Abstract
Purpose
The anesthesiologist-directed sedation service has not been well established in Japan partly due to reimbursement issue. In this study, we compared the cost-effectiveness of sedation by non-anesthesiologists with that of sedation or general anesthesia by anesthesiologists under the Japanese medical fee schedule.
Methods
We conducted a single-center observational study with patients who required sedation or general anesthesia for magnetic resonance imaging (MRI) during a 12-month period. Costs per patient and failure rates of imaging were modeled in a decision analysis tree with sensitivity analysis. Costs were estimated from the health-care sector perspective.
Results
A total of 1546 patients were analyzed. The failure rate of sedation by non-anesthesiologists was 17.5% (264 out of 1506), whereas all the sedation and general anesthesia by anesthesiologists were successful. The cost-effectiveness analysis with setting successful sedation as outcomes showed that the mean cost per patient was 84.2 USD for sedation by anesthesiologists, followed by 74.2–92.7 USD for intravenous sedation by non-anesthesiologists, 112.1–458.3 USD for oral or rectal sedation by non-anesthesiologists, and 605.4 USD for general anesthesia by anesthesiologists. The one-way sensitivity analysis demonstrated that the cost per patient of sedation by a non-anesthesiologist would remain higher than that of sedation by an anesthesiologist, provided that the failure rate is over 11.3% for sedation via oral or rectal route, or over 3.6% for intravenous route, respectively.
Conclusions
Anesthesia-directed sedation would be more cost-effective than oral or rectal sedation by non-anesthesiologists for children undergoing MRI in the Japanese medical fee schedule.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Historically, anesthesiologists have been involved directly in sedating or anesthetizing children as well as have taken active role in establishing guidelines and standards for sedation of children outside the operating room over several decades in the world [1,2,3]. In North America and Europe, the anesthesiologist-directed sedation service has been established [1,2,3]. In contrast, such service has not been well established in Japan. A shortage of anesthesiologists has been cited as the most common barrier to development of such an anesthesiologist-directed sedation service [4]. On top of that, small reimbursement benefits for the anesthesiologist-directed sedation service would also account for underdevelopment of the service in Japan.
The underdevelopment of the anesthesiologist-directed sedation service has caused to reduce several aspects of the quality in pediatric procedural sedation, which includes the following; rate of adverse events associated with sedation and general anesthesia (S/GA) [5, 6]; rate of successful S/GA for imaging examinations [7]; effect of S/GA on imaging flow [8]; effect of S/GA on imaging quality [6, 9].
The continued improvement of pediatric sedation practice depends on the involvement of qualified professionals such as anesthesiologists. This involvement can be guaranteed through proper reimbursement [1]. No clinical researches remain to be published to compare sedation by non-anesthesiologists and S/GA by anesthesiologists from the viewpoint of cost-effectiveness based on the national medical fee schedule in Japan.
The aims of this study was to compare the cost-effectiveness of sedation by non-anesthesiologists with that of S/GA by anesthesiologists for children undergoing magnetic resonance imaging (MRI) with setting successful sedation as the outcome measure.
Methods
Study design and subjects
The protocol used to gather data related to patients requiring S/GA for MRI at Saitama Children’s Medical Center was approved by the institutional review board (IRB). Informed assent or consent from patients or guardians was secured on an ‘opt out’ basis which was approved by the IRB because the study was part of a quality improvement project.
We conducted a single-center observational study with review of the charts of patients who received S/GA for MRI by an anesthesiologist or a non-anesthesiologist during a 12-month period between August 1, 2017 and July 30, 2018. With the shift from use of computed tomography (CT) to MRI in infants and children due to concerns about ionizing radiation, there is an increasing need for sedation because of the longer duration of MRI as well as the increased need for patient immobility and cooperativeness [10]. Saitama Children’s Medical Center is a public quaternary care children’s hospital in Japan that provides inpatient and outpatient procedural sedation for approximately 1500 children requiring MRI scans per year. Since the introduction of an anesthesiologist-directed sedation service in 2014, S/GA has been provided by non-anesthesiologists or anesthesiologists.
The study inclusion criterion was age of less than 21 years. Patient characteristics, including sex, age, weight, height, American Society of Anesthesiologists Physical Status (ASA-PS) classification, and presence of cognitive developmental delay were obtained from a standardized pre-sedation history and physical evaluation form. Cognitive developmental delay was broadly defined in this study, which includes chromosomal abnormalities, autistic spectrum disorder, attention deficit hyperactivity disorder, and cerebral palsy with cognitive developmental delay. The S/GA start and end times, types of S/GA administered, and routes and types of medications initially administered were obtained from standard S/GA records which was entered by nurses in the MRI suite or by the anesthesiologists in charge. Patients who had been already sedated or anesthetized before scanning were excluded.
Sedation, which is basically classified into three levels from mild, moderate and deep, and general anesthesia were defined based on the definitions by the American Society of Anesthesiologists [11]. Successful sedation was defined as completion of imaging in a sedated or anesthetized patient without requirement for cancelation or cessation of the scanning followed by rescheduling.
Sedation or general anesthesia practices
Our institution basically recommends that a patient of ASA-PS class III and over should be referred to anesthesiologists for sedation outside the operating room, as several sedation guidelines recommend [2, 3]. It, however, is finally up to non-anesthesiologists’ discretion which patient should be referred to anesthesiologists and how sedation would be conducted. The joint commission of three academic societies in Japan (the Japan Pediatric Society, the Japanese Society of Pediatric Anesthesiology, and the Japanese Society of Pediatric Radiology) has issued guidelines for sedation of children undergoing MRI scans, which have not recommended specific regimens or doses of sedative or anesthetic agents [12]. Hence, a contributing physician at each department individually chooses a drug regimen which she or he deems most appropriate. Most non-anesthesiologists do not use propofol for sedation at our institution.
Assumptions and calculations of the costs and cost-effectiveness analysis
Based on expert clinical opinions and the published literature [13], we estimated the costs related to S/GA for MRI scans from the viewpoint of this modality being a limited social resource. The calculations focused on drug cost, procedure cost, and opportunity cost (cost of rescheduled MRI scan and GA procedure), and did not take into account consumables and facility costs, given that these are the same regardless of whether the S/GA is administered by an anesthesiologist or a non-anesthesiologist. All the costs were calculated based on the national medical fee schedule in Japan on the assumption that the duration of S/GA required for an MRI scan is one hour per patient (aged 3 years and weighing 15 kg). For international readers to understand, the local currency, Japanese yen, was converted to the US dollar as 1 JPY = 0.0089 USD in this study.
In the Japanese national medical fee schedule as of September 2019, even when a certified anesthesiologist provides deep sedation outside the operating room, the institution can claim only 71 US dollars (USD) (8000 Japanese yen (JPY)) for reimbursement of a sedation practice fee per patient, which is only 18 USD (2000 JPY) more than when non-anesthesiologists provide sedation. The reimbursement of a sedation practice per patient is much less than that of a general anesthetic practice per surgery in the operating room which is at least 534 USD (60,000 JPY).
The largest component of the medical costs was staff time (particularly that of the physician, surgeon, and anesthesiologist), determined from the literature on cost-effectiveness analysis [14]. However, the Japanese national medical fee schedule only reimburses hospital fees, but not doctors’ fees. Hence, the assumption of staff time was substituted with the associated procedure cost based on the medical remuneration points. We assumed that if imaging were aborted, GA would be administered on another subsequent day to enable the imaging to be completed. In such a case, the cost of MRI scanning and GA procedure fee for an anesthesiologist would be added on the assumption that another patient would have been able to undergo MRI instead. If imaging under intravenous (IV) sedation provided by a non-anesthesiologist failed, the outpatient consulting fee (assumed as four patients per hour) for a non-anesthesiologist would be added as an opportunity cost on top of addition of the cost of MRI scanning and GA procedure fee for an anesthesiologist.
Hence, the mean total cost per patient by sedative medication was calculated as the following, respectively:
(First-line treatment cost) = (medication cost) + (procedure cost).
(Mean cost of second-line treatment) = {(opportunity cost on failure) + (additional cost on failure)} × (failure rate [%])/100.
(Mean total cost per patient) = (first-line treatment cost) + (mean cost of second-line treatment).
The results of the cost-effectiveness analysis were presented as the mean cost per patient.
A change in the cost relative to the sedation failure rate was calculated using one-way sensitivity analysis of the probability of failure of sedation by a non-anesthesiologist or an anesthesiologist [13].
Results
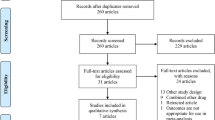
During the study period, 1575 patients required S/GA for MRI. Twenty-nine patients were excluded: 27 patients who had already been sedated or anesthetized before scanning, one whose MRI scan was stopped because of a technical malfunction of the MRI apparatus, and one who refused the MRI examination after a sedative had been administered. Of the 1546 patients eligible for the study, 1506 were sedated by non-anesthesiologists and 40 were sedated or anesthetized by anesthesiologists. When provided by non-anesthesiologists, sedation was successful in 1242 patients and failed in 264. S/GA was provided by anesthesiologists in 40 patients, all of whose MRI scans were successfully completed (Fig. 1). Tables 1 and 2 summarize the patient demographics, routes and types of initial medications administered, the departments to which the pediatricians, surgeons, or anesthesiologists in charge of administering S/GA belonged, regions imaged, and image-acquisition times. Types of medications administered were classified into six groups for the purpose of data analysis based on the initial medication used for S/GA, because more than 20 medications were identified to have been administered, with a variety of additional medications which included triclofos sodium, pentobarbital calcium, chloral hydrate, hydroxyzine, and midazolam. 1064 out of 1506 sedations by non-anesthesiologists (72.6%) were done with the use of oral or rectal medications. Anesthesiologists exclusively used sevoflurane and/or propofol for sedation and anesthesia. The non-anesthesiology department-in-charge varied and was also classified into seven categories.
All the reasons of failed sedation by non-anesthesiologists were inadequate sedation. The failure rates according to the initial medications administered were as follows: triclofos sodium (oral) 18.5% (157/848), pentobarbital calcium (oral) 33.3% (71/213), chloral hydrate (rectal) 60.0% (18/30), hydroxyzine (IV) 2.2% (2/91), and midazolam (IV) 4.6% (14/307). These sedation failure rates of sedation by non-anesthesiologists and the 0% failure rate of S/GA by anesthesiologists were modeled in a cost-effectiveness analysis (Table 3).
The cost-effectiveness analysis demonstrated that the mean total cost per patient according to the route of initial medication administered and provider were as it follows: oral or rectal route by a non-anesthesiologist, 112.1–253.8 USD; IV route by a non-anesthesiologist, 74.2–92.7 USD; sedation by an anesthesiologist, 84.2 USD; and GA by an anesthesiologist, 605.4 USD. Table 3 summarizes how the cost per patient was calculated.
Figure 2 illustrates how the cost of sedation varied depending on the failure rate as determined by one-way sensitivity analysis. This analysis demonstrated that the cost per patient of sedation by a non-anesthesiologist would remain higher than that of sedation by an anesthesiologist, provided that the failure rate is over 11.3% for sedation via oral or rectal route, or over 3.6% for IV route, respectively.
Change in cost relative to failure rate of sedation. The cost per patient of sedation by a non-anesthesiologist would remain higher than that of sedation by an anesthesiologist, provided that the failure rate is over 11.3% for sedation via oral or rectal route, or over 3.6% for intravenous route, respectively
Discussion
This study showed that the failure rate of sedation by non-anesthesiologists for children undergoing MRI was markedly higher than that of S/GA by anesthesiologists. Our cost-effectiveness analysis demonstrated that sedation by anesthesiologists for MRI would be more cost-effective than sedation via oral or rectal route by non-anesthesiologists.
Japan formally introduced a cost-effectiveness analysis to inform health-care decision making, mainly when it comes to the pricing of new pharmaceuticals and technologies, although the target of the analysis remains limited and has not yet included health technologies and procedures in the field of S/GA in April 2019 [15]. Our analysis was the first study to compare the cost-effectiveness of sedation by non-anesthesiologists with S/GA by anesthesiologists under the Japanese national medical fee schedule, which suggested that sedation by anesthesiologists would be more cost-effective than sedation via oral or rectal route by non-anesthesiologists. The cost-effectiveness in sedation by anesthesiologists would be derived from significantly lower reimbursement compared to that for general anesthesia, and higher failure rates of sedation by non-anesthesiologists compared to that by anesthesiologists. In the field of pediatric and adolescent sedation, cost-effectiveness analyses in the UK demonstrated that the cost of sedation and general anesthesia for several common procedures (dental procedures, short painful procedures, painless imaging, and endoscopy) varied depending on the failure rate [14]. The analyses showed that the cost per patient for sedation with intravenous midazolam for adolescent dental care and esophago-gastroscopy would remain higher than that of general anesthesia, provided that the failure rate of sedation with intravenous midazolam goes over 37% and 25%, respectively [14]. The NICE costing report estimated the costs from the perspective of health-care systems and personal social services in the UK. Generally cost-effectiveness analysis itself is based on the estimation of the pertinent costs from the perspective of health-care systems and personal social services in each country or region. Hence, we did not make a direct comparison of cost-effectiveness analyses in the field of pediatric sedation between our analysis under Japanese medical fee schedule and others in other countries or regions.
Successful sedation was set as the outcome of cost-effectiveness analysis in this study. Historically, most children who receive sedation outside the operating room had a good outcome in terms of reduced anxiety during a procedure [16, 17]. As far as sedation for pediatric MRI was concerned, combinations of benzodiazepines, barbiturates, hypnotics, and narcotics were typically used, with variable success and a risk of upper airway obstruction and respiratory depression [2, 4]. Previously reported studies showed chloral hydrate, midazolam, and pentobarbital were the drugs of choice by non-anesthesiologists for pediatric sedation in most imaging examinations, with failure rates in the range of 2–15% [2, 9, 18, 19]. The result of our study was consistent with these previously reported ones. Raising the effectiveness, or the success rate, of sedation by non-anesthesiologists may be possible by several ways such as developing guidelines [20, 21], institutional protocols, and training systems for non-anesthesiologists [22]. Even in North America, the majority of the hospitalists surveyed perceived they had not achieved competency in sedation [23]. Anesthesiologists are expected to take initiative and responsibility for improvement of sedation by non-anesthesiologists [1]. Training and supporting pediatric hospitalists to provide procedural sedation, however, has the potential to avoid unnecessary referrals to anesthesiologists and to decrease painful procedures done without sedation [24].
Limitations
The limitations of this study include its observational study design, the single-center experience with possibly low generalizability, and the theoretical and limited evaluation by assumptions in the cost-effectiveness analysis with deterministic one-way sensitivity analysis [25]. In terms of estimating and calculating costs, it is important to note that this study only took into account direct health-care costs, and did not include indirect costs such as transportation costs to and from hospital and opportunity cost of productivity loss in forfeited wages of the guardians who accompanied patients from the societal perspective. The cost-effective analysis from the societal perspective will be our tasks and challenges of the future. Then, we did not estimate quality-adjusted life years, but we think this is unlikely to affect our conclusions. There will be some disutility (reduced health-related quality of life) associated with sedation failure. These changes, however, will occur over a short period of time and therefore differences in mean quality-adjusted life years between strategies are likely to be negligible [14].
Conclusion
The failure rate of sedation provided by non-anesthesiologists for children undergoing MRI was considerably higher than that of S/GA provided by anesthesiologists. Our study was the first comparison of the cost-effectiveness of sedation administered by non-anesthesiologists with that of S/GA provided by anesthesiologists under the Japanese national medical fee schedule, which suggested that sedation provided by anesthesiologists for children undergoing MRI would be more cost-effective than sedation via oral or rectal route provided by non-anesthesiologists and GA provided by anesthesiologists. The cost-effectiveness in sedation by anesthesiologists would be derived from significantly lower reimbursement compared to that for general anesthesia and higher failure rates of sedation by non-anesthesiologists compared to that by anesthesiologists.
References
Cravero JP, Blike GT. Review of pediatric sedation. Anesth Analg. 2004;99:1355–64.
Coté CJ, Wilson S. American Academy of Pediatrics; American Academy of Pediatric. Dentistry guidelines for monitoring and management of pediatric patients before, during, and after sedation for diagnostic and therapeutic procedures. Pediatrics. 2019;143:e20191000.
Wilson SR, Shinde S, Appleby I, Boscoe M, Conway D, Dryden C, Ferguson K, Gedroyc W, Kinsella SM, Nathanson MH, Thorne J, White M, Wright E. Guidelines for the safe provision of anaesthesia in magnetic resonance units 2019: guidelines from the Association of Anaesthetists and the Neuro Anaesthesia and Critical Care Society of Great Britain and Ireland. Anaesthesia. 2019;74:638–50.
Lalwani K, Michel M. Pediatric sedation in North American Children’s hospitals: a survey of anesthesia providers. Pediatr Anesth. 2005;15:209–13.
Kamat PP, McCracken CE, Simon HK, Stormorken A, Mallory M, Chumpitazi CE, Cravero JP. Trends in outpatient procedural sedation: 2007–2018. Pediatrics. 2020;145:e20193559.
Malviya S, Voepel-Lewis T, Eldevik OP, Rockwell DT, Wong JH, Tait AR. Sedation and general anaesthesia in children undergoing MRI and CT: adverse events and outcomes. Br J Anaesth. 2000;84:743–8.
Couloures KG, Beach M, Cravero JP, Monroe KK, Hertzog JH. Impact of provider specialty on pediatric procedural sedation complication rates. Pediatrics. 2011;127:e1154–60.
Vanderby SA, Babyn PS, Carter MW, Jewel SM, McKeever PD. Effect of anesthesia and sedation on pediatric MR imaging patient flow. Radiology. 2010;256:229–37.
Ucisik-Keser FE, Chi TL, Hamid Y, Dinh A, Chang E, Ferson DZ. Impact of airway management strategies on magnetic resonance image quality. Br J Anaesth. 2016;117:i97–102.
Masaracchia MM, Tsapakos MJ, McNulty NJ, Beach ML. Changing the paradigm for diagnostic MRI in pediatrics: don’t hold your breath. Paediatr Anaesth. 2017;27:880–4.
American Society of Anesthesiologists. Continuum of depth of sedation: definition of general anesthesia and levels of sedation/analgesia. https://www.asahq.org/standards-and-guidelines/continuum-of-depth-of-sedation-definition-of-general-anesthesia-and-levels-of-sedationanalgesia. [Accessed on 15 February 2022]
The Joint Commission of the Japanese Society of Pediatrics, the Japanese Society of Pediatric Anesthesiology and the Japanese Society of Pediatric Radiology. The guidelines of sedation for children undergoing magnetic resonance imaging (in Japanese). Nihon Shonika Gakkai Zasshi (J Jap Pediatr Soc). 2020;124:771–805.
Sanders GD, Neumann PJ, Basu A, Brock DW, Feeny D, Krahn M, Kuntz KM, Meltzer DO, Owens DK, Prosser LA, Salomon JA, Sculpher MJ, Trikalinos TA, Russell LB, Siegel JE, Ganiats TG. Recommendations for conduct, methodological practices, and reporting of cost-effectiveness analyses: second panel on cost-effectiveness in health and medicine. JAMA. 2016;316:1093–103.
National Institute for Health and Clinical Excellence. NICE clinical guideline 112. Sedation in children and young people: sedation for diagnostic and therapeutic procedures in children and young people. appendix F cost-effectiveness analysis. https://www.ncbi.nlm.nih.gov/books/NBK82223/ [Accessed on 15 February 2022]
Hasegawa M, Komoto S, Shiroiwa T, Fukuda T. Formal implementation of cost-effectiveness evaluations in Japan: a unique health technology assessment system. Value Health. 2020;23:43–51.
King WK, Stockwell JA, Deguzman MA, Simon HK, Khan NS. Evaluation of a pediatric sedation service for common diagnostic procedures. Acad Emerg Med. 2006;13:673–6.
Pena BM, Krauss B. Adverse events of procedural sedation and analgesia in a pediatric emergency department. Ann Emerg Med. 1999;34:483–91.
Green SM, Rothrock SG, Lynch EL, Ho M, Harris T, Hestdalen R, Hopkins GA, Garrett W, Westcott K. Intramuscular ketamine for pediatric sedation in the emergency department: safety profile in 1022 cases. Ann Emerg Med. 1998;31:688–97.
Malviya S, Voepel-Lewis T, Tait AR. Adverse events and risk factors associated with the sedation of children by nonanaesthesiologists. Anesth Analg. 1997;85:1207–13.
American Society of Anesthesiologists. Advisory on granting privileges for deep sedation to non-anesthesiologist physicians. https://www.asahq.org/standards-and-guidelines/advisory-on-granting-privileges-for-deep-sedation-to-non-anesthesiologist-physicians [Accessed on 15 Feb 2022]
American Society of Anesthesiologists Task Force on Sedation and Analgesia by Non-Anesthesiologists. Practice guidelines for sedation and analgesia by non-anesthesiologists. Anesthesiology. 2002;96:1004–17.
Komasawa N, Fujiwara S, Atagi K, Ueki R, Haba M, Ueshima H, Kaminoh Y, Minami T. Effects of a simulation-based sedation training course on non-anesthesiologists’ attitudes toward sedation and analgesia. J Anesth. 2014;18:785–9.
Librizzi J, Winer JC, Banach L, Davis A. Perceived core competency achievements of fellowship and non-fellowship-trained early career pediatric hospitalists. J Hosp Med. 2015;10:373–9.
Srinivasan M, Bhaskar S, Carlson DW. Variation in procedural sedation practices among children’s hospitals. Hosp Pediatr. 2015;5:148–53.
McCabe C, Paulden M, Awotwe I, Sutton A, Hall P. One-way sensitivity analysis for probabilistic cost-effectiveness analysis: conditional expected incremental net benefit. Pharmacoeconomics. 2020;38:135–41.
Acknowledgements
The authors thank and acknowledge Norifumi Kuratani, MD, PhD, MPH, at Saitama Children’s Medical Center, and nurses and technicians at the radiology department for assisting the authors with data collection.
Funding
This study was conducted with a research fund from Saitama Children’s Medical Center. Otherwise this study did not receive any specific grant from funding agencies in the public or commercial sectors.
Author information
Authors and Affiliations
Contributions
All authors were involved in study design, data interpretation, and data analysis. All authors critically revised the report, commented on drafts of the manuscript, and approved the final report.
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest.
Informed consent
Informed consent was waived as the study was determined to be exempt.
IRB approval
IRB approval was obtained through Saitama Children’s Medical Center.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Obara, S., Nakata, Y. & Yamaoka, K. Cost-effectiveness analysis of sedation and general anesthesia regimens for children undergoing magnetic resonance imaging in Japan. J Anesth 36, 359–366 (2022). https://doi.org/10.1007/s00540-022-03051-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00540-022-03051-w