whole cord angle was 3.6° and 12.7°, respectively (d). As shown in figure d–f, the most compression at C6/7 disappeared completely, which indicated a satisfactory recovery of local spinal cord expansion (d–f). In addition, his spinal cord was kept lordotic after decompression. His JOA score improved to 16 at the final follow-up, with the improvement rate of 85.7%. There was no CSF leakage or other complications during the follow-up. OPLL ossification of the posterior longitudinal ligament
Case 2
A 69-year-old male patient presented with numbness in his bilateral upper extremity for 2 years, worsening for 1 month. Preoperative imaging revealed mixed cervical OPLL at C2-T7, with the loss of Cobb angle (Fig. 4a, b). The patient underwent posterior laminectomy with instrumented fusion (Fig. 4c). His local and whole cord angle was 33.5° and 14.6°, respectively, with preoperative JOA score of 9 (Fig. 4d). Postoperative MRI showed the cord shifted posteriorly (Fig. 4e). However, his local cord angle recovered badly (26.8°) compared to preoperation, which indicated the decompression of the cord was not sufficient, especially at C3/4 level (Fig. 4e). The whole cord angle decreased to 4.6°, which suggested that his spinal cord alignment became from preoperative lordosis to postoperative straight condition (Fig. 4e). Three days after operation, the patient experienced C5 nerve palsy. However, he recovered significantly after administration of glucocorticoid for 4 days. At the final follow-up, his JOA score was 12 and the improvement rate was 50.0%.
Case 2 A 69-year-old male patient presented with numbness in his bilateral upper extremity for 2 years, worsening for 1 month. Preoperative imaging revealed mixed cervical OPLL at C2-T7, with the loss of Cobb angle (a, b). The patient underwent posterior laminectomy with instrumented fusion (c). His local and whole cord angle was 33.5° and 14.6°, respectively, with preoperative JOA score of 9 (d). Postoperative MRI showed the cord shifted posteriorly (e). However, his local cord angle recovered badly (26.8°) compared to preoperation, which indicated the decompression of the cord was not sufficient, especially at C3/4 level (e). The whole cord angle decreased to 4.6°, which suggested that his spinal cord alignment became from preoperative lordosis to postoperative straight condition (e). Three days after operation, the patient experienced C5 nerve palsy. However, he recovered significantly after administration of glucocorticoid for 4 days. At the final follow-up, his JOA score was 12 and the improvement rate was 50.0%. OPLL ossification of the posterior longitudinal ligament
Case 3
A 70-year-old female patient exhibited neck pain, numbness, and weakness at her bilateral upper extremities for nearly 4 years. Preoperative imaging showed cervical OPLL from C4 to C6, with the occupying rate of the spinal cord of 76.8% at the narrowest level (Fig. 5a–c). She underwent posterior C3-7 hemilaminectomy and unilateral fixation (Fig. 5d, e). However, her symptoms did not improve after surgery. Her local cord angle at C4/5 level was 59.2°, without significant improvement compared with preoperation (61.1°) (Fig. 5f). Her whole cord angle was − 3.8° at the final follow-up, which indicated a kyphotic spinal cord alignment. Axial MRI also suggested that the spinal cord was not decompressed sufficiently (Fig. 5g).
Case 3 A 70-year-old female patient exhibited neck pain, numbness, and weakness at her bilateral upper extremities for nearly 4 years. Preoperative imaging showed cervical OPLL from C4 to C6, with the occupying rate of the spinal cord of 76.8% at the narrowest level (a–c). She underwent posterior C3-7 hemilaminectomy and unilateral fixation (d, e). However, her symptoms did not improve after surgery. Her local cord angle at C4/5 level was 59.2°, without significant improvement compared with preoperation (61.1°) (f). Her whole cord angle was − 3.8° at the final follow-up, which indicated a kyphotic spinal cord alignment. Axial MRI also suggested that the spinal cord was not decompressed sufficiently (g). OPLL: ossification of the posterior longitudinal ligament
Discussion
Cervical myelopathy due to OPLL has been characterized by the chronic and long-term compression of the spinal cord, and therefore, the functional recovery of spinal cord is the key point for surgical decompression. Several surgical options for cervical OPLL have been well established, which involve mainly anterior or posterior surgery. However, the optimum method for multilevel OPLL remains controversial. Actually, both anterior and posterior routine surgeries have disadvantages. Theoretically, an optimal surgical procedure for OPLL should release the compression of the spinal cord, reconstruct the volume of spinal canal and its physiological curvature, restore the intervertebral height, and acquire fewer complications via only one unitary surgical approach [20]. ACAF, as a novel surgical technique, can achieve all these goals by hoisting the vertebrae with ossified mass anteriorly in our previous studies [21, 22].
Many studies have investigated the condition of the spinal cord before and after surgery, such as intramedullary signal changes [23, 24]. However, the clinical value of these parameters remains unclear. In this current study, we proposed a novel parameter, spinal cord angle, to evaluate the local and whole morphological change of the spinal cord. A previous study by Lee et al. [25] ever reported the compression angle based on the morphology of the OPLL at the maximum compressed level and found higher compression angles of OPLL had more deleterious effects on the spinal cord and more decreased preoperative JOA scores. However, Lee’s study only focused on the ossified mass itself. In fact, the morphological change of spinal cord is not always consistent with that of the ossified mass. In addition, the authors did not analyze the prognosis effect of compression angle. Previous studies suggested that patients who underwent ACAF acquired good recovery of the expansion of the spinal cord and spinal cord alignment, which we think also is significantly important for the recovery of neurological function of the spinal cord [15, 19, 26]. Therefore, the local cord angle was proposed in this study to better evaluate the expansion of the spinal cord, and the smaller the local cord angle was, the better the expansion of the spinal cord would be. In this study, except those whose spinal cord was damaged significantly before surgery, the majority of patients acquired satisfactory expansion of the cord, especially in patients with ACAF. Additionally, the whole cord angle in group A was also better improved at the final follow-up. We believe there were two reasons to explain the results. Firstly, anterior approach had better therapeutic efficiency than posterior approach in treating multilevel OPLL in a prospective study by Hou et al. [27]. In the procedure of ACAF, the ossified mass was not resected like ACCF. Instead, it was hoisted anteriorly with the vertebral bodies. As an anterior approach, ACAF can restore the spinal canal to its normal morphology directly, which we believe was significant for the sufficient expansion of the cord. Secondly, for patients with dura ossification (DO), the dura mater may also be elevated due to the adhesion to OPLL after ACAF, which facilitated to decrease the pressure of cerebrospinal fluid exerted on the cord. However, posterior laminectomy depended largely on the posterior floating of the cord, and the depression effect may be limited in case with larger ossified mass or bad cervical curvature after operation (Figs. 3, 4). A correlation analysis in this study revealed the strong correlation between local cord angle and the expansion of the spinal cord which has been reported affects largely the neurological function [28]. Taken together, these results showed that ACAF can achieve better local expansion of the spinal cord than posterior approach.
Increasing studies have indicated that abnormal or excessive motion of the cervical spine could result in increased strain and shear forces within the spinal cord [29]. Kuwazawa et al. [30] also reported the mean length of the cervical cord in extension was shorter than in neutral and flexion at the anterior, middle, and posterior line, which may suggest the lordotic condition of the spinal cord may be the optimum condition for its function. Yang et al. [19] have divided the abnormal spinal cord alignment into several types, including straight with or without shifting, sigmoid, and kyphosis, and suggested the patients abnormal alignment of the cord frequently had worse neurological outcome. Therefore, we speculated that the lengthening of the cord during motion may disrupt the cerebrospinal fluid and the blood in the cord and further lead to progressive damage to the cord. However, Yang’s classification was relatively subjective, and there were no baseline criteria to evaluate the spinal cord alignment. In this present study, we first proposed the S line (yellow line) and whole cord angle to evaluate the spinal cord alignment and redefine the types of spinal cord alignment (Fig. 6). When the affected spinal cord did not exceed the S line, it was considered as lordotic (Fig. 6a). Conversely, if the affected exceeded the S line, it was kyphotic (Fig. 6b). If the affected spinal cord was parallel and near to the S line, we defined it as straight (Fig. 6c). Finally, if the affected spinal cord intercrossed with the S line like the $, we defined it as S-type spinal cord (Fig. 6d). In addition, the whole cord angle was used to quantitate the spinal cord alignment, and the smaller the angle is, the worse the spinal cord alignment would be. In this present study, all patients with ACAF were restored to lordotic spinal cord. However, due to the nature of posterior decompression, the majority of patients had straight, kyphotic, or even S-type alignment of the spinal cord. In fact, the restoration of the whole spinal cord alignment depends largely on the restoration of physiological curvature of the spinal canal and the sufficient decompression of the spinal cord. ACAF could sufficiently enlarge the volume of the bony spinal canal and maintain its sagittal alignment within the physiologically permissible flexural tension zone, which could facilitate the recovery of spinal cord lordosis. In addition, the preserving of posterior structures of the cervical spine can limit the posterior floating of the cord and restore cervical stability. And thus, the spinal cord was kept in the normal position with a good curvature after ACAF. Although no study has claimed that the spinal cord must be kept in normal position with even physical lordosis, in theory it should be. However, whether the types of spinal cord alignment have a predictive effect of postoperative recovery of neurological function still requires further study.
Representative types of spinal cord alignment after decompression surgery. a Lordotic spinal cord alignment after ACAF; b straight spinal cord alignment after posterior laminectomy; c kyphotic spinal cord alignment after posterior laminectomy; d S-type spinal cord alignment after posterior laminectomy. The S line was indicated by yellow line. ACAF anterior controllable antedisplacement and fusion
Cobb angle (C2-7 angle) was also reported to correlate with clinical outcomes [10, 31, 32]. The results in this study also revealed that patients who underwent ACAF acquired satisfactory restoration of cervical lordosis at the final follow-up, whereas for patients in group P, the Cobb angle decreased at the final follow-up, which is consistent with previous study [33]. Considering the better clinical recovery in group A, we reckoned that the worsening of Cobb angle after posterior laminectomy may also contribute the lower improvement rate of JOA score. The current study also demonstrated that patients who underwent ACAF had fewer complications such as C5 palsy, which is consistent with our previous study [34]. C5 nerve palsy is the most common complication after posterior surgery [35]. The lower occurrence of C5 nerve palsy in group A may be attributed to the better recovery of cervical lordosis and the spinal cord rotation [15].
This study has several limitations. Firstly, the samples size was small and the duration of follow-up was short. Studies with more patients and longer-term follow-up should be conducted in the future. Secondly, because the majority of patients who had cervical OPLL were multilevel, it is quite difficult to select enough patients with only one-level ossified mass. Therefore, we evaluated the patients with anterior multilevel OPLL rather than single-level lesion, which may bring several disruptions. Future study with enough patients who have only local one-level ossified mass will be conducted to confirm the clinical significance of the local cord angle. Thirdly, due to small sample size, we could not conduct the multivariable regression technique to study the predictive effect of cord angle on the surgical outcomes of ACAF. However, we identified the close correlation between cord angle and the expansion of the spinal cord, which is important to neurological recovery. Thirdly, this was only a retrospective study in our institution; a multicenter, prospective study should be performed.
Conclusion
This present study revealed that ACAF can achieve better recovery of the expansion of the spinal cord, spinal cord alignment, and Cobb angle, with better postoperative JOA score and less complications, compared with posterior laminectomy in treating severe cervical OPLL.
References
Fujimori T, Le H, Hu SS, Chin C, Pekmezci M, Schairer W, Tay BK, Hamasaki T, Yoshikawa H, Iwasaki M (2015) Ossification of the posterior longitudinal ligament of the cervical spine in 3161 patients: a ct-based study. Spine (Phila Pa 1976). https://doi.org/10.1097/BRS.0000000000000791
Hou Y, Liang L, Shi GD, Xu P, Xu GH, Shi JG, Yuan W (2017) Comparing effects of cervical anterior approach and laminoplasty in surgical management of cervical ossification of posterior longitudinal ligament by a prospective nonrandomized controlled study. Orthop Traumatol Surg Res 103:733–740. https://doi.org/10.1016/j.otsr.2017.05.011
Pham MH, Attenello FJ, Lucas J, He S, Stapleton CJ, Hsieh PC (2011) Conservative management of ossification of the posterior longitudinal ligament. A review. Neurosurg Focus 30(3):E2. https://doi.org/10.3171/2011.1.FOCUS10273
Yoshii T, Sakai K, Hirai T, Yamada T, Inose H, Kato T, Enomoto M, Tomizawa S, Kawabata S, Arai Y, Okawa A (2016) Anterior decompression with fusion versus posterior decompression with fusion for massive cervical ossification of the posterior longitudinal ligament with a ≥ 50% canal occupying ratio: a multicenter retrospective study. Spine J 16(11):1351–1357. https://doi.org/10.1016/j.spinee.2016.07.532
Smith ZA, Buchanan CC, Raphael D, Khoo LT (2011) Ossification of the posterior longitudinal ligament: pathogenesis, management, and current surgical approaches. A review. Neurosurg Focus 30:E10. https://doi.org/10.3171/2011.1.FOCUS10256
Abiola R, Rubery P, Mesfin A (2016) Ossification of the posterior longitudinal ligament: etiology, diagnosis, and outcomes of nonoperative and operative management. Glob Spine J 6:195–204. https://doi.org/10.1055/s-0035-1556580
Lee CH, Jahng TA, Hyun SJ, Kim KJ, Kim HJ (2016) Expansive laminoplasty versus laminectomy alone versus laminectomy and fusion for cervical ossification of the posterior longitudinal ligament: is there a difference in the clinical outcome and sagittal alignment? Clin Spine Surg 29:E9–E15. https://doi.org/10.1097/BSD.0000000000000058
Ahsan MK, Awwal MA, Khan SI, Zaman N, Haque MH, Zahangiri Z (2017) Open-door laminoplasty for multilevel cervical spondylotic myelopathy and ossification of the posterior longitudinal ligament (OPLL) using titanium reconstruction miniplate and screws. Mymensingh Med J 26:558–568
Ma L, Liu FY, Huo LS, Zhao ZQ, Sun X, Li F, Ding WY (2018) Comparison of laminoplasty versus laminectomy and fusion in the treatment of multilevel cervical ossification of the posterior longitudinal ligament: a systematic review and meta-analysis. Medicine (Baltimore) 97:e11542. https://doi.org/10.1097/MD.0000000000011542
Liu H, Li Y, Chen Y, Wu W, Zou D (2013) Cervical curvature, spinal cord MRIT2 signal, and occupying ratio impact surgical approach selection in patients with ossification of the posterior longitudinal ligament. Eur Spine J 22:1480–1488. https://doi.org/10.1007/s00586-013-2707-7
Rajshekhar V, Kumar GS (2005) Functional outcome after central corpectomy in poor-grade patients with cervical spondylotic myelopathy or ossified posterior longitudinal ligament. Neurosurgery 56:1279–1284 (discussion 1284-5)
Phan K, Scherman DB, Xu J, Leung V, Virk S, Mobbs RJ (2017) Laminectomy and fusion vs laminoplasty for multi-level cervical myelopathy: a systematic review and meta-analysis. Eur Spine J 26:94–103
Kennamer BT, Arginteanu MS, Moore FM, Steinberger AA, Yao KC, Gologorsky Y (2019) Complications of poor cervical alignment in patients undergoing posterior cervicothoracic laminectomy and fusion. World Neurosurg 122:e408–e414. https://doi.org/10.1016/j.wneu.2018.10.062
Sun J, Shi J, Xu X, Yang Y, Wang Y, Kong Q, Yang H, Guo Y, Han D, Jiang J, Shi G, Yuan W, Jia L (2018) Anterior controllable antidisplacement and fusion surgery for the treatment of multilevel severe ossification of the posterior longitudinal ligament with myelopathy: preliminary clinical results of a novel technique. Eur Spine J 27:1469–1478. https://doi.org/10.1007/s00586-017-5437-4
Sun K, Wang S, Sun J, Wang H, Huan L, Sun X, Lv H, Wang Z, Zou W, Shi J (2019) Surgical outcomes after anterior controllable antedisplacement and fusion (ACAF) compared to single open-door laminoplasty: preliminary analysis of postoperative changes of the spinal cord displacement. World Neurosurg 127:e288–e298. https://doi.org/10.1016/j.wneu.2019.03.108
Liu X, Chen Y, Yang H, Li T, Xu B, Chen D (2017) Expansive open-door laminoplasty versus laminectomy and instrumented fusion for cases with cervical ossification of the posterior longitudinal ligament and straight lordosis. Eur Spine J 26:1173–1180. https://doi.org/10.1007/s00586-016-4912-7
Lau D, Winkler EA, Than KD, Chou D, Mummaneni PV (2017) Laminoplasty versus laminectomy with posterior spinal fusion for multilevel cervical spondylotic myelopathy: influence of cervical alignment on outcomes. J Neurosurg Spine 27:508–517. https://doi.org/10.3171/2017.4.SPINE16831
Lee SE, Jahng TA, Kim HJ (2015) Correlation between cervical lordosis and adjacent segment pathology after anterior cervical spinal surgery. Eur Spine J 24:2899–2909. https://doi.org/10.1007/s00586-015-4132-6
Yang H, Sun J, Shi J, Yang Y, Guo Y, Zheng B, Wang Y (2018) In situ decompression to spinal cord during anterior controllable antedisplacement fusion treating degenerative kyphosis with stenosis: surgical outcomes and analysis of C5 nerve palsy based on 49 patients. World Neurosurg 15:e501–e508. https://doi.org/10.1016/j.wneu.2018.04.078
Grosso MJ, Hwang R, Mroz T, Benzel E, Steinmetz MP (2013) Relationship between degree of focal kyphosis correction and neurological outcomes for patients undergoing cervical deformity correction surgery. J Neurosurg Spine 18(6):537–544. https://doi.org/10.3171/2013.2.SPINE12525
Yang H, Sun J, Shi J, Guo Y, Zheng B, Wang Y, Xu X, Shi G (2018) Anterior controllable antedisplacement fusion as a choice for 28 patients of cervical ossification of the posterior longitudinal ligament with dura ossification: the risk of cerebrospinal fluid leakage compared with anterior cervical corpectomy and fusion. Eur Spine J. https://doi.org/10.1007/s00586-018-5813-8
Sun J, Sun K, Wang Y, Shi J, Yang H, Guo Y, Xu X, Kong Q, Chen K, Zheng B, Shi G, Wang Y (2018) Quantitative anterior enlargement of the spinal canal by anterior controllable antedisplacement and fusion for the treatment of cervical ossification of the posterior longitudinal ligament with myelopathy. World Neurosurg 120:e1098–e1106. https://doi.org/10.1016/j.wneu.2018.08.233
Chen CJ, Lyu RK, Lee ST, Wong YC, Wang LJ (2001) Intramedullary high signal intensity on T2-weighted MR images in cervical spondylotic myelopathy: prediction of prognosis with type of intensity. Radiology 221:789–794
Vedantam A, Rajshekhar V (2014) Change in morphology of intramedullary T2-weighted increased signal intensity after anterior decompressive surgery for cervical spondylotic myelopathy. Spine (Phila Pa 1976) 39:1458–1462. https://doi.org/10.1097/BRS.0000000000000440
Lee N, do Yoon H, Kim KN, Shin HC, Shin DA, Ha Y (2018) Compression angle of ossification of the posterior longitudinal ligament and its clinical significance in cervical myelopathy. J Korean Neurosurg Soc 59:471–477. https://doi.org/10.3340/jkns.2016.59.5.471
Sun K, Wang S, Sun J, Huan L, Xu X, Sun X, Zhang B, Wang Y, Shi J, Guo Y (2018) Analysis of the correlation between cerebrospinal fluid space and outcomes of anterior controllable antedisplacement and fusion for cervical myelopathy due to ossification of the posterior longitudinal ligament. World Neurosurg 122:e358–e366. https://doi.org/10.1016/j.wneu.2018.10.051
Hou Y, Liang L, Shi GD, Xu P, Xu GH, Shi JG, Yuan W (2017) Comparing effects of cervical anterior approach and laminoplasty in surgical management of cervical ossification of posterior longitudinal ligament by a prospective nonrandomized controlled study. Orthop Traumatol Surg Res 103:733–740. https://doi.org/10.1016/j.otsr.2017.05.011
Fujimori T, Iwasaki M, Okuda S, Takenaka S, Kashii M, Kaito T, Yoshikawa H (2014) Long-term results of cervical myelopathy due to ossification of the posterior longitudinal ligament with an occupying ratio of 60% or more. Spine 39:58–67. https://doi.org/10.1097/BRS.0000000000000054
Henderson FC, Geddes JF, Vaccaro AR, Woodard E, Berry KJ, Benzel EC (2005) Stretch-associated injury in cervical spondylotic myelopathy: new concept and review. Neurosurgery 56:1101–1113 (discussion 1101-1113)
Kuwazawa Y, Pope MH, Bashir W, Takahashi K, Smith FW (2006) The length of the cervical cord: effects of postural changes in healthy volunteers using positional magnetic resonance imaging. Spine (Phila Pa 1976) 31:E579–E583
Sun JC, Zhang B, Shi J, Sun KQ, Huan L, Sun XF, Liu N, Zheng B, Wang HB (2018) Can K-line predict the clinical outcome of anterior controllable antedisplacement and fusion surgery for cervical myelopathy caused by multi-segmental ossification of the posterior longitudinal ligament? World Neurosurg 116:e118–e127. https://doi.org/10.1016/j.wneu.2018.04.128
Lau D, Winkler EA, Than KD, Chou D, Mummaneni PV (2017) Laminoplasty versus laminectomy with posterior spinal fusion for multilevel cervical spondylotic myelopathy: influence of cervical alignment on outcomes. J Neurosurg Spine 27:508–517. https://doi.org/10.3171/2017.4.SPINE16831
Kim B, Yoon DH, Ha Y, Yi S, Shin DA, Lee CK, Lee N, Kim KN (2016) Relationship between T1 slope and loss of lordosis after laminoplasty in patients with cervical ossification of the posterior longitudinal ligament. Spine J 16:219–225. https://doi.org/10.1016/j.spinee.2015.10.042
Yang H, Guo Y, Shi J, Sun J, Wang Y, Xu X, Zheng B, Shi G (2018) Surgical results and complications of anterior controllable antedisplacement fusion as a revision surgery after initial posterior surgery for cervical myelopathy due to ossification of the posterior longitudinal ligament. J Clin Neurosci 56:21–27. https://doi.org/10.1016/j.jocn.2018.08.014
Takase H, Murata H, Sato M, Tanaka T, Miyazaki R, Yoshizumi T, Tateishi K, Kawahara N, Yamamoto T (2018) Delayed C5 palsy after anterior cervical decompression surgery: preoperative foraminal stenosis and postoperative spinal cord shift increase the risk of palsy. World Neurosurg 120:e1107–e1119. https://doi.org/10.1016/j.wneu.2018.08.240
Acknowledgements
The study was supported by the National Natural Science Foundation of China, Grant/Award Numbers: No. 81871828, No. 81702141.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
No conflicts of interest need to be explained.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sun, K., Wang, S., Huan, L. et al. Analysis of the spinal cord angle for severe cervical ossification of the posterior longitudinal ligament: comparison between anterior controllable antedisplacement and fusion (ACAF) and posterior laminectomy. Eur Spine J 29, 1001–1012 (2020). https://doi.org/10.1007/s00586-019-06216-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-019-06216-6