Abstract
A topic of confusion over the interactions between arbuscular mycorrhizal (AM) fungi and plants is the mycorrhizal status of some plant families such as Cyperaceae, which is generally considered to be non-mycorrhizal. Here, we conducted experiments to explore how the abiotic environmental conditions and AM network influence the interactions between AM fungi and Carex capillacea. We grew Carex capillacea alone or together with a mycorrhizal host species Medicago sativa in the presence or absence of AM fungi (soil inoculum from Mount Segrila and Rhizophagus intraradices from the Chinese Bank of the Glomeromycota, BGC). Plants were grown in a growth chamber and at two elevational sites of Mount Segrila, respectively. The results indicate that mycorrhizal host plants ensured the presence of an active AM fungal network whether under growth chamber or alpine conditions. The AM fungal network significantly depressed the growth of C. capillacea, especially when native inocula were used and the plants grew under alpine site conditions, although root colonization of C. capillacea increased in most cases. Moreover, the colonization level of C. capillacea was much higher (≤ 30%) when growing under alpine conditions compared with growth chamber conditions (< 8.5%). Up to 20% root colonization by Rhizophagus intraradices was observed in monocultures under alpine conditions. A significant negative relationship was found between shoot phosphorus concentrations in M. sativa and shoot dry mass of C. capillacea. These results indicate that growing conditions, AM network, and inoculum source are all important factors affecting the susceptibility of C. capillacea to AM fungi, and growing conditions might be a key driver of the interactions between AM fungi and C. capillacea.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Arbuscular mycorrhizal (AM) fungi belong to the phylum Glomeromycota and are ubiquitous root symbionts that form mutualistic associations with c. 70% of terrestrial vascular plant species (Brundrett 2009). These obligate biotrophs are important as they supply growth-limiting nutrients to the host plants (Kiers et al. 2011), protect them from soil pathogens (Smith and Read 2008), and alleviate environmental stress in exchange for plant-assimilated carbon (C) (Augé 2001). In addition, AM fungi can also influence a number of important ecosystem functions such as plant productivity, plant diversity, nutrient cycling, and soil structure (Grime et al. 1987; van der Heijden et al. 1998; Li et al. 2015; Powell and Rillig 2018). Still, an estimated 18% of vascular plants do not form mycorrhizal associations (Brundrett 2009). Members of the Cyperaceae, along with the Brassicaceae, Amaranthaceae, Caryophyllaceae, Chenopodiaceae, and Juncaceae are described as characteristically “non-mycorrhizal, NM” species (Tester et al. 1987). However, under certain conditions, some presumed NM plants can be colonized by AM fungi and form rudimentary AM (RAM) phenotype structures like hyphae and vesicles (Muthukumar and Udaiyan 2002; Veiga et al. 2013; Cosme et al. 2018). These typically non-mycorrhizal species are sometimes referred to as non-host or “non-mycotrophic” species (Janos 2007; Lekberg et al. 2015).
The Cyperaceae have an estimated 5500 species in c. 98 genera, are cosmopolitan in distribution, and represent the 10th most species-rich family among the angiosperms (Govaerts and Simpson 2007; Waterway et al. 2015; Spalink et al. 2016). Cyperaceae are usually regarded as non-mycorrhizal but a number have been observed to form mycorrhizal associations (Muthukumar et al. 2004; Veselkin et al. 2014). Early reports regarding mycorrhizal incidence in the Cyperaceae have been reviewed by Newman and Reddell (1987). More information has subsequently been acquired on the widespread occurrence of mycorrhiza in the Cyperaceae. Muthukumar et al. (2004) summarized data from 221 members of the Cyperaceae and found that the proportion considered to be mycorrhizal species had increased from 11 to 40% with a considerable decline in non-host species from 74 to 49% since 1987. Based on published reports, the Cyperaceae have been repeatedly declared NM by many authors (Powell 1975; Brundrett and Kendrick 1988) while others consider the Cyperaceae to be NM-AM, which means plants with variable AM or NM roots (Cooke and Lefor 1998; Miller et al. 1999; Ruotsalainen and Aikio 2004; Gai et al. 2006; Konoplenko et al. 2017). Current interest is mainly focused on how the mycorrhizal status of this plant family is variable and on the ecological function of the RAM phenotype.
Several hypotheses have been generated to explain the variable mycorrhizal status in roots of the Cyperaceae. One widely accepted idea is that the mycorrhizal status of its members might be influenced strongly by environmental conditions (Muthukumar et al. 2004). The rarely mycorrhizal or non-mycorrhizal status of the Cyperaceae is often attributed to the wet and waterlogged or disturbed habitats they inhabit (Tester et al. 1987). Another hypothesis is that the colonization experienced by the Cyperaceae is a consequence of being part of a community mycelial network (as in Fitter et al. 1998) and the hyphal and vesicular colonization within the plant root systems of the Cyperaceae are likely to indicate the plant is a passive participant (Miller 2005). This hypothesis was supported by the evidence that an arbuscular structure is rarely observed or very low in frequency (Miller et al. 1999; Johnson et al. 2003b). However, key experiments remain to be conducted, chiefly to test what are the most important drivers influencing the AM colonization of the Cyperaceae.
Despite never or rarely establishing AM symbiosis, non-host plants can also be affected by AM fungi and most studies indicate a negative effect of the presence of AM fungi on the growth of non-host species (Allen et al. 1989; Francis and Read 1994, 1995; Sanders and Koide 1994; Johnson 1998; Ruotsalainen and Aikio 2004; Rinaudo et al. 2010). Some AM fungi may penetrate the roots of non-host plants, particularly when the mycorrhizal network is supported by neighboring mycorrhizal host plants. This interaction with host-supported AM fungal hyphae often reduces the growth of the infected non-host plants (Cosme et al. 2018). Evidence is therefore accumulating to suggest that non-host species can be suppressed in the presence of AM fungi (Veiga et al. 2012, 2013). Some possible mechanisms explaining the deleterious effects of AM fungi on non-host plants are as follows. (1) The plant actively rejects infection (Allen et al. 1989). Invaded non-host root segments have been observed to turn brown in color and the fungus has disappeared from the root (Allen et al. 1989; Veiga et al. 2013). (2) Non-host plants are at a competitive disadvantage compared with mycorrhizal plants (Sanders and Koide 1994). For example, both mycorrhizal plants and AM fungi produce toxic compounds that suppress the growth of non-host species (Francis and Read 1994; Veiga et al. 2012), or extension of AM mycelium inhibits non-host root development (Francis and Read 1994).
The Cyperaceae often dominate arctic and alpine vegetation where AM fungi are often absent and the plants are often designated non-hosts (Tester et al. 1987; Harley and Harley 1987). Some studies have found that Carex species grown in monoculture are unable to support an active mycorrhizal mycelial network or propagule production (Ruotsalainen and Aikio 2004; Johnson et al. 2003a). However, numerous workers have observed fungal colonization in roots of the Cyperaceae grown with mycotrophic host plants in the field (Brundrett 2009; Li et al. 2015; Muthukumar et al. 2004; Gai et al. 2006). The factors affecting AM colonization of Carex species and the growth responses of Carex spp. to AM fungi remain unknown. To explore these questions, we focused on Carex capillacea, a species that is widely distributed on the Tibetan plateau, to evaluate the relative importance of AM networks, and growth conditions (e.g., growth chamber conditions and alpine conditions) in impacting the mycorrhizal status of C. capillacea and the interactions between AM fungi and C. capillacea. Our hypotheses were that the low mycotrophy observed in C. capillacea was mainly caused by the abiotic environmental conditions, and the growth response of C. capillacea to AM fungi was affected by AM fungal networks together with growth conditions.
In the present study, we set out to explore the hypotheses using two experiments. In experiment 1, C. capillacea and M. sativa plants were grown together (with no mesh) to assess the effect of AM fungal networks on the growth of C. capillacea in a growth chamber (9 °C night and 21 °C day). In experiment 2, C. capillacea was grown with or without the host species M. sativa at two elevations (3000 and 3904 m a.s.l.) on Mount Segrila to assess the effect of AM fungal networks and abiotic environmental conditions on the interactions between AM fungi and C. capillacea.
Materials and methods
Plant material, fungal inoculum, and soil mixture
In this study, we used seeds of C. capillacea obtained from about 200 individual plants in September 2014 at 3300 m a.s.l. on Mount Segrila (29° 21′–29° 50′ N, 94° 28′–94° 51′ E), Nyingchi region, Tibet. Seeds of M. sativa were obtained from the Beijing Crovo Grass Industry Technology Development Center. Rhizophagus intraradices (BGC JX04B) inoculum was supplied by the Chinese Bank of the Glomeromycota (BGC) then was propagated on Sorghum vulgare for 5 months in pots containing a sterilized mixture of quartz sand and 20% low-phosphorus loam soil.
In experiment 1, soil AM fungal inoculum was collected in the form of rhizosphere soil of C. capillacea at 3300 m a.s.l., then transported in coolers and stored at 4 °C. At the same time, 20 widely separated (10 m apart) collection points were selected to collect bulk soil. Approximately 200 g of soil was collected from each sampling point, mixed thoroughly and passed through a 2-mm sieve to remove large roots and stones. The composite soil samples were stored at room temperature for use as the growth medium. In experiment 2, we collected the rhizosphere soil of C. capillacea at two elevations (3000 m and 3904 m a.s.l.) on Mount Segrila then transported it in coolers and stored it at 4 °C. The rhizosphere soils from the low and high site were mixed in the ratio of 1:1 as AM fungal inoculum. The bulk soil also was collected from each elevation site using the same methods as in experiment 1 above, then mixed thoroughly and passed through a 2-mm sieve to remove large roots and stones. The growth medium was a mixture of the bulk soil from the two elevations in the ratio of 1:1, then stored at room temperature.
Experiment 1: effects of AM fungi on the interaction of C. capillacea and M. sativa in a growth chamber
C. capillacea and M. sativa plants were grown in 0.7-L pots, with three treatments: non-mycorrhizal control inoculum (NM), with soil inoculum (S), or with R. intraradices (R.i). Growth medium (105 g) and quartz sand (15 g) were mixed after γ-irradiation (25 kGγ for 48 h to kill the indigenous soil microflora). Each pot received 300 g of soil substrate including the following: (1) 90-g sterile soil AM fungal inoculum and 90-g sterile R. intraradices inoculum (control, NM); (2) 90-g live soil AM fungal inoculum and 90-g sterile R. intraradices inoculum (treatment S); (3) 90-g sterile soil AM fungal inoculum and 90-g live R. intraradices inoculum (treatment R.i.). Thirty milliliters of non-sterile inoculum filtrate was added to all pots in order to compensate for the other soil organisms but without AM fungal propagules. The filtrate was prepared by mixing the inoculum and distilled water (1:2 w/v) and passing the mixture through a 38-μm sieve (Ji et al. 2013). There was a total of three treatments each replicated four times, making a total of 12 pots.
M. sativa seeds (0.5 g) were grown in the pot and simultaneously C. capillacea seeds were sown in a germination disk. Four weeks after the germination of M. sativa, about 30 uniform seedlings were selected and the same numbers (about 30) of fine C. capillacea seedlings were transplanted to the pot. Thus, every pot had about 60 seedlings, including about half C. capillacea and half M. sativa.
The experiment was conducted in a controlled environment chamber (Yiheng, MGC-450HP-2, Shanghai, China) in 2017. The temperature setting was 9 (night) and 21 °C (day) and was chosen based on the mean temperature during the growing season of C. capillacea (May to September). The relative humidity (70%) was based on the average air humidity on the mountain (data from the National Climate Station at 3300 m). Artificial light was used to provide photosynthetically active radiation (PAR) of 432 mmol m−2 s−1 for 16 h every day. The pots were randomly assigned in the controlled environment chamber and moved every 2 weeks to avoid environmental gradients. The plants were watered twice or three times per week.
Experiment 2: effects of AM fungi on the interaction of C. capillacea and M. sativa under alpine conditions
This experiment was set up during the plant growing season at two sites of Mount Segrila, a low elevation site (3000 m a.s.l.) and a high elevation site (3904 m a.s.l.) in 2017. Plants were grown in a 1.5-L dual-compartment microcosms which were divided into two equal parts with a 30-μm nylon mesh (permeable to AM hyphae but not to roots) to minimize the effects of direct root competition, set upon the soil surface. At each elevation site, the microcosms were designed with a randomized block with three factors. One factor, neighbor treatment, contained two levels: C. capillacea grown alone and C. capillacea grown in combination with M. sativa in the other half of the microcosm (monocultures and mixtures, respectively). The other factor, AM fungal inoculation, comprised three levels: without AM fungi (NM), with soil AM fungal inoculum (S), and with R. intraradices (R.i). Growth medium (230 g) and quartz sand (150 g) were added after autoclave sterilization (120 min at 121 °C). Each half microcosm received 500 g of soil substrate including the following: (1) 80-g live soil inoculum and 40-g sterile R. intraradices inoculum (treatment S); (2) 80-g sterile soil inoculum and 40-g live R. intraradices inoculum (treatment R.i); and (3) 80-g sterile AM fungal soil inoculum and 40-g sterile R. intraradices inoculum (control, NM). There were a total of six treatments at each site. Each treatment was replicated five times, making a total of 60 dual-compartment microcosms.
According to the neighbor treatments, about 1.5-g M. sativa seeds were grown in one-half of the microcosms (mixture) or the microcosms were left unsown (monoculture), and at the same time, C. capillacea seeds were sown in seed germination disks. Upon germination of M. sativa, smaller seedlings were removed and about 60 uniform seedlings remained. Four weeks after the germination of C. capillacea, about 60 uniform seedlings were transplanted in the other half of each microcosm.
Harvest and sample analysis
All plants were harvested after 12 weeks. At harvest the shoots of all plants were cut at the soil surface, oven-dried (65 °C) and weighed to determine the aboveground biomass. Oven-dried shoots were ground and the shoot phosphorus (P) concentration of C. capillacea in experiment 2 was determined. Shoot P was determined spectrophotometrically after calcination and extraction with hydrochloric acid (Thomas et al. 1967). Roots were carefully removed from the soil substrate and washed with distilled water then cut into 1-cm-long pieces and stored at − 80 °C until analysis. The mixed soil substrates for analysis of physicochemical properties in each experiment were air-dried and sieved (< 2 mm). Soils were analyzed for pH (1:2.5 soil/water, w/v). Soil organic carbon (SOC) was determined by wet oxidation followed by titration with ferrous ammonium sulfate. Total nitrogen (TN) in soils was determined with an elemental analyzer (EA1108, Fisons Instruments SpA, Milan, Italy). Available phosphorus was extracted with 0.5 M NaHCO3. (Soil analyses are presented in Table S1.)
Half amounts of roots in a pot were randomly chosen as a sample for estimation of AM colonization. One-centimeter root segments were cleared with 10% KOH and stained with 0.05% (v/v) trypan blue in lactic acid (modified method of Phillips and Hayman 1970). Root colonization was estimated using the magnified-intersections method (McGonigle et al. 1990) with at least 300 intersections per root sample. The hyphal length density in soil was determined using the gridline intersect method at × 200 magnification (Miller et al. 1995).
Statistical analysis
All measured variables were tested for normality and homogenous variance and the colonization of C. capillacea in experiment 1 were ln(x + 1) transformed for meeting the model assumptions. In experiment 1, differences in AM fungal colonization, hyphal length density, and shoot dry mass of two plant species among inoculation treatments were assessed by one-way ANOVA, and significant differences among treatments were tested using Turkey’s honestly significant difference (HSD) test at the 95% confidence level. In experiment 2, we used three-way ANOVA to test the effects of site, neighbor, inoculum, and their interactions on colonization, hyphal length density, and shoot dry mass of C. capillacea. The significant differences of each variable of C. capillacea and shoot dry mass of M. sativa among treatments were determined using Tukey’s honesty significant difference (HSD) test at the 95% confidence level. Pearson’s correlation analysis was used to explore the relationship between shoot phosphorus concentration of M. sativa and shoot dry mass of C. capillacea. Statistical analysis was conducted using the SPSS 20.0 software package (SPSS Inc., Chicago, IL).
Results
Experiment 1: effects of AM fungi on the interaction of C. capillacea and M. sativa in the growth chamber
The colonization of C. capillacea was significantly higher with R.i. than that with no inoculum (Table 1). Soil colonization (hyphal length density) was much greater when inoculated with R. intraradices than with soil AM fungal inoculum (S) (Table 1). Soil AM fungal inoculum (S) significantly reduced the biomass of C. capillacea compared with the NM inoculum. However, R. intraradices had no significant effect on the growth of C. capillacea. In contrast to C. capillacea, M. sativa plants inoculated with AM fungi had significantly higher colonization levels and shoot dry mass than the NM control plants (Table 1).
Experiment 2: effects of AM fungi on the interaction of C. capillacea and M. sativa under alpine conditions
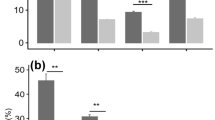
The percentage of root length colonized by AM fungi depended on site, neighboring host plants, inoculum, and their second- and third-order interactions (Table 2). Roots of C. capillacea were barely colonized when grown without M. sativa in the local soil inoculant (S) treatment at both elevations (Fig. 1a). In the presence of M. sativa, C. capillacea plants had significantly higher colonization than the respective controls at the low elevation (Fig. 1a). Colonization of C. capillacea reached > 30% when inoculated with R. intraradices (Fig. 1a, Site-Low). However, the presence of M. sativa did not increase the colonization of C. capillacea at the high elevation (Fig. 1a). We observed few arbuscules (0.35 ± 0.24%) in the roots of C. capillacea inoculated with R. intraradices in the presence of M. sativa. We measured a significant site × neighbor × inoculum interaction for hyphal length density (Table 2). The neighboring host increased the hyphal length on the C. capillacea side of the microcosms only at the high elevation site (Fig. 1b, Site-High). There were no significant differences in hyphal length between the absence and presence of neighboring host treatments at the low elevation. The root and soil colonization of M. sativa were both higher than those of C. capillacea, reaching about 40% (Fig. 1a) and 2 m g−1 soil, respectively (Fig. 1b).
Root AM colonization of C. capillacea (a) and hyphal length density of AM fungi (b) in microcosms with (mix) and without (mono) M. sativa (experiment 2). Inner graphs present the M. sativa data. White bars (Site-Low) and black bars (Site-High) mean that plants were grown at the low elevation site and at the high elevation site, respectively. S refers to mixture (1:1) of soil AM fungal inoculum from the low and high elevation sites; R.i. refers to Rhizophagus intraradices. Error bars are standard errors of the mean (SEM). Bars topped by the same letter do not differ significantly at P ≤ 0.05 by Turkey’s honestly significant difference (HSD) test
Biomass of C. capillacea was significantly affected by site, neighboring host plants, and inoculum (Table 2). When the plants grew at low or high elevation, AM fungal inoculation significantly reduced the biomass of C. capillacea, especially in the soil AM fungal inoculum treatments (Fig. 2a). No discernible growth response of M. sativa between inoculation treatments was observed at the low elevation, but M. sativa plants inoculated with R. intraradices had significantly higher shoot dry mass than the other inoculation treatments at the high elevation (Fig. 2b).
Shoot dry mass of C. capillacea (a) grown in microcosms without (mono) and with (mix) mycorrhizal plants and shoot dry mass of M. sativa (b) (experiment 2). White bars (Site-Low) and black bars (Site-High) mean that plants were grown at the low elevation site and at the high elevation site, respectively. NM refers to without AM fungi; S refers to mixture (1:1) of soil inoculum from low and high elevation sites; R.i. refers to Rhizophagus intraradices. Error bars are standard errors of the mean (SEM). Bars topped by the same letter do not differ significantly at P ≤ 0.05 by Turkey’s honestly significant difference (HSD) test
There were no significant differences in shoot P concentration of C. capillacea between the treatments with and without host species at the low site (Fig. S1a). However, the effect of AM fungi on shoot P concentration of C. capillacea depended on the presence or absence of the host species at high elevation (Fig. S1a). In the presence of M. sativa, AM fungi significantly reduced the shoot P concentration of C. capillacea compared with the NM controls (Fig. S1a). Despite the lack of a growth response, mycorrhizal M. sativa plants grown with C. capillacea showed significantly higher shoot P concentrations compared with the NM controls (Fig. S1b). Moreover, there were significant negative relationships between the shoot P concentrations of M. sativa and shoot dry mass (r = − 0.619, P < 0.001) (Fig. 3).
Discussion
AM colonization of C. capillacea
Two experiments were conducted in the present study to explore whether the low mycotrophy observed in C. capillacea is caused by the abiotic environmental conditions. We found that the AM colonization level of C. capillacea varied with growing conditions. Low colonization (up to 7%) without arbuscules was found in the growth chamber (experiment 1) and reached 30% in the alpine conditions together with a small number of arbuscules (0.35 ± 0.24%) (experiment 2). The higher AM colonization in experiment 2 compared to experiment 1 might be due to the response of the AM fungi and the plant to the alpine conditions (Yang et al. 2018). In the cold edaphic conditions of the high alpine region, the physiological activity of AM fungi, along with their interactions with non-host plants, and hence their ecological functions, may differ considerably from their interactions under temperate conditions (Menzel et al. 2016; Bueno et al. 2017). Shi et al. (2015) provided direct evidence that AM fungi would have high intraradical colonization under low temperature conditions. Yang et al. (2017) found that temperature may be an important driver mediating the interaction between host plants and AM fungi in an alpine ecosystem. The plant possibly allocates more carbon to the belowground AM fungi to ensure their survival in the stressful conditions (Hodge 1996; Johnson et al. 2002; Barrett et al. 2014).
Results from these two experiments also emphasize the importance of a neighboring host species in increasing the AM colonization of C. capillacea roots. The host plants in the system ensured the presence of an active AM fungal network. When C. capillacea was grown together with a host plant, considerable root colonization levels were observed in the presence of AM fungi. This is in accordance with previous reports showing that non-host plants can be colonized, although usually at low levels, in the presence of a host (Francis and Read 1995; Veiga et al. 2012, 2013; Ocampo et al. 1980). Some reports have demonstrated that non-host plants are unable to recognize bioactive molecules released by AM fungi and which may be important for the establishment of a functional AM symbiosis (Navazio et al. 2007; Genre et al. 2013). However, some bioactive molecules released by host plants permeate the void of the soil to the root surfaces of non-host plants, inducing AM fungal colonization of the roots of the non-host plant. Lekberg et al. (2015) therefore consider non-host species to be passively colonized by AM fungi. In this context, neighboring host plants established an active AM mycelial network which extensively colonized the roots of C. capillacea and developed rudimentary AM (RAM) phenotypes, albeit without arbuscules. Furthermore, a considerable level of AM colonization of C. capillacea was found in the absence of a host species when plants were grown in two elevation conditions with R.i. This evidence strongly indicates that the AM structures in the roots were not always part of a community mycelial network. One explanation for the occurrence of higher initial colonization at the high altitude might be that stressful environments reduce the ability of C. capillacea to resist colonization by AM fungi (García-Garrido and Ocampo 2002).
The growth response of C. capillacea to AM fungi
As far as we know, this study is the first to demonstrate that the growth of C. capillacea is reduced in the presence of a mycorrhizal plant. Results from both experiments demonstrate that the biomass of C. capillacea declined in the presence of a previously established and active AM mycelium. This evidence is consistent with a range of former reports in which this interaction between non-host plants and host-supported AM fungi often reduces the growth of the colonized non-host plant (Lekberg et al. 2015; Veiga et al. 2013; Yang et al. 2017; Hirrel et al. 1978; Ocampo 1986). It is noteworthy that we found that AM fungi significantly depress the biomass of C. capillacea in the absence of host plants when grown in alpine conditions. This growth reduction may be a consequence of a photosynthate drain by mycorrhizal fungi (Allen and Allen 1990; Raven et al. 2018). Lekberg et al. (2015) conducted a 13CO2 pulse-chase labeling on seven Dianthus individuals in a Danish coastal grassland and found that some C can be delivered from a non-host plant to AM fungi. Another possibility is that AM fungi may benefit indirectly through increasing the competitive advantage of neighboring host plants (Francis and Read 1994; Ocampo 1986) or by removing nutrients, especially P (Eason et al. 1991; Lekberg et al. 2015; Mikkelsen et al. 2008) from the C. capillacea side of the microcosm via AM hyphae to the coexisting host plants. We found a significant relationship between biomass and decline in P content of C. capillacea and the P concentration of M. sativa in experiment 2. It is likely that the host species benefited partially from the AM fungi at the expense of the neighboring M. sativa.
We also found differences in the reduction of C. capillacea biomass due to different inoculum sources. It is possible that the different community composition of AM fungi was responsible for the different effects. Numerous reports indicate that AM fungal diversity and assemblages both play important functional roles in ecosystems (Powell and Rillig 2018; Jiang et al. 2018). Another possible factor may be the selection of host plants. There is some experimental evidence that plants can shape root-associating AM fungal communities by favoring beneficial AM fungal taxa and disfavoring others (Kiers et al. 2011; Bever et al. 2009).
Currently, non-host plant families occupying widely contrasting habitats are divided into two groups, the Brassicaceae type and the Proteaceae type (Lambers and Teste 2013). The Cyperaceae belong to the second group and dominate soils with very low P availability. These species have a range of root specializations that allow them to mine soil P, for example using dauciform roots in various Carex species growing in widely different habitats (Playsted et al. 2006; Lambers et al. 2006). However, there have been no reports of dauciform structures of Carex in China and we did not find dauciform roots in C. capillacea in the present study.
In summary, we found that AM fungi had some ability to colonize the roots of C. capillacea through hyphal networks. In particular, C. capillacea can be extensively colonized by AM fungi under alpine conditions, even in the absence of neighboring mycorrhizal plants. As a consequence, the most important driver of AM colonization of C. capillacea may not be the hyphal network from the host, but the alpine environmental conditions. This seems to disagree with previous reports of other non-host plants (Veiga et al. 2012, 2013). The mechanisms involved still require further study. Negative mycorrhizal effects on the non-host plant C. capillacea are consistent, and AM inoculum source, neighboring host, and growth conditions were all important factors influencing the effect.
References
Allen MF, Allen EB (1990) Carbon source of VA mycorrhizal fungi associated with Chenopodiaceae from a semiarid shrub-steppe. Ecology 71:2019–2021. https://doi.org/10.2307/1937610
Allen MF, Allen EB, Friese CF (1989) Responses of the non-mycotrophic plant Salsola kali to invasion by vesicular-arbuscular mycorrhizal fungi. New Phytol 111:45–49. https://doi.org/10.1111/j.1469-8137.1989.tb04216.x
Augé RM (2001) Water relations, drought and vesicular-arbuscular mycorrhizal symbiosis. Mycorrhiza 11:3–42. https://doi.org/10.1007/s005720100097
Barrett G, Campbell CD, Hodge A (2014) The direct response of the external mycelium of arbuscular mycorrhizal fungi to temperature and the implications for nutrient transfer. Soil Biol Biochem 78:109–117. https://doi.org/10.1016/j.soilbio.2014.07.025
Bever JD, Richardson SC, Lawrence BM, Holmes J, Watson M (2009) Preferential allocation to beneficial symbiont with spatial structure maintains mycorrhizal mutualism. Ecol Lett 12:13–21. https://doi.org/10.1111/j.1461-0248.2008.01254.x
Brundrett MC (2009) Mycorrhizal associations and other means of nutrition of vascular plants: understanding the global diversity of host plants by resolving conflicting information and developing reliable means of diagnosis. Plant Soil 320:37–77. https://doi.org/10.1007/s11104-008-9877-9
Brundrett MC, Kendrick WB (1988) The mycorrhizal status, root anatomy, and phenology of plants in a sugar maple forest. Can J Bot 66:1153–1173. https://doi.org/10.1139/b88-166
Bueno CG, Moora M, Gerz M, Davison J, Öpik M, Pärtel M, Helm A, Ronk A, Kühn I, Zobel M (2017) Plant mycorrhizal status, but not type, shifts with latitude and elevation in Europe. Glob Ecol Biogeogr 26:690–699. https://doi.org/10.1111/geb.12582
Cooke JC, Lefor MW (1998) The mycorrhizal status of selected plant species from Connecticut wetlands and transition zones. Restor Ecol 6:214–222. https://doi.org/10.1111/j.1526-100X.1998.00628.x
Cosme M, Fernández I, van der Heijden MGA, Pieterse CMJ (2018) Non-mycorrhizal plants: the exceptions that prove the rule. Trends Plant Sci 23:577–587. https://doi.org/10.1016/j.tplants.2018.04.004
Eason WR, Newman EI, Chuba PN (1991) Specificity of interplant cycling of phosphorus: the role of mycorrhizas. Plant Soil 137:267–274. https://doi.org/10.1007/BF00011205
Fitter AH, Graves JD, Watkins NK, Robinson D, Scrimgeour C (1998) Carbon transfer between plants and its control in networks of arbuscular mycorrhizas. Funct Ecol 12:406–412. https://doi.org/10.1046/j.1365-2435.1998.00206.x
Francis R, Read DJ (1994) The contributions of mycorrhizal fungi to the determination of plant community structure. Plant Soil 159:11–25. https://doi.org/10.1007/BF00000091
Francis R, Read DJ (1995) Mutualism and antagonism in the mycorrhizal symbiosis, with special reference to impacts on plant community structure. Can J Bot 73:S1301–S1309. https://doi.org/10.1139/b95-391
Gai JP, Cai XB, Feng G, Christie R, Li XL (2006) Arbuscular mycorrhizal fungi associated with sedges on the Tibetan plateau. Mycorrhiza 16:151–157. https://doi.org/10.1007/s00572-005-0031-8
García-Garrido JM, Ocampo JA (2002) Regulation of the plant defence response in arbuscular mycorrhizal symbiosis. J Exp Bot 53:1377–1386. https://doi.org/10.1093/jexbot/53.373.1377
Genre A, Chabaud M, Balzergue C, Puech-Pagès V, Novero M, Rey T, Fournier J, Rochange S, Bécard G, Bonfante P, Barker DG (2013) Short-chain chitin oligomers from arbuscular mycorrhizal fungi trigger nuclear Ca2+ spiking in Medicago truncatula roots and their production is enhanced by strigolactone. New Phytol 198:190–202. https://doi.org/10.1111/nph.12146
Govaerts R, Simpson DA (2007) World checklist of Cyperaceae. The Board of Trustees of the Royal Botanic Gardens, Kew
Grime JP, Mackey JML, Hillier SH, Read DJ (1987) Floristic diversity in a model system using experimental microcosms. Nature 328:420–422. https://doi.org/10.1038/328420a0
Harley JL, Harley EL (1987) A check-list of mycorrhiza in the British flora-addenda, errata and index. New Phytol 107:741–749. https://doi.org/10.1111/j.1469-8137.1987.tb00912.x
Hirrel MC, Mehravaran H, Gerdemann JW (1978) Vesicular-arbuscular mycorrhizae in the Chenopodiaceae and Cruciferae: do they occur? Can J Bot 56:2813–2817. https://doi.org/10.1139/b78-336
Hodge A (1996) Impact of elevated CO2 on mycorrhizal associations and implications for plant growth. Biol Fert Soils 23:388–398. https://doi.org/10.1007/bf00335912
Janos DP (2007) Plant responsiveness to mycorrhizas differs from dependence upon mycorrhizas. Mycorrhiza 17:75–91. https://doi.org/10.1007/s00572-006-0094-1
Ji BM, Gehring CA, Wilson GWT, Miller RM, Flores-Renteria L, Johnson NC (2013) Patterns of diversity and adaptation in Glomeromycota from three prairie grasslands. Mol Ecol 22:2573–2587. https://doi.org/10.1111/mec.12268
Jiang SJ, Liu YJ, Luo JJ, Qin M, Johnson NC, Öpik M, Vasar M, Chai Y, Zhou X, Mao L, du G, An L, Feng H (2018) Dynamics of arbuscular mycorrhizal fungal community structure and functioning along a nitrogen enrichment gradient in an alpine meadow ecosystem. New Phytol 220:1222–1235. https://doi.org/10.1111/nph.15112
Johnson NC (1998) Responses of Salsola kali and Panicum virgatum to mycorrhizal fungi, phosphorus and soil organic matter: implications for reclamation. J Appl Ecol 35:86–94. https://doi.org/10.1046/j.1365-2664.1998.00277.x
Johnson D, Leake JR, Read DJ (2002) Transfer of recent photosynthate into mycorrhizal mycelium of an upland grassland: short-term respiratory losses and accumulation of 14C. Soil Biol Biochem 34:1521–1524. https://doi.org/10.1016/S0038-0717(02)00126-8
Johnson D, Booth RE, Whiteley AS, Bailey MJ, Read DJ, Grime JP, Leake JR (2003a) Plant community composition affects the biomass, activity and diversity of microorganisms in limestone grassland soil. Eur J Soil Sci 54:671–677. https://doi.org/10.1046/j.1351-0754.2003.0562.x
Johnson D, Vandenkoornhuyse PJ, Leake JR, Gilbert L, Booth RE, Grime JP, Young JPW, Read DJ (2003b) Plant communities affect arbuscular mycorrhizal fungal diversity and community composition in grassland microcosms. New Phytol 161:503–515. https://doi.org/10.1046/j.1469-8137.2003.00938.x
Kiers ET, Duhamel M, Beesetty Y, Mensah JA, Franken O, Verbruggen E, Fellbaum CR, Kowalchuk GA, Hart MM, Bago A, Palmer TM, West SA, Vandenkoornhuyse P, Jansa J, Bucking H (2011) Reciprocal rewards stabilize cooperation in the mycorrhizal symbiosis. Science 333:880–882. https://doi.org/10.1126/science.1208473
Konoplenko MA, Güsewell S, Veselkin DV (2017) Taxonomic and ecological patterns in root traits of Carex (Cyperaceae). Plant Soil 420:37–48. https://doi.org/10.1007/s11104-017-3292-z
Lambers H, Teste FP (2013) Interactions between arbuscular mycorrhizal and non-mycorrhizal plants: do non-mycorrhizal species at both extremes of nutrient availability play the same game? Plant Cell Environ 36:1911–1915. https://doi.org/10.1111/pce.12117
Lambers H, Shane MW, Cramer MD, Pearse SJ, Veneklaas EJ (2006) Root structure and functioning for efficient acquisition of phosphorus: matching morphological and physiological traits. Ann Bot 98:693–713. https://doi.org/10.1093/aob/mcl114
Lekberg Y, Rosendahl S, Olsson PA (2015) The fungal perspective of arbuscular mycorrhizal colonization in ‘nonmycorrhizal’ plants. New Phytol 205:1399–1403. https://doi.org/10.1111/nph.13118
Li XL, Zhang JL, Gai JP, Cai XB, Christie P, Li XL (2015) Contribution of arbuscular mycorrhizal fungi of sedges to soil aggregation along an altitudinal alpine grassland gradient on the Tibetan Plateau. Environ Microbiol 17:2841–2857. https://doi.org/10.1111/1462-2920.12792
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular arbuscular mycorrhizal fungi. New Phytol 115:495–501. https://doi.org/10.1111/j.1469-8137.1990.tb00476.x
Menzel A, Hempel S, Manceur AM, Götzenberger L, Moora M, Rillig MC, Zobel M, Kühn I (2016) Distribution patterns of arbuscular mycorrhizal and non-mycorrhizal plant species in Germany. Perspect Plant Ecol 21:78–88. https://doi.org/10.1016/j.ppees.2016.06.002
Mikkelsen BL, Rosendahl S, Jakobsen I (2008) Underground resource allocation between individual networks of mycorrhizal fungi. New Phytol 180:890–898. https://doi.org/10.1111/j.1469-8137.2008.02623.x
Miller RM (2005) The nonmycorrhizal root a strategy for survival in nutrient-improverished soils. New Phytol 165:655–658. https://doi.org/10.1111/j.1469-8137.2005.01331.x
Miller RM, Reinhardt DR, Jastrow JD (1995) External hyphal production of vesicular arbuscular mycorrhizal fungi in pasture and tallgrass prairie communities. Oecologia 103:17–23. https://doi.org/10.1007/BF00328420
Miller RM, Smith CR, Jastrow JD, Bever JD (1999) Mycorrhizal status of the genus Carex (Cyperaceae). Am J Bot 86:547–553. https://doi.org/10.2307/2656816
Muthukumar T, Udaiyan K (2002) Seasonality of vesicular-arbuscular mycorrhizae in sedges in a semi-arid tropical grassland. Acta Oecol 23:337–347. https://doi.org/10.1016/S1146-609X(02)01165-7
Muthukumar T, Udaiyan K, Shanmughavel P (2004) Mycorrhiza in sedges - an overview. Mycorrhiza 14:65–77. https://doi.org/10.1007/s00572-004-0296-3
Navazio L, Moscatiello R, Genre A, Novero M, Baldan B, Bonfante P, Mariani P (2007) A diffusible signal from arbuscular mycorrhizal fungi elicits a transient cytosolic calcium elevation in host plant cells. Plant Physiol 144:673–681. https://doi.org/10.1104/pp.106.086959
Newman EI, Reddell P (1987) The distribution of mycorrhizas among families of vascular plants. New Phytol 106:745–751. https://doi.org/10.1111/j.1469-8137.1987.tb00175.x
Ocampo JA (1986) Vesicular-arbuscular mycorrhizal infection of “host” and “non-host” plants: effect on the growth responses of the plants and competition between them. Soil Biol Biochem 18:607–610. https://doi.org/10.1016/0038-0717(86)90083-0
Ocampo JA, Martin J, Hayman DS (1980) Influence of plant interactions on vesicular-arbuscular mycorrhizal infections. I. Host and non-host plants grown together. New Phytol 84:27–35. https://doi.org/10.1111/j.1469-8137.1980.tb00746.x
Phillips JM, Hayman DS (1970) Improved procedure for clearing roots and staining parasitic and vesicular-arbuscular fungi for rapid assessment of infection. Trans Br Mycol Soc 55:158–161. https://doi.org/10.1016/S0007-1536(70)80110-3
Playsted CWS, Johnston ME, Ramage CM, Edwards DG, Cawthray GR, Lambers H (2006) Functional significance of dauciform roots: exudation of carboxylates and acid phosphatase under phosphorus deficiency in Caustis blakei (Cyperaceae). New Phytol 170:491–500. https://doi.org/10.1111/j.1469-8137.2006.01697.x
Powell CL (1975) Rushes and sedges are non-mycotrophic. Plant Soil 42:481–484. https://doi.org/10.1007/BF00010023
Powell JR, Rillig MC (2018) Biodiversity of arbuscular mycorrhizal fungi and ecosystem function. New Phytol 220:1059–1075. https://doi.org/10.1111/nph.15119
Raven JA, Lambers H, Smith SE, Westoby M (2018) Costs of acquiring phosphorus by vascular land plants: patterns and implications for plant coexistence. New Phytol 217:1420–1427. https://doi.org/10.1111/nph.14967
Rinaudo V, Barberi P, Giovannetti M, van der Heijden MGA (2010) Mycorrhizal fungi suppress aggressive agricultural weeds. Plant Soil 333:7–20. https://doi.org/10.1007/s11104-009-0202-z
Ruotsalainen AL, Aikio S (2004) Mycorrhizal inoculum and performance of nonmycorrhizal Carex bigelowii and mycorrhizal Trientalis europaea. Can J Bot 82:443–449. https://doi.org/10.1139/b04-011
Sanders IR, Koide RT (1994) Nutrient acquisition and community structure in co-occurring mycotrophic and non-mycotrophic old-field annuals. Funct Ecol 8:77–84. https://doi.org/10.2307/2390114
Shi LJ, Yang R, Zhang JL, Cai XB, Christie P, Li XL, Gai JP (2015) Evidence for functional divergence in AM fungal communities from different montane altitudes. Fungal Ecol 16:19–25. https://doi.org/10.1016/j.funeco.2015.03.009
Smith SE, Read DJ (2008) Mycorrhizal symbiosis. Academic Press, Cambridge
Spalink D, Drew BT, Pace MC, Zaborsky JG, Starr JR, Cameron KM, Givnish TJ, Sytsma KJ (2016) Biogeography of the cosmopolitan sedges (Cyperaceae) and the area-richness correlation in plants. J Biogeogr 43:1893–1904. https://doi.org/10.1111/jbi.12802
Tester M, Smith SE, Smith FA (1987) The phenomenon of “nonmycorrhizal” plants. Can J Bot 65:419–431. https://doi.org/10.1139/b87-051
Thomas RL, Sheard RW, Moyer JR (1967) Comparison of conventional and automated procedures for nitrogen, phosphorus, and potassium analysis of plant material using a single digestion. Agron J 59:240–243. https://doi.org/10.2134/agronj1967.00021962005900030010x
van der Heijden MGA, Klironomos JN, Ursic M, Moutoglis P, Streitwolf-Engel R, Boller T, Wiemken A, Sanders IR (1998) Mycorrhizal fungal diversity determines plant biodiversity, ecosystem variability and productivity. Nature 396:69–72. https://doi.org/10.1038/23932
Veiga RSL, Howard K, van der Heijden MGA (2012) No evidence for allelopathic effects of arbuscular mycorrhizal fungi on the non-host plant Stellaria media. Plant Soil 360:319–331. https://doi.org/10.1007/s11104-012-1256-x
Veiga RSL, Faccio A, Genre A, Pieterse CM, Bonfante P, van der Heijden MGA (2013) Arbuscular mycorrhizal fungi reduce growth and infect roots of the non-host plant Arabidopsis thaliana. Plant Cell Environ 36:1926–1937. https://doi.org/10.1111/pce.12102
Veselkin DV, Konoplenko MA, Betekhtina AA (2014) The ability to form mycorrhiza in the genus Carex L. (Cyperaceae): the published data analysis. Plant Life Asian Russia 16:26–35
Waterway MJ, Bruhl JJ, Wilson KL et al (2015) Making Carex monophyletic (Cyperaceae, tribe Cariceae): a new broader circumscription. Bot J Linn Soc 179:1–42. https://doi.org/10.1111/boj.12298
Yang R, Cai XB, Li XL, Christie P, Zhang JL, Gai JP (2017) Temperature-mediated local adaptation alters the symbiotic function in arbuscular mycorrhiza. Environ Microbiol 19:2616–2628. https://doi.org/10.1111/1462-2920.13737
Yang R, Li SM, Qin ZF, Cai X, Li X, Christie P, Zhang J, Feng G, Gai J (2018) Importance of AM fungi and local adaptation in plant response to environmental change: field evidence at contrasting elevations. Fungal Ecol 34:59–66. https://doi.org/10.1016/j.funeco.2018.04.006
Funding
This work was supported by the National Key Research and Development Program of China (2017YFD0200200/2017YFD0200203) and the National Natural Science Foundation of China (NSFC, Projects 41877049 and 41271269).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 32 kb)
Rights and permissions
About this article
Cite this article
Zhang, H., Qin, Z., Chu, Y. et al. Interactions between arbuscular mycorrhizal fungi and non-host Carex capillacea. Mycorrhiza 29, 149–157 (2019). https://doi.org/10.1007/s00572-019-00882-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-019-00882-6