Abstract
Fibroblast growth factor 21 (FGF21), known for its metabolic effects, is a potential target for treating diabetes. This study aimed to assess the FGF21 levels and insulin resistance index after resistance and endurance training in the male diabetic rat’s heart tissue. Twenty-four streptozotocin-induced diabetic rats have been divided into four groups randomly: endurance training (6), resistance training (6), sham (6), and healthy (6). The endurance training group trained on the treadmill 5 sessions/week for 4 weeks, while the resistance training group trained 6 sessions/week on the ladder. Before and following the first and last training sessions, blood samples had been collected to measure FBS, FBI, and HOMA. Following the final training session, heart tissue was lysed, and we determined the level of FGF21 by ELISA technique. Within- and between-group differences were measured in FBS, FBI, and HOMA-IR through analyzing data using pairwise samples T tests and repeated-measure ANOVA test respectively and the differences in FGF21 levels between groups through a one-way ANOVA test through SPSS software. The resistance training group had more FGF21 protein than the other groups. The endurance and resistance training groups did not differ significantly (P = 0.249). However, there was a significant difference from the sham group (P = 0.008). In addition, fasting blood sugar level and insulin resistance index reduced significantly in both groups of endurance training (P = 0.001, P = 0.005) and resistance training (P = 0.004, P = 0.01), and there were no significant differences between groups (P > 0.05). Both endurance and resistance training reduce fasting blood sugar and insulin resistance index. Furthermore, resistance training increases the amount of FGF21 in the heart tissue. However, there are no differences with endurance training.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Diabetes affects metabolism and causes hyperglycemia. It has a significant role in cardiovascular disorders, physical injuries, mortality, and health costs. Diabetes is a widespread risk factor for cardiovascular disorders and contributes to mortality rates (Sharma et al. 2020; Joseph et al. 2022). In the long run, diabetes leads to complications in the kidneys, eyes, heart, and nerves (Lopez et al. 2006). Cardiovascular disease is the primary reason for mortality among diabetics, not merely hypertension, and CAD (coronary artery disease), but mainly because of the direct diabetes influences on the heart, regardless of any pathological conditions (Ernande and Derumeaux 2012). Type 2 diabetes could be treated with FGF21, according to another study (Dostalova et al. 2009; Woo et al. 2013). The family of the fibroblast growth factor FGF21 is produced in the pancreas, adipose tissue, and liver. Glucose regulation and lipid metabolism depend on FGF21 (Dutchak et al. 2012). It increases glucose uptake through the glucose transporter 4 (GLUT4) (Wente et al. 2006), participates in insulin production, inhibits glucagon secretion, and induces pancreatic beta cell expression, which help maintain cell function and decrease blood glucose (Zhang et al. 2008).
Exercise is a proven approach to preventing and managing type 2 diabetes, its associated cardiovascular complications, and mortality (Powers et al. 2002; Riddell and Iscoe 2006). Exercise training can increase GLUT4 in the muscle sarcolemma. Furthermore, exercise training causes fatty acid oxidation and prevents their accumulation in cells of the muscle and promotes the transfer of GLUT4 to the sarcolemma of muscle cells (Ku et al. 2010). Endurance training increases the aerobic capacity of these tissues, allowing for more glucose utilization (Ignarro et al. 2007). This action is partly due to the increase in GLUT4 expression in the muscle sarcolemma. The activation of the AMPK signaling pathway stimulates the translocation of GLUT4 to the membrane of the cell, facilitating insulin activity and decreasing the need for insulin, consequently reducing the HOMA-IR index.
Previous studies investigated the influences of resistance, endurance, and concurrent training on FGF21 and the resistance index of insulin. Matsui et al. (2022) researched the effect of aerobic exercise training on FGF in correlation with glucose tolerance and concluded that aerobic training reduces FGF and increases glucose tolerance in overweight and obese men (Matsui et al. 2022). Gharakhanlou and Bonab (2022) investigated the impact of water training for 12 weeks on FGF21, glycemic index, and lipid profile within patients with diabetes type 2, an increase in FGF21, a decrease in resistance of insulin, and an improvement in the lipid profile (Gharakhanlou and Bonab 2022). Taniguchi et al. showed that 3 weeks of endurance training reduced serum FGF21 and hepatic fat levels without affecting weight in patients with fatty liver (Taniguchi et al. 2016). Resistance training increased GLUT4 protein content, protein kinase B, insulin receptors, and glycogen synthase. Kim and Sang (2017) demonstrated that resistance training for 12 weeks increased irisin and FGF21 in diabetic rats’ skeletal muscles (Kim and Sang 2017). Some researchers have studied the impacts of concurrent training, with some showing that resistance training is more effective than endurance training on insulin sensitivity and glucose homeostasis. Keihanian et al. (2019) showed the influence of 8 weeks of concurrent training on FGF21 in diabetic men and showed that resistance training had a more significant impact (Keihanian et al. 2019). However, Astinchap et al. (2021) investigated the effect of 8 weeks of concurrent training on insulin resistance index (HOMA), FGF21, and beta Klotho (BKL) demonstrated a reduction in insulin resistance, FGF21, and an increase in BKL by no distinction among resistance and endurance training (Astinchap et al. 2021). García-Hermoso et al. (2023) in their review manuscript reported exercise training is an anti-inflammation therapy and metabolism-improving strategy with minimal side effects for type 2 diabetes mellitus patients (García-Hermoso et al. 2023).
Considering the positive exercise training influence on insulin resistance index, FGF21, and the lack of consensus about the resistance and endurance training influences on these parameters, the purpose of this study was to determine the impact of 4 weeks of resistance and endurance training on fasting blood sugar, insulin resistance index (HOMA-IR), and FGF21 levels in the male Wistar diabetic rat’s heart tissue.
Material and methods
All procedures in this study have been endorsed by the Ethics Committee of Shahid Rajaee Teacher Training University of Tehran (IRSRTTU.SSF.2020.111). According to statistical reference (Hill et al. 2006) and according to the ethical recommendation of the local ethics committee, 30 male Wistar rats with 200 to 250 g of weight and ages between 8 and 12 weeks were selected randomly, six rats died and 24 rats remained, which were randomized to four groups: endurance training (6), resistance training (6), sham (6), and healthy (6). The rats were housed in polycarbonate cages made by Razi Rad exclusive company, with dimensions 15 × 15 × 30 cm. They were housed in a room maintained at 18–23 °C, 50% relative humidity, with 12-h day-night cycles. (The natural day-and-night division through the timer adjusted the darkness and brightness of the laboratory environment.) All rats were provided ad libitum accessibility to nutritional compacts and PVC drinking containers filled with urban water. Lee index was calculated by measuring the rats’ weight and height (weight 0.33/Naso-anal length). Fasting blood sugar (FBS) and insulin resistance index were assessed through a pre-test by taking blood from rats’ tails. Diabetes induced by 30 mg/kg injecting a single dose of streptozotocin dissolved in sodium citrate buffer with pH = 4.5 after a 4-week available high-fat diet (diet with 45% animal fat) was assigned to them (Holmes et al. 2015). A glucometer was used to measure diabetes affirmed by fasting blood glucose levels higher than 300 mg/dL, 4 days following the injection (Nourzad et al. 2022). Insulin levels in the rats’ plasma were measured using the ELISA method with the Insulin kit of Raybiotech company and the ELISA reader DA-3200. The insulin resistance index (HOMA-IR index) has been computed using the formula below:
where FBS refers to fasting blood sugar, and FBI refers to fasting blood insulin.
Following the pre-test, the rats underwent a familiarization and adaptation program—initially, they became familiar with the training room and environment. After 1 week, endurance training rats used the stationary treadmill without activating it, while resistance training rats were placed on a ladder to become familiar with the training equipment. The rats of the sham group were only brought to the training room instead of being placed in the training programs. Two days before the start of the exercise, the treadmill was turned on at the speed of 5 m/min, the endurance training rats were placed on the treadmill, and the resistance training rats were directed to climb a ladder without any added weights to become familiar with the exercise. After the familiarization phase, the exercise training programs began. The endurance training rats run on the treadmill for 5 sessions/week for 4 weeks. The endurance training protocol included a warm-up phase (speed of activity = 7 m/min on the treadmill for 3 min), followed by a desired speed of 15 m/min for 25 min at the first session. The speed was then increased by 1 m per min weekly, and after the training, the speed was gradually decreased to reach the initial speed for a cool-down phase (Tanoorsaz, et al. 2017). In addition, the resistance training rats trained on the ladder for 6 sessions/week for 4 weeks. In this program, in each session, the rats had to climb the ladder for three sets of 6, the rest between each round was 1 min, and the rest between each set was 3 min. Finally, after the training, they climbed the ladder twice without weights to cool down. In each session, weighing tied to the rats’ tails was based on a percentage of their body weight (Karimian et al. 2015). After 4 weeks of training, the rats were given food and water following their last training session. After 1 day, food and water were removed to induce fasting for 10 to 12 h. The rats were anesthetized with xylazine (10 mg/kg) and ketamine (100 mg/kg) and then killed by moving the neck (Nourzad et al. 2022). Blood was collected from their hearts, and blood sugar and levels of insulin were recalculated to assess the index of HOMA-IR as a post-test. The heart tissue was carefully separated and stored in a special kit in a freezer − 80 °C. The heart tissue was then lysed according to the instructions of the cell lysis solution and then centrifuged for 15 min at 4 °C. The supernatant was analyzed for protein measurement (FGF21 level) using an ELISA reader DA-3200. Paired sample T tests and repeated-measure ANOVA tests were used to measure within- and between-group differences in FBS, FBI, and HOMA-IR, respectively. Differences in FGF21 levels through groups were analyzed by the one-way ANOVA and LSD post hoc tests through SPSS (version 25) software.
Results
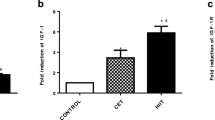
The study subjects’ information is described in Table 1, including their weight, Lee index, and height. The resistance training rats had higher levels of FGF21 protein than the other groups, and no notable distinction was found among the endurance and resistance training rats (P = 0.249). However, a considerable distinction was found among the sham and resistance training rats (P = 0.008). Moreover, both the resistance and endurance training rats showed a notable reduction in FBS level and HOMA-IR (P < 0.05). The FGF21 levels and the differences in fasting blood sugar level, insulin, and HOMA-IR between the groups from the pretest to the post-test are presented in Table 2 and Fig. 1.
A The differences in FGF21 levels between groups through one-way ANOVA and LSD post hoc test. ns, no significant difference; ET, endurance training group; RT, resistance training group; SH, Sham group; H, health group; *p ≤ 0.05; **p < 0.01. B The within- and between-group differences in fasting blood sugar level (FBS), through paired-sample T test and repeated-measure ANOVA test. ns, no significant difference; ET, endurance training group; RT, resistance training group; SH, Sham group; H, health group; *p ≤ 0.05; **p < 0.01; ***p < 0.001. C The within- and between-group differences in insulin level, through paired sample T test and repeated-measure ANOVA test. ns, no significant difference; ET, endurance training group, RT, resistance training group; SH, Sham group; H, health group. D The within- and between-group differences in insulin resistance index (HOMA-IR), through paired-sample T test and repeated-measure ANOVA test. ns, no significant difference; ET, endurance training group; RT, resistance training group; SH, Sham group; H, health group; **p < 0.01
Discussion
This study induced type 2 diabetes by streptozotocin injection and increased fasting blood glucose and HOMA-IR index. Four weeks of resistance and endurance training reduced fasting plasma glucose concentration and HOMA-IR index significantly compared to before exercise training. Exercise training can increase GLUT4 in muscle sarcolemma (Wente et al. 2006). In addition, exercise training causes fatty acid oxidation and prevents their accumulation in cells of muscle and eases the GLUT4 transfer to the surface of the cell (Jonker et al. 2013). The findings of this research agreed with the outcomes of the Messi et al. investigation (Massi-Benedetti 1996) and disagreed with the data of Cauza et al. and Segal et al. Among the reasons for the difference between studies, it should be mentioned the difference in the type of subjects, training, and the distinction in the severity of the exercise training (Cauza et al. 2005 and Segal et al.1991). For example, the samples in Cauza’s study included elderly men and women (Cauza et al. 2005). Four weeks of resistance and endurance training raised the amount of insulin, but it was not significant, and it was distinct from the research results of Duncan et al. (2003). Of course, when diabetes is induced with STZ, pancreatic β-cells are destroyed and insulin is not secreted ( Duncan et al.2003; Sharma et al. 2023). In this case, the amount of insulin secretion is increased after exercise, and the reason for not being significant can be due to the small number of rats in the groups. In addition, the descriptive findings of the current investigation demonstrated that the FGF21 protein was greater in the resistance training rats compared to other groups, but in inferential analysis, the resistance and endurance training rats did not show any notable distinction, while only the resistance training rats exhibited a considerable difference when compared to the sham group. Studies have shown that FGF-21 contributes significantly to regulating lipid and glucose homeostasis (Gharakhanlou and Bonab 2022; Keihanian et al. 2019; Astinchap et al. 2021). FGF-21 can potentially increase glucose uptake by upregulating GLUT4 and serve as a treatment for diabetes type 2. Keihanian et al. (2019) have shown that both endurance and resistance training can improve some symptoms of type 2 diabetes, for example, high LDL, HOMA-IR, and FBS, as well as hepatokines (FGF-21 and fetuin-A-B), through the upstream regulation of inhibitor tyrosine kinase receptor of insulin in the liver and increase insulin sensitivity by increasing GLUT4 protein, glycogen and hexokinase production, and glucose delivery to muscle (Keihanian et al. 2019). In addition, we found no notable distinction among the endurance and resistance training rats in the amount of FGF21. This case is consistent with the absence of significant distinction in levels of insulin resistance and FBS in both resistance and endurance training groups. The decrease in blood sugar caused by endurance training can be due to the development in glucose uptake with consumer tissues like muscle and fat tissue and the increase in glucose transporters (GLUT4), membrane transport of this carrier, and the increase in glucose oxidation in the consumer tissue (Nourzad et al. 2022). Following endurance training and in parallel with increasing the aerobic capacity of consumer tissues, a suitable platform is provided for more use of glucose. Part of this action is due to the increase of GLUT4 in the sarcolemma of the muscle. Triggering of the AMPK signaling trajectory increases the GLUT4 translocation to the membrane of the cell. The increase in GLUT4 expression in the muscle level causes the proper activity of insulin in taking glucose into the tissue. And the consequence of this issue is the reduction of the need for high levels of insulin and as a result its reduction (Gharakhanlou and Bonab 2022; Taniguchi et al. 2016). Furthermore, in resistance training, the effect of stronger muscle contractions can be mentioned. Muscle contractions activate the PI3K/Akt signaling axis leading to the FGF21 production and its increase. The activation of the PI3K/Akt signaling axis in skeletal muscles, which is mediated by insulin, is responsible for the positive regulation of FGF-21 (Astinchap et al. 2021). It has also been reported that FGF21 is a factor that increases insulin sensitivity and is downstream of the PPAR-γ receptor, which regulates lipid metabolism and is associated with reducing hyperglycemia. Recently, the PPAR γ-FGF axis has been identified to maintain metabolic homeostasis and insulin sensitivity in adipose tissue. In addition, FGF21 reduces blood sugar by maintaining the function of B cells and stimulating the increase of hepatic glucose uptake by increasing the activity of hepatic glucokinase. Galandari et al. (2021) have shown a significant relationship between increasing FGF and decreasing insulin resistance (Ghalandari et al. 2021). In this line, long-term moderate-intensity endurance training likewise regulates the content of liver fat and the AMPK/SIRT1 axis, thus reducing resistance to FGF21, which may result from increased FGF21 receptor expression (Astinchap et al. 2021). In this regard, due to the possible effect of the level of FGF21 receptors in reducing insulin resistance in diabetic patients and the lack of examination of their level in this study (which is a constraint of this study), it is suggested future studies examine the simultaneous changes of FGF21 and its receptors due to resistance and endurance training. Furthermore, the small number of rats in the groups was another limitation of this study. We suggest that future researchers study more diabetic rats.
References
Astinchap A, Monazzami A, Fereidoonfara K, Rahimi Z, Rahimi M (2021) Modulation of fibroblast growth factor-21 and βklotho proteins expression in type 2 diabetic women with non-alcoholic fatty liver disease following endurance and strength training. Hepat Mon 21(7)
Cauza E, Hanusch-Enserer U, Strasser B, Ludvik B, Metz-Schimmerl S, Pacini G, Haber P (2005) The relative benefits of endurance and strength training on the metabolic factors and muscle function of people with type 2 diabetes mellitus. Arch Phys Med Rehabil 86(8):1527–1533
Dostalova I, Haluzikova D, Haluzik M (2009) Fibroblast growth factor 21: a novel metabolic regulator with potential therapeutic properties in obesity/type 2 diabetes mellitus. Physiol Res 58(1)
Duncan GE et al (2003) Exercise training, without weight loss, increases insulin sensitivity and postheparin plasma lipase activity in previously sedentary adults. Diabetes Care 26(3):557–562
Dutchak PA, Katafuchi T, Bookout AL, Choi JH, Ruth TY, Mangelsdorf DJ, Kliewer SA (2012) Fibroblast growth factor-21 regulates PPARγ activity and the antidiabetic actions of thiazolidinediones. Cell 148(3):556–567
Ernande L, Derumeaux G (2012) Diabetic cardiomyopathy: myth or reality? Arch Cardiovasc Dis 105(4):218–225
García-Hermoso A, Ramírez-Vélez R, Díez J, González A, Izquierdo M (2023) Exercise training-induced changes in exerkine concentrations may be relevant to the metabolic control of type 2 diabetes mellitus patients: a systematic review and meta-analysis of randomized controlled trials. J Sport Health Sci 12(2):147–157
Ghalandari F, Nikooie R, Moflehi D (2021) The effect of 8 weeks of endurance training on serum levels of fibroblast growth factor 1 and insulin resistance in type 2 diabetic women. J Appl Exerc Physiol 17(34):95–108
Gharakhanlou BJ, Bonab SB (2022) The effect of 12 weeks of training in water on serum levels of SIRT1 and FGF-21, glycemic index, and lipid profile in patients with type 2 diabetes. Int J Diabetes Dev C 1–8
Hill T, Lewicki P, Lewicki P (2006) Statistics: methods and applications: a comprehensive reference for science, industry, and data mining. StatSoft, Inc.
Holmes A, Coppey LJ, Davidson EP (2015) Yorek MA (2015) Rat models of diet-induced obesity and high fat/low dose streptozotocin type 2 diabetes: effect of reversal of high fat diet compared to treatment with enalapril or menhaden oil on glucose utilization and neuropathic endpoints. J Diabetes Res 1:307285
Ignarro LJ, Balestrieri ML, Napoli C (2007) Nutrition, physical activity, and cardiovascular disease: an update. Cardiovasc Res 73(2):326–340
Jonker JT, de Mol P, de Vries ST, Widya RL, Hammer S, van Schinkel LD, ... & Lamb HJ (2013) Exercise and type 2 diabetes mellitus: changes in tissue-specific fat distribution and cardiac function. Radiology 269(2):434–442
Joseph, J. J., Deedwania, P., Acharya, T., Aguilar, D., Bhatt, D. L., Chyun, D. A., … & American Heart Association Diabetes Committee of the Council on Lifestyle and Cardiometabolic Health; Council on Arteriosclerosis, Thrombosis and Vascular Biology; Council on Clinical Cardiology; and Council on Hypertension (2022) Comprehensive management of cardiovascular risk factors for adults with type 2 diabetes: a scientific statement from the American Heart Association. Circulation 145(9):e722–e759
Karimian J, Khazaei M, Shekarchizadeh P (2015) Effect of resistance training on capillary density around slow and fast twitch muscle fibers in diabetic and normal rats. Asian J Sports Med 6(4)
Keihanian A, Arazi H, Kargarfard M (2019) Effects of aerobic versus resistance training on serum fetuin-A, fetuin-B, and fibroblast growth factor-21 levels in male diabetic patients. Physiol Int 106(1):70–80
Kim HJ, Song W (2017) Resistance training increases fibroblast growth factor-21 and irisin levels in the skeletal muscle of Zucker diabetic fatty rats. J Exerc Nutr Biochem 21(3):50
Ku YH, Han KA, Ahn H, Kwon H, Koo BK, Kim HC, Min KW (2010) Resistance exercise did not alter intramuscular adipose tissue but reduced retinol-binding protein-4 concentration in individuals with type 2 diabetes mellitus. J Int Med Res 38(3):782–791
Lopez AD, Mathers CD, Ezzati M, Jamison DT, Murray CJ (2006) Measuring the global burden of disease and risk factors, 1990–2001. GBD Risk Factors 1:1–14
Massi-Benedetti M, Herz M, Pfeiffer C (1996) The effects of acute exercise on metabolic control in type II diabetic patients treated with glimepiride or glibenclamide. Horm Metab Res 28(09):451–455
Matsui M, Kosaki K, Myoenzono K, Yoshikawa T, Park J, Kuro-o M, Maeda S (2022) Effect of aerobic exercise training on circulating fibroblast growth factor-21 response to glucose challenge in overweight and obese men: a pilot study. Exp Clin Endocrinol Diabetes 130(11):723–729
Nourzad F, Shahidi F, Saleh Pourpour M (2022) The effect of aerobic and resistance training on insulin resistance index (HOMA-IR) and BCL-2/BAX ratio in apoptotic pathway in the heart tissue of male wistar diabetic rats. J Sport Exerc Physiol 15(1):69–82
Powers SK, Lennon SL, Quindry J, Mehta JL (2002) Exercise and cardioprotection. Curr Opin Cardiol 17(5):495–502
Riddell MC, Iscoe KE (2006) Physical activity, sport, and pediatric diabetes. Pediatr Diabetes 7(1):60–70
Segal KR, Edano ALBERT, Abalos A, Albu JEANINE, Blando LORNA, Tomas MB, Pi-Sunyer FX (1991) Effect of exercise training on insulin sensitivity and glucose metabolism in lean, obese, and diabetic men. J Appl Physiol 71(6):2402–2411
Sharma A, Mittal S, Aggarwal R, Chauhan MK (2020) Diabetes and cardiovascular disease: inter-relation of risk factors and treatment. FJPS 6:1–19
Sharma M, Chan HK, Lavilla CA Jr, Uy MM, Froemming GRA, Okechukwu PN (2023) Induction of a single dose of streptozotocin (50 mg) in rat model causes insulin resistance with type 2 diabetes mellitus. Fundam Clin Pharmacol 37(4):769–778
Taniguchi H, Tanisawa K, Sun X, Kubo T, Higuchi M (2016) Endurance exercise reduces hepatic fat content and serum fibroblast growth factor 21 levels in elderly men. J Clin Endocrinol 101(1):191–198
Tanoorsaz S, Behpoor N, Tadibi V (2017) Changes in cardiac levels of caspase-8, Bcl-2 and NT-proBNP following 4 weeks of aerobic exercise in diabetic rats. IJBSM 2(4):172–177
Wente W, Efanov AM, Brenner M, Kharitonenkov A, Koster A, Sandusky GE, ... & Gromada J (2006) Fibroblast growth factor-21 improves pancreatic β-cell function and survival by activation of extracellular signal–regulated kinase 1/2 and Akt signaling pathways. Diabetes 55(9):2470–2478
Woo YC, Xu A, Wang Y, Lam KS (2013) Fibroblast growth factor 21 as an emerging metabolic regulator: clinical perspectives. Clin Endocrinol 78(4):489–496
Zhang X, Yeung DC, Karpisek M, Stejskal D, Zhou ZG, Liu F, ... & Xu A (2008) Serum FGF21 levels are increased in obesity and are independently associated with the metabolic syndrome in humans. Diabetes 57(5):1246–1253
Acknowledgements
This study has been performed in the exercise physiology laboratory of the Shahid Rajaee Teacher Training University of Tehran. So, I would like to thank all individuals for their help in collecting information for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This study was not supported by any funding.
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures in this study have been endorsed by the Ethics Committee of Shahid Rajaee Teacher Training University of Tehran (IRSRTTU.SSF.2020.111).
Informed consent
For this type of study, informed consent is not required.
Consent for publication
The author consents for publication of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zare Karizak, S. Impact of 4-week endurance and resistance training on level of insulin resistance and FGf21 in heart tissue of diabetic male rats. Comp Clin Pathol 33, 665–670 (2024). https://doi.org/10.1007/s00580-024-03584-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-024-03584-4