Abstract
The accumulation of phosphorus (P) in plants increases their biomass and resistance/tolerance to organic pollutants. Both characteristics are mandatory for the utilization of plants in phytoremediation. Arbuscular mycorrhizal (AM) fungi improve plant P nutrition, and thus growth. However, only a few studies have focused on the dynamics of inorganic P (Pi) uptake in AM fungal-colonized plants in the presence of organic pollutants. Indeed, most of the results so far were obtained after harvesting the plants, thus by evaluating P concentration and content at a single time point. Here, we investigated the effects of the AM fungus Rhizophagus irregularis MUCL 41833 on the short-term Pi uptake dynamics of Medicago truncatula plants grown in the presence of benzo[a]pyrene (B[a]P), a polyaromatic hydrocarbon (PAH) frequently found in polluted soils. The study was conducted using a non-destructive circulatory semi-hydroponic cultivation system to investigate the short-term Pi depletion from a nutrient solution and as a corollary, the Pi uptake by the AM fungal-colonized and non-colonized plants. The growth, P concentration, and content of plants were also evaluated at harvest. The presence of B[a]P neither impacted the development of the AM fungus in the roots nor the plant growth and Pi uptake, suggesting a marked tolerance of both organisms to B[a]P pollution. A generally higher Pi uptake coupled with a higher accumulation of P in shoots and roots was noticed in AM fungal-colonized plants as compared to the non-colonized controls, irrespective of the presence or absence of B[a]P. Therefore, fungal-colonized plants showed the best growth. Furthermore, the beneficial effect provided by the presence of the AM fungus in roots was similar in presence or absence of B[a]P, thus opening the door for potential utilization in phytomanagement of PAH-polluted soils.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Polyaromatic hydrocarbons (PAHs) produced by combustion of petroleum hydrocarbons (Ball and Truskewycz 2013) are mainly released in the environment by anthropogenic activities (industry, transport, wastewater and sewage sludge, biochar amendments…) (see review by Plouznikoff et al. 2016). Airborne PAHs leave the atmosphere and accumulate in soils via dry or wet deposition (Ball and Truskewycz 2013), polluting soils, affecting their inhabitants, and thus increasing the risks of food chain contamination. Consequently, the Environmental Protection Agency of the United States of America (US-EPA 1978) listed 16 priority PAHs to eliminate from polluted soils, among which is benzo[a]pyrene (B[a]P). The European Chemical Agency (ECHA) listed B[a]P as well as seven other PAHs on Annex XII entry 50 (restrictions on the manufacture, placing on the market and use of certain dangerous substances, mixtures and articles). B[a]P is a high molecular weight PAH that has been classified since 1983 as a “carcinogenic to humans (Group 1)” by the International Agency for Research on Cancer (IARC). This substance “may cause genetic defects, may cause cancer, may damage fertility and may damage the unborn child, is very toxic to aquatic life with long lasting effects and may cause an allergic skin reaction” (ECHA 2016).
Among the numerous methods to remediate organic-polluted soils is the use of plants (so-called phytoremediation; see review by Alkorta and Garbisu 2001). This method is cost-effective and environmental-friendly, and offers a number of side-added values such as bioenergy, biofuel, building and construction materials, or timber (Berthelot et al. 2018). However, phytoremediation also has its drawbacks since only a restricted number of plant species can naturally tolerate and/or accumulate high concentrations of pollutants (Glick 2010) and their biomass production is most often limited under these harsh conditions (Gerhardt et al. 2009). Among the plants frequently reported to tolerate relatively high amounts of organic pollutants and to metabolize them are those belonging to the genus Medicago (Gao et al. 2011; Zhou et al. 2013; Agnello et al. 2016; Lenoir et al. 2017). However, their growth may be limited or slowed in the presence of pollutants, thereby restricting their potential for phytoremediation. Boosting their biomass production, via for instance their association with plant growth promoting microorganisms, can therefore substantially increase their phytoremediation potential.
Arbuscular mycorrhizal (AM) fungi are an attractive option for plant-based environmental remediation. They are critical in the fitness of plants in severely disturbed environments, including those contaminated by organic pollutants (Plouznikoff et al. 2016). These soil fungi are obligate root symbionts which colonize nearly 80% of terrestrial plants (Smith and Read 2008). Their benefits for the growth and survival of plants in soils/substrates contaminated by organic pollutants have been reported in numerous studies (Leyval and Binet 1998; Verdin et al. 2006; Debiane et al. 2008, 2009; Lenoir et al. 2017). For instance, in studies conducted in vitro with root organs of Cichorium intybus and Medicago truncatula, reduced oxidative stress and induction of the expression of genes encoding antioxidant enzymes, as well as regulation of activities of antioxidant enzymes, were found in roots of AM fungal-colonized plants in comparison to non-colonized controls (Debiane et al. 2008, 2009; Lenoir et al. 2017).
Arbuscular mycorrhizal fungi improve plant mineral nutrition (in particular that of inorganic phosphorus, Pi) (Smith and Read 2008). Phosphorus is an essential macronutrient for plant growth, but is rather immobile in soil, and therefore poorly available for plants, limiting crop production in many regions of the world (Holford 1997). Consequently, plants have developed different strategies to increase their access to P in soil, such as the release of carboxylate, changes in root architecture, and production of root clusters (Ryan et al. 2001; Lambers et al. 2003; Lynch and Ho 2005; Shane and Lambers 2005; Javot et al. 2007). A widely distributed strategy is the association with AM fungi, which in some conditions may provide up to 90% of the plant Pi requirements (van der Heijden et al. 2008). Indeed, even if plant roots have access to sufficient Pi, all mycorrhizal plants are likely to obtain at least some Pi via their AM fungal associates (Smith et al. 2003, 2004). This is particularly true for M. truncatula L. c.v. Jemalong which was demonstrated to acquire between 60 and 80% of its Pi from Rhizophagus irregularis BEG87, associated with a positive growth response (Smith et al. 2004).
Although the benefits provided by AM fungi have been demonstrated frequently on several plant species, studies focusing on the role of these microorganisms on the growth and Pi uptake of plants (e.g., M. truncatula) in presence of organic pollutants such as B[a]P are rare (e.g., Calonne et al. 2014). In addition, the beneficial contribution of AM fungi to P accumulation in plants was most often evaluated destructively (i.e., at harvest), thus by quantification of P at a single time point. Therefore, the objectives of the present study were to determine, under a circulatory semi-hydroponic cultivation system, the impact of R. irregularis MUCL 41833 on (1) the short-term dynamics of Pi uptake and (2) the growth, P concentration and content, and biomass, of colonized M. truncatula plants grown in the presence of B[a]P as compared to non-colonized plants.
Materials and methods
Biological materials
Seeds of Zea mays cv. ES Ballade were supplied by the Centre Indépendant de Promotion Fourragère (CIPF—http://www.cipf.be/). The seeds were surface-disinfected as described by Garcés-Ruiz et al. (2017a), placed on wet paper, and germinated during 4 days in plastic containers (Aarts Plastics, Netherlands) placed in the dark at room temperature (~ 20 °C).
Seeds of Medicago truncatula L., cv. Jemalong A17 (SARDI, Australia), were surface-disinfected by immersion in bleach (8% active chloride) for 15 min and rinsed 3 times with sterile (121 °C for 15 min) deionized water (Dupré de Boulois et al. 2006).
The AM fungus Rhizophagus irregularis (Błaszk., Wubet, Renker & Buscot) C. Walker & A. Schüßler comb. nov. MUCL 41833 was supplied by the Glomeromycota in vitro collection (GINCO) (www.mycorrhiza.be/ginco-bel) on the modified Strullu-Romand (MSR) medium (Declerck et al. 1998). The fungus was cultured in vitro as detailed in Cranenbrouck et al. (2005) and subsequently mass-produced during several months on maize.
Colonization of M. truncatula plants
One 13.2 l (56.5 × 36 × 6.5 cm) plastic tray containing a growing medium composed of a mixture of sterilized (121 °C for 15 min) vermiculite and sand (w/w, 1/1, Euroquartz/Agra-Vermiculite Pull Rhenen) was inoculated with spores and colonized root pieces of the mass-produced AM fungus R. irregularis MUCL 41833. The inoculum was placed between two layers of the substrate. A second plastic tray was prepared with the same substrate without AM fungus inoculum. Ten four-day-old maize seedlings were planted in each plastic box for 2 months to produce mycorrhizal (M) and non-mycorrhizal (NM) plants. Maize plants were maintained under controlled conditions at 22 °C/18 °C (day/night), a relative humidity (RH) of 70%, a photoperiod of 16 h day−1, and a photosynthetic photon flux (PPF) of 120 μmol m−2 s−1. The plants received weekly 1 l of modified Hoagland (Hoagland and Arnon 1950) solution (i.e., 90% P-impoverished solution: P = 6.245 mg l−1) designated as “Hoagland low-P” throughout the text.
After 6 weeks, root colonization was estimated on two randomly selected maize plants and had reached 90.6 ± 13.3% (see root colonization protocol below). No colonization was noticed in the NM maize plants. At that time, 100 M. truncatula plants were planted in the two trays and maintained under the same conditions as above. After 1 month, four M. truncatula plants were harvested randomly from each of the two trays and total colonization (%TC) in roots was estimated. The total colonization of the M plants reached 90.8 ± 1.7%, while no colonization was noticed in the NM plants.
Preparation of B[a]P-contaminated substrate
Perlite was first 1 mm-sieved and then washed with deionized water before drying in an oven at 50 °C during 48 h. B[a]P was added in the containers to reach a quantity per container relatively close to some high values reported in the literature (Alarcón et al. 2006; Smith et al. 2006). Twenty-five grams of dry perlite was mixed with 75 ml of acetone containing B[a]P (Sigma-Aldrich, Saint-Louis, USA) in order to obtain 3.2 mg of B[a]P per gram of perlite (i.e., 80 mg per container). Acetone was then evaporated during 48 h in a chemical hood before transfer into the containers. Twenty-five grams of perlite was also mixed with 75 ml of acetone without B[a]P and evaporated as above (i.e., the −B[a]P treatment).
Experimental set-up
The containers consisted of inverted 75 mm diameter wash bottles (500 ml - VWR, USA) cut at the base. A mesh of 100 µm size pore (Prosep B.V.B.A., Belgium) was glued on the top of the bottle to avoid losses of the growing substrate and to prevent roots from growing outside the bottle. Each container was filled with 25 g perlite (i.e., 80 mg B[a]P).
After 1 month of colonization in the planting trays with maize, four M. truncatula plants were transferred in each individual container containing the perlite amended or not with B[a]P. Roots of the M and NM plants were rinsed with deionized water to eliminate the substrate and seed debris prior to transfer into the containers. The containers with plants were randomly disposed in holes made in flex foam supports on two separate tables. A control without plants (four containers for each B[a]P treatment) was also included and similarly randomly disposed. Each container was wrapped with aluminum foil and the surface of the perlite was covered with black quartz (Dekoline, Belgium) to prevent algae development.
Before starting the experiment, the M and NM M. truncatula plants were acclimatized during 14 days in the perlite substrate amended or not with B[a]P. The plants received 200 ml of Hoagland low-P solution every 48 h. The old solution was discarded and replaced by fresh solution.
The experiment was designed as a two-way factorial combination of AM fungal inoculation treatment (NM and M) * B[a]P treatment (absence or presence). Thus, it comprised four treatments, each with six containers, containing each four plants. At transfer, the number of leaves was counted and reached 13 ± 1.2 and 12.7 ± 1 for the M plants and 11.2 ± 0.8 and 11.2 ± 1 for the NM plants in the −B[a]P and +B[a]P containers, respectively. The plants in the M and NM treatments had different numbers of leaves at the start of the experiment according to an ANOVA (p ≤ 0.001). Four non-vegetated containers were added per B[a]P condition and served as negative controls.
Monitoring Pi depletion in Hoagland low-P solution and determining P concentration in biological material
After acclimatization, a circulatory semi-hydroponic cultivation system was set up to monitor the short-term depletion of Pi in the Hoagland low-P solution, according to the protocol described by Garcés-Ruiz et al. (2017a). The solution circulated via a peristaltic pump through the containers with M or NM M. truncatula plants. From the Pi depletion in the circulating solution, Pi uptake rates were deduced for the M and NM M. truncatula plants. Prior to the Pi-uptake dynamics experiment, a flushing was performed to establish an equal nutrient concentration in all containers as described by Garcés-Ruiz et al. (2017a). A preliminary test demonstrated the absence of significant loss (loss not greater than 5%) of B[a]P introduced in perlite during the flushing (results not shown).
After flushing, 1 l of Hoagland low-P solution per plant and NV (non-vegetated) containers was prepared. Twenty ml of the solution was sampled from each bottle before the start of the circulatory system [considered as time 0 (T0)] to determine initial Pi concentration in the glass bottles. The circulating system was then initiated at a speed of 7.4 ml min−1. At defined intervals, 20 ml of the nutrient solution was sampled in Falcon tubes of 50 ml and was then stored at 4 °C in the dark before Pi analysis by inductive coupled plasma atomic emission spectrometer (ICP-AES).
The duration of the experiment was 51 days. At three defined intervals [i.e., at day 14 (1st bio-assay), 28 (2nd bio-assay), and 49 (3rd bio-assay)], the Pi-uptake dynamics were analyzed, and the production of leaves per container enumerated. The leaves were enumerated to calculate the relative growth rate (RGR) as follows: RGR = (Ltx-Ltx-1)/(tx-tx-1), where L corresponds to the number of leaves and tx and tx-1 correspond to each measurement interval (between 0 and 14 days, 14 and 16 days, 16 and 28 days, 28 and 30 days, 30 and 49 days, and 49 and 51 days).
Pi concentration was assessed in the samples of Hoagland low-P solution. Two milliliters of each sample was diluted five times with ultrapure water (Millipore, France). The solution was then acidified with 20 ml of HNO3 at 65% (Merck, Germany) and subsequently analyzed by ICP-AES (ICAP 6500, Thermo-Scientific,USA) (Garcés-Ruiz et al. 2017a). Data obtained (in ppm) were converted in milligram per liter. The Pi depletion values obtained from the medium were standardized according to those obtained by their respective blanks and Pi concentration at time 0 h, following the formula below:
where:
- [P]:
-
Pi concentration in the solution
- x:
-
sample
- NV:
-
non-vegetated containers (i.e., perlite control) respective to the sample analyzed
- T:
-
time considered (12, 24, or 42 h after the start of the circulatory system)
- T0:
-
0 h before the start of the circulatory system
Net Pi uptake was determined from the depletion of Pi in the Hoagland low-P solution circulating through the plants containers. Data were then converted as percentage of Pi depletion which is the percentage of the Pi concentration remaining in the bottles at time 12, 24, and 42 h relative to the initial Pi concentration in the bottles at time T0 = 100%.
At the end of the experiment (i.e., 51 days), the shoots (SDW), roots (RDW), and total (TDW) dry weights were determined after drying in an oven at 50 °C for 48 h. One gram of the root system was used to evaluate AM fungal root colonization (see below) and 100 mg of shoots or roots to estimate P concentration (Garcés-Ruiz et al. 2017a). Phosphorus concentration was converted from ppm to mg kg−1 and content of P was determined according to the dry weights of shoots and roots.
AM fungal root colonization
The root samples were cleared in KOH (10%) and stained with blue ink (Walker 2005) to quantify total (%TC), arbuscular (%AC) and spores/vesicles (%VC) colonization rates using the magnified intersect method (Mc Gonigle et al. McGonigle et al. 1990). Fifteen ca. 1–1.5 cm root fragments were mounted on slides and 3 slides per treatment were observed under a dissecting microscope (Olympus BH2–RFCA, Japan) at 10 X magnification. Three intersections per root fragment were observed for estimation of root colonization.
Data analyses
The effects of “AM fungal inoculation” and “B[a]P treatment” on the RGR, the number of leaves produced, and the percentage of Pi depletion over time were analyzed with a mixed model for repeated measurements, where “time” and a factor resulting from the combination (= “groups”) of “AM fungal inoculation” and “B[a]P treatment” (= four conditions) were regarded as fixed factors. To fulfill the assumptions of normality and homoscedasticity, the RGR and number of leaves responses were transformed using a log10. When the interaction was significant (p ≤ 0.05), meaning that the effect of the “groups” factor depends on the time; slices in the mixed model were used to focus on the effect of the “groups” factor at each time. When the interaction was not significant, we computed Tukey’s test as post-hoc test (p ≤ 0.05) on each significant principal effect (“time” and/or “groups” factors). The factor “table” (2 different tables in the growth chamber) was regarded as a random factor. Data were analyzed with SAS 9.4. statistical software (SAS Institute Inc., USA).
The P concentration and content and the SDW, RDW, and TDW were subjected to a standard least squares test (two-way ANOVA) with REML estimation where “AM fungal inoculation” and “B[a]P treatment” were regarded as fixed factors and “Table” as a random factor. When the random factor “Table” was not significant, it was excluded from the model. The two-way ANOVA was followed by contrast analyses to discriminate among the means between the four treatments. The four treatments were analyzed using the following contrasts: (a) M/−B[a]P vs NM/−B[a]P, (b) M/−B[a]P vs M/+B[a]P, (c) M/+B[a]P vs NM/+B[a]P, and (d) NM/−B[a]P vs NM/+B[a]P. Normal distribution of residuals was checked before analyses. Data were analyzed using the JMP Pro 13 statistical software (SAS Institute Inc., USA).
A one-way ANOVA was used to test the difference in the number of leaves of M. truncatula plants before transfer to the containers and the root colonization rate between M plants grown in the presence and absence of B[a]P after arcsin transformation. Data were analyzed using the JMP Pro 13 statistical software (SAS Institute Inc., USA).
Results
Short-term Pi depletion in the Hoagland low-P nutrient solution
Short-term dynamics of Pi depletion (throughout the text expressed as the percentage of Pi depletion - see definition in materials and method section) was conducted at day 14 (1st bio-assay, Fig. 1(A)), 28 (2nd bio-assay, Fig. 1(B)), and 49 (3rd bio-assay, Fig. 1(C)), and measurements made at three time-points (12, 24, and 42 h).
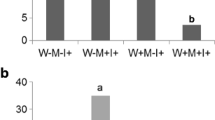
Short-term Pi depletion analysis (expressed as the percentage of Pi concentration remaining in the nutrient solution at times 12, 24 and 42 h relative to the initial Pi concentration in the nutrient solution at T0) in Rhizophagus irregularis MUCL 41833 mycorrhizal (M) or non-mycorrhizal (NM) Medicago truncatula plants grown in a circulatory semi-hydroponic container system fed with a P-impoverished Hoagland solution supplemented (+B[a]P) or not (−B[a]P) with B[a]P at the concentration of 3.2 mg g−1 perlite. Measurements were made after (A) 14 days, (B) 28 days and (C) 49 days of growth. Data are presented as means (N = 6 for the M/+B[a]P, N = 5 for the other treatments) ± SD. For each time, means accompanied by the same letter do not differ significantly, according to the mixed model for repeated measurements and slices post-hoc tests (p ≤ 0.05). The absence of lower case letters at each time of analysis means no differences between the analyzed treatments
The percentage of Pi depletion in the nutrient solution significantly decreased (p < 0.001) over time for the 3 bio-assays.
In the 1st bio-assay, the initial Pi concentration in the nutrient solution reached 6.26 mg l−1. The plants took around 30% (i.e., 1.9 mg) of the initial Pi concentration in the nutrient solution. An interaction was determined between the factors “time” and “groups” (p = 0.021), suggesting differences between the four conditions at different times of analysis. Indeed, no differences in percentage of Pi depletion was noticed between M and NM plants at 12 and 24 h, irrespective of the presence or absence of B[a]P (p = 0.762 and 0.31 respectively), while at 42 h, the M plants grown in presence of the pollutant took up 86% more Pi than the NM plants (p = 0.002). The presence of B[a]P did not impact the percentage of Pi uptake by the M and NM plants.
During the 2nd bio-assay, the plants took between 20 and 70% of the initial Pi concentration in the nutrient solution (i.e., 6.35 mg l−1), which corresponded to 1.27 and 4.45 mg of Pi. A significant interaction was also noticed between “time” and “groups” (p < 0.001). Indeed, whereas plants from the four treatments took up similar Pi quantity in the nutrient solution at 12 h (p = 0.136), Pi uptake differed between treatments at 24 and 42 h (p = 0.004 and < 0.001 respectively). A higher percentage of Pi depletion was measured in the nutrient solution of the M/−B[a]P treatment suggesting a higher uptake of Pi by the plants in the M/−B[a]P treatment as compared to those in the NM/−B[a]P treatment at 24 h (p = 0.026). To the contrary, M and NM plant grown in presence of the B[a]P took up similar amount of Pi (p = 0.135). At 42 h, M/−B[a]P and M/+B[a]P took up 77.7 and 66.4% more Pi than NM/−B[a]P and NM/+B[a]P, respectively (p = 0.002 and 0.003 respectively). The presence of B[a]P did not impact the percentage of Pi uptake by the M and NM plants.
For the 3rd bio-assay, the plants took around 76% (i.e., 4.81 mg Pi) of the initial Pi concentration (i.e., 6.35 mg l−1) in the nutrient solution. Contrarily to the 1st and 2nd bio-assays, plants took up similar Pi concentration in the nutrient solution, irrespective of the presence or absence of the AM fungus and of the pollutant.
Plant and fungal development
The impact of B[a]P in the growth medium and root colonization by the AM fungus were evaluated on the development of M. truncatula plants by enumerating the number of leaves, as well as determining the RDW, SDW, and TDW.
The number of leaves produced after transfer into the containers at day 14, 16, 28, 30, 49, and 51 by the NM or M M. truncatula plants grown in absence or presence of B[a]P is shown in Fig. 2 and the relative growth rate (RGR) in Table 1. Plants developed well throughout the experiment as evidenced by the significant increase in number of leaves produced (p < 0.001). An interaction was noticed between the factors “time” and “groups” (p = 0.006), suggesting differences between the four treatments over time. Indeed, a significantly higher number of leaves was enumerated in the M versus NM plants, irrespective of the presence or absence of B[a]P at 14 and 16 days (p < 0.001 for 14 and 16 h), whereas this difference was not observed later. The number of leaves produced per container was 87.9 and 76.8% lower for NM/−B[a]P and NM/+B[a]P plants in comparison with M/−B[a]P and M/+B[a]P plants respectively, 14 days after transfer of the plants to the containers. The RGR was low during the first weeks of growth in the containers, until day 16. It then increased until day 30. After the 2nd bio-assay (i.e., 30 days after transfer into containers), the RGR decreased until the beginning of the last bio-assay (49 days after transfer into containers), meaning that the number of new leaves produced was slow between the 2nd and the 3rd bio-assay. The RGR strongly increased during the 3rd bio-assay. No interaction between the factors “time” and “groups” was observed, but the presence of the fungus consistently increased the RGR, irrespective of the time of observation (p < 0.001), and also irrespective of the presence or absence of B[a]P. This suggested that the presence of the fungus improved the plant leave production per day along the experiment.
Number of leaves produced by Rhizophagus irregularis MUCL 41833 mycorrhizal (M) or non-mycorrhizal (NM) Medicago truncatula plants after 14, 16, 28, 30, 49 and 51 days of growth in a circulatory semi-hydroponic container system fed with a P-impoverished Hoagland solution supplemented (+B[a]P) or not (−B[a]P) with B[a] P at the concentration of 3.2 mg g−1 perlite. Data are presented as means (N = 6 for the M/+B[a]P treatment; N = 5 for the other treatments) ± SD. For each analyzed time, means accompanied by the same letter do not differ significantly, according to the mixed model for repeated measurements and slices post-hoc tests (p ≤ 0.05). $ indicates that M/−B[a]P and NM/−B[a]P (p = 0.001), M/+B[a]P and NM/+B[a]P (p = 0.001), M/+B[a]P and NM/−B[a]P (p < 0.001) and M/−B[a]P and NM/+B[a]P (p = 0.006) are significantly different, but there was no difference between M/−B[a]P and M/+B[a]P (p = 0.959) and NM/−B[a]P and NM/+B[a]P (p = 0.864). + indicates that M/−B[a]P and NM/−B[a]P are significantly different (p < 0.001), M/+B[a]P and NM/+B[a]P (p = 0.021), M/+B[a]P and NM/−B[a]P (p < 0.001), as well as M/−B[a]P and NM/+B[a]P (p = 0.011), but there was no difference between M/−B[a]P and M/+B[a]P (p = 0.993) and NM/−B[a]P and NM/+B[a]P (p = 0.176)
At harvest, plant weight per container ranged between 2.8 and 4.6 g. The presence of the AM fungus significantly affected RDW, SDW and TDW, which were higher for M plants in comparison with NM ones (Fig. 3) irrespective of the presence of B[a]P (p = 0.004, 0.002, and ≤ 0.001 for RDW, SDW, and TDW respectively). The effect of the presence of the AM fungus was larger for the RDW of plants grown in the presence of B[a]P than in its absence, according to the contrasts test.
(a) Shoots dry weight (SDW - g), (b) roots dry weight (RDW - g; shown as positive values below the abscissa) and (c) total dry weight (TDW - g) in Rhizophagus irregularis MUCL 41833 mycorrhizal (M) or non-mycorrhizal (NM) Medicago truncatula plants grown in a circulatory semi-hydroponic container system fed with a P-impoverished Hoagland solution supplemented (+B[a]P) or not (−B[a]P) with B[a]P at the concentration of 3.2 mg g−1 perlite, 51 days after transfer into containers. Data are presented as means (N = 6 for M/+B[a]P, N = 5 for the other treatments) ± SD. The SDW, RDW and TDW were analyzed by a two-way ANOVA followed by contrasts post-hoc test (p ≤ 0.05). Different lower case letters indicate significant differences between NM/−B[a]P and NM/+B[a]P; different upper case letters indicate significant differences between M/−B[a]P and M/+B[a]P and the presence of * indicates a significant difference between M and NM plants, according to the contrasts test (p ≤ 0.05)
Colonization rates of M. truncatula roots after 51 days of growth in the absence or presence of B[a]P are presented in Table 2. The %TC, %AC, and %VC of M. truncatula roots remained similar in the presence or absence of B[a]P.
P concentration and total P content in plants
The P concentration and content of shoots and roots of the plants were evaluated at the end of the experiment (day 51) (Table 3). Whereas the P concentration in shoots remained similar irrespective of the treatment, the P concentration in roots and the P content in shoots, roots and in the whole plant were significantly affected by the presence of R. irregularis. Indeed, the M plants contained more P in shoots (p = 0.043), roots (p = 0.001) and in the whole plant (p = 0.005), than the NM plants. On the other hand, B[a]P did not affect any of these parameters. The interaction “AM fungal inoculation * B[a]P treatment” had an effect on the P concentration in roots (p = 0.029). Whereas −B[a]P roots accumulated similar P concentrations (p = 0.922), M plants grown in presence of the pollutant accumulated 47% more P than the NM ones (p = 0.005).
Discussion
The circulatory semi-hydroponic cultivation system developed for maize plants by Garcés-Ruiz et al. (2017a) was used for M. truncatula plants in our experiment. High root colonization (42.7 ± 15.1% TC) and production of leaves (increased by 17 times in 51 days of culture) were obtained for the plants grown in absence of B[a]P, supporting the suitability of the cultivation system for growing the plant and fungus associates.
M. truncatula and the AM fungus were chosen in our study for their numerous advantages in term of phytomanagement strategies. Indeed, M. truncatula and more generally plants from the Medicago genus are able to accumulate and metabolize organic pollutants (Gao et al. 2011; Zhou et al. 2013; Lenoir et al. 2017). As reviewed by Agnello et al. (2016), these plants grow fast, produce large biomass above and below ground, develop an extensive tap root system with considerable soil depth exploration ability, associate with symbiotic microorganisms (i.e. Rhizobium and AM fungi) and are widely distributed and well adapted to different climatic conditions. Similarly, R. irregularis is widely distributed over the world and has been demonstrated to grow in petroleum, crude oil and PAH-disturbed environments (De la Providencia et al. 2015; Lenoir et al. 2016a; Garcés-Ruiz et al. 2017b). R. irregularis has been reported to improve PAH dissipation in the mycorrhizosphere, reduce PAH accumulation in mycorrhizal hosts and mitigate PAH-induced stresses in host plant (as oxidative stress) (Verdin et al. 2006; Alarcón et al. 2008; Cheung et al. 2008; Debiane et al. 2008, 2009; Liu and Dalpé 2009; Lenoir et al. 2016b, 2017). It was further shown to stimulate the establishment of microbial communities, particularly PAH degrading bacteria, in the mycorrhizosphere (Corgié et al. 2006; Zhou et al. 2009). Finally, this fungus is well-known to transport P to plants, even under environmental stress conditions, and thus improve P nutrition of plants (Smith et al. 2004; De Jaeger et al. 2011; Zocco et al. 2011; Calonne et al. 2014; Garcés-Ruiz et al. 2017a).
The perlite substrate with circulating Hoagland low-P nutrient solution was adequate for growing the AM fungus-colonized and non-colonized M. truncatula plants and thus for measuring over time the percentage Pi depletion in the nutrient solution and by corollary the percentage Pi uptake in the plant systems, in the presence as well as absence of B[a]P. Indeed, the dynamics of Pi uptake by plants increased over time and was associated with increasing growth of plants in the absence as well as in the presence of the AM fungus or B[a]P.
As expected, the B[a]P at concentration considered in our study (3.2 mg of B[a]P per g of perlite or 80 mg per container) did not affect the production of leaves, the RGR and the Pi uptake and accumulation by NM and M plants confirming the tolerance of M. truncatula to high concentrations of B[a]P. In addition, the presence of the fungus, which also appeared unaffected by the pollutant, was beneficial for the plants in presence as well as in absence of B[a]P. The uptake of Pi was significantly higher in the M plants as compared to the NM plants at 42 h for plants grown in presence of the pollutant for the 1st and the 2nd bio-assay, but did not differ in the 3rd bio-assay. In absence of the pollutant, M plants also took up more Pi after 24 h during the 2nd bio-assay.
The absence of AM fungal effect on Pi uptake at 12 h in the 1st and the 2nd bio-assay and at 24 h in the 1st bio-assay possibly could be ascribed to the slow growth of the M and NM plants at that time. Thus, a reduced Pi uptake was noticed in both treatments and no significant differences could be detected. This was corroborated by the limited production of leaves during the 1st bio-assay. For the 3rd bio-assay, the absence of differences in Pi uptake between the plants in the M and NM treatments irrespective of the presence of absence of the pollutant could be the result of an identical requirement of P for their growth, because the number of leaves produced was similar between both treatments.
Conversely, the significantly higher Pi uptake in the M plants as compared to the NM plants at the end of the 1st bio-assay (i.e. at 42 h for the +B[a]P plants) and during the 2nd bio-assay (at 24 h for the –B[a]P plants and at 42 h for the –B[a]P and + B[a]P plants) suggested an active contribution of the AM fungus to the uptake of Pi in absence but also in presence of B[a]P. Interestingly, Calonne et al. (2014) showed with root organ cultures (ROC) that the extraradical mycelium (ERM) of the AM fungus was able to transport Pi to its host plant even if the amount transported was reduced when ERM developed in presence of B[a]P. This suggested that the mechanism of transport was still active. Because the root colonization of M. truncatula plants in our study appeared not affected by the presence of B[a]P, an active transport of P from the fungus to the roots could be expected. Garcés-Ruiz et al. (2017a) with R. irregularis as well as Colpaert et al. (1999) with ectomycorrhizal fungi, hypothesized that the increased uptake of Pi could be ascribed to an improved nutrient foraging in the substrate by the fungus. In our study, the ERM density was not measured, but was expected to be close to the control because the root colonization was not affected by B[a]P. Consequently, the sum of area covered by the hyphae and roots in contact with the percolating solution was probably much higher in the M plants as compared to the roots alone of the NM plants, increasing the Pi depletion in the nutrient solution. Indeed, a better growth of the roots was measured in the M plants as compared to the NM ones, as shown by the RDW. Therefore, a possibly greater number of Pi transporters might operate in the M root systems (Pi transporters of the roots and of the fungus) than in the NM roots as suggested by Colpaert et al. (1999) and Garcés-Ruiz et al. (2017a). In the presence of the pollutant, the increased Pi uptake by M plants in comparison with NM ones could be explained by a possibly better B[a]P degradation in the rhizosphere of M plants, as earlier reported (Joner et al. 2001; Cheung et al. 2008; Liu and Dalpé 2009; Wu et al. 2011; Aranda et al. 2013; Zhou et al. 2013; Lenoir et al. 2016b, 2017). A lower PAH accumulation in the M plants in comparison with the NM ones has also been hypothesized (Lenoir et al. 2016a, 2017), coupled with an accumulation of B[a]P in lipid bodies of the AM fungus (Verdin et al. 2006), decreasing the B[a]P concentration in the substrate. Such dissipation of B[a]P in the rhizosphere of roots could lead to a better Pi uptake by M plants grown in the presence of B[a]P. Finally, it also has been extensively described in the literature that the association with an AM fungus changes the metabolism of sugars, polyamines and lipids in plants as well as other aspects of plant physiology (photosynthesis, osmotic balance …) and reduces PAH-induced oxidative stress (Plouznikoff et al. 2016; Lenoir et al. 2017), which could lead to an improved Pi uptake and accumulation.
The ability of mycorrhizal plants to take up and accumulate proportionally more P than NM plants has been frequently suggested or observed when grown in the presence of PAHs (Joner and Leyval 2001; Rabie 2004; Liu and Dalpé 2009; Wu et al. 2011; Yu et al. 2011; Lu and Lu 2015; Lenoir et al. 2016a), diesel (Tang et al. 2009) and crude oil (Nwoko 2014). Our results confirm these studies, as shown by the P concentration and content in roots and shoots of M plants grown in the presence of the pollutant, resulting from better Pi uptake after 42 h at the 1st and the 2nd bio-assays by M plants in comparison with NM plants. Indeed, the concentration of P in the roots of the M plants grown in presence of the pollutant was significantly higher as compared to the roots of the NM plants, as earlier reported by Liu and Dalpé (2009) with leek. In addition, P content in roots and in whole plants was significantly higher in the M plants as compared to the NM ones, irrespective of the presence or absence of B[a]P. As mentioned by Olsson et al. (2002), the sink for P is normally the intraradical mycelium. An accumulation of P in intraradical structures could be directly available to the host, a non-negligible advantage for the mycorrhizal host.
As a consequence, the higher Pi uptake by M plants and their higher P accumulation was associated with better growth of the M plants than the NM ones along the experiment, as shown by the higher RGR during this period. Therefore, the SDW was higher in the M versus NM plants at harvest, and M shoots accumulated more P than NM shoots, irrespective of the presence of absence of the pollutant.
In conclusion, M. truncatula did not suffer from the concentration of B[a]P used in the present experiment, as expected. Interestingly, its association with R. irregularis MUCL 41833 increased its biomass as compared to the NM controls in absence as well as presence of B[a]P, a result most probably related to a better Pi uptake and a larger P accumulation in plant tissues. The AM response of the plant was similar in the absence or presence of B[a]P. In addition, the fungus seemed not impacted by the pollutant since root colonization parameters were similar in absence or presence of B[a]P. Knowing that such “growth-benefit” is one of the most important reasons for considering AM fungi in phytoremediation strategies (Joner et al. 2001; Joner and Leyval 2003; Wu et al. 2011), and that these symbionts present remarkable advantages in term of phytoremediation, the pairing M. truncatula/R. irregularis MUCL 41833 may potentially represent promising associates for phytomanagement of B[a]P-polluted soils and possibly other PAHs. An in-depth study of their capacity to accumulate and degrade high molecular weight PAHs thus merits further attention.
References
Agnello AC, Bagard M, Van Hullebusch E, Esposito G, Huguenot D (2016) Comparative bioremediation of heavy metals and petroleum hydrocarbons co-contaminated soil by natural attenuation, phytoremediation, bioaugmentation and bioaugmentation-assisted phytoremediation. Sci Tot Environ 563:693–703. https://doi.org/10.1016/j.scitotenv.2015.10.061
Alarcón A, Delgadillo-Martínez J, Franco-Ramírez A, Davies FT Jr, Ferrera-Cerrato R (2006) Influence of two polycyclic aromatic hydrocarbons on spore germination, and phytoremediation potential of Gigaspora margarita-Echynochloa polystachya symbiosis in benzo [a]pyrene-polluted substrate. Rev Int Contam Ambient 22:39–47
Alarcón A, Davies FT Jr, Autenrieth RL, Zuberer DA (2008) Arbuscular mycorrhiza and petroleum-degrading microorganisms enhance phytoremediation of petroleum-contaminated soil. Int J Phytoremed 10:251–263. https://doi.org/10.1080/15226510802096002
Alkorta I, Garbisu C (2001) Phytoremediation of organic contaminants in soils. Bioresour Technol 79:273–276. https://doi.org/10.1016/S0960-8524(01)00016-5
Aranda E, Scervino JM, Godoy P, Reina R, Ocampo JA, Wittich RM, García-Romera I (2013) Role of arbuscular mycorrhizal fungus Rhizophagus custos in the dissipation of PAHs under root-organ culture conditions. Environ Pollut 181:182–189. https://doi.org/10.1007/s00572-003-0285-y
Ball A, Truskewycz A (2013) Polyaromatic hydrocarbon exposure: an ecological impact ambiguity. Environ Sci Pollut Res 20:4311–4326. https://doi.org/10.1007/s11356-013-1620-2
Berthelot C, Blaudez D, Beguiristain T, Chalot M, Leyval C (2018) Co-inoculation of Lolium perenne with Funneliformis mosseae and the dark septate endophyte Cadophora sp. in a trace element-polluted soil. Mycorrhiza 28:301–314. https://doi.org/10.1007/s00572-018-0826-z
Calonne M, Fontaine J, Tisserant B, Dupré de Boulois H, Grandmougin-Ferjani A, Declerck S, Lounès-Hadj Sahraoui A (2014) Polyaromatic hydrocarbons impair phosphorus transport by the arbuscular mycorrhizal fungus Rhizophagus irregularis. Chemosphere 104:97–104. https://doi.org/10.1016/j.chemosphere.2013.10.070
Cheung KC, Zhang JY, Deng HH, Ou YK, Leung HM, Wu SC, Wong MH (2008). Interaction of higher plant (jute), electrofused bacteria and mycorrhiza on anthracene biodegradation. Biores Technol99:2148–2155. https://doi.org/10.1016/j.biortech.2007.05.037
Colpaert JV, Van Tichelen KK, Van Assche JA, Van Laere A (1999) Short-term phosphorus uptake rates in mycorrhizal and non-mycorrhizal roots of intact Pinus sylvestris seedlings. New Phytol 143:589–597. https://doi.org/10.1046/j.1469-8137.1999.00471.x
Corgié SC, Fons F, Beguiristain T, Leyval C (2006) Biodegradation of phenanthrene, spatial distribution of bacterial populations and dioxygenase expression in the mycorrhizosphere of Lolium perenne inoculated with Glomus mosseae. Mycorrhiza16:207–212. https://doi.org/10.1007/s00572-006-0049-6
Cranenbrouck S, Voets L, Bivort C, Renard L, Strullu DG, Declerck S (2005) Methodologies for in vitro cultivation of arbuscular mycorrhizal fungi with root organs. In: Declerck,S, Strullu DG, Fortin A (eds) In vitro culture of mycorrhizas. Springer-Verlag Berlin, Heidelberg, pp 341–375. https://doi.org/10.1007/3-540-27331-x_18
De Jaeger N, de la Providencia IE, Dupré de Boulois H, Declerck S (2011) Trichoderma harzianum might impact phosphorus transport by arbuscular mycorrhizal fungi. FEMS Microbiol Ecol77:558–567. https://doi.org/10.1111/j.1574-6941.2011.01135.x
Debiane D, Garçon G, Verdin A, Fontaine J, Durand R, Grandmougin-Ferjani A, Shirali P, Lounès-Hadj Sahraoui A (2008) In vitro evaluation of the oxidative stress and genotoxic potentials of anthracene on mycorrhizal chicory roots. Environ Exp Bot64:120–127. https://doi.org/10.1016/j.envexpbot.2008.04.003
Debiane D, Garçon G, Verdin A, Fontaine J, Durand R, Shirali P, Grandmougin-Ferjani A, Lounès-Hadj Sahraoui A (2009) Mycorrhization alleviates benzo [a]pyrene-induced oxidative stress in an in vitro chicory root model. Phytochemistry 70:1421–1427. https://doi.org/10.1016/j.phytochem.2009.07.002
Declerck S, Strullu DG, Plenchette C (1998) Monoxenic culture of the intraradical forms of Glomus sp. isolated from a tropical ecosystem: a proposed methodology for germplasm collection. Mycologia 90:579–585. https://doi.org/10.2307/3761216
van der Heijden MG, Bardgett RD, van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310. https://doi.org/10.1111/j.1461-0248.2007.01139.x
Dupré de Boulois H, Voets L, Delvaux B, Jakobsen I, Declerck S (2006) Transport of radiocaesium by arbuscular mycorrhizal fungi to Medicago truncatula under in vitro conditions. Environ Microbiol 8:1926–1934. https://doi.org/10.1111/j.1462-2920.2006.01070.x
ECHA (2016). https://echa.europa.eu/documents/10162/176064a8-0896-4124-87e1-75cdf2008d59
Gao Y, Li Q, Ling W, Zhu X (2011) Arbuscular mycorrhizal phytoremediation of soils contaminated with phenanthrene and pyrene. J Hazard Mater 185(2–3):703–709
Garcés-Ruiz M, Calonne-Salmon M, Plouznikoff K, Misson C, Navarrete-Mier M, Cranenbrouck S, Declerck S (2017a) Dynamics of short-term phosphorus uptake by intact mycorrhizal and non-mycorrhizal maize plants grown in a circulatory semi-hydroponic cultivation system. Front plant Sci 8:1471. https://doi.org/10.3389/fpls.2017.01471
Garcés-Ruiz M, Senés-Guerrero C, Declerck S, Cranenbrouck S (2017b) Arbuscular mycorrhizal fungal community composition in Carludovica palmata, Costus scaber and Euterpe precatoria from weathered oil ponds in the Ecuadorian Amazon. Front Microbiol 8:2134. https://doi.org/10.3389/fmicb.2017.02134
Gerhardt KE, Huang XD, Glick BR, Greenberg BM (2009) Phytoremediation and rhizoremediation of organic soil contaminants: potential and challenges. Plant Sci 176:20–30. https://doi.org/10.1016/j.plantsci.2008.09.014
Glick BR (2010) Using soil bacteria to facilitate phytoremediation. Biotechnol Adv 28:367–374. https://doi.org/10.1016/j.biotechadv.2010.02.001
Hoagland DR, Arnon DI (1950) The water-culture method for growing plants without soil. Circular - California Agricultural Experiment Station, 347, 2nd ed.
Holford ICR (1997) Soil phosphorus: its measurement, and its uptake by plants. Austral J Soil Res 35:227–240. https://doi.org/10.1071/s96047
Javot H, Pumplin N, Harrison MJ (2007) Phosphate in the arbuscular mycorrhizal symbiosis: transport properties and regulatory roles. Plant Cell Environ 30:310–322. https://doi.org/10.1111/j.1365-3040.2006.01617.x
Joner EJ, Leyval C (2001) Influence of arbuscular mycorrhiza on clover and ryegrass grown together in a soil spiked with polycyclic aromatic hydrocarbons. Mycorrhiza10:155–159. https://doi.org/10.1007/s005720000071
Joner EJ, Leyval C (2003) Rhizosphere gradients of polycyclic aromatic hydrocarbon (PAH) dissipation in two industrial soils and the impact of arbuscular mycorrhiza. Environ Sci Technol 37:2371–2375. https://doi.org/10.1021/es020196y
Joner EJ, Johansen A, Loibner AP, dela Cruz MA, Szolar OH, Portal JM, Leyval C (2001) Rhizosphere effects on microbial community structure and dissipation and toxicity of polycyclic aromatic hydrocarbons (PAHs) in spiked soil. Environ Sci Technol 35:2773–2777. https://doi.org/10.1021/es000288s
de la Providencia IE, Stefani FO, Labridy M, St-Arnaud M, Hijri M (2015) Arbuscular mycorrhizal fungal diversity associated with Eleocharis obtusa and Panicum capillare growing in an extreme petroleum hydrocarbon-polluted sedimentation basin. FEMS Microbiol Lett 362:fnv081. https://doi.org/10.1093/femsle/fnv081
Lambers H, Cramer MD, Shane MW, Wouterlood M, Poot P, Veneklaas EJ (2003) Introduction—structure and functioning of cluster roots and plant responses to phosphate deficiency. Plant Soil 248:ix–xix. https://doi.org/10.1023/a:1025561812696
Lenoir I, Lounès-Hadj Sahraoui A, Laruelle F, Dalpé Y, Fontaine J (2016a) Arbuscular mycorrhizal wheat inoculation promotes alkane and polycyclic aromatic hydrocarbon biodegradation: microcosm experiment on aged-contaminated soil. Environ Pollut 213:549–560. https://doi.org/10.1016/j.envpol.2016.02.056
Lenoir I, Lounès-Hadj Sahraoui A, Fontaine J (2016b) Arbuscular mycorrhizal fungal-assisted phytoremediation of soil contaminated with persistent organic pollutants: a review. Eur J Soil Sci 67:624–640. https://doi.org/10.1111/ejss.12375
Lenoir I, Fontaine J, Tisserant B, Laruelle F, Lounès-Hadj Sahraoui A (2017) Beneficial contribution of the arbuscular mycorrhizal fungus, Rhizophagus irregularis, in the protection of Medicago truncatula roots against benzo [a] pyrene toxicity. Mycorrhiza 27:465–476. https://doi.org/10.1007/s00572-017-0764-1
Leyval C, Binet P (1998) Effect of polyaromatic hydrocarbons in soil on arbuscular mycorrhizal plants. J Environ Qual 27:402–407. https://doi.org/10.2134/jeq1998.00472425002700020022x
Liu A, Dalpé Y (2009) Reduction in soil polycyclic aromatic hydrocarbons by arbuscular mycorrhizal leek plants. Int J Phytoremed 11:39–52. https://doi.org/10.1080/15226510802363444
Lu YF, Lu M (2015) Remediation of PAH-contaminated soil by the combination of tall fescue, arbuscular mycorrhizal fungus and epigeic earthworms. J Hazard Mater 285:535–541. https://doi.org/10.1016/j.jhazmat.2014.07.021
Lynch JP, Ho MD (2005) Rhizoeconomics: carbon costs of phosphorus acquisition. Plant Soil 269:45–56. https://doi.org/10.1007/s11104-004-1096-4
McGonigle TP, Miller MH, Evans DG, Fairchild GL, Swan JA (1990) A new method which gives an objective measure of colonization of roots by vesicular-arbuscular mycorrhizal fungi. New Phytol 115:495–501. https://doi.org/10.1111/j.1469-8137.1990.tb00476.x
Nwoko CO (2014) Effect of arbuscular mycorrhizal (AM) fungi on the physiological performance of Phaseolus vulgaris grown under crude oil contaminated soil. J Geosci Environ Prot 2:9–14. https://doi.org/10.4236/gep.2014.24002
Olsson PA, van Aarle IM, Allaway WG, Ashford AE, Rouhier H (2002) Phosphorus effects on metabolic processes in monoxenic arbuscular mycorrhiza cultures. Plant Physiol 130:1162–1171. https://doi.org/10.1104/pp.009639
Plouznikoff K, Declerck S, Calonne-Salmon M (2016) Mitigating abiotic stresses in crop plants by arbuscular mycorrhizal fungi. In: Vos C, Kazan K (eds) Belowground defence strategies in plants. Springer International Publishing, Switzerland, pp 341–400. https://doi.org/10.1007/978-3-319-42319-7_15
Rabie GH (2004) Using wheat-mungbean plant system and arbuscular mycorrhiza to enhance in-situ bioremediation. Food Agri Environ 2:381–390. https://doi.org/10.1234/4.2004.234
Ryan PR, Delhaize E, Jones DL (2001) Function and mechanism of organic anion exudation from plant roots. Annu Rev Plant Biol 52:527–560. https://doi.org/10.1146/annurev.arplant.52.1.527
Shane MW, Lambers H (2005) Cluster roots: a curiosity in context. Plant Soil 274:101–125. https://doi.org/10.1007/1-4020-4099-7_5
Smith SE, Read DJ (2008) Mycorrhizal Symbiosis, 3rd Ed, Academic Press
Smith SE, Smith FA, Jakobsen I (2003) Mycorrhizal fungi can dominate phosphate supply to plants irrespective of growth responses. Plant Physiol 133:16–20. https://doi.org/10.1104/pp.103.024380
Smith SE, Smith FA, Jakobsen I (2004) Functional diversity in arbuscular mycorrhizal (AM) symbioses: the contribution of the mycorrhizal P uptake pathway is not correlated with mycorrhizal responses in growth or total P uptake. New Phytol 162:511–524. https://doi.org/10.1111/j.1469-8137.2004.01039.x
Smith MJ, Flowers TH, Duncan HJ, Alder J (2006) Effects of polycyclic aromatic hydrocarbons on germination and subsequent growth of grasses and legumes in freshly contaminated soil and soil with aged PAHs residues. Environ Pollut 141:519–525. https://doi.org/10.1016/j.envpol.2005.08.061
Tang M, Chen H, Huang JC, Tian ZQ (2009) AM fungi effects on the growth and physiology of Zea mays seedlings under diesel stress. Soil Biol Biochem 41:936–940. https://doi.org/10.1016/j.soilbio.2008.11.007
U.S. EPA. Federal Register (43 FR 4108), (Washington, DC: U.S. EPA. January 1978). List of priority pollutants. https://www.epa.gov/sites/production/files/2015-09/documents/priority-pollutant-list-epa.pdf
Verdin A, Lounès-Hadj Sahraoui A, Fontaine J, Grandmougin-Ferjani A, Durand R (2006) Effects of anthracene on development of an arbuscular mycorrhizal fungus and contribution of the symbiotic association to pollutant dissipation. Mycorrhiza 16:397–405. https://doi.org/10.1007/s00572-006-0055-8
Walker C (2005) A simple blue staining technique for arbuscular mycorrhizal and other root-inhabiting fungi. Inoculum 56:68–69
Wu FY, Yu XZ, Wu SC, Lin XG, Wong MH (2011) Phenanthrene and pyrene uptake by arbuscular mycorrhizal maize and their dissipation in soil. J Hazard Mater 187:341–347. https://doi.org/10.1016/j.jhazmat.2011.01.024
Yu XZ, Wu SC, Wu FY, Wong MH (2011) Enhanced dissipation of PAHs from soil using mycorrhizal ryegrass and PAH-degrading bacteria. J Hazard Mater 186:1206–1217. https://doi.org/10.1016/j.jhazmat.2010.11.116
Zhou XB, Cébron A, Béguiristain T, Leyval C (2009) Water and phosphorus content affect PAH dissipation in spiked soil planted with mycorrhizal alfalfa and tall fescue. Chemosphere 77:709–713. https://doi.org/10.1016/j.chemosphere.2009.08.050
Zhou XB, Zhou J, Xiang X, Cébron A, Béguiristain T, Leyval C (2013) Impact of four plant species and arbuscular mycorrhizal (AM) fungi on polycyclic aromatic hydrocarbon (PAH) dissipation in spiked soil. Pol J Environ Stud 22:1239–1245
Zocco D, Van Aarle IM, Oger E, Lanfranco L, Declerck S (2011) Fenpropimorph and fenhexamid impact phosphorus translocation by arbuscular mycorrhizal fungi. Mycorrhiza 21:363–374. https://doi.org/10.1007/s00572-010-0344-0
Acknowledgements
The research leading to these results has received funding from the Seventh Framework Programme [FP7/PEOPLE-2013-IEF] under grant agreement n°623425. The authors thank Anne Iserentant (Environmental Sciences, Earth and Life Institute, UCL) for ICP-AES analysis, Loup Damman for technical assistance, and Catherine Rasse (SMCS, UCL) for support in statistical analyses.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Calonne-Salmon, M., Plouznikoff, K. & Declerck, S. The arbuscular mycorrhizal fungus Rhizophagus irregularis MUCL 41833 increases the phosphorus uptake and biomass of Medicago truncatula, a benzo[a]pyrene-tolerant plant species. Mycorrhiza 28, 761–771 (2018). https://doi.org/10.1007/s00572-018-0861-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-018-0861-9