Abstract
The aim of a 3-year study was to investigate whether inoculation of Pinus sylvestris L. and Picea abies (L.) Karst. seedlings with mycorrhizas of Cenococcum geophilum Fr., Piceirhiza bicolorata, and Hebeloma crustuliniforme (Bull.) Quel. has any impact on: 1) survival and growth of outplanted seedlings on abandoned agricultural land, and 2) subsequent mycorrhizal community development. For inoculation, the root system of each plant was wrapped in a filter paper containing mycelium, overlaid with damp peat–sand mixture and wrapped in a paper towel. In total, 8,000 pine and 8,000 spruce seedlings were planted on 4-ha of poor sandy soil in randomized blocks. Already after the first year natural mycorrhizal infections prevailed in the inoculated root systems, and introduced mycorrhizas were seldom found. Yet, the seedlings that had been pre-inoculated with C. geophilum and the P. bicolorata during the whole 3-year period showed significantly higher survival and growth as compared to controls. Moreover, the independent colonization of roots by C. geophilum and the P. bicolorata from natural sources was also observed. A diverse mycorrhizal community was detected over two growing seasons in all treatments, showing low impact of inoculation on subsequent fungal community development. A total of 19 additional ectomycorrhizal morphotypes was observed, which clustered into two well-separated groups, according to host tree species (pine and spruce). In conclusion, the results showed limited ability to increase tree survival and growth, and to manipulate the mycorrhizal community even by extensive pre-inoculations, indicating that fungal community formation in root systems is governed mainly by environmental factors.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The establishment and performance of outplanted seedlings has often been reported to depend on ectomycorrhizal (ECM) fungi (Perry et al. 1987; Kropp and Langlois 1990; Stenström et al. 1990; Le Tacon et al. 1994; Pera et al. 1999; Baum et al. 2002; Dunabeitia et al. 2004), which may enhance uptake of water and nutrients (Smith and Read 1997) and lengthen the life and increase the growth of roots (Chilvers and Gust 1982; Wilcox 1996) by protecting them against drought, pathogens, and heavy metal pollution (Chakravarty and Unestam 1985; Colpaert and Vanassche 1992; Morin et al. 1999; Van Tichelen et al. 2001; Ortega et al. 2004). Because of the absence of host trees, a farmland is often deficient in natural ectomycorrhizal inoculum (Hacskaylo 1973), thus mycorrhization of seedling roots at outplanting might be beneficial. However, seedlings used in afforestation are often cultivated as bare root seedlings in forest nurseries and reportedly are only colonized by ECM fungi to a lesser extent (Dunabeitia et al. 2004; Menkis et al. 2005). Consequently, failure in afforestation has been hypothesized to be caused by the absence of mycorrhizas (Bjorkman 1970; Mikola 1970; Marx 1980). One way of overcoming this problem would be preinoculation of seedlings with selected ECM fungi. As this is a highly resource-consuming effort, the seedlings should be inoculated with ECM fungi best suited to host tree species and that rapidly colonize their roots, and which are well adapted to the environmental conditions of the planting site (Perry et al. 1987).
The present work was carried out with three ECM fungi: Cenococcum geophilum Fr., Piceirhiza bicolorata (Brand et al. 1992), and Hebeloma crustuliniforme (Bull.) Quel., which possess characteristics that might be of importance for successful establishment of conifer seedlings on a farmland.
P. bicolorata is a distinct ECM morphotype formed by an ascomycete of the Hymenoscyphus ericae aggregate (Vrålstad et al. 2000), which occurs on a variety of conifers and hardwoods. The ability of this fungus to form mycorrhizas with both ectomycorrhizal and ericoid hosts (Villarreal-Ruiz et al. 2004) may provide access to a broad belowground network and consequently larger nutrient sources. Association of P. bicolorata with P. sylvestris and P. abies seedlings in numerous forest nurseries (Menkis et al. 2005) indicate its suitability to the managed environment, e.g., arable land. This fungus has also been found in association with pioneer tree species inhabiting heavy metal polluted soils, and thus might be involved in nutrient mobilization and substrate detoxification processes (Vrålstad et al. 2000). Although it was hypothesized that P. bicolorata aggregate might be beneficial for plants in marginal habitats (Vrålstad et al. 2000), this was not tested in the field inoculation experiments.
C. geophilum is known as an extremely drought-tolerant ECM ascomycete (Mexal and Reid 1973; Trappe 1977; Pigott 1982), which also grows and forms mycorrhizas over the pH range 2.4–7.5 and at high salt concentrations (Mikola 1948; Saleh-Rastin 1976). C. geophilum is associated with a wide range of tree species in forest nurseries and in forest ecosystems (Trappe 1988; Dahlberg and Stenström 1991; Menkis et al. 2005). It was found as the dominant species at the boundary of forest and agricultural land both near and distant (20 m) from the trees, and thus may play an important role in natural establishment of tree seedlings (Dickie and Reich 2005).
The basidiomycete H. crustuliniforme is an efficient root colonizer of young trees (Dighton and Mason 1985). Its survival and function on alkaline-saline substrates (Senior et al. 1993; Kernaghan et al. 2002; Muhsin and Zwiazek 2002) and the ability to protect plants against root pathogens (Perrin and Garbaye 1983) might be important for performance of outplanted seedlings. However, the reports on its effect on seedling growth are contradictory, as both the stimulation and retarding of growth have been observed (Theodorou and Bowen 1970; Bledsoe et al. 1982; Le Tacon and Bouchard 1986).
The main aim of the present work was to determine whether artificial inoculation of root systems with the three ECM fungi has any effect on survival and growth of the seedlings after outplanting under field conditions of abandoned farmland. At the same time, we investigated the impact the treatment of seedling roots with fungal inocula might have on subsequent mycorrhizal community development.
Materials and methods
Study sites and experimental design
The study area comprised 4 ha of abandoned farmland at Pocelonys in southern Lithuania (54°20′ N, 24°14′ E, 150–160 m above sea level). Within the area, mean annual precipitation is about 620 mm and the length of the growing season is ca. 190 days. Average temperature during the growth season is ca. 15°C. The site is characterized as low-productivity agricultural land and had been cultivated for many decades until the beginning of the 1990s. Large applications of fertilizers were repeatedly applied during the last three decades of cultivation, which have likely resulted in low background levels of ECM fungi. Before our study, the land was abandoned for more than 10 years and in the meantime turned to natural grassland lacking woody vegetation. The site is characterized by sandy soils, corresponding to vaccinio-myrtilliosa forest type.
Planting was carried out in April 2003 with 1-year-old pine (Pinus sylvestris) and 2-year-old spruce (Picea abies) seedlings, which were greenhouse-cultivated as bare root seedlings in a local forest nursery. A previous study had shown that in similarly cultivated seedlings from this nursery only ca. 20% of seedling roots were mycorrhizal (Menkis et al. 2005). Within tree species, seedlings were of similar height—ca. 11 cm for pine and 15 cm for spruce. For each tree species, three different ECM treatments and the non-inoculated control treatment were established in a randomized block design with four replicate blocks containing each of the four treatments. Before seedling outplanting, each 0.125-ha plot (25 × 50 m) was ploughed in 16 rows to ca. 12 cm deep with a forestry plough and 500 trees per plot were planted in rows at 1.6 × 1.5 m spacing. In total, 8,000 P. sylvestris and 8,000 P. abies seedlings were planted at an initial density of 4,000 seedlings per hectare. The distance between the pine and spruce sites was ca. 150 m.
Soil characteristics
Soil samples were collected in June 2003. Each of the 16 pine and 16 spruce plots was divided into two subplots from which soil cores were taken in the bed of ploughed rows to a depth of 20 cm at five different locations. Within each subplot, all sampled cores were pooled and analyzed as a bulk sample. Soil analyses were carried out at the Forest Soils Testing Laboratory, Lithuanian Forest Tree Breeding and Seed Farming Centre, Girionys, Kaunas reg., Lithuania. Soil pH in H2O and the basic nutrients (mg/100 g of soil) as mineralizable N, P2O5, and K2O were determined. The results are summarized in Table 1. Among the plots with the same tree species differences in tested soil properties were, in most cases, not significant; however, significant differences in soil pH, P2O5, and K2O content were found between the pine and spruce plots (Table 1). As the soil samples were taken a mere 1.5 months after the seedlings were planted, we assume that the estimated soil properties of the site were preexisting before the plantation was established.
Inoculum preparation and inoculation
Cenococcum geophilum (isolate UP162), a Piceirhiza bicolorata fungal isolate (isolate aurim678) and Hebelomacrustuliniforme (isolate UP184) isolates were obtained from the culture collection of the Department of Forest Mycology & Pathology, Swedish University of Agricultural Sciences, Uppsala. Fungal stock cultures were maintained in darkness at 21°C on half-strength modified Melin–Norkrans (MMN) medium (Marx 1969).
Initially, the ability of selected fungi to form ectomycorrhizas was determined on sterile, 2-week-old P. sylvestris and P. abies seedlings, which were aseptically inoculated with agar plugs from the fungal stock cultures in Petri dishes with growth substrate of fine sphagnum peat, vermiculate, 1/10 strength liquid MMN mixture in the ratio 1:4:2 (Rosling et al. 2004). After 6 weeks incubation in a growth chamber, the fungi colonized the majority of seedling fine roots producing distinctive ectomycorrhizas. All fungi were re-isolated from the inoculation trials onto modified MMN media to maintain high vigor of stock cultures.
Vegetative inoculum was produced using a modification of the paper-sandwich technique (Chilvers et al. 1986). Initially, 14–16 agar plugs from the actively growing fungal stock cultures were aseptically placed in empty 14-cm-diameter Petri dishes. These were overlaid with five sheets of sterile, 13-cm-diameter filter paper (Munktell Analytical Filter Paper, Grycksbo, Sweden), which were moistened with 30 ml of liquid MMN media. The layering procedure was repeatedly carried out until the Petri dishes were filled. The final layer was mycelial plugs on the uppermost filter papers. The filter papers were situated in between the mycelial plugs, which allowed mycelial growth from both directions and consequently better establishment. Filter papers used in the control treatment were inoculated in the same manner, but only with sterile MMN agar plugs. The inoculated Petri dishes were incubated in darkness at 21°C for 30 days.
Filter papers with established ECM mycelia (Fig. 1a) and sterile control treatment sheets were soaked three times in sterile deionized water to remove any remaining nutrients. The filter papers were then transported submerged in sterile water to the afforestation site (Marx and Daniel 1976). Standardized inoculations (one filter paper per seedling containing identical fungal genotype) were carried out immediately before planting. The root system of each seedling was shortened to a size similar to that of the filter paper (ca. 13 cm diameter) and wrapped in the filter paper containing ECM mycelium (Fig. 1b) or in the filter paper of the control treatment. In addition, a layer of damp sphagnum peat–sand mixture 3:1 was placed on the top of filter paper, and this was then wrapped in a double layer of paper towel (Tork, SCA, Sweden) (Fig. 1c). In total, 12,000 of seedlings inoculated with three ECM fungi and 4,000 control seedlings were immediately outplanted after inoculation. To check vitality of the inocula, after the planting was completed remaining inoculated filter papers were brought back to the laboratory and from each treatment the respective fungus was re-isolated into pure culture. Moreover, to check that mycorrhization of roots is not restricted by the inoculation system, inoculated seedlings of each treatment in triplicate were planted in plastic pots with non-sterile sand and grown for 8 weeks in the growth chamber at 21°C and photoperiodicity 16 h light and 8 h dark.
Method used for inoculation of Pinus sylvestris and Picea abies seedlings: a vegetative inoculum—mycelium of Piceirhiza bicolorata growing on MMN moistened filter paper; b components and inoculation procedure—roots wrapped in filter paper containing ectomycorrhizal mycelium are covered with damp sand–peat mixture and everything is wrapped in paper towel; c inoculated seedling before outplanting
Morphotyping and measurements
In the field, inoculation success and the composition of the ECM communities were studied twice, at the end of the growing seasons of 2003 and 2004. The number of investigated plants and roots are shown in Table 2. After random collection of seedlings in each plot, the root systems were excised from the stems, individually packed into plastic bags, transported to the laboratory, and kept at 4°C for a maximum of 4 weeks. Before investigation, each root system was washed in tap water and 20 single root tips from each plant were randomly collected from different parts of the root system using forceps. Mycorrhizal tips were morphotyped as described by Menkis et al. (2005). For identification, these were compared with published illustrations (Agerer 1986–1988; Agerer et al. 1996–1998). Only morphotypes that matched the descriptions were given taxonomic names. The morphotypes that did not match any of those from available publications were classed as unidentified, grouped accordingly to morphological characters, and given a descriptive name (e.g., Unidentified no. 1 [white reddish]), as presented in Table 3.
Survival of plants was determined three times, at the end of growing seasons of 2003, 2004, and 2005, by counting all living trees. The height increment was measured for all living trees at the end of the second and third growth seasons (2004 and 2005).
Statistical analyses
The impact of treatment on seedling survival and mycorrhizal colonization was analyzed using chi-square (χ 2) tests (Conover 1980; Mead and Curnow 1983). The aim was to answer the question: how strong is the evidence that a given treatment has an impact on seedling survival, and on seedling mycorrhization, when compared to control or another treatment? The comparisons were calculated from the actual number of observations in respective treatment pairs (living vs dead trees, and mycorrhizal vs non-mycorrhizal plants/root tips).
The survival of seedlings was evaluated separately for each respective year (2003, 2004 and 2005) and tree species (pine and spruce). The proportions of living and dead plants were pairwise compared to the treatments in all possible combinations (e.g., pine, year 2003: C. geophilum vs P. bicolorata; C. geophilum vs H. crustuliniforme; C. geophilum vs control; P. bicolorata vs H. crustuliniforme; P. bicolorata vs control; H. crustuliniforme vs control; similar for years 2004 and 2005; and then the same for spruce). This allowed us to estimate the relative impact of each treatment on the survival of each tree species during the 3-year period. As each of the datasets was subjected to three comparisons, confidence limits for p values of the chi-squared tests were reduced three times (p/3), as required by the Bonferroni correction (Sokal and Rohlf 1995).
The extent of mycorrhizal colonization of pine and spruce seedlings was at first evaluated separately for the years 2003 and 2004. At this stage, the proportions of mycorrhizal and non-mycorrhizal plants/root tips were also compared between the treatment pairs in all possible combinations (e.g. pine, year 2003: C. geophilum vs P. bicolorata; C.g. vs H. crustuliniforme; C. g. vs control; P. b. vs H.c.; P. b. vs control; H.c. vs control; similar for year 2004; and then the same for spruce). This allowed to estimate the relative impact of each treatment on mycorrhization of plants during the 2-year period. Furthermore, the values from the same treatment were compared between different years (e.g., pine, C. geophilum for 2003 vs 2004; similar for P. bicolorata, H. crustuliniforme and control; and then the same for spruce). This provided the information on the possible influence of each treatment on the subsequent development of mycorrhizal communities in the field. Finally, the comparisons of mycorrhizal colonization were done between pine and spruce, within the same respective treatment and year (e.g., C. geophilum, year 2003: pine vs spruce; C. geophilum, year 2004: pine vs spruce; similar for P. bicolorata, H. crustuliniforme, and control). Here, the respective susceptibility of pine and spruce to colonization by both inoculated and natural mycorrhizas was evaluated. As the five comparisons were made with each dataset, confidence limits for p values of the chi-squared tests were reduced five times (p/5), as required by the Bonferroni correction.
Height increment of the seedlings was analyzed by one-way analysis of variance (ANOVA) (Chalmers and Parker 1989; Fowler et al. 2001). Pearson correlation coefficients were calculated to examine the relationship between soil chemical properties and survival and height increment of plants (p < 0.05). The statistics were computed using Minitab® Statistical Software (Minitab® 2003). Ectomycorrhizal community structures were analyzed using principal component analysis (PCA) (Fowler et al. 2001) in CANOCO 4.5 (Ter Braak and Smilauer 1998).
Results
Survival and growth of plants
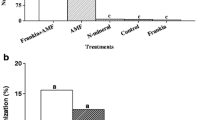
The establishment of planted stock in our experimental plantations was rather high, and, after the first growing season, consisted of 67–78% and 77–88% for pine and spruce, respectively (Fig. 2). Yet, its dynamics during study period in both tree species and in all treatments has revealed a similar trend of slightly increasing mortality. During the first three growing seasons, pine seedlings inoculated with C. geophilum and P. bicolorata showed survival rates 10–13% higher than control seedlings (chi-squared test, p < 0.0001). By contrast, the survival of seedlings inoculated with H. crustuliniforme did not differ significantly from the control (p > 0.065; Fig. 2a). In spruce, during the first three growing seasons only C. geophilum inoculated seedlings had significantly higher survival than the control seedlings (ca. 9%; p < 0.0001). P. bicolorata had no effect on survival, while the seedlings treated with H. crustuliniforme had significantly lower survival (ca. 7%; p < 0.0001; Fig. 2b) than the control seedlings.
a Survival of Pinus sylvestris seedlings during three growing seasons. Within the same year, statistically significant differences between the treatments (Cg—Cenococcum geophilum, Pb—Piceirhiza bicolorata, Hc—Hebeloma crustuliniforme, Co—non-inoculated control) in chi-squared tests are designated by different letters. Error bars indicate standard error of the mean. b Survival of Picea abies seedlings during three growing seasons. Within the same year, statistically significant differences between the treatments (Cg—Cenococcum geophilum, Pb—Piceirhiza bicolorata, Hc—Hebeloma crustuliniforme, Co—non-inoculated control) in chi-squared tests are designated by different letters. Error bars indicate standard error of the mean
At outplanting, seedlings of each species were similar in height and in root system size; however, one-way ANOVA revealed certain differences in height increment among some treatments after second and third growing seasons (Fig. 3). After the third season, pine seedlings inoculated with C. geophilum had significantly greater height increment compared to the control (p < 0.03), while those inoculated with H. crustuliniforme were not significantly different (p > 0.09), and seedlings inoculated with P. bicolorata were significantly lower that the control (p < 0.0001).
Height increment of Pinus sylvestris and Picea abies seedlings after the second and third growing seasons. Within tree species and the same year, statistically significant differences between the treatments (Cg—Cenococcum geophilum, Pb—Piceirhiza bicolorata, Hc—Hebeloma crustuliniforme, Co—non-inoculated control) in one-way ANOVA are designated by different letters. Error bars indicate standard error of the mean
By contrast, spruce seedlings inoculated with the P. bicolorata had significantly greater height increment than that of non-inoculated plants (p < 0.001) after the second growing season, and this was also observed for both P. bicolorata and H. crustuliniforme after the third growing season. The inoculation of spruce with C. geophilum did not enhance height increment of trees to a significant extent (Fig. 3). For both tree species, a significant increase in height increment was observed in corresponding treatments compared to the second and third growing seasons (p < 0.0001). Generally, pine seedlings in each corresponding year and treatment exhibited significantly greater height increment than spruce seedlings (p < 0.001) (Fig. 3). No significant correlation was found between tested soil chemical properties and the survival and height increment of seedlings of either pine or spruce (p > 0.095).
Ectomycorrhizal colonisation
Table 2 shows that after the first growing season, mycorrhizal colonization was observed on 95–100% of inoculated pine seedlings and on 75% of control pines, and on 75–85% of inoculated spruce and 65% of control spruce. However, the differences in colonization of inoculated and control plants in each tree species were not statistically significant. After the first season, the pooled mycorrhizal colonization of all pine seedlings appeared to be significantly higher than that of all spruce (by 14%; p < 0.02). After the second growing season, mycorrhizal colonization of inoculated pines remained ca. 95–100%, but the mycorrhization of control plants increased to 100% (Table 2). For spruce seedlings, the second growing season resulted in a sharp increase in mycorrhization: up to 95–100% in each treatment and the control. Consequently, after two growing seasons no significant differences in the proportion of mycorrhizal plants were observed either among different treatments of spruce (including control) or between the two tree species (Table 2).
After the first season, the proportion of colonized roots (66–69%) of inoculated pine seedlings did not differ between the different mycorrhizal treatments. In non-inoculated controls, the mycorrhization was 14–17% lower, and in the comparisons with inoculation treatments the differences were statistically significant (p < 0.0003; Table 2). After the second year, mycorrhizal colonisation of root systems in P. bicolorata and H. crustuliniforme treatments, and in the control increased by 8–12%, while in C. geophilum treatment it remained about the same (+1%). Consequently, at this stage pine seedlings inoculated with H. crustuliniforme and P. bicolorata showed significantly higher (14–15%) colonization of roots than non-inoculated seedlings (p < 0.0001), but the difference between the C. geophilum treatment and the control was not significant (Table 2).
By contrast, root systems of spruce seedlings after the first season showed comparatively low ECM colonization: in each treatment and the control only 21–35% of root tips were mycorrhizal; less than half the level on pine (when pooled, 29% vs 64%; p < 0.0001; Table 2). Moreover, C. geophilum and P. bicolorata treatments were not significantly different from the control, and in the H. crustuliniforme treatment root colonization was significantly lower than in the control and in the other two treatments (by 8–14%; p < 0.035). However, the second year resulted in a sharp increase in mycorrhization of root systems of spruce, and the gap between the two tree species narrowed considerably. Thus, in non-inoculated control seedlings the extent of root system colonization by ECM fungi increased almost twofold (up to 54% of roots), in C. geophilum and P. bicolorata treatments increased by more than twice (to 70%), and in H. crustuliniforme treatment increased more than three times (to 63%; Table 2). However, despite these increases, a significant difference remained after the second year between mycorrhization of pine and spruce root systems (when pooled, 72 vs 64%; p < 0.0001). At this stage, in C. geophilum and P. bicolorata treatments ECM colonization of spruce root systems was significantly higher than in the control (by 16%; p < 0.0001). In H. crustuliniforme treatment mycorrhization was higher than in control, but the difference was not statistically significant (Table 2).
After the first growing season, mycorrhizal colonization of roots was in all treatments (including control) significantly higher for pine than for spruce (p < 0.0001). After the second season, colonization of roots remained higher for pine (p < 0.03), with the exception of the C. geophilum treatment where mycorrhization did not significantly differ between the tree species (p > 0.32).
Ectomycorrhizal community structure
Examination of the inoculated systems incubated in the growth chamber showed that the majority of fine roots of pine and spruce seedlings were colonized by the target fungi. H. crustuliniforme was the most efficient colonizer inhabiting ca. 75% of fine roots, while C. geophilum and P. bicolorata mycobiont colonized ca. 50% of fine roots. It was also observed that roots readily grew out through the layer of paper towel, and these were already colonized by the introduced fungi.
By contrast, after the first growing season the majority of observed mycorrhizas in the field were not the ones that had been inoculated (Table 3). Moreover, at this stage morphotypes of C. geophilum and P. bicolorata were also observed on roots of control and H. crustuliniforme-inoculated seedlings, but in all cases at significantly lower frequencies (p < 0.0001). After the second year, C. geophilum and P. bicolorata morphotypes were recorded at variable frequencies throughout all treatments, except for the pine control, but H. crustuliniforme disappeared completely from both tree species (Table 3).
Many other mycorrhizas were recorded on our study sites both after the first and the second growing seasons. A total of ten distinct morphotypes were observed after the first growing season and 19, including ten new, after the second season. When pooled, 20 morphotypes were detected, 11 of which were unique for pine, four unique for spruce, and five common to both tree species. Eleven morphotypes (55%) could not be matched to published descriptions and remained unidentified (Table 3).
The PCA of ectomycorrhizal communities clustered the morphotypes observed on pine and spruce into two well-separated groups (Fig. 4). The first axis explained 45.2% of the variation, while axis 2 explained a further 30.0%. A tight cluster of P. sylvestris indicated that there were no obvious differences in fungal community structure either between different treatments or growing seasons. The most common ECM morphotypes in different treatments/seasons were largely the same and included Suillus luteus (L.) Gray, Rhizopogon sp., Thelephora terrestris Ehrh. ex Fr., P. bicolorata, and Unidentified no. 3 (Fig. 4; Table 3). On the other hand, the elongated cluster of P. abies indicated the dynamic variation in ECM community structure between the two growing seasons (Fig. 4). One of the main reasons for this was sharply increased frequency of roots colonized by Amphinema byssoides (Pers.: Fr.) J. Erikss. and P. bicolorata (Table 3).
First and second axes of a principal component analysis of ectomycorrhizal communities in roots of Pinus sylvestris and Picea abies seedlings from different inoculation treatments (Cg—Cenococcum geophilum, Pb—Piceirhiza bicolorata, Hc—Hebeloma crustuliniforme, Co—non-inoculated control) and different growing seasons (Cg—2003; Cg—2004). Taxonomic names correspond to a position in the ordination (centred), and show the ten most common ectomycorrhizal fungi detected by mycorrhizal morphotyping
The differences in ECM community structure of P. abies were also observed between the different treatments after the first growing season. However, after the second season, all treatments were more or less similar to each other and clustered together (Fig. 4). In most cases, the most common taxa on spruce were A. byssoides, T. terrestris, P. bicolorata, C. geophilum, and Unidentified no. 2. However, the frequencies of these morphotypes differed considerably between different treatments and growing seasons (Table 3).
Discussion
The results of the present work showed that: 1) the persistence of inoculated mycorrhiza in the field and its effect on tree host depended on the fungus; 2) in two out of three fungal treatments (C. geophilum and P. bicolorata), inoculation resulted in positive effects on seedling survival and growth; 3) although significant, such effects were rather limited, but persisted at least for 3 years (Figs. 2 and 3); 4) the persistence of inoculated strains was generally low and natural colonization by indigenous ectomycorrhizas was abundant (Table 3).
There are a number of studies in which high persistence of inoculated mycorrhizal symbionts has been reported in forest plantations, but those involved different fungal and tree species than those used in the present work, and were carried out under different ecological conditions. For example, introduced Pisolithus spp. was shown to persist for up to 3 years in Chinese eucalypt plantations (Dell et al. 2002), and inoculated Laccaria bicolor (Maire) Orton persisted for 3–10 years in Douglas fir plantations in France (Selosse et al. 1998a,b; Di Battista et al. 2002). In Mediterranean pine plantations, inoculated Suillus collinitus persisted for 4 years (El Karkouri et al. 2006), and in Australia inoculated Amanita muscaria strains persisted for more than 30 years (Sawyer et al. 2001). By contrast, Heinonsalo et al. (2004) did not detect the inoculated L. bicolor strains in a forest plantation after 4 years and no treatment-related impact was observed in soil microbial communities. Despite that, they recorded significantly increased shoot growth in the mycorrhiza-inoculated plots compared with the non-inoculated plots, which corresponds well to the results of the present work. Yet, such cases when significant growth effects have been observed in spite of the fact that the introduced fungi did not persist are hard to explain, but, on the other hand, the positive impact might be initiated during more successful establishment of mycorrhiza-inoculated seedlings immediately after outplanting. Therefore, the possibility cannot be excluded that the growth conditions at the very early stages after planting have persisting effects for subsequent development of a tree seedling in the field.
It is known that mycorrhizal colonization under field conditions is to a large extent governed by ecological factors such as soil characteristics, tree species, and fungi (McAfee and Fortin 1985; Hedlund and Gormsen 2002). Thus, as our sites were apparently suitable for C. geophilum and P. bicolorata, a number of independent colonizations of seedlings by these fungi were observed in non-treated sets even after the first growing season, and substantially increased after the second (Table 3). Simultaneously, there seemingly was a high number of well-adapted indigenous mycorrhizas in the area, and after 2 years 19 additional morphotypes were observed on our seedlings (Table 3). Consequently, the formation of natural ectomycorrhizal communities was to a large extent observed in our plots, which proceeded much faster in pine than in spruce plantations. After the first growing season, in all treatments, the pine ECM community was far more diverse and showed a higher proportion of colonized roots than did the spruce ECM community, while during the second year the mycorrhization of spruce increased sharply (Tables 2 and 3). Moreover, the ectomycorrhizal communities of pine and spruce were clearly different (Fig. 4). Besides the tree species, an additional contributing factor could be the soil properties that differed markedly between the plantations (Table 1).
Therefore, the different rates of survival and growth of trees pre-inoculated with mycorrhizas observed in preceding studies (Baum et al. 2002; Beckjord and McIntosh 1984; Cram et al. 1999; Danielson and Visser 1989; Dunabeitia et al. 2004; Le Tacon et al. 1994; Loopstra 1988; Pera et al. 1999; Riffle and Tinus 1982; Stenström and Ek 1990; Stenström et al. 1990; Teste et al. 2004) could probably be explained by the diversity of factors (fungus, soil, tree species) involved in mycorrhizal establishment. In our recent work, we also demonstrated that ECM colonization rates and community structure differ significantly in forest nurseries under different cultivation systems (Menkis et al. 2005), and that different pathogens and endophytes attack conifer seedling roots planted in nurseries, forest clear-cuts, and abandoned farmland (Menkis et al. 2006), which might also be of importance to seedling survival and growth.
Finally, although significantly positive effects of mycorrhizal inoculations on tree performance and growth are reported in the present work, these were not dramatic and were associated with large investment of resources. One reason for the rather slight impact could be the good survival and growth, and an extensive natural mycorrhization of our trees on control plots. The artificial mycorrhization of seedlings, therefore, might be more useful for afforestation of marginal sites with harsh conditions where the first months or even weeks are crucial for tree establishment. Indeed, the improved seedling survival is encouraging because it can prove decisive in, e.g., dry years and, when cumulated over a long period of time, could result in a significant difference in terms of silviculture. More observations and measurements are, therefore, necessary to elucidate this, and will be done in our experimental plantations in the future.
References
Agerer R (1986–1988) Colour atlas of ectomycorrhizae. Einhorn-Verlag, Schwäbisch Gmünd, München, Germany
Agerer R, Danielson RM, Egli S, Ingleby K, Luoma D, Treu R (1996–1998) Description of ectomycorrhizae. Einhorn-Verlag, Schwäbisch Gmünd, München, Germany
Baum C, Stetter U, Makeschin F (2002) Growth response of Populus trichocarpa to inoculation by the ectomycorrhizal fungus Laccaria laccata in a pot and a field experiment. For Ecol Manag 163:1–8
Beckjord PR, McIntosh MS (1984) Growth and fungal persistence by Quercus rubra inoculated with ectomycorrhizal fungi and planted on a clear-cutting and strip mine. Can J Bot 62:1571–1574
Bjorkman E (1970) Forest tree mycorrhiza—the conditions for its formation and the significance for tree growth and afforestation. Plant Soil 32:589–610
Bledsoe CS, Tennyson K, Lopushinsky W (1982) Survival and growth of outplanted Douglas-fir seedlings inoculated with mycorrhizal fungi. Can J For Res 12:720–723
Brand F, Gronbach E, Taylor AFS (1992) Piceirhiza bicolorata. In: Agerer R (ed) Colour atlas of ectomycorrhizae. Einhorn-Verlag, Schwäbich Gmünd, München, Germany, pp 73
Chakravarty P, Unestam T (1985) Role of mycorrhizal fungi in protecting damping-off of Pinus sylvestris L. seedlings. In: Gianinazzi-Pearson V, Gianinazzi S (eds) Physiological and genetical aspects of mycorrhizae. Institut national de le recherche agronomique, Dijon, France, Proceedings of the 1st European Symposium on Mycorrhizae, pp 811–814
Chalmers N, Parker P (1989) Fieldwork and statistics for ecological projects, second edition. The Open University, Dorchester
Chilvers GA, Gust WL (1982) Comparison between the growth rates of mycorrhizas, uninfected roots and a mycorrhizal fungus of Eucalyptus st. johnii R.T. Bak. New Phytol 91:453–466
Chilvers GA, Douglass AP, Lapeyrie FF (1986) A paper-sandwich technique for rapid synthesis of ectomycorrhizas. New Phytol 103:397–402
Colpaert JV, Vanassche JA (1992) Zinc toxicity in ectomycorrhizal Pinus sylvestris. Plant Soil 143:201–211
Conover WJ (1980) Practical nonparametric statistics, second edition. Wiley, New York
Cram MM, Mexal JG, Souter R (1999) Successful reforestation of South Carolina sandhills is not influenced by seedling inoculation with Pisolithus tinctorius in the nursery. South J Appl For 23:46–52
Dahlberg A, Stenström E (1991) Dynamic changes in nursery and indigenous mycorrhiza of Pinus sylvestris seedlings planted out in forest and clearcuts. Plant Soil 136:73–86
Danielson RM, Visser S (1989) Host response to inoculation and behaviour of introduced and indigenous ectomycorrhizal fungi of jack pine grown on oil-sands tailings. Can J For Res 19:1412–1421
Dell B, Malajczuk N, Dunstan WA (2002) Persistence of some Australian Pisolithus species introduced into eucalypt plantations in China. For Ecol Manag 169:271–281
Di Battista C, Bouchard D, Martin F, Genere B, Amirault JM, Le Tacon F (2002) Survival after outplanting of the ectomycorrhizal fungus Laccaria bicolor S238N inoculated on Douglas fir (Pseudotsuga menziesii (Mirb.) Franco) cuttings. Ann For Sci 59:81–92
Dickie IA, Reich PB (2005) Ectomycorrhizal fungal communities at forest edges. J Ecol 93:244–255
Dighton J, Mason PA (1985) Mycorrhizal dynamics during forest tree development. In: Moore D, Casselton LA, Wood DA, Frankland JC (eds) Developmental biology of higher fungi. Cambridge University Press, Cambridge, pp 163–171
Dunabeitia M, Rodriguez N, Salcedo I, Sarrionandia E (2004) Field mycorrhization and its influence on the establishment and development of the seedlings in a broadleaf plantation in the Basque Country. For Ecol Manag 195:129–139
El Karkouri K, Selosse MA, Mousain D (2006) Molecular markers detecting an ectomycorrhizal Suillus collinitus strain on Pinus halepensis roots suggest successful inoculation and persistence in Mediterranean nursery and plantation. FEMS Microbiol Ecol 55:146–158
Fowler J, Cohen L, Jarvis P (2001) Practical statistics for field biology, second edition. Wiley, Chichester
Hacskaylo E (1973) Dependence of mycorrhizal fungi on hosts. J Torrey Bot Soc 100:217–223
Hedlund K, Gormsen D (2002) Mycorrhizal colonization of plants in set-aside agricultural land. Appl Soil Ecol 19:71–78
Heinonsalo J, Frey-Klett P, Pierrat JC, Churin JL, Vairelles D, Garbaye J (2004) Fate, tree growth effect and potential impact on soil microbial communities of mycorrhizal and bacterial inoculation in a forest plantation. Soil Biol Biochem 36:211–216
Kernaghan G, Hambling B, Fung M, Khasa D (2002) In vitro selection of boreal ectomycorrhizal fungi for use in reclamation of saline-alkaline habitats. Restor Ecol 10:43–51
Kropp BR, Langlois CG (1990) Ectomycorrhizae in reforestation. Can J Forest Res 20:438–451
Le Tacon F, Bouchard D (1986) Effects of different ectomycorrhizal fungi on growth of larch, Douglas fir, Scots pine and Norway spruce seedlings in fumigated nursery soils [Laccaria laccata, Hebeloma crustuliniforme]. Acta Oecol Oecol Appl 7:389–402
Le Tacon F, Alvarez IF, Bouchard D, Henrion B, Jackson MR, Luff S, Parlade JI, Pera J, Stenström E, Villeneuve N, Walker C (1994) Variations in field response of forest trees to nursery ectomycorrhizal inoculation in Europe. In: Read DJ, Lewis DH, Fitter AH, Alexander IJ (eds) Mycorrhizas in ecosystems. CAB International, Wallingford, UK, pp 119–134
Loopstra LM (1988) Ectomycorrhizal inoculation fails to improve performance of Sitka spruce seedlings on clearcuts in southeastern Alaska. West J Appl For 3:110–112
Marx DH (1969) The influence of ectotrophic ectomycorrhizal fungi on the resistance of pine roots to pathogenic infections. I. Antagonism of mycorrhizal fungi to pathogenic fungi and soil bacteria. Phytopathology 59:153–163
Marx DH (1980) Ectomycorrhiza fungus inoculations: a tool to improve forestation practices. In: Mikola P (ed) Tropical mycorrhiza research. Oxford University Press, Oxford, pp 13–71
Marx DH, Daniel WJ (1976) Maintaining cultures of ectomycorrhizal and plant pathogenic fungi in sterile cold water storage. Can J Microbiol 22:338–341
McAfee BJ, Fortin JA (1985) Competitive interactions of ectomycorrhizal mycobionts under field conditions. Can J Bot 64:848–852
Mead R, Curnow RN (1983) Statistical methods in agriculture and experimental biology. Chapman & Hall, London
Menkis A, Vasiliauskas R, Taylor AFS, Stenlid J, Finlay R (2005) Fungal communities in mycorrhizal roots of conifer seedlings in forest nurseries under different cultivation systems, assessed by morphotyping, direct sequencing and mycelial isolation. Mycorrhiza 16:33–41
Menkis A, Vasiliauskas R, Taylor AFS, Stenström E, Stenlid J, Finlay R (2006) Fungi in decayed roots of conifer seedlings from forest nurseries, afforested clearcuts and abandoned farmland. Plant Pathol 55:117–129
Mexal J, Reid CPP (1973) The growth of selected mycorrhizal fungi in response to induced water stress. Can J Bot 51:1579–1588
Mikola P (1948) On the physiology and ecology of Cenococcum geophilum especially as a mycorrhizal fungus on birch. Commun Inst for Fenn 36:1–101
Mikola P (1970) Mycorrhizal inoculation for afforestation. Int Rev For Res 3:123–196
Minitab® Inc. (2003) Minitab Statistical Software. Release 14.1. Minitab Inc., Pennsyllvania
Morin C, Samson J, Dessureault M (1999) Protection of black spruce seedlings against Cylindrocladium root rot with ectomycorrhizal fungi. Can J Bot 77:169–174
Muhsin TM, Zwiazek JJ (2002) Colonization with Hebeloma crustuliniforme increases water conductance and limits shoot sodium uptake in white spruce (Picea glauca) seedlings. Plant Soil 238:217–225
Ortega U, Dunabeitia M, Menendez S, Gonzalez-Murua C, Majada J (2004) Effectiveness of mycorrhizal inoculation in the nursery on growth and water relations of Pinus radiata in different water regimes. Tree Physiol 24:65–73
Pera J, Alvarez IF, Rincon A, Parlade J (1999) Field performance in northern Spain of Douglas-fir seedlings inoculated with ectomycorrhizal fungi. Mycorrhiza 9:77–84
Perrin R, Garbaye J (1983) Influence of ectomycorrhizae on infectivity of Pythium-infested soil and substrates. Plant Soil 71:345–351
Perry AD, Molina R, Amaranthus PM (1987) Mycorrhizae, mycorrhizospheres, and reforestation: current knowledge and research needs. Can J For Res 17:929–940
Pigott CD (1982) Survival of mycorrhiza formed by Cenococcum geophilum Fr. in dry soils. New Phytol 92:513–517
Riffle JW, Tinus RW (1982) Ectomycorrhizal characteristics, growth, and survival of artificially inoculated ponderosa and Scots pine in a greenhouse and plantation. For Sci 28:646–660
Rosling A, Lindahl BD, Finlay R (2004) Carbon allocation to ectomycorrhizal roots and mycelium colonising different mineral substrates. New Phytol 162:795–802
Saleh-Rastin N (1976) Salt tolerance of the mycorrhizal fungus Cenococcum graniforme (Sow.) Ferd. Eur J For Pathol 6:184–187
Sawyer NA, Chambers SM, Cairney JWG (2001) Distribution and persistence of Amanita muscaria genotypes in Australian Pinus radiata plantations. Mycol Res 105:966–970
Selosse MA, Jacquot D, Bouchard D, Martin F, Le Tacon F (1998a) Temporal persistence and spatial distribution of an American inoculant strain of the ectomycorrhizal basidiomycete Laccaria bicolor in European forest plantations. Mol Ecol 7:561–573
Selosse MA, Martin F, Le Tacon F (1998b) Survival of an introduced ectomycorrhizal Laccaria bicolor strain in a European forest plantation monitored by mitochondrial ribosomal DNA analysis. New Phytol 140:753–761
Senior E, Smith JE, Watsoncraik IA, Tosh JE (1993) Ectomycorrhizae and landfill site reclamations—fungal selection criteria. Lett Appl Microbiol 16:142–146
Smith SE, Read DJ (1997) Mycorrhizal Symbiosis. Academic, London, UK
Sokal RR, Rohlf (1995) Biometry, third edition. WH Freeman, New York
Stenström E, Ek M (1990) Field growth of Pinus sylvestris following nursery inoculation with mycorrhizal fungi. Can J For Res 20:914–918
Stenström E, Ek M, Unestam T (1990) Variation in field response of Pinus sylvestris to nursery inoculation with four different ectomycorrhizal fungi. Can J For Res 20:1796–1803
ter Braak CJF, Smilauer P (1998) Canoco reference manual and user’s guide to Canoco for Windows: software for canonical community ordination, Version 4. Microcomputer Power, Ithaca, NY, USA
Teste FP, Schmidt MG, Berch SM, Bulmer C, Egger KN (2004) Effects of ectomycorrhizal inoculants on survival and growth of interior Douglas-fir seedlings on reforestation sites and partially rehabilitated landings. Can J For Res 34:2074–2088
Theodorou C, Bowen GD (1970) Mycorrhizal responses of radiata pine in experiments with different fungi. Aust For 34:183–193
Trappe JM (1977) Selection of fungi for ectomycorrhizal inoculation in nurseries. Annu Rev Phytopathol 15:203–222
Trappe J (1988) Lessons from alpine fungi. Mycologia 80:1–10
Van Tichelen KK, Colpaert JV, Vangronsveld J (2001) Ectomycorrhizal protection of Pinus sylvestris against copper toxicity. New Phytol 150:203–213
Villarreal-Ruiz L, Anderson IC, Alexander IJ (2004) Interaction between an isolate from the Hymenoscyphus ericae aggregate and roots of Pinus and Vaccinium. New Phytol 164:183–192
Vrålstad T, Fossheim T, Schumacher T (2000) Piceirhiza bicolorata—the ectomycorrhizal expression of the Hymenoscyphus ericae aggregate. New Phytol 145:549–563
Wilcox HE (1996) Mycorrhizae. In: Wasel Y, Eshel A, Kafkafi U (eds) Plant roots: the hidden half. Marcel Dekker, New York, pp 689–721
Acknowledgements
This research was funded by the European Commission Marie Curie Host Fellowships Project No 042622 LITCOAST and the Royal Swedish Academy of Agriculture and Forestry (KSLA).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Menkis, A., Vasiliauskas, R., Taylor, A.F.S. et al. Afforestation of abandoned farmland with conifer seedlings inoculated with three ectomycorrhizal fungi—impact on plant performance and ectomycorrhizal community. Mycorrhiza 17, 337–348 (2007). https://doi.org/10.1007/s00572-007-0110-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00572-007-0110-0