Abstract
We examined differences in fine root morphology, mycorrhizal colonisation and root-inhabiting fungal communities between Picea abies individuals infected by Heterobasidion root-rot compared with healthy individuals in four stands on peat soils in Latvia. We hypothesised that decreased tree vitality and alteration in supply of photosynthates belowground due to root-rot infection might lead to changes in fungal communities of tree roots. Plots were established in places where trees were infected and in places where they were healthy. Within each stand, five replicate soil cores with roots were taken to 20 cm depth in each root-rot infected and uninfected plot. Root morphological parameters, mycorrhizal colonisation and associated fungal communities, and soil chemical properties were analysed. In three stands root morphological parameters and in all stands root mycorrhizal colonisation were similar between root-rot infected and uninfected plots. In one stand, there were significant differences in root morphological parameters between root-rot infected versus uninfected plots, but these were likely due to significant differences in soil chemical properties between the plots. Sequencing of the internal transcribed spacer of fungal nuclear rDNA from ectomycorrhizal (ECM) root morphotypes of P. abies revealed the presence of 42 fungal species, among which ECM basidiomycetes Tylospora asterophora (24.6 % of fine roots examined), Amphinema byssoides (14.5 %) and Russula sapinea (9.7 %) were most common. Within each stand, the richness of fungal species and the composition of fungal communities in root-rot infected versus uninfected plots were similar. In conclusion, Heterobasidion root-rot had little or no effect on fine root morphology, mycorrhizal colonisation and composition of fungal communities in fine roots of P. abies growing on peat soils.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Heterobasidion spp. are among the most widespread root-rot pathogens in boreal and northern temperate forests where they cause huge economic losses (Woodward et al. 1998). Heterobasidion includes five species, three occurring in Europe, H. annosum, H. parviporum and H. abietinum, and two in North America, H. irregulare and H. occidentale (Dalman et al. 2010). Although the host species may partly overlap, among European species, H. annosum is mainly found on pine, H. parviporum on spruce and H. abietinum on fir (Dalman et al. 2010 and references therein). The fungi enter stands mainly through the colonisation of fresh conifer stumps; mycelia grow down to root systems and infect neighbouring healthy trees via root contacts (Rishbeth 1951). Heterobasidion can persist and remain active in root systems for decades and infect trees of the next forest generation planted on infested sites (Rishbeth 1951; Stenlid 1987). Heterobasidion generally causes extensive heart-rot in roots and stem of Norway spruce (Picea abies), resulting in reduced diameter/volume growth (Bendz-Hellgren and Stenlid 1995, 1997), but it may also infect functional sapwood and cause tree mortality (Piri 2003; Piri and Korhonen 2001).
In Latvia, P. abies stands constitute 17 % (5374 km2) of the total forest area. Of the P. abies area, 38 % (2067 km2) is on drained peat soils (Jansons 2011). In Latvia, ca. 16 % of P. abies stands on peat soils are root-rot infected (Arhipova et al. 2011), which causes up to 20 % of timber loss (Gaitnieks et al. 2008). Often occurrence of Heterobasidion root-rot in P. abies stands on peat soils was suggested to be associated with development of rich vegetation and moist conditions (Stivriņa et al. 2010). Besides, it might also be promoted by alkaline and nutrient-rich groundwater, which occurs in at least 80 % of the total drained peat forest area (Zālītis 2006).
Ectomycorrhizal (ECM) fungi are commonly associated with fine roots of P. abies and depend on their host for nutrition (Smith and Read 1997). Some ECM fungi can protect roots against certain pathogens and promote plant growth (Sinclair et al. 1982). Several ECM fungi were also able to reduce growth of Heterobasidion when confronted in vitro (Napierala-Filipiak and Werner 2000). Although Heterobasidion is not a fine-root pathogen and it is not able to freely grow in the soil (Garbelotto and Gonthier 2013), we hypothesise that overall reduction in tree vitality as a result of root-rot infection might lead to alterations in ECM communities in tree roots. For example, Bendz-Hellgren and Stenlid (1995) suggested that observed 50 % of the growth loss could be due to allocation of resources to cope with the Heterobasidion infection. Furthermore, in cases where functional sapwood is infected, transport of photosynthetically derived carbohydrates to roots and ultimately to ECM symbionts might be limited, potentially leading to even more pronounced alterations in communities of associated fungi. In Latvia, fine root morphological parameters and the abundance of ECM morphotypes were compared in Heterobasidion root-rot infected and uninfected stands of P. abies growing on mineral and peat soils (Gaitnieks 2005; Gaitnieks et al. 2000). Gaitnieks (2005) observed some differences in ECM communities between root-rot infected and uninfected trees but also a significant site effect. However, in that study there was a need for more reliable identification of ECM species.
The objective of the present study was to compare fine root morphology, mycorrhizal colonisation and associated fungal communities in Heterobasidion root-rot infected and uninfected P. abies growing on peat soils. The rationale of the study was to get better understanding on whether and how Heterobasidion root-rot may affect indirectly morphology of fine roots and their colonisation by ECM fungi. This was expected to demonstrate that, in addition to the direct negative effect to tree health, Heterobasidion as other biotic stress factors (Saravesi et al. 2015; Treu et al. 2014) may also indirectly determine health and sustainability of forest ecosystems.
Material and methods
Study sites
For study, we selected four P. abies stands (K, O, S and M) in Latvia (Table 1). Information on the health status (defoliation) of each stand was obtained from the Latvian State Forest Research Institute “Silava” and is presented in Table 2. Defoliation was evaluated using the method described by Eichhorn et al. (2010). The presence/absence of Heterobasidion root-rot in each stem was determined by taking wood samples ca. 30–40 cm above the ground using an increment borer. Surface-sterilised wood samples were aseptically incubated on malt-agar media, and outgrowth of Heterobasidion spp. was scored after a week. Both H. annosum and H. parviporum were recorded in the samples. Based on scoring results, three or four plots, each possessing a group of three to five infected trees, and a corresponding number of closely situated plots with three to five uninfected trees were selected randomly in each site (Table 1).
Sampling
In July 2013, soil with roots was sampled from the uppermost 20-cm soil layer using a soil core 12 cm in diameter. Within each site, five replicate soil cores were taken from each of Heterobasidion root-rot infected and uninfected plots resulting in 140 soil cores in total (Table 1). Soil cores were placed separately in plastic bags, transported to the laboratory and stored at +4 °C until processing. P. abies roots were separated from soil and rinsed under tap water. Cleaned roots were cut into 1-cm segments and placed in water in Petri dishes with diameter 14 cm and a 7 × 7 mm grid on the bottom. ECM morphotypes were identified in 100-grid squares using a Leica MZ-7.5 stereomicroscope (Leica Microsystems, Wetzlar, Germany). ECM morphotypes were grouped based on colour, form and texture, and the presence and patterns of rhizomorphs or external mycelia (Agerer 1986–2006). Living and dead fine roots were identified based on their colour and elasticity of the central cylinder (Vogt and Persson 1991). Morphological parameters of fine roots including length, volume, surface area and number of root tips were determined using an Epson Perfection V750Pro scanner (Epson, Tokyo, Japan) and WinRHIZO 2005 C (Regent Instruments Inc., Canada) software. After scanning, roots were dried at 50 °C for 12 h and weighed. Soil was dried 1 week at room temperature (ca. 21 °C) and sieved (mesh size 2 × 2 mm) to separate larger fractions, which were discarded. Chemical analyses of soil were conducted using established standard methods (International Organization for Standardization (ISO) standard and Latvian State standards (LVS)). Samples were prepared for analysis according to ISO 11464:2005. Soil pH (KCl) was potentiometrically measured in the deionised water suspension according to LVS ISO 10390. Total carbon content was determined using elemental analyser ELTRA CS530 (Eltra GmbH, Haan, Germany) according to LVS ISO 10694. The content of total N was determined using modified Kjeldahl method (ISO 11261:1995), and P was assayed by spectrophotometry using the ammonium molybdate spectrometric method in 1 M HCl extract (LVS 398). Concentration of K, Ca and Mg was determined by atomic absorption spectrophotometer with an acetylene-air flame in 1 M HCl extract.
Molecular analysis
In each site, up to five root tips of each distinct ECM morphotype were collected and used for molecular identification of fungal species. DNA extraction from individual root tips was done as described by Vainio et al. (1998). Amplification of the internal transcribed spacer (ITS) region of fungal nuclear rDNA using primers ITS1F (Gardes and Bruns 1993) and ITS4 (White et al. 1990) and electrophoresis and sequencing was done as described by Klavina et al. (2013). Raw sequence data were analysed using the SeqMan version 5.07 software from DNASTAR package (DNASTAR, Madison, WI, USA) and BioEdit v. 7.0.9.0 (Hall 1999). Databases at GenBank (Altschul et al. 1997) and UNITE https://unite.ut.ee/ (Kõljalg et al. 2013) were used to determine the identity of ITS rDNA sequences. The criteria used for identification were as follows: sequence coverage >80 %; similarity to species level 97–100 %; and similarity to genus level 94–96 %. The sequences are available from GenBank under accession numbers KR019832–KR019874.
Statistical analyses
Differences in defoliation, root morphological parameters, mycorrhizal colonisation and soil chemical properties between Heterobasidion root-rot infected and uninfected plots of each respective study site were compared using one-way analysis of variance (ANOVA) in Minitab v.16.2.4 (Minitab, Inc., Coventry, UK). Richness of fungal species in root-rot infected and uninfected plots of each study site was compared by non-parametric chi-square test (Magurran 1988). The Shannon diversity index was used to characterise diversity of fungal communities (Shannon 1948). Fungal community structure in roots of P. abies and possible Heterobasidion root-rot effect was analysed using canonical correspondence analysis (CCA) in CANOCO 4.5 (ter Braak and Smilauer 1998). Significance of the environmental variables was tested using a generalised linear model and Gaussian distribution. MANOVA was used to evaluate degree of separation (along CCA axis 1 and 2) between the fungal communities in Heterobasidion root-rot infected and uninfected plots.
Results
Defoliation was significantly higher in all Heterobasidion root-rot infected plots than in corresponding uninfected plots (Table 2). Within O, S and M sites (Table 1), root morphological parameters were similar between Heterobasidion root-rot infected and uninfected plots (Table 3). In site K (Table 1), in contrast, all (except number of dead root tips) root morphological parameters in root-rot infected plots were significantly lower than those in uninfected plots (Table 3). Root mycorrhizal colonisation was high in all sites and plots (ranging between 86.4 and 100 %), and no significant differences were found between corresponding root-rot infected and uninfected plots (Table 3). Soil chemical properties varied among the sites (Table 4), but were similar between root-rot infected and uninfected plots within each site (Table 4) with some exceptions: in site K, pH and concentration of N and Ca were significantly lower in root-rot infected plots than in uninfected plots, while the C/N ratio was significantly higher in root-rot infected versus uninfected plots (Table 4).
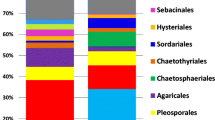
In total, 42 fungal species were detected by ITS rDNA sequencing of analysed fine root samples of P. abies. Most of the species (38) were ECM (Table 5), but also saprotrophic or weakly pathogenic fungal species such as Cryptococcus sp., Ilyonectria sp. and Trichosporon sp. were sequenced. Within each site, a chi-square test showed that richness of fungal species did not significantly differ between root-rot infected and uninfected plots (Table 5). The Shannon diversity index was also similar between root-rot infected versus uninfected plots of each site (Table 5). The most common fungi were Tylospora asterophora (24.6 %), Amphinema byssoides (14.5 %), Russula sapinea (9.7 %) and Tomentella stuposa (7.8 %) (Table 5).
CCA ordination (Fig. 1) of fungal communities explained 27.3 % of the variation on axis 1 and 23.1 % on axis 2. CCA showed that composition of fungal communities was mainly determined by soil factor C/N ratio (p < 0.002); pH (p < 0.001) and K (p < 0.02) present at each site (Fig. 1). MANOVA showed that separation of fungal communities between Heterobasidion root-rot infected and uninfected plots was not supported significantly along axis 1 and axis 2. Consequently, root-rot infected and uninfected plots of each site were more or less well grouped together on axis 1, while their position in the ordination showed relative importance of environmental variables. C/N ratio had a significant effect on fungal community structure and consequently on position of root-rot infected (MR) and uninfected (MH) plots of M site, pH on position of K and S sites, while potassium had a significant effect on position of root-rot infected (OR) and uninfected (OH) plots of O site and their separation along axis 2.
Ordination diagram based on canonical correspondence analysis (CCA) of fungal communities in roots of Picea abies from Heterobasidion root-rot infected (dark-grey circles) and uninfected (light-grey circles) stands. Names of the plots (KR, KH, OR, OH, SR, SH, MR, MH) are as in Table 1. Arrows show environmental variables (pH, C/N and K) that were found to be of significant importance. Taxonomic names are shown in the ordination for the ten most common fungal species of a study and correspond to their position in the ordination
Discussion
Despite the significantly higher defoliation in root-rot infected plots, the results demonstrated that Heterobasidion root-rot had little or no effect on fine root morphology, root mycorrhizal colonisation (Table 3) and composition of fungal communities in fine roots of P. abies (Table 5, Fig. 1), thereby rejecting the hypothesis that reduction in tree vitality as a result of root-rot infection might lead to alterations in fine root morphology and mycorrhizal colonisation. In the K site, differences in root morphological parameters between root-rot infected versus uninfected plots were likely due to the significant differences in soil chemical properties between these plots (Table 4). In agreement, it was shown that pH and concentration of N in the soil may have a major effect on root morphological parameters of P. abies (Helmisaari et al. 2009). Previous studies suggested that mycorrhizal colonisation of Scots pine (Pinus sylvestris) is lower in Heterobasidion root-rot infected stands (Коротков 1974) and that root-rot may also affect fine root morphology of P. abies (Gaitnieks 2005). However, the present study did not show such effects for P. abies on peat soils though these effects may appear later, i.e. in older trees and after Heterobasidion root-rot becomes extensively established (Swedjemark and Stenlid 1993). In support, establishment and spread of Heterobasidion in living trees of P. abies is generally much slower than in non-living trees or stumps due to active defence responses and build-up of a metabolite-rich reaction zone (Bendz-Hellgren et al. 1999; Oliva et al. 2012). Furthermore, the results suggest that in the investigated P. abies stands, root-rot did not restrict the flow of photosynthates belowground, as fungal communities in roots and root mycorrhizal colonisation remained largely unchanged (Fig. 1, Table 3). In contrast, tree girdling, which terminates the supply of photosynthates to roots, has been shown to have a profound effect on activity and composition of fungal communities in roots (Högberg et al. 2001). Bergemann et al. (2013) showed a significant reduction of ECM soil hyphal biomass following tree girdling while simulating the potential impact of the invasive pathogen Phytophthora ramorum. The latter may suggest that Heterobasidion root-rot may also firstly affect production and abundance of ECM hyphae in the soil while changes in abundance and composition of ECM communities in roots occur at a later stage.
Our study suggests that differences/similarities in soil properties among the different study sites were the major factors determining the abundance and composition of fungal communities in fine roots of P. abies (Table 4, Fig. 1). We observed large dominance of a few ECM species (Table 5), and a similar pattern was shown in a study of Heterobasidion root-rot infected stands in Germany (Bücking 1979). Among the dominant fungi, T. asterophora is one of the most constant and abundant ECM fungi on spruce roots (Eberhardt et al. 1999). T. stuposa, another commonly detected ECM species, has been described as one of the most commonly encountered tomentelloid fungi in Swedish spruce stands (Kõljalg et al. 2000), and A. byssoides is an efficient root coloniser of P. abies (Menkis et al. 2011; Menkis et al. 2007; Vaario et al. 2009). Better outplanting performance of abundantly colonised seedlings (Menkis et al. 2011) and common occurrence in forest plantations (Kranabetter 2004; Menkis et al. 2007; Vaario et al. 2009) suggest that A. byssoides may be of particular importance to health and sustainability of P. abies forest stands. Thus, possibility should not be excluded that through increased supply of mineral nutrients and water to the host (Smith and Read 1997), these ECM fungi mitigated the negative effect of Heterobasidion root-rot, thereby contributing to similar growth parameters of P. abies roots in both root-rot infected and uninfected plots.
References
Agerer R (1986–2006) Colour atlas of ectomycorrhizae. Einhorn-Verlag, Schwäbisch Gmünd, München, Germany
Altschul SF, Madden TL, Schäffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Arhipova N, Gaitnieks T, Donis J, Stenlid J, Vasaitis R (2011) Butt rot incidence, causal fungi, and related yield loss in Picea abies stands of Latvia. Can J For Res 41:2337–2345. doi:10.1139/x11-141
Bendz-Hellgren M, Stenlid J (1995) Long-term reduction in the diameter growth of butt rot affected Norway spruce, Picea abies. Forest Ecol Manag 74:239–243. doi:10.1016/0378-1127(95)03530-N
Bendz-Hellgren M, Stenlid J (1997) Decreased volume growth of Picea abies in response to Heterobasidion annosum infection. Can J For Res 27:1519–1524. doi:10.1139/x97-104
Bendz-Hellgren M, Brandtberg P-O, Johansson M, Swedjemark G, Stenlid J (1999) Growth Rate of Heterobasidion annosum in Picea abies established on forest land and arable land. Scand J For Res 14:402–407. doi:10.1080/02827589950154104
Bergemann SE, Kordesch NC, VanSant-Glass W, Garbelotto M, Metz TA (2013) Implications of tanoak decline in forests impacted by Phytophthora ramorum: girdling decreases the soil hyphal abundance of ectomycorrhizal fungi associated with Notholithocarpus densiflorus. Madroño 60:95–106. doi:10.3120/0024-9637-60.2.95
Bücking E (1979) Fichten-Mykorrhizen auf Standorten der Schwäbischen Alb und ihre Beziehung zum Befall durch Fomes annosus. Eur J Forest Pathol 9:19–35
Dalman K, Olson Å, Stenlid J (2010) Evolutionary history of the conifer root rot fungus Heterobasidion annosum sensu lato. Mol Ecol 19:4979–4993. doi:10.1111/j.1365-294X.2010.04873.x
Eberhardt U, Walter L, Kottke I (1999) Molecular and morphological discrimination between Tylospora fibrillosa and Tylospora asterophora mycorrhizae. Can J Botany 77:11–21. doi:10.1139/b98-182
Eichhorn J, Roskams P, Ferretti M, Mues V, Szepesi A, Durrant D (2010) Visual assessment of crown condition and damaging agents. Manual part IV. Manual on methods and criteria for harmonized sampling, assessment, monitoring and analysis of the effects of air pollution on forests. In. UNECE ICP Forests programme co-ordinating centre, Hamburg, pp 1–49 (available at: http://icp-forests.net/page/icp-forests-manual)
Gaitnieks T (2005) Vitality of Norway spruce fine roots in stands infected by Heterobasidion annosum. In: Solheim H, Hietala AM (eds) Proceedings of the SNS meeting in forest pathology: Forest pathology research in Nordic and Baltic countries 2005. Skogbrukets Kursinstitutt, Biri, Norway, pp 79–82 (available at: http://www.skogoglandskap.no/filearchive/a-2006-1.pdf)
Gaitnieks T, Liepa I, Rokjanis B, Indriksons A (2000) Development of Norway spruce mycorrhiza in mixed Norway spruce and grey alder stands infected by Heterobasidion annosum. Metsanduslikud Uurimused 34:44–51
Gaitnieks T, Arhipova N, Donis J, Stenlid J, Vasaitis R (2008) Butt rot incidence and related losses in Latvian Picea abies (L.) Karst. stands. Proceedings of 12th international conference on root and butt rots, Berkley, California, pp 177–179
Garbelotto M, Gonthier P (2013) Biology, epidemiology, and control of Heterobasidion species worldwide. Annu Rev Phytopathol 51:39–59. doi:10.1146/annurev-phyto-082712-102225
Gardes M, Bruns T (1993) ITS primers with enhanced specificity for basidiomycetes—application to the identification of mycorrhizae and rusts. Mol Ecol 2:113–118
Hall T (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for windows 95/98/NT. Nucleic Acid S 41:95–98
Helmisaari HS, Saarsalmi A, Kukkola M (2009) Effects of wood ash and nitrogen fertilization on fine root biomass and soil and foliage nutrients in a Norway spruce stand in Finland. Plant Soil 314:121–132. doi:10.1007/s11104-008-9711-4
Högberg P, Nordgren A, Buchmann N, Taylor AFS, Ekblad A, Högberg MN, Nyberg G, Ottosson-Lofvenius M, Read DJ (2001) Large-scale forest girdling shows that current photosynthesis drives soil respiration. Nature 411:789–792. doi:10.1038/35081058
Jansons J (2011) Statistical inventory of Latvia’s forest resources (results of cycle I), Latvian State Forest Research Institute “Silava”. (In Latvian), (available at: http://www.silava.lv/22/section.aspx/View/13)
Klavina D, Gaitnieks T, Menkis A (2013) Survival, growth and ectomycorrhizal community development of container- and bare-root grown Pinus sylvestris and Picea abies seedlings outplanted on a forest clear-cut. Balt For 19:39–49
Kõljalg U, Dahlberg A, Taylor AFS, Larsson E, Hallenberg N, Stenlid J, Larsson KH, Fransson PM, Karen O, Jonsson L (2000) Diversity and abundance of resupinate thelephoroid fungi as ectomycorrhizal symbionts in Swedish boreal forests. Mol Ecol 9:1985–1996
Kõljalg U, Nilsson RH, Abarenkov K, Tedersoo L, Taylor AFS, Bahram M, Bates ST, Bruns TD, Bengtsson-Palme J, Callaghan TM, Douglas B, Drenkhan T, Eberhardt U, Dueñas M, Grebenc T, Griffith GW, Hartmann M, Kirk PM, Kohout P, Larsson E, Lindahl BD, Lücking R, Martín MP, Matheny PB, Nguyen NH, Niskanen T, Oja J, Peay KG, Peintner U, Peterson M, Põldmaa K, Saag L, Saar I, Schüßler A, Scott JA, Senés C, Smith ME, Suija A, Taylor DL, Telleria MT, Weiss M, Larsson K-H (2013) Towards a unified paradigm for sequence-based identification of fungi. Mol Ecol 22:5271–5277. doi:10.1111/mec.12481
Kranabetter J (2004) Ectomycorrhizal community effects on hybrid spruce seedling growth and nutrition in clearcuts. Can J Botany 82:983–991. doi:10.1139/x05-095
Коротков ГП (1974) Распространение микориз в сосновых насаждениях, зараженных корневой губкой [Distribution of mycorrhiza in the pine stands damaged by Heterobasidion root-rot]. Лесоведение 5:63–67
Magurran AE (1988) Ecological diversity and its measurement. Princeton University Press, Princeton
Menkis A, Vasiliauskas R, Taylor AFS, Stenlid J, Finlay R (2007) Afforestation of abandoned farmland with conifer seedlings inoculated with three ectomycorrhizal fungi—impact on plant performance and ectomycorrhizal community. Mycorrhiza 17:337–348. doi:10.1007/s00572-007-0110-0
Menkis A, Bakys R, Lygis V, Vasaitis R (2011) Mycorrhization, establishment and growth of outplanted Picea abies seedlings produced under different cultivation systems. Silva Fenn 45:283–289. doi:10.14214/sf.118
Napierala-Filipiak A, Werner A (2000) Antagonism of higher fungi to Heterobasidion annosum (Fr.) Bref. in laboratory conditions. Dendrobiology 45:65–81
Oliva J, Julio Camarero J, Stenlid J (2012) Understanding the role of sapwood loss and reaction zone formation on radial growth of Norway spruce (Picea abies) trees decayed by Heterobasidion annosum s.l. Forest Ecol Manag 274:201–209. doi:10.1016/j.foreco.2012.02.026
Piri T (2003) Early development of root rot in young Norway spruce planted on sites infected by Heterobasidion in southern Finland. Can J For Res 33:604–611. doi:10.1139/x02-200
Piri T, Korhonen K (2001) Infection of advance regeneration of Norway spruce by Heterobasidion parviporum. Can J For Res 31:937–942. doi:10.1139/x01-021
Rishbeth J (1951) Observations on the biology of Fomes annosus, with particular reference to East Anglian pine plantations. III. Natural and experimental infection of pines, and some factorsaffecting severity of the disease. Ann Bot 15:221–246
Saravesi K, Aikio S, Wali PR, Ruotsalainen AL, Kaukonen M, Huusko K, Suokas M, Brown SP, Jumpponen A, Tuomi J, Markkola A (2015) Moth outbreaks alter root-associated fungal communities in subarctic mountain birch forests. Microb Ecol 69:788–797. doi:10.1007/s00248-015-0577-8
Shannon CE (1948) A mathematical theory of communication. Bell Syst Tech J 27:379–423
Sinclair WA, Sylvia DM, Larsen AO (1982) Disease suppression and growth promotion in Douglas-fir seedlings by the ectomycorrhizal fungus Laccaria laccata. Forest Sci 28:191–201
Smith SE, Read DJ (1997) Mycorrhizal symbiosis. Academic, London
Stenlid J (1987) Controlling and predicting the spread of Heterobasidion annosum from infected stumps and trees of Picea abies. Scand J For Res 2:187–198. doi:10.1080/02827588709382457
Stivriņa B, Kenigsvalde K, Korhonen K, Gaitnieks T (2010) Importance of large dimension decaying logging residues of spruce in the spread of Heterobasidion root rot. Mežzinātne 22:88–102 (In Latvian)
Swedjemark G, Stenlid J (1993) Population dynamics of the root rot fungus Heterobasidion annosum following thinning of Picea abies. Oikos 66:247–254
ter Braak CJF, Smilauer P (1998) Canoco reference manual and user’s guide to Canoco for Windows: software for canonical community ordination, Version 4. Microcomputer Power, Ithaca
Treu R, Karst J, Randall M, Pec GJ, Cigan PW, Simard SW, Cooke JEK, Erbilgin N, Cahill JF (2014) Decline of ectomycorrhizal fungi following a mountain pine beetle epidemic. Ecology 95:1096–1103. doi:10.1890/13-1233.1
Vaario LM, Tervonen A, Haukioja K, Haukioja M, Pennanen T, Timonen S (2009) The effect of nursery substrate and fertilization on the growth and ectomycorrhizal status of containerized and outplanted seedlings of Picea abies. Can J For Res 39:64–75. doi:10.1139/x08-156
Vainio J, Korhonen K, Hantula J (1998) Genetic variation in Phlebia gigantea as detected with random amplified microsatellite (RAMS) markers. Mycol Res 102:187–192
Vogt KA, Persson H (1991) Root methods. In: Lassoie JP, Hinckley TM (eds) Techniques and approaches in forest tree ecophysiology. CRC Press, Boca Raton, pp 477–502
White TJ, Bruns T, Lee S, Taylor J (1990) Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In: Innis MA, Gelfand DH, Sninsky JJ, White TJ (eds) PCR protocols: a guide to methods and applications. Academic Press, Inc, San Diego, pp 315–322
Woodward S, Stenlid J, Karjalainen R, Hüttermann A (1998) Heterobasidion annosum: biology, ecology, impact and control. CAB International, Wallingford
Zālītis P (2006) Preconditions for forest management. SIA, Riga (In Latvian)
Acknowledgments
The study was supported by the JSC “Latvian State Forests”, ERDF funded project (no. L-KC-11-0004) “Methods and technologies for increasing forest capital value”, research direction “Investigation of the factors limiting the spread of root rot”, grant project no.426/2012 of Latvian Council of Science “Evaluation of factors affecting the efficacy of Phlebiopsis gigantea against Heterobasidion root rot” and the Swedish Energy Agency (Energimyndigheten).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Gaitnieks, T., Klavina, D., Muiznieks, I. et al. Impact of Heterobasidion root-rot on fine root morphology and associated fungi in Picea abies stands on peat soils. Mycorrhiza 26, 465–473 (2016). https://doi.org/10.1007/s00572-016-0685-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-016-0685-4