Abstract
Serum samples from 489 male and female camels were tested for antibodies against Coxiella burnetii using indirect enzyme-linked immunosorbent assay (ELISA). Antibodies to C. burnetii were recorded in sera of 252 (51.64 %) camels. Significant differences in prevalence were found between male and female camels, juvenile and adult camels, and different ecotypes and different sampling locations. Three hundred seven camels were simultaneously tested for C. burnetii antibodies by ELISA and indirect immunofluorescence (IFA) tests. Close agreement was found between the results of the two tests. A high prevalence of C. burnetii antibodies was also recorded in milk samples tested by ELISA. Clinical samples from serologically positive camels were subjected to PCR analysis using primers which amplify the repetitive transposon-like and transposase gene regions of C. burnetii. Positive DNA amplification was obtained from both regions, with the highest shedding of C. burnetii in fecal samples (27.59 %) followed, in descending order, by urine (23.81 %), blood (15.85 %), and milk (6.5 %). The present results indicate that camels are a major reservoir of C. burnetii in Saudi Arabia. High prevalence of C. burnetii in camels, poor sanitary standards under which these animals are kept, and the consumption of raw camel milk indicate that camels can also be a major source of Q fever transmission to humans in Saudi Arabia.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The world population of the Arabian one-humped camel (Camelus dromedarius) is about 20 million. Of these, more than 800,000 of the finest camel ecotypes are found in Saudi Arabia. For thousands of years, the camel has been closely associated with the lives of inhabitants of the Arabian Peninsula. It has been mentioned in holy books and in countless anthologies and verses from ancient Arabic poetry. Today, the camel remains a highly valued beast for its meat, milk, wool, skin, and folk medicine. It also serves as a means of transportation and sport and a source of pride and wealth. Efforts to improve camel health and productivity are, however, limited as most camels are still reared by nomads under poor management and sanitary conditions. Several infectious diseases are known to affect camels, including a number of zoonotic infections (Abbas et al. 1987; Wernery and Kadan 1995). Among the zoonoses, Coxiella burnetii, the causative agent of Q fever, appears to be one of the most widespread infections in camels. It has been reported in camels from almost all parts of the Middle East and North and East Africa where these animals are reared (Wernery and Kadan 1995). Very high prevalence of Q fever antibodies was reported in the sera of camels in some countries, namely, 66 % in Egypt (Soliman et al. 1992), 80 % in Chad (Schelling et al. 2003), 62 % in Saudi Arabia (Hussein et al. 2008) and up to 100 % in nomadic camels in southeastern Ethiopia (Gumi et al. 2013).
The objective of the present study was to investigate the prevalence of anti-C. burnetii antibodies in the blood and milk of camels in Saudi Arabia and to determine the shedding routes of the organism in infected camels.
Materials and methods
Four hundred eighty nine camels of either sex were tested for antibodies against C. burnetii. The animals were selected randomly at camel enclosures, markets, slaughter houses, and free-ranged herds in five geographical locations. They were broadly divided into juvenile or sexually premature (1–2 years) and adult (≥3 years) animals. The camels belonged to Maghater, Majahim, and mixed ecotypes. All camels were clinically normal, but some harbored the camel tick, Hyalomma dromedarii. None of the adult females was pregnant while some were lactating.
Serological tests
Ten-milliliter jugular blood samples were drawn from each camel into a plain vacutainer tube and allowed to clot at room temperature for 3 h. Serum was separated by centrifugation at 1500g for 15 min and stored at −20 °C. The sera were diluted at 1:100 and tested using an indirect enzyme-linked immunosorbent assay (ELISA) designed to detect antibodies against phases I and II C. burnetii antigens (CHEKIT-Q fever enzyme immunoassay; IDEXX laboratories, Bommeli Diagnostics, AG, Bern, Switzerland). Tests were performed in microtiter plates pre-coated with inactivated C. burnetii antigens (Nine Mile reference strain) according to the manufacturer’s protocol. However, instead of using peroxidase-conjugated monoclonal anti-ruminant IgG supplied with the test kits, a specific goat anti-camel IgG peroxidase conjugate was used (Triple J. Farms, 777 Jorgensen Place, Bellingham, WA 98226, USA). The optical density (OD) was determined at 450 nm using a microtiter plate reader. OD percentage (OD%), corresponding to the intensity of color change and hence antibody concentration, was determined as follows:
where S, N, and P are the OD values of the tested, negative control, and positive control sera, respectively. A good visual cutoff was observed at OD% = 40, and samples with OD values ≥40 were considered positive while samples with OD values <40 were considered negative.
Ninety two female camel sera were tested simultaneously by both ELISA and indirect immunofluorescence (IFA). The latter assay was performed using a commercial kit designed to detect IgG antibodies against phase II C. burnetii (Nine Mile strain) antigen (Vircell, S. L. Pza. Dominguez Ortiz I. Poligono Industrial Dose de Octubre. 18320 Santa Fe, Granada, Spain). Specific FITC–conjugated goat anti-camel IgG (Triple J. Farms, 777 Jorgensen Place, Bellingham, WA 98226, USA) was used. The first positive serum dilution was recorded at 1:64. Positive sera were further titrated by serial twofold dilutions up to a maximum titer of 1:8192. The results were determined using fluorescence microscopy (Axioskop 2 Plus; Zeiss, Gttingen, Germany) at ×400 magnification. Positive sera showed bright apple green fluorescence against a dark background.
ELISA test was also applied to detect C. burnetii antibodies in defatted milk samples from 48 lactating females. Sera from the same camels were tested simultaneously by ELISA. For collecting milk samples, the udders were thoroughly washed and dried and the teats were cleaned using disinfectant swipes. The first two streams of milk were discarded. Ten-milliliter milk samples were then collected from each camel into clean, sterile polystyrene tubes and transferred in ice to the laboratory. The samples were centrifuged, the fat-fraction was removed and discarded, and the nonfat fraction was frozen at −20 °C until tested. Each milk sample was diluted 1:5 in PBS solution and tested in the same manner as serum.
DNA extraction and PCR assay
Polymerase chain reaction (PCR) was applied to detect C. burnetii DNA in clinical specimens from camels. A total of 209 samples, comprising 82 whole blood, 77 milk, 21 urine, and 29 fecal samples, were collected from seropositive camels. All samples were kept frozen at −80 °C until tested. DNA was extracted from blood, milk, and urine using Qiagen DNeasy blood and tissue extraction kit (GmbH, Hilden, Germany). DNA extraction from fecal samples was carried out using a Bioline extraction kit (Bioline, Humber Rd, London, UK). Extractions were performed following the manufacturer’s instructions. Nested PCR was performed for DNA amplification using primers which amplify transposon-like region and transposase gene. Positive control C. burnetii DNA was provided by Professor Klaus Henning of the Institute of Epidemiology, Friedrich-Loeffler-Institute in Germany. The PCR was carried out in DNA thermal cycler (Bioline, Humber Road, London, UK). Following initial denaturation at 95 °C for 8 min, rapid PCR program was performed consisting of 35 cycles of denaturation at 95 °C for 15 s, annealing at 52 °C for 5 s, and extension at 72 °C for 18 s. Re-amplification was performed using 35 cycles of denaturation at 95 °C for 15 s, annealing at 48 °C for 5 s, and extension at 72 °C for 18 s. Amplification products were separated by agarose gel electrophoresis, stained with ethidium bromide and visualized under UV transilluminator (Mohammed et al. 2014).
Statistical analysis
Data were analyzed with the incidence of C. burnetii coded as a binary dependant variable (0 for sero-negative and 1 for seropositive animals). Frequencies and means of C. burnetii prevalence and titres were computed using Statistical Analysis System V. 9.1 software for Windows. A probability value of p ≤ 0.05 was considered significant. The relationship between ELISA and IFA tests was achieved using the PROC CORR procedure.
Results
Serology
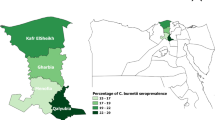
Proc frequency and independency test results for the prevalence of Q fever antibodies in camels using chi-square are summarized in Table 1. Out of 489 camels tested for anti-C. burnetii antibodies by indirect ELISA, 252 camels were found to be serologically positive for antibodies against the organism, giving an overall prevalence of 51.53 % (Table 1). The highest prevalence was recorded in Magahim camels, which are the main camels raised for milk production in Saudi Arabia. The prevalence rate was more than three times in female as compared to male camels and more than twice in adult compared to juvenile camels. The prevalence also varied significantly in camels from different localities.
Comparison between ELISA and IFA tests for detection of C. burnetii antibodies in camels’ sera is given in Table 2. The prevalence rates revealed by ELISA and IFA tests were 69.57 and 61.96 %, respectively. There was a high level of agreement (Kappa = 86 %) between the results of the two tests, indicating that either of them can be used for serological surveys of Q fever in camels or to confirm each other. High IFA titers equal to or exceeding 1:8192 were recorded in some camels, probably indicating recent infection.
The results of ELISA tests in serum and milk also agreed closely (Table 3). Out of the 48 female camels tested, 30 camels revealed antibodies against C. burnetii in milk and 32 were positive by the serum test. The latter animals included all the camels shown to be positive by ELISA milk tests. Spearman’s correlation analysis showed highly significant positive correlation (0.61) between the test results both in serum and milk (p < 0.0001).
PCR
Positive PCR amplification was obtained from camels’ blood, feces, milk, and urine samples using primers which amplify repetitive transposon-like and transposase gene regions of C. burnetii (Fig. 1; Table 4). As the table shows, the shedding of C. burnetii by these animals was highest in fecal samples and urine (27.59 and 23.81 %, respectively), followed by blood (15.85 %) and least in milk (6.5 %).
Discussion
There is a striking paucity of information on the status of Q fever in man and animals in Saudi Arabia, although the disease has been described as hyperendemic among the Kingdom’s inhabitants since the 1960s (Gelpi 1966; Lippi et al. 1968). It was only in 2008 that camels were screened for Q fever for the first time in the Kingdom (Hussein et al. 2008). The results of that study and the present results indicate an alarmingly high prevalence of C. burnetii among Saudi indigenous camels (62 and 51.53 %, respectively). This is the highest prevalence among all species of farm animals recently screened for Q fever in Saudi Arabia (unpublished report, 2014). Such high prevalence is clearly attributable to the poor management and hygienic conditions under which camels are kept in that country, in addition to their frequent exposure to heavy sandstorms and dust contaminated with airborne infections, presumably including C. burnetii, during desert grazing. Another contributing factor could be the common infestation of camels with the camel tick, H. dromedarii. This tick is known to harbor C. burnetii and to secrete it in its feces and saliva (Rehácek and Brezina 1968).
The present study showed that serological prevalence of camel coxiellosis was more than three times higher in female than male camels. This higher susceptibility of females, especially adult females, is due to the remarkable affinity of C. burnetii to the udder, placenta, and other female reproductive organs of ruminants (Aitkin 1989; Marrie 1990; Kilic et al. 2005). Up to one billion organisms per gram may be found in these tissues (CDC 1997).
The study also showed that the prevalence of C. burnetii in camels was significantly affected by age, the prevalence in adult camels being more than double the prevalence in juvenile camels. Age has been recognized as an important risk factor for C. burnetii seropositivity in other species of animals—in general, the older the animal is, the higher is the risk of it being exposed to infection and of becoming seropositive (Klaasen et al. 2014; Jung et al. 2014). The same is true for humans where the prevalence of Q fever increases with age (Psaoulaki et al. 2006; Kardeňosa et al. 2006).
Significant differences in the prevalence of C. burnetii in animals are known to occur between different countries and different geographical locations within the same country. In the current study, the detection of a higher prevalence of C. burnetii in camels in Hait versus Riyadh city could be attributed to a number of factors. Hait is a small agricultural village about 25 km from Riyadh, with high densities of camels and small ruminants. The animals are kept by nomadic herdsmen under poor hygienic and husbandry conditions. Besides, the area is heavily polluted by sewage water from Riyadh, and numerous rats roam about near camel sites. It is also an open area surrounded by sand dunes and exposed to frequent sandstorms. At the same time, the village adjoins a historical water spring and mountain caves that attract many visitors. All of these factors might contribute to the higher risk of coxiellosis in the area.
No records are found of molecular detection of C. burnetii in farm animals in Saudi Arabia, apart from our own (Mohammed et al. 2014). However, several studies have been carried out in other countries which documented the presence of C. burnetii DNA in various clinical samples, such as blood, milk, birthing fluids, aborted fetuses, and tissue samples of domestic and wild animals (Rodolakis et al. 2007; Fretz et al. 2007; Garcia-Perez et al. 2009; Rousset et al. 2009; Rahimi 2009; Rahimi et al. 2010; Angen et al. 2011; Jung et al. 2014). Our study also constitutes the first world record of direct identification of C. burnetii by PCR in feces, urine, and milk, and the second in the blood, of camels. To our knowledge, the only other record of direct detection of C. burnetii DNA from camels was from Iran, where partial DNA of 16S ribosomal RNA gene of C. burnetii was detected in 10.8 % of blood samples from local camels (Doosti et al. 2014). The present PCR results showed that the camel is capable of shedding C. burnetii in its feces, urine, milk, and blood, with the highest percentage of shedding being recorded in fecal samples (27.6 %) suggesting that feces might be the preferred shedding route of this organism in camels. Since C. burnetii tends to localize in the reproductive tract of infected female animals, specimens of vaginal mucus and birthing fluids of female camels should also be investigated as additional sources of transmission of the Q fever agent, especially to herders assisting these animals during parturition or abortion (Berri et al. 2000, 2007).
The PCR analysis of camel samples in the present study was not designed for comparison with serological results but to investigate shedding routes of C. burnetii in camels, which showed feces to be more important in transmitting C. burnetii than urine, blood, and mil. However, this does not preclude the fact that PCR is becoming increasingly used in diagnosis and other studies of coxiellosis in animals, not only because of its higher specificity and sensitivity compared to conventional serology but also more importantly because it measures current infection, unlike conventional serological methods which do not differentiate between past and ongoing infections. On the other hand, PCR is more laborious and more costly than conventional serology and requires laboratories with PCR capabilities which are not available in many laboratories in developing countries. For this reason, serological methods, particularly ELISA and IFA, are still more widely used than PCR for large-scale surveys of C. burnetii in animals.
Some authors consider ELISA to be more specific and sensitive than IFA for serological surveys of C. burnetii (Soliman et al. 1992; Kovakova et al. 1987; Field et al. 2000). In the present study, however, we compared the seroprevalence of C. burnetii in camels using both ELISA and IFA and the results of the two tests agreed closely. Therefore, either of these tests can be used to screen anti-C. burnetii antibodies in animals. The infectivity of C. burnetii is so high that with some strains of this organism, only one microorganism may be sufficient initiate infection (Waag 1997). Human infection with C. burnetii is usually acquired from farm animals (Guatteo et al. 2011; Schimmer et al. 2014). Therefore, the prospects of transmission of this organism from camels to humans in Saudi Arabia are high, not only because of the exceptionally high prevalence of the infection in camels and their capability of shedding C. burnetii in their secretions and excretions but also because of the widespread tradition, throughout the Arabian Peninsula, of consuming raw camel milk (Alshaikh et al. 2007) and the belief held by many desert dwellers that consuming diluted camel urine (usually diluted with milk) was an effective remedy for many human ailments, including abdominal disorders, liver diseases, and cancer (Khorshid 2011; Al-Bashan 2011).
In conclusion, the high prevalence of C. burnetii in camels and the lack of basic sanitary knowledge by most camel herders as well as the consumption of unpasteurized camel milk clearly indicate that camels in Saudi Arabia might play a major role as a reservoir of C. burnetii and a significant source for the transmission of Q fever to humans.
References
Abbas B, Yassin TTM, Elzubir AEA (1987) Survey for certain zoonotic diseases in camels in the Sudan. Rev Elev Med Vet Pays Trop 40:231–233
Aitkin ID (1989) Clinical aspects and prevention of Q fever in animals. Eur J Epidemiol 5(4):420–424
Al-Bashan MM (2011) In vitro assessment of the antimicrobial activity and biochemical properties of camel’s urine against some human pathogenic microbes. Middle-East J Sci Res 7(6):947–958
Alshaikh MA, Al-Haidary A, Aljumaah RS, Mohammed O, Alkurashi M, Gar Elnabi AR, Hussein MF (2007) Camel Brucellosis in Riyadh Region. Saudi Arabia J Camel Practice Res 14(2):113–117
Angen O, Stahl M, Agerholm J, Christoffersen AB, Agger JF (2011) Dynamics of relationship between the presence of Coxiella burnetii DNA antibodies and intrinsic variables in cow milk and bulk tank milk from Danish dairy cattle. J Dairy Sci 94:5750–5759
Berri M, Larcoucau K, Rodolakis A (2000) The detection of Coxiella burnetti from ovine genital swabs, milk and faecal samples by the use of a single touchdown polymerase chain reaction. Vet Microbiol 72:285–293
Berri M, Rousset E, Champion JL, Russo P, Rodokalis A (2007) Goats may experience reproductive failures and shed Coxiella burnetii at two successive parturitions after Q fever infection. Res Vet Sci 83:47–52
CDC (Centers for Disease Control and Prevention) (1997) Q fever outbreak—Switzerland. MMWR 33:355–356
Doosti A, Arshi A, Sadeghi M (2014) Investigation on Coxiella burnetii in Iranian camels. Comp Clin Pathol 23:43–46
Field P, Mitchell J, Santiago A, Dickeson D, Chan S, Ho D, Murphy A, Cuzzubbo A, Devine P (2000) Comparison of a commercial enzyme-linked immunosorbent assay with immunofluorescence and complement fixation tests for detection of Coxiella burnetii (Q fever). J Clin Microbiol 38(4):645–1647
Fretz R, Schaeren W, Tanner M, Baumgartner A (2007) Screening of various foodstuffs for occurrence of Coxiella burnetii in Switzerland. Int J Food Microbiol 116:414–418
Garcia-Perez AL, Astobiza JF, Atxaerandio R, Hurtado A, Juste RA (2009) Investigation of Coxiella occurrence in dairy sheep flocks by bulk-tank milk analysis and antibody level determination. J Dairy Sc 92:1581–1584
Gelpi AP (1966) Q fever in Saudi Arabia. Am J Trop Med Hyg 15:784–798
Guatteo R, Seegers H, Taurel AF, Joly A, Beaudeau F (2011) Prevalence of Coxiella burnetii infection in domestic ruminants: a critical review. Vet Microbiol 149(1–2):1–16
Gumi B, Ferdessa R, Yamuah L, Sori T, Tolosa T, Assefa A, Zinsstag J, Schelling E (2013) Seroprevalence of brucellosis and Q fever in southeast Ethiopian pastoral livestock. J Vet Sci Med Diag 2:1
Hussein MF, Alshaikh M, Gad El-Rab MO, Aljumaah RS, Gar El Nabi AR, Abdel Bagi AM (2008) Serological prevalence of Q fever and chlamydiosis in camels in Saudi Arabia. J Anim Vet Adv 7:685–688
Jung BY, Seo M, Lee S, Byun J, Oem J, Kwak D (2014) Molecular and serologic detection of C. burnetii in native Korean goats (Capra hircus coreanae). Vet Microbiol 204:152–155
Kardeňosa N, Sanfeliu I, Font B, Muňoz T, Nogueras MM, Segura F (2006) Short report: Seroprevalence of human infection by Coxiella burnetii in Barcelona (northeast of Spain). Am J Trop Med Hyg 75(1):33–35
Khorshid FA (2011) The cytotoxic effect of PM 701 and its fractions on cell proliferation of breast cancer cells, MCF7. Am J Drug Disc Dev 1:200–208
Kilic S, Pasa S, Babur C, Ozlem MB (2005) Investigation of Coxiella burnetii antibodies in sheep in Aydin region. Turkey Rev Med Vet 156(6):336–340
Klaasen M, Roest H, van der Hoek W, Goossens B, Secka A (2014) Coxiella burnetii seroprevalence in small ruminants in the Gambia. Plos One 9(1):e85424
Kovakova E, Gallo J, Shrameck S, Kazar J, Bergina R (1987) Coxiella burnetii antigens for detection Q fever antibodies by ELISA in human sera. Viro 31:254–259
Lippi M, Sebastiani A, El-Mutabakani H (1968) Detection of serum antibodies against reoviruses, adenovirus and C. burnetii in group inhabitants of Riyadh, Saudi Arabia. Arch Ital Sc Med Trop 49:129–136
Marrie TJ (1990) Epidemiology of Q fever. In: Marrie TJ (ed) The disease, vol 1. Fla: CRC Press, Boca Raton, pp pp 49–70
Mohammed OB, Jarelnabi A, Aljumaah RS, Alshaikh MA, Bakhiet Amel O, Omer Sawsan O, Alagaili AN, Hussein MF (2014) Coxiella burnetii, the causative agent of Q fever in Saudi Arabia: molecular detection from camel and other domestic livestock. Asian Pac J Trop Med 2014:715–719
Psaoulaki A, Hadjichristodoulou C, Loukaides F, Soteriades E, Konstantinidis A, Papastergiou P, Ioannidou MC, Tselentis Y (2006) Epidemiological study of Q fever in humans, ruminant animals and ticks in Cyprus using a geographical distribution system. Eur J Clin Microbiol Infect Dis 35:576–586
Rahimi E (2009) Coxiella burnetii in goat bulk milk samples in Iran. J Dairy Sci 92:1581–1584
Rahimi E, Doosti A, Ameri M, Kabiri E, Sharifian B (2010) Detection of coxiella burnetii by nested PCR in bulk milk samples from dairy bovine, ovine and caprine herds in Iran. Zoonoses Public health 57:38–41
Rehácek J, Brezina R (1968) Detection of Coxiella burnetii in saliva of experimentally infected ticks. Hyalomma dromedarii Koch. Bull World Hlth Org 39(6):974–977
Rodolakis A, Berri M, Hechard C, Caudron C, Souriau A, Bodler CC, Blanchard B, Camuset P, Devillechaise P, Natrop JC, Vadet JP, Arricau-Bouvery N (2007) Comparison of Coxiella brunetii shedding in milk of dairy bovine, caprine and ovine herds. J Dairy Sci 90:5352–5360
Rousset E, Berri M, Durnad B, Dafour P, Prigent M, Delcroix T, Touratier A, Rodolakis A (2009) Coxiella burnetii shedding routes and antibody response after outbreak of Q fever-induced abortion in dairy goat herds. Appl Environ Microbiol 75:428–433
Schelling E, Diguimbay C, Daoud S, Nicolet P, Boierlin P, Tanner M, Zinsstag J (2003) Brucellosis and Q fever seroprevalences of nomadic pastoralist and their livestock in Chad. Prev Vet Med 61:279–293
Schimmer B, Schotten N, van Engelen E, Hautvast ILA, Schneeberger PM, van Duijnhoven YTHP (2014) Coxiella burnetii Seroprevalence and Risk for Humans on Dairy Cattle Farms, the Netherlands, 2010–2011. Emerg Infect Dis 20:3
Soliman A, Boulos A, Botros M, Watts D (1992) Evaluation of a competitive enzyme immunoassay for detection of Coxiella burnetii antibody in animal sera. J Clin Microbiol 30(5):1595–1597
Waag DM (1997) Q fever. Lenhart, M. K. Medical Aspects of Biological Warfare. Washington D.C, In, pp 199–213
Wernery U, Kadan OR (1995) Infectious disease of camelids. Blackwell Wissenschafts Verlag, Berlin
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hussein, M.F., Alshaikh, M.A., Al-Jumaah, R.S. et al. The Arabian camel (Camelus dromedarius) as a major reservoir of Q fever in Saudi Arabia. Comp Clin Pathol 24, 887–892 (2015). https://doi.org/10.1007/s00580-014-2002-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00580-014-2002-y