Abstract
Objectives
To investigate the cervical spinal canal diameters variance under positional MRI, and also the relationship between cervical canal diameter variance rate and grade of degeneration.
Methods
From January 2013 to January 2015, a consecutive of 273 symptomatic patients (166 males and 207 females) with an average age of 44.6 years (range 21–89 years) underwent positional cervical MRI. T2-weighted sagittal images of 1638 cervical intervertebral discs from 273 subjects were classified into five grades. The canal diameter and canal diameter variance rate at three positions and their comparison among five grade of degeneration were evaluated. The measurements were tabulated and analyzed using SPSS. 13.0. p values less than 0.05 were considered to indicate a statistically significant difference.
Results
The sagittal cervical canal diameter at the C5/6 level were the smallest compared with the other levels regardless of neutral, flexion or extension positions, C5/6 level had the largest canal diameter variance rate in both flexion and extension (8.14 ∓ 0.38 and 7.81 ∓ 0.31 %, respectively), second was C4/5 level (7.65 ∓ 0.39 and 7.67 ∓ 0.32 %, respectively). A total of 1638 discs were classified into 5 groups, each level showed the similar tendency that no matter what position, with the increasing grade of degenerative disc degree, spine canal diameter decreased gradually. For C5/6 under extension and flexion position, significant difference was also noted between grade 2 and 3; For C4/5 under extension position, significant difference existed between grade 1 and 2, grade 1 and 3, while under flexion position, significant difference existed between grade 2 and 4, and the results also showed no significant difference at the same degree of degeneration on both levels between extension and flexion position.
Conclusions
C5/6 and C4/5 is of higher risk of suffering SCI than other levels, C4/5 level predispose SCI at earlier stage than C5/6, patients presenting with intermediate signal and slight decreased disc height on T2 weighted MRI at C4/5 level should be paid attention.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
To the best of our knowledge, few reports have thus far described the differences between the pathogenetic mechanisms of cervical canal stenosis at different segments using positional MRI [1–4]. In this study, we investigated the cervical spinal canal diameters changing under positional MRI, and also the relationship between cervical canal diameter changing under positional MRI and degree of degeneration was examined.
Materials and methods
From January 2013 to January 2015, a consecutive of 273 symptomatic patients (166 males and 207 females) with an average age of 44.6 years (range 21–89 years) underwent positional cervical MRI. All these patients presented with neck pain with or without neurogenic symptoms. None of the subjects had previously undergone any spinal surgery. Most patients were in a chronic process, and showed a progressive exacerbation. The duration of the disease ranged from 5 months to 9 years, with a mean of 4.3 years. After the onset of the disease, all subjects experienced neck and shoulder discomfort, numbness of limbs, unsteady gait and zonesthesia in chest or abdomen. Physical examination showed that compression was mainly on lower cervical spinal cord, and different degrees of incomplete paralysis were detected in limbs. Cervical spinal cord function of all patients with CSM was measured using Japanese Orthopaedic Association (JOA) scoring method. JOA score of all CSM patients ranged from 6 to 14, with a mean score of 12.21.
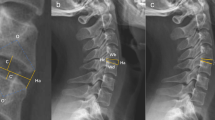
Positional MRI
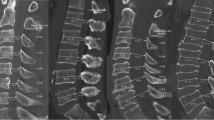
The measurements of the cervical spine in flexion, neutrality and extension were performed with the patient in the supine position. Cervical flexion was initiated by placing a 20 cm pillow below the head. Cervical extension was initiated by placing the pillow below the upper thoracic vertebrae. The MRI machine used is a 1.5T MRI scanner (Siemens Magnetom Espree Pink 1.5T MRI System). The technical details included XJ Gradients (33 mT/m @ 125 T/m/s), XQ Gradients (45 mT/m @ 200 T/m/s), Tim [204 × 24], [204 × 48], [204 × 64]. All sequences were acquired without fat saturation. The data obtained from the MR images were recorded on a computer for subsequent calculations. Sagittal MR images were analyzed in three positions—flexion, neutral, and extension. The sagittal diameter of cervical canal at six disc levels (C2/3, C3/4, C4/5, C5/6, C6/7, C7/T1) of each position were recorded. Radiographs were accessed by a DICOM-based Radworks 5.1 (Applicare Medical Imaging BV, Zeist, The Netherlands) computer software program. The measurements of T2-weighted disc degree and sagittal diameter of spinal canal were recorded by a spine surgeon and a radiographer, reliability assessment was based on intraclass correlation (ICC), which was an appropriate statistical tool for this analysis, and the final results were the means of both measurements.
Cervical intervertebral disc degeneration
A comprehensive grading system for cervical disc degeneration was obtained by previously reported systems of classifying cervical intervertebral disc degeneration based on degenerative changes in the cervical functional spinal unit (FSU) [5]. Accordingly, T2-weighted sagittal images of 1638 cervical intervertebral discs from 273 subjects were classified into five grades which defined as following: grade 0: normal disc; grade 1: nucleus hyperintensive signal with normal disc height; grade 2: nucleus intermediate signal with slight decreased disc height; grade 3: nucleus hypointensive signal with decreased disc height and disc herniation/osteophyte; grade 4: nucleus hypotintensive signal with collapsed disc height and disc herniation/osteophyte.
We defined flexion variance rate of spine canal as (flexion spine canal diameter at disc level−neutral spine diameter at same disc level)/neutral spine diameter at same disc level, and extension variance rate of spine canal as (extension spine canal diameter at disc level−neutral spine diameter at same disc level)/neutral spine diameter at same disc level association between disc degree and spine canal diameter variance rate at each disc level under three different positions were evaluated.
Data were expressed as mean ± standard deviation. The measurements were tabulated and analyzed using JMP ver. 8.0 (SAS Institute Inc., Cary, NC, USA). Differences between groups were examined for statistical significance using the Mann–Whitney test. p values less than 0.05 were considered to indicate a statistically significant difference.
Results
Our results showed good reliability of measurements of cervical spine canal diameter and disc degeneration grade with the ICC was 0.86 (95 % CI 0.81–0.92) and 0.97 (95 % CI 0.95–0.99).
Sagittal cervical canal diameter
The sagittal cervical canal diameter at the C5/6 level was the smallest compared with the other levels regardless of neutral, flexion or extension positions (11.92 ∓ 0.11, 12.39 ∓ 0.11, 11.97 ∓ 0.10, respectively), and second was C4/5 level (12.18 ∓ 0.10, 12.64 ∓ 0.10 and 11.70 ∓ 0.11 for neutral, flexion and extension, respectively). The cervical canal diameter increased from C5/6 to C7/T1 and from C5/6 to C2/3 regardless of positions, and C2/3 level had the largest canal diameter at flexion position, while C7/T1 had the largest canal diameter at neutral and extension position (Table 1).
We measured the extension and flexion variance rate at each disc level (Table 2). C5/6, C7/T1 reached the largest extension variance (0.91 ∓ 0.04) and flexion variance (0.92 ∓ 0.05), respectively, followed them was C4/5 (0.91 ∓ 0.04) at extension and C5/6 (0.91 ∓ 0.04) at flexion, however, when considering the variance rate, C5/6 level had the largest value in both flexion and extension (8.14 ∓ 0.38 and 7.81 ∓ 0.31 %, respectively), followed them was C4/5 level (7.65 ∓ 0.39 and 7.67 ∓ 0.32 %, respectively). We noted that although C2/3 had larger flexion and extension variance rate than C5/6, the standard deviation were larger significantly than that of C5/6, which revealed C2/3 variance rate had the characteristics of discrete distribution. We concluded from Tables 1 and 2 that C5/6 level was the most prevalent level of developing spinal cord injury without radiographic abnormalities due to its largest extension and flexion variance rate and narrowest canal diameter, and second was C4/5 level, which corroborate the clinical findings.
To evaluate the association between degree of degeneration and canal diameter variance rate of each level, a total of 1638 discs were classified into 5 groups (group 0-normal, group 4-severe degeneration). Table 3 listed number of each group under every level. Group 2 had largest number of discs (n = 871), and group 1 had second largest number of discs (n = 635). Table 4 listed the each cervical canal diameter in each group under three different positions. Each level showed the similar tendency that no matter what position, with the increasing grade of degenerative disc degree, spine canal diameter decreased gradually, normal disc had the largest canal diameter and grade 4 disc had the smallest canal diameter.
For every level, we compared the flexion and extension variance rate between each two different degenerative degree (0/1, 1/2, 2/3, 3/4, 0/2, 1/3, 2/4, 0/3, 1/4, 0/4), Table 5 showed extension variance rate comparison, Table 6 showed flexion variance rate comparison. For C5/6 under extension position, significant difference was noted between grade 2 and 3, no difference between grade 0 and 1, grade 1 and 2, grade 0 and 2, grade 3 and 4; while under flexion position, significant difference also existed between grade 2 and 3, no difference between grade 0 and 1, grade 1 and 2, grade 0 and 2, grade 3 and 4. For C4/5 under extension position, significant difference existed between grade 1 and 2, grade 1 and 3, no difference between grade 0 and 1, grade 2 and 3, grade 3 and 4; while under flexion position, significant difference existed between grade 2 and 4, no difference between grade 0 and 1, grade 1 and 2, 2 and 3, 3 and 4, 1 and 3, 0 and 2. From these data, we concluded for C5/6 level, degenerative degree of grade 3 is a demarcation, grade ≥3 had larger disc canal diameter variance rate under flexion–extension movement than grade <3; and for C4/5 level, under extension position, grade 2 is a demarcation, while under flexion position, grade 4 is a demarcation. Considering the canal diameter variance at C4/5 level under extension position was a little bit larger than flexion position (Table 2), to evaluate whether this difference is significant, we compared the difference of variance rate between extension and flexion position at each degree of degeneration for C4/5 and C5/6, the results showed no significant difference at the same degree of degeneration on both levels (Table 7), which indicate extension position exert similar impact on canal diameter with flexion position. As a result, we considered grade 2 was a demarcation for C4/5, grade ≥2 had larger disc canal diameter variance rate under flexion–extension movement than grade <2. The process of degeneration was an age-related phenomenon, so C4/5 tended to suffer spinal cord injury at earlier stage than C5/6.
Discussion
Previous studies revealed that flexion and extension movement changed the cervical spinal canal diameter and its components [6–8]. Our study obtained more accurate results using positional MRI, unlike previous studies that used conventional roentgenography or neutral MRI.
Ruegg et al. [9] investigate the relevance of 2D MRI measurements for the diagnosis of critical cervical spinal canal stenosis. They found that the cord-canal-area ratio (>0.8) or the space available for the cord (<1.2 mm) measured on MR images can be used to reliably identify patients at risk for acute CSCI after a minor trauma to the cervical spine. Ono et al. [10] reported that the AP compression ratio of <0.4, which was measured by dividing the sagittal diameter by the transverse diameter of the spinal cord, indicates an abnormal neurological function. Matsuura et al. [11] in their comparative study using CT scans, reported significant differences between normal patients and those with an injury to the spinal cord with regard to the shape and transverse diameter of the spinal canal rather than the area of the spinal cord itself. However, these methods used to determine the shape of the spinal cord and compression ratio can be useful when assessing the severity of a lesion caused by a disc protrusion or osteophytes. However, their use is limited when evaluating traumatic spinal stenosis without severe compression. In Kyung’s [12] study, the neurological outcome was not affected by the sagittal diameter of the spinal canal and the spinal cord area measured on the MRI images (p = 0.539, 0.317) while there was a significant correlation between Pavlov’s ratio on the plain radiographs and the development of clinical symptoms (p = 0.006). Although the width of the spinal canal area was inversely proportion to the severity of the neurological injury, no statistical significance was found (p = 0.158). Torg et al. [13] reported that a ratio of 0.80 or less in spinal canal diameter to the vertebral body diameter indicated a high sensitivity for transient cervical neuropraxia. However, in this study, the diameters and ratios were derived from lateral radiographs, and the obtained measurements were significantly smaller than those obtained from MRI images. Lee et al. [14] used MRI to measure the canal diameter at disc level and disc diameter in 20 SCI patients, and draw the conclusion of complete SCI is associated with a small canal diameter at disc level to disc diameter ration at C4/5 and with extensive high signal intensity in MRI. In our studies, we undertook the positional MRI to document the extension-flexion canal diameter of cervical spine, and found the cervical canal diameter at disc level reached minimum at C5/6 level regardless of positions, and C5/6 also had largest canal diameter variance rate, the second was C4/5 level, which means C5/6 and C4/5 have higher risk of suffering SCI than other levels. This was corroborated by Guo’s [15] epidemiological study, in their study, they found the C4/5 segment was the most common level, accounting for 48.7 % of all SCI, C5/6 was the next, which accounted for 30.5 %. For the adult population, the most prevalent predominant level of cervical myelopathy or spondylosis is C4/5, followed by C5/6 and C3/4 [16, 17]. Meanwhile, we noted whether C7/T1 or C2/3 showed no association between degree of degeneration and canal diameter variance rate, this partially explained few incidence of SCI at these 2 levels. The characteristics of injury level distribution due to the age group indirectly reveal that, secondary degeneration may participate in the onset of adult SCI.
Morishita et al. [18] in their papers used degenerative grade as a parameter, and compared the grade between different canal diameter groups, however, they did not evaluate the association between degenerative grade and AP diameter. For cervical spine myelopathy suffering SCI, the most important thing is canal diameter variance rate, so in our study, we evaluated the association between disc degenerative degree and canal diameter variance rate under extension and flexion position, and found different level predisposed SCI at different disc degenerative degree, for C5/6, the disc-level canal diameter variance rate of disc degenerative degree ≥3 showed significant difference compared with that of disc degenerative degree <2, grade 3 showed following characteristics such, as nucleus hypointensive signal with decreased disc height and disc herniation/osteophyte, while grade 2 presented with the features of nucleus intermediate signal with slight decreased disc height, so C5/6 with degenerative degree ≥3 has larger canal diameter variance rate and is of high risk of suffering SCI than other degree. For C4/5, grade ≥2 has larger canal diameter variance rate and i is high risk of suffering SCI. Furthermore, disc degeneration is an age-related process, grade 2 degeneration predispose C4/5 at SCI, while grade 3 degeneration predispose C5/6 at SCI, we draw the conclusion that C4/5 level predispose SCI at earlier stage than C5/6, we should pay attention to patients presenting with intermediate signal and slight decreased disc height in T2 weighted MRI at C4/5 level, they should be cautious for their daily life, especially the neck movement.
This study had some limitations. It is known that the segmental range of motion changes depending on the cervical curves [19]. This type of research should consider the differences in the original cervical curve. Second, this study focused mainly on the MRI findings and statistical analysis of symptomatic patients, the results need clinical test in SCI patients. Third, due to the limitation of MRI machine, patients in our study can not be placed in upright position and the flexion–extension movement is passive, we are unable to consider the effect of gravity and spinal curve on measurement, so the kinematic study on upright patients and active flexion and extension movement should performed during the future study. Despite these limitations, we consider the current results obtained from MRI measurements clearly defined predispotisions of SCI. In future studies, it may be helpful for clinician to predict the risk factors of SCI and correspondent intervention.
In conclusion, C5/6 and C4/5 have higher risk of suffering SCI than other levels due to their lowest canal diameter and largest canal diameter variance rate, C5/6 with degenerative degree ≥3 is of high risk of suffering SCI, while for C4/5, grade ≥2 has high risk of suffering SCI, C4/5 tended to suffer SCI at earlier stage than C5/6.
References
Dolan RS, Butler JS et al (2016) Mechanical and cellular processes driving cervical myelopathy. World J Orthop 7(1):20–29
Baptiste DC, Fehlings MG (2006) Pathophysiology of cervical myelopathy. Spine J 6:190S–197S
Henderson FC, Geddes JF, Vaccaro AR, Woodard E, Berry KJ, Benzel EC (2005) Stretch-associated injury in cervical spondylotic myelopathy: new concept and review. Neurosurgery 56:1101–1103
Yu WD, Williams SL (2006) Spinal imaging: radiographs, computed tomography, and magnetic resonance imaging. Spine 3. AAOS 6:57–67
Kettler A, Rohlmann F, Neidlinger-Wilke C, Werner K, Claes L, Wilke HJ (2006) Validity and interobserver agreement of a new radiographic grading system for intervertebral disc degeneration: part 2. Cervical spine. Eur Spine J 15:732–741
Hayashi T, Daubs MD, Suzuki A et al (2014) Effect of Modic changes on spinal canal stenosis and segmental motion in cervical spine. Eur Spine J 23(8):1737–1742
Emrah S, Bayan A et al (2015) The occupancy of the components in the cervical spine and their changes with extension and flexion. Glob Spine J 5:396–405
Fulong D, Cailiang S et al (2012) Measurement of voume-occupying rate of cervical spinal canal and its role in cervical spondylotic myelopathy. Eur Spine J 22:1152–1157
Ruegg TB, Wicki AG et al (2015) The diagnostic value of magnetic resonance imaging measurements for assessing cervical spinal canal stenosis. J Neurosurg Spine 22(3):230–236
Ono K, Ota H, Tada K, Yamamoto T (1977) Cervical myelopathy secondary to multiple spondylotic protrusion: a clinico-pathologic study. Spine 2(2):109–125
Matsuura P, Waters RL, Adkins RH, Rothman S, Gurbani N, Sie I (1989) Comparison of computerized tomography parameters of the cervical spine in normal control subjects and spinal cord-injured patients. J Bone Joint Surg Am 71(2):183–188
Song KJ, Choi BW et al (2009) The relationship between spinal stenosis and neurological outcome in traumatic cervical spine injury: an analysis using Pavlov’s ratio, spinal cord area, and spinal canal area. Clin Orthop Surg 1:11–18
Torg JS, Naranja RJ Jr, Pavlov H, Galinat BJ, Warren R, Stine RA (1996) The relationship of developmental narrowing of the cervical spinal canal to reversible and irreversible injury of the cervical spinal cord in football players. J Bone Joint Surg Am 78(9):1308–1314
Lee SE, Chung CK (2015) Risk prediction for development of traumatic cervical spinal cord injury without spinal instability. Global Spine J 3(12):315–321
Guo H, Liu J, Qi X et al (2012) Epidemiological characteristics of adult SCIWORA in Tianjin, China: a preliminary study. Eur Spine J 21:165–171
Lu J, Wu X, Li Y, Kong X (2008) Surgical results of anterior corpectomy in the aged patients with cervical myelopathy. Eur Spine J 17:129–135
Sieh KM, Leung SM, Lam JS et al (2009) The use of average Pavlov ratio to predict the risk of post operative upper limb palsy after posterior cervical decompression. J Orthop Surg Res 4:24
Morishita Y, Naito M et al (2009) The relationship between the cervical spinal canal diameter and the pathological changes in the cervical spine. Eur Spine J 18:877–883
Takeshima T, Omokawa S, Takaoka T, Araki M, Ueda Y, Takakura Y (2002) Sagittal alignment of cervical flexion and extension: lateral radiographic analysis. Spine (Phila Pa 1976) 27:E348–E355
Acknowledgments
This study received no funding and Grants from any institutional or governmental support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Jiang, X., Chen, D., Lou, Y. et al. Kinematic analysis of cervical spine canal diameter and its association with grade of degeneration. Eur Spine J 25, 2166–2172 (2016). https://doi.org/10.1007/s00586-016-4624-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00586-016-4624-z