Abstract
Objective
To assess the independent and combined associations of tumor-related psychiatric symptoms (TRPS) with dynamic health-related quality of life (HRQL) in patients with hepatocellular carcinoma (HCC) after hepatectomy and to identify related patterns of health behaviors.
Methods
This prospective study included patients with HCC who underwent hepatectomy between September 2021 and May 2022. Independent and combined associations between TRPS and HRQL were identified by generalized linear model and weighted quantile sum model, respectively. Trajectories of HRQL were identified by latent class mixed model.
Results
Among the 205 patients, 174 (84.9%) were male. For the outcome of HRQL at 6 months: Anxiety, depression, fatigue, and sleep disorder were independently associated with a decrease of HRQL (all P < 0.05). A negative combined effect of TRPS was also found (β = − 5.07, 95% CI, − 10.01 to − 0.13), with depression emerged as the predominant contributor (49%). The health behaviors of body mass index, smoking, drinking, or physical exercise were not significantly modified the associations between combined TRPS and HRQL (all P > 0.05 for interaction). Similar results were also found for the HRQL at baseline and at 1 and 3 months. Three HRQL trajectory groups were identified: recover (44.9%), poor (44.4%), and deteriorating (10.7%). Deteriorating group was associated with higher incidence of TRPS (all P < 0.05).
Conclusions
TRPS were associated with a decrease of HRQL regardless of healthy behaviors in HCC patients. Therefore, healthy behaviors promotion alone might not substantially increase HRQL associated with TRPS, and other measures tackling TRPS are warranted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hepatocellular carcinoma (HCC) is a widely prevalent primary liver cancer that causes substantial cancer-related morbidity and mortality on a global scale [1]. Hepatectomy represents the preferred therapeutic approach for patients with HCC who satisfy the established criteria for operability and has demonstrated efficacy in prolonging patient survival [2, 3]. Nevertheless, the overall prognosis for HCC patients remains suboptimal owing to the condition’s significant heterogeneity and high recurrence rate [4, 5]. Hence, there is an urgent need to prioritize health-related quality of life (HRQL) in the management of these patients. Measuring HRQL as an endpoint has become a customary practice in clinical trials evaluating the safety and effectiveness of novel chemotherapeutic agents in nearly all cancer types, including HCC [6, 7].
Patients with cancer usually present with a wide range of tumor-related psychiatric symptoms (TRPS), such as sleep disorder, fatigue, anxiety, and depression prior to cancer treatments [8,9,10]. Study has revealed that preexisting TRPS can lead to unfavorable outcomes during and after active treatments, such as poor treatment tolerability (i.e., the extent to which overt adverse effects can be tolerated by the patient [8]), functional decline, and reduced quality of life [11, 12]. Consequently, TRPS, as reported by patients, have increasingly been used as patient-reported outcomes (PROs) to capture symptom burden in cancer patients [13, 14]. Monitoring and managing symptoms using PROs have demonstrated the potential to improve HRQL, decrease emergency department visits, and ensure the continuation of cancer treatments [8, 14]. Despite this progress, the association between TRPS and HRQL in HCC patients after hepatectomy remains unclear. However, considerable interindividual variability in the longitudinal trajectory of PROs, which may be obscured by a description of the average population level [15]. Few studies comprehensively evaluated dynamic PROs after treatment. Some patient subgroups may follow trajectories at high risk of short-term deterioration in functional health and symptom burden.
Most previous studies [16,17,18] examining the association between TRPS and HRQL has primarily utilized a binary classification system (e.g., depressed versus non-depressed) or total scores. This approach assumes that all symptoms have equal weight as severity indicators [16], despite the absence of empirical evidence. Furthermore, traditional statistical models have evaluated the impact of individual TRPS without accounting for their combined effects, interactions with other TRPS, and nonlinear association with the outcome variable.
Early identification of high-risk groups for HRQL deterioration is crucial for timely, patient-specific supportive care interventions, including those facilitating a healthy lifestyle [19]. This study was conducted among HCC patients who received hepatectomy, with the following aims: (1) to describe dynamics of patient-reported HRQL over 6 months after hepatectomy; (2) to identify independent and combined associations between TRPS and patient-reported HRQL; and (3) to focus on how modifiable health behaviors are associated with distinct patterns of HRQL.
Methods
Study design and patients
This prospective cohort study recruited patients who underwent hepatectomy for hepatocellular carcinoma (HCC) at Guangxi Medical University Cancer Hospital in Nanning, China, between September 2021 and May 2022. The inclusive criteria comprise patients diagnosed with HCC through postoperative pathology, who have undergone surgical treatment for the first time in the hospital, aged between 18 and 75 years, with an estimated survival time of at least 12 months. Additionally, patients must be able to complete the questionnaire follow-up independently or under the guidance of the investigator. Conversely, patients who suffer from other types of malignant tumors, severe heart, brain, lung, or kidney diseases, previous mental illness or consciousness disturbances, and those who experience non-illness-related stress factors such as significant family changes during the study period, as well as those who refuse to participate or are lost to follow-up or have died, are excluded from the study.
Prior to commencing the study, the study protocol was reviewed and approved by the Ethics Review Committee of Guangxi Medical University Cancer Hospital, and it was conducted in accordance with the principles of the Declaration of Helsinki. Informed consent in writing was obtained from all patients prior to hepatectomy. Moreover, the study was designed and reported according to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. [20]
Assessments of TRPS
We focuses on the TRPS that arise in patients with tumors, encompassing sleep disorder, fatigue, anxiety, and depression. The selection of these particular symptoms was informed by their high prevalence among HCC patients [21,22,23,24,25,26,27,28], as well as the availability of psychometric scales with established reliability and validity to effectively capture and measure their respective impacts [29,30,31,32]. TRPS data were collected before hepatectomy.
Sleep disorder was determined through the use of the Pittsburgh Sleep Quality Index (PSQI), a widely utilized, internationally recognized questionnaire designed to assess sleep quality based on a set of 19 self-rated items and five external queries [29]. The PSQI score, which ranges from 0 to 21, is a composite of seven subscales that evaluate different aspects of sleep, including sleep quality, sleep latency, sleep duration, sleep efficiency, sleep disturbances, sleep remedies, and daytime fatigue, with each subscale ranging from 0 to 3 points. The instrument has demonstrated high levels of both reliability and validity [29, 33]. The study utilized a PSQI cutoff score of 8 or higher to identify individuals with elevated sleep disorder symptoms [33].
Fatigue was assessed using the Cancer Fatigue Scale (CFS), a 15-item questionnaire that measures fatigue as a construct on a sum scale consisting of three subscales: physical, cognitive, and affective fatigue [30]. The CFS demonstrates robust reliability and validity and captures the multidimensionality of fatigue using a five-point Likert scale, where higher scores indicate greater levels of fatigue (range 0–60) [30]. Participants were classified as having elevated fatigue if their total score on the CFS was 6 or higher [30].
Anxiety and depression were determined through the use of the Hospital Anxiety and Depression Scale (HADS). HADS consists of two subscales: anxiety (HADS-A) and depression (HADS-D). Each score consists of seven questions, ranging from 0 to 3 (each score is 0–21); depending on the probability of the psychological problem described, patients with a score of 8 or higher can be classified as possibly significant symptoms [31].
Medical coping modes determined through the use of the Medical coping modes questionnaire (MCMQ), which was compiled by Feifel et al. [32] and is used to investigate the psychological and behavioral correlates of patients using primary coping strategies in the treatment of illness. MCMQ has 20 items in 3 dimensions, including confrontation (8 items; score is 0–32), avoidance (7 items; score is 0–28), and resignation (5 items; score is 0–20); each item adopts Likert 4 score method [32]. The scores of three dimensions can reflect the tendency of the patient to adopt coping strategies when facing treatment [32].
Outcomes
The primary outcome was the HRQL at 6 months follow-up after hepatectomy. The secondary outcomes were HRQL at baseline (prior to hepatectomy), and at 1 month and 3 months follow-up after hepatectomy. HRQL data were collected prospectively and were determined through the use of the Functional Assessment of Cancer Therapy-Hepatobiliary questionnaire (FACT-Hep). The 45-item FACT-Hep consists of five subscales: physical well-being, social and family well-being, emotional well-being, functional well-being, and the hepatobiliary cancer subscale (Hep-CS). The Hep-CS includes 18 items that assess specific symptoms of hepatobiliary carcinoma and side-effects of its treatment [19]. Aggregate scores can also be formed, from 0 to 180, higher scores on all scales of the FACT-Hep reflect better quality of life or fewer symptoms [19].
Covariates
At baseline, trained interviewers collected information on sociodemographic status and health-related factors using a structured questionnaire, including age, gender, body mass index, educational level, the night shift due to work requirements, occupational status, self-reported economic pressure, caregiver, and siesta (taking a short afternoon nap at least one to two times per week). Healthy behaviors included self-reported smoking status, drinking status (12 episodes of drinking a year), and physical exercise. Clinical characteristics included comorbidities, cirrhosis, surgery, tumor size, tumor number, large vessel invasion, postoperative complications, and BCLC stage. Educational level was classified as primary school or below, middle or high school, and college or above. Occupational status was classified as on the job, laid-off or unemployed, and retirement. Economic pressure was classified as very heavy, heavy, generally, and light. Caregiver was classified as spouse, parent or child, and relatives or friends. Comorbidities was classified into two groups: no and yes (diabetes, hypertension, dyslipidemia, and chronic kidney disease). Body mass index was calculated as weight in kilograms divided by height in meters squared; height and weight were measured by a trained nurse. Surgery and postoperative complications were not baseline characteristics (prior to hepatectomy).
Statistical analyses
In this study, categorical variables were described using frequency and percentage, while continuous variables were described using means and standard deviations (SDs) or medians and interquartile ranges (IQRs). To compare quality of life between groups, we employed the Kruskal–Wallis rank sum test. Additionally, we used the Spearman test to assess the correlation between TPRS and social support.
To examine the independent associations between TRPS and HRQL at baseline, and at 1, 3, and 6 months after hepatectomy, we used generalized linear models (GLMs) to estimate the coefficient (β) with 95% confidence intervals (CIs). To conduct these analyses, the score of each TPRS was split into tertiles and included in GLMs, with the first tertile serving as the reference group. Alternatively, for sleep disorder, fatigue, anxiety, and depression, we coded the scores as dichotomous variables using a cutoff. The models were adjusted for age, gender, body mass index, educational level, night shift, occupational status, economic pressure, caregiver, smoking history, drinking history, siesta, physical exercise, comorbidities, cirrhosis, tumor size, tumor number, large vessel invasion, and BCLC stage. We excluded surgery and postoperative complications at baseline since this information was unavailable. To explore potential nonlinear associations, we also employed a restricted cubic spline (RCS) regression model with four knots at the 5th, 35th, 65th, and 95th percentiles of each TRPS score (with the reference being the 5th percentile).
We employed the weighted quantile sum (WQS) regression model to investigate the combined associations between TRPS and HRQL. This analytical approach is designed to estimate the relative contribution of mixed exposures to health outcomes [34]. Specifically, the WQS model assumes a linear relationship between TRPS and HRQL across quantiles of each exposure and calculates a weighted index that represents the overall burden of these symptoms. The weight index reflects the contribution of each psychiatric symptom to the entire effect and is constrained between 0 and 1, with a sum of 1 [35]. To determine the weights, we used bootstrap resampling methods for 1000 iterations. To identify potential modifying variables of healthy behaviors, stratified analyses were conducted for body mass index, smoking history, drinking history, and physical exercise. We evaluated P-values for interaction using interaction terms and likelihood ratio tests.
We employed latent class mixed-effect models (LCMM) to evaluate longitudinal variations in FACT-Hep summary score [36,37,38]. This analytical approach facilitated the identification of unobserved clusters, or latent trajectory groups, of individuals exhibiting similar patterns of HRQL outcomes over time. Polynomial trajectories were defined through model estimation, with model selection involving the iterative determination of the optimal (1) number of trajectory groups and (2) shape/order of each group, as determined by maximum likelihood methods. To evaluate trajectory groups, we categorized time into monthly intervals. The ideal model should satisfy the following conditions simultaneously: (1) The average posterior probability of each class is greater than 0.7; (2) the proportion of people in a potential category is greater than 5%; (3) entropy > 0.800. Each latent trajectory group was assigned a descriptive label to provide a succinct summary of the associated HRQL outcome pattern. Subsequently, participant characteristics were described for each identified group. Finally, mean scores for all scales included in the FACT-Hep questionnaire were summarized for each trajectory group.
Sensitivity analyses were conducted as follows: (1) non-adjusting for healthy behaviors of body mass index, smoking history, drinking history, and physical exercise, to avoid the impact of potential mediation effects, (2) repeating all analyses using the generalized estimating equation model, and (3) using the g-computation approach [39] to account for the combined associations. All analyses were performed using R statistical software version 4.2.2 (R Foundation), and the R packages “gWQS” and “lcmm” were utilized for combined associations analyses and LCMM analyses, respectively. Two-sided P < 0.05 was considered as statistically significant (Figs 1, 2, 3, 4 and 5).
Nonlinear association between individual tumor-related psychiatric symptoms score and health-related quality of life. Graphs show estimate (β) for quality of life according to anxiety score (A–D), depression (E–H) score, fatigue score (I–L), and sleep disorder score (M–P) at baseline and at 1, 3, and 6 months after hepatectomy. Data were fitted by a restricted cubic spline linear regression model adjusted for age, gender, body mass index, educational level, night shift, occupational status, economic pressure, caregiver, smoking history, drinking history, siesta, physical exercise, comorbidities, cirrhosis, tumor size, tumor number, large vessel invasion, and BCLC stage, but exclude surgery and postoperative complications at baseline, and the models were conducted with 4 knots at the 5th, 35th, 65th, 95th percentiles of individual tumor-related psychiatric symptoms score (reference is the 5th percentile). Solid lines indicate estimate (β), and shadow shapes indicate 95% CIs. Abbreviation: CI, confidence interval
Estimated weights assigned to individual tumor-related psychiatric symptoms with the WQS model. Weights are in a negative direction and obtained when the effect parameter of WQS model was constrained to the negative direction with 1000 repeated holdout validations. WQS model was adjusted for age, gender, body mass index, educational level, night shift, occupational status, economic pressure, caregiver, smoking history, drinking history, siesta, physical exercise, comorbidities, cirrhosis, tumor size, tumor number, large vessel invasion, and BCLC stage, but exclude surgery and postoperative complications at baseline. Abbreviation: WQS, weighted quantile sum
Trajectory groups according to best-fitting model. A Trajectory groups of health-related quality of life measured by the 45-item of FACT-Hep Scale. Mean physical well-being scores (B), mean social well-being scores (C), mean emotional well-being scores (D), mean functional well-being scores (E), and mean hepatobiliary cancer scores (F) by trajectory group and by time point. Respective SDs for the means are available in the eTable 4. Solid lines represent the predicted trajectories, dashed lines represent the mean scores at baseline. Abbreviation: FACT-Hep, Functional Assessment of Cancer Therapy-Hepatobiliary
Results
Cohort characteristics
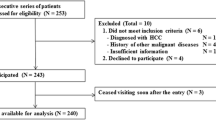
A total of 226 patients were initially recruited for the study. After excluding 13 non-HCC patients, 6 who died, 1 who refused evaluation, and 1 who was lost to follow-up, a final sample of 205 patients was included for analysis, with follow-up assessments conducted at 1, 3, and 6 months (Fig. 6). The mean (SD) age of the included patients at baseline was 52.8 (10.9) years, with an average body mass index of (23.2 ± 2.9) kg/m2. Of these patients, 174 (84.9%) were male. Table 1 presents the demographic and clinical characteristics of the study patients.
Association between combined tumor-related psychiatric symptoms and health-related quality of life stratified by different health behaviors. Graphs show estimate (β) and 95% CIs for the associations between negative WQS index and health-related quality of life at baseline (A), at 1 month (B), at 3 months (C), and at 6 months (D) adjusted for age, gender, body mass index, educational level, night shift, occupational status, economic pressure, caregiver, smoking history, drinking history, siesta, physical exercise, comorbidities, cirrhosis, tumor size, tumor number, large vessel invasion, and BCLC stage, but exclude surgery and postoperative complications at baseline. Estimates were interpreted as the effect of increasing the combined tumor-related psychiatric symptoms by one quantile on the health-related quality of life. Abbreviation: WQS, weighted quantile sum
Independent association between TRPS and HRQL
At baseline, the prevalence of anxiety, depression, fatigue, and sleep disorders were found to be 44.9% (92/205), 40.5% (83/205), 56.1% (115/205), and 56.1% (115/205), respectively. The detailed scores for all scales can be found in e Table 1. Additionally, the quality of life at baseline and postoperative follow-ups of 1, 3, and 6 months were assessed, and the mean (SD) scores were reported as 135.34 ± 17.96 points, 106.92 ± 17.85 points, 119.40 ± 20.88 points, and 129.65 ± 24.68 points, respectively. The detailed scores for all scales can be found in e Table 2.
Table 2 presents the association of individual TRPS and HRQL at baseline and postoperative follow-ups of 1, 3, and 6 months. For the primary outcome of HRQL at 6 months, after adjusting for potential confounders, the presence of symptoms related to anxiety was found to be significantly associated with a decrease of 11.85 points (β = − 11.85, 95% CI, − 18.11 to − 5.58; P < 0.001) on HRQL when compared to patients without such symptoms. Additionally, the presence of symptoms related to depression, fatigue and sleep disorder were also found to be significantly associated with a decrease in HRQL of 14.53 points (β = − 14.53, 95% CI, − 20.58 to –8.47; P < 0.001), 13.61 points (β = − 13.61, 95% CI, − 19.91 to − 7.32; P < 0.001), and 12.14 points (β = − 12.14, 95% CI, − 18.49 to − 5.79; P < 0.001), respectively, when compared to individuals without these symptoms. Similar results were found when modeling individual TRPS score as tertiles, by comparing tertile 3 with tertile 1, the adjusted β were − 13.26 (95% CI, − 21.14 to − 5.38) for anxiety, − 14.83 (95% CI, − 23.35 to − 6.32) for depression, − 13.38 (95% CI, − 21.01 to − 5.76) for fatigue, − 13.68 (95% CI, − 21.93 to − 5.43) for sleep disorder. Similar results were also found for the HRQL at baseline and at 1 and 3 months.
Figure 1 shows the nonlinear association of individual TRPS and HRQL using RCS regression model. A negative and linear association between depression score, fatigue score, and sleep disorder score with HRQL were found at all time points (for nonlinearity, all P > 0.05, except for depression and fatigue at baseline). Notably, anxiety score demonstrates an inverse S-shaped association with HRQL at all time points (for nonlinearity, all P < 0.05).
Combined associations between TRPS and HRQL
Table 2 presents the combined effect of TRPS with HRQL, and WQS negative index represent the negative combined effects of TRPS. After adjusting for potential confounders, the WQS negative index was negatively associated with HRQL at all time points (at baseline, β = − 14.69, 95% CI, − 17.27 to − 12.11; at 1 month, β = − 4.55, 95% CI, − 8.16 to − 0.94; at 3 months, β = − 4.88, 95% CI, − 8.67 to − 1.09; at 6 months, β = − 5.07, 95% CI, − 10.01 to − 0.13). Figure 2 exhibits the weights allocated to individual components comprising the amalgamated impact of TRPS in the WQS regression model. Notably, depression emerged as the predominant contributor in the negative direction at all time points (39% at baseline, 57% at 1 month, 59% at 3 months, 49% at 6 months)..
Health behaviors and combined effect
The distribution of health behaviors, as presented in Table 1. Specifically, the proportion of patients with a body mass index less than 23 was found to be 47.8%, while 61.0% of patients reported being non-smokers. Additionally, 72.2% of patients reported being non-drinkers, and 49.8% reported engaging in physical exercise. However, after conducting statistical analyses, we did not find evidence to suggest that the health behaviors of body mass index, smoking, drinking, or physical exercise significantly modified the associations between combined TRPS and HRQL at all time points (all P > 0.05 for interaction; Fig. 3).
HRQL trajectory groups and TRPS
The final model generated by our study delineated three trajectory groups (Fig. 4A). Model selection metrics are available in the Table 3. The predominant trajectory group, which comprised a considerable proportion of patients (n = 92; 44.9%), exhibited high scores of FACT-Hep at baseline, with some additional negative inflections, and subsequently experienced slow and partial recovery until month 6 (recover group). The second trajectory group (n = 91; 44.4%), with overall low scores at baseline, demonstrated similar negative inflections and slowly and partially recovered, yet remained below the average score at month 6 (poor group). The third trajectory group (n = 22; 10.7%), displaying very low baseline scores, had a persistent decline in their HRQL throughout the study duration (deteriorating group). The poor trajectory group and deteriorating trajectory group reported mean values below average level at baseline across multiple HRQL domains (Fig. 4A–F).
Table 1 shows patient characteristics by trajectory group. Patients in deteriorating group were more likely to have a lower body mass index, have primary school education, have a laid-off or unemployed occupational status, have a very heavy economic pressure, have a larger tumor size, have postoperative complications, have no siesta, have large vessel invasion, and have C stage of BCLC (all P < 0.05).
Figure 5 shows the distribution of TRPS by HRQL trajectory group. Among these groups, the highest prevalence rates of anxiety, depression, fatigue, and sleep disorders were observed in the deteriorating group, with rates of 81.8%, 77.3%, 90.9%, and 81.8%, respectively. The poor group also exhibited notable prevalence rates, which were 62.6%, 61.5%, 72.5%, and 76.9% for anxiety, depression, fatigue, and sleep disorders, respectively. Scores for all scales by HRQL trajectory group are shown in e Table 3.
Sensitivity analyses
The results of sensitivity analyses were consistent with main findings when unadjusted for healthy behaviors (e Table 4) or using the generalized estimating equation model (e Table 5). Particularly, the combined associations were also consistent with the WQS model when using the quantile G-computation approach (e Tables 6, 7).
Discussion
In this prospective study, we found that TRPS, including anxiety, depression, fatigue, and sleep disorders, were independently associated with a decrease in HRQL, and a negative combined effect of TRPS was also found by the WQS model, with depression being the predominant contributor. However, health behaviors such as body mass index, smoking, drinking, or physical exercise did not significantly modify the associations between TRPS and HRQL. Using a latent-class analysis, we identified three different trajectories among HCC patients receiving hepatectomy characterized by recover (44.9%), poor (44.4%), and deteriorating (10.7%) patient-reported HRQL patterns, with the deteriorating group being associated with a higher incidence of TRPS. In addition, body mass index, educational level, occupational status, economic pressure, siesta, tumor size, large vessel invasion, postoperative complications, and BCLC stage may be the trajectory group membership.
HCC is a type of liver cancer that can have a significant impact on patients’ physical and psychological well-being [40]. Anxiety, depression, fatigue, and sleep disorders are common among HCC patients and can further exacerbate the burden of the disease [18, 41,42,43]. Studies show that up to 24.1 ~ 66.7% of HCC patients experience depression [21,22,23,24,25], while up to 24.3 ~ 51.8% experience anxiety [21, 22, 24, 25]. Anxiety and depression can be caused by a variety of factors, including the diagnosis of cancer, the fear of recurrence, and the impact of the disease on daily life [26]. Fatigue is also common among HCC patients; it can be caused by various factors such as treatment side effects, pain, and sleep disturbances [27, 28, 44]. The severity of fatigue can range from mild to severe, and it can have a significant impact on a patient’s daily activities and overall quality of life [28]. Sleep disorders are also prevalent in HCC patients, including difficulty falling asleep, staying asleep, and waking up too early. These sleep disorders can result in fatigue, reduced concentration, and decreased cognitive function, which can negatively affect patients’ daily activities and overall quality of life [45,46,47]. In this study, the incidence of anxiety, depression, fatigue, and sleep disturbance found to be 44.9% (92/205), 40.5% (83/205), 56.1% (115/205), and 56.1% (115/205), respectively, which were consistent with the previous research results. Moreover, a strong association between these symptoms and inferior quality of life has been demonstrated.
Considering the effects of various TRPS on health outcomes, the potential nonlinear associations and the simultaneous exposure to multiple TRPS are more consistent with real-world situations. The combined impact of these factors must therefore be considered. We also utilized RCS regression and WQS regression for the analyses. The RCS regression show that there was a negative and linear association between depression score, fatigue score, and sleep disorder score with HRQL. Notably, there was an inverse S-shaped association of anxiety with HRQL; these seem not to simply explain that it is beneficial when anxiety are at the lowest and highest scores, although we do not yet know the mechanism of this nonlinear association. The WQS models, based on a combination of covariates and interactions among TRPS, are capable of both estimating the overall effect of multiple TRPS on HRQL, as well as identifying the TRPS in mixed exposures that have a significant impact on outcomes. According to the results of this study, the WQS indices of TRPS were negatively associated with HRQL, which was consistent with our results in the GLMs analysis and supports the robustness of the analyses to some extent. However, the WQS regression model has a drawback in that the cumulative effect of TRPS with diverse modes of action cannot be concurrently examined. Notably, if TRPS have a negative association with HRQL in the WQS regression model, the model deems them to be positively associated and provides them inconsequential weights in the WQS weights [34, 35].
Engaging in healthy behaviors has been associated with improved HRQL in patients with HCC [48, 49]. However, one major finding of this study was that healthy behaviors were not significant modifiers of the associations between TRPS and HRQL. A healthy behaviors, which includes regular exercise, a healthy diet, and avoiding smoking and excessive alcohol consumption, among other things, can help people maintain their physical and mental health [50]. However, when individuals suffer from anxiety, depression, fatigue, and sleep disorders, it can be challenging for them to maintain a healthy behavior. One reason for this is that anxiety, depression, and fatigue can all have a negative impact on individuals’ mental and emotional states, making them more prone to unhealthy behaviors [51]. For instance, individuals experiencing these issues may be more likely to engage in emotional eating, and avoid exercise. Additionally, anxiety, depression, and fatigue can all cause individuals to lack the motivation and energy necessary to establish and maintain a healthy lifestyle [51]. Sleep disorders, on the other hand, can have a direct impact on an individual’s ability to engage in healthy behaviors [52, 53]. Individuals experiencing sleep disturbances, such as insomnia or sleep apnea, may find it challenging to establish a regular exercise routine, prepare healthy meals, or maintain a consistent sleep schedule. In summary, anxiety, depression, fatigue, and sleep disorders can all make it difficult for individuals to maintain a healthy behavior. To address these issues, it is crucial to develop an individualized plan that incorporates mental health interventions, such as therapy and medication, and lifestyle changes, such as regular exercise, healthy eating, and sleep hygiene practices, to support individuals in achieving and maintaining a healthy lifestyle.
Previous literature tried to describe posthepatectomy changes in HRQL; however, most of it only captured population averages or was focused on specific symptoms [54,55,56,57]. Here, we present a number of novel findings regarding HRQL trajectories in the 6 months following hepatectomy. We provide a detailed assessment of overall and specific hepatectomy metrics, offering insight into the characteristics of subpopulations that experience persistent deterioration. Although the majority of patients exhibit a return to preoperative HRQL levels by the 6-month mark, we identified two specific patient clusters for whom short-term HRQL dynamics were notably worse. The group experiencing deteriorating HRQL was most affected by primary treatment, with a significant drop observed from baseline to month 3 that did not recover. This segment exhibited sharp deterioration across multiple functions, including physical, social and family, emotional, functional, and the hepatobiliary cancer subscale. In nearly all cases, median scores for the deteriorating trajectory group were lower than the baseline median level, indicating a need for clinician attention and dedicated supportive care.
Our study additionally revealed that the deteriorating HRQL group had a higher incidence of anxiety, depression, fatigue, and sleep disorders. One possible explanation for this observation is that chronic stress may contribute to immune decline [58,59,60]. Chronic stress is characterized by a persistent non-specific adaptive response of the body, resulting from the activation of the hypothalamic–pituitary–adrenal axis and sympathetic nervous system in response to physiological and psychological stressors, and is typically manifested as anxiety and depression. Chronic stress can impact the immune system, as prolonged anxiety and depression can negatively affect immune function, leading to a highly immunosuppressed tumor microenvironment (TME). For cancer patients, treatment confidence, treatment side effects, and financial pressures can all contribute to long-term psychological stress, further exacerbating immune suppression [61].
This study has several strengths, including its prospective design and the construction of a combined TRPS variable to comprehensively evaluate the complex relations between healthy behaviors and TRPS with dynamic HRQL. Sensitivity analyses were also conducted to demonstrate the robustness of the findings. However, there are also limitations that must be acknowledged. First, the measurements of TRPS and healthy behaviors were mainly self-reported and were only taken once, resulting in the possibility of measurement errors. Additionally, the long-term trajectories of HRQL and changes in healthy behaviors could not be captured. Secondly, disease status could have influenced both healthy behaviors and TRPS, and while adjustments were made for comorbidities at baseline, the possibility of reverse causation and residual confounding cannot be entirely eliminated. Third, only patients from China were involved in this study; thus, the findings may not fully generalize to other countries. Fourth, we fitted the latent class model with a smaller sample size. Although our sample size may be relatively modest for latent class modeling, but our model evaluation metrics support the suitability of the fitted latent trajectory model, we also conducted several assessments to ensure the robustness of our findings. Fifth, due to the small sample size of the deteriorating group, a multivariate model could not be utilized to analyze the association between TRPS and health behaviors with HRQL trajectories; thus, future studies with multivariable multinomial logistic regression are preferred. Finally, despite controlling for key personal characteristics and comorbidities, residual confounding remains possible, and causal inference cannot be made due to the nature of observational studies.
Conclusions
Based on this cohorts, TRPS were found to be significantly associated with a decrease of dynamic HRQL, with this association partially mediated by healthy behavior factors. These findings highlight the critical importance of personalized interventions, particularly given the absence of standardized lifestyle-change programs and inconsistent third-party reimbursement of behavioral interventions for patients with cancer [62,63,64]. To optimize resource allocation, researchers are increasingly exploring the efficacy of targeted lifestyle-promotion interventions and individualized patient support strategies [63, 64]. The current study provides valuable insights into the identification of patient-level risk factors and the development of tailored interventions for the preservation of HRQL, including the early identification of behavioral concerns and the provision of healthy lifestyle support programs.
Data availability
Some or all data generated or used during the study are available from the corresponding author by request.
References
Siegel RL, Miller KD, Wagle NS, Jemal A (2023) Cancer statistics, 2023. CA Cancer J Clin 73(1):17–48
Cucchetti A, Zhong J, Berhane S et al (2020) The chances of hepatic resection curing hepatocellular carcinoma. J Hepatol 72(4):711–717
Yuan BH, Zhu YK, Zou XM, Zhou HD, Li RH, Zhong JH (2022) Repeat hepatic resection versus percutaneous ablation for the treatment of recurrent hepatocellular carcinoma: meta-analysis. BJS Open 6(2):1–8
Omata M, Cheng AL, Kokudo N et al (2017) Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: a 2017 update. Hepatol Int 11(4):317–370
Zhong JH, Ke Y, Wang YY, Li LQ (2015) Liver resection for patients with hepatocellular carcinoma and macrovascular invasion, multiple tumours, or portal hypertension. Gut 64(3):520–521
Kim J, Singh H, Ayalew K et al (2018) Use of PRO Measures to inform tolerability in oncology trials: implications for clinical review, IND safety reporting, and clinical site inspections. Clin Cancer Res 24(8):1780–1784
Steel JL, Eton DT, Cella D, Olek MC, Carr BI (2006) Clinically meaningful changes in health-related quality of life in patients diagnosed with hepatobiliary carcinoma. Ann Oncol 17(2):304–312
Flannery MA, Culakova E, Canin BE, Peppone L, Ramsdale E, Mohile SG (2021) Understanding treatment tolerability in older adults with cancer. J Clin Oncol 39(19):2150–2163
Deshields TL, Potter P, Olsen S, Liu J (2014) The persistence of symptom burden: symptom experience and quality of life of cancer patients across one year. Support Care Cancer 22(4):1089–1096
Drageset J, Corbett A, Selbaek G, Husebo BS (2014) Cancer-related pain and symptoms among nursing home residents: a systematic review. J Pain Symptom Manag 48(4):699-710 e691
Omran S, McMillan S (2018) Symptom severity, anxiety, depression, self- efficacy and quality of life in patients with cancer. Asian Pac J Cancer Prev 19(2):365–374
Lage DE, El-Jawahri A, Fuh CX et al (2020) Functional impairment, symptom burden, and clinical outcomes among hospitalized patients with advanced cancer. J Natl Compr Canc Netw 18(6):747–754
Whisenant MS, Bamidele O, Cleeland C, Williams LA (2021) Preferences of individuals with cancer for patient-reported outcome measures. Oncol Nurs Forum 48(2):173–183
Basch E, Deal AM, Kris MG et al (2016) Symptom monitoring with patient-reported outcomes during routine cancer treatment: a randomized controlled trial. J Clin Oncol 34(6):557–565
Vitale A, Svegliati-Baroni G, Ortolani A et al (2023) Epidemiological trends and trajectories of MAFLD-associated hepatocellular carcinoma 2002–2033: the ITA.LI.CA database. Gut 72(1):141–152
Kim EH, Park JH, Lee SM, Gwak MS, Kim GS, Kim MH (2016) Preoperative depressed mood and perioperative heart rate variability in patients with hepatic cancer. J Clin Anesth 35:332–338
Fan SY, Eiser C, Ho MC (2010) Health-related quality of life in patients with hepatocellular carcinoma: a systematic review. Clin Gastroenterol Hepatol 8(7):559–564
Tan DJH, Quek SXZ, Yong JN et al (2022) Global prevalence of depression and anxiety in patients with hepatocellular carcinoma: systematic review and meta-analysis. Clin Mol Hepatol 28(4):864–875
Heffernan N, Cella D, Webster K et al (2002) Measuring health-related quality of life in patients with hepatobiliary cancers: the functional assessment of cancer therapy-hepatobiliary questionnaire. J Clin Oncol 20(9):2229–2239
von Elm E, Altman DG, Egger M et al (2007) The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: guidelines for reporting observational studies. Epidemiology 18(6):800–804
Llovet JM, Kelley RK, Villanueva A et al (2021) Hepatocellular carcinoma. Nat Rev Dis Primers 7(1):6
Tan DJH, Ng CH, Lin SY et al (2022) Clinical characteristics, surveillance, treatment allocation, and outcomes of non-alcoholic fatty liver disease-related hepatocellular carcinoma: a systematic review and meta-analysis. Lancet Oncol 23(4):521–530
Wolf E, Rich NE, Marrero JA, Parikh ND, Singal AG (2021) Use of hepatocellular carcinoma surveillance in patients with cirrhosis: a systematic review and meta-analysis. Hepatology 73(2):713–725
Huang DQ, Fowler KJ, Liau J et al (2022) Comparative efficacy of an optimal exam between ultrasound versus abbreviated MRI for HCC screening in NAFLD cirrhosis: a prospective study. Aliment Pharmacol Ther 55(7):820–827
Lee KT, Lin JJ, Shi HY (2018) Anxiety and depression are associated with long-term outcomes of hepatocellular carcinoma: a nationwide study of a cohort from Taiwan. World J Biol Psychiatry 19(6):431–439
Verma M, Paik JM, Younossi I, Tan D, Abdelaal H, Younossi ZM (2021) The impact of hepatocellular carcinoma diagnosis on patients’ health-related quality of life. Cancer Med 10(18):6273–6281
Lai YH, Shun SC, Hsiao YL et al (2007) Fatigue experiences in hepatocellular carcinoma patients during six weeks of stereotactic radiotherapy. Oncologist 12(2):221–230
Sun V, Ferrell B, Juarez G, Wagman LD, Yen Y, Chung V (2008) Symptom concerns and quality of life in hepatobiliary cancers. Oncol Nurs Forum 35(3):E45-52
Buysse DJ, Reynolds CF 3rd, Monk TH, Berman SR, Kupfer DJ (1989) The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res 28(2):193–213
Okuyama T, Akechi T, Kugaya A et al (2000) Development and validation of the cancer fatigue scale: a brief, three-dimensional, self-rating scale for assessment of fatigue in cancer patients. J Pain Symptom Manage 19(1):5–14
Zigmond AS, Snaith RP (1983) The hospital anxiety and depression scale. Acta Psychiatr Scand 67(6):361–370
Feifel H, Strack S, Nagy VT (1987) Coping strategies and associated features of medically ill patients. Psychosom Med 49(6):616–625
Tzeng JI, Fu YW, Lin CC (2012) Validity and reliability of the Taiwanese version of the Pittsburgh Sleep Quality Index in cancer patients. Int J Nurs Stud 49(1):102–108
Tanner EM, Bornehag CG, Gennings C (2019) Repeated holdout validation for weighted quantile sum regression. MethodsX 6:2855–2860
Carrico C (2021) gWQS: An R package for linear and generalized weighted quantile sum (WQS) regression [EB/OL]. R package version 2.3.2. https://CRAN.R-project.org/package=gWQS
Proust-Lima C, Philipps V, Liquet B (2017) lcmm: An R package for estimation of latent class mixed models and joint latent class models [EB/OL]. R package version 1.9.5. https://CRAN.R-project.org/package=lcmm
Ahn J, Liu S, Wang W, Yuan Y (2013) Bayesian latent-class mixed-effect hybrid models for dyadic longitudinal data with non-ignorable dropouts. Biometrics 69(4):914–924
Lennon H, Kelly S, Sperrin M et al (2018) Framework to construct and interpret latent class trajectory modelling. BMJ Open 8(7):e020683
Keil AP, Buckley JP, O’Brien KM, Ferguson KK, Zhao S, White AJ (2020) A quantile-based g-computation approach to addressing the effects of exposure mixtures. Environ Health Perspect 128(4):47004
Villanueva A (2019) Hepatocellular carcinoma. N Engl J Med 380(15):1450–1462
Sun VC, Sarna L (2008) Symptom management in hepatocellular carcinoma. Clin J Oncol Nurs 12(5):759–766
Poort H, Jacobs JM, Pirl WF, Temel JS, Greer JA (2020) Fatigue in patients on oral targeted or chemotherapy for cancer and associations with anxiety, depression, and quality of life. Palliat Support Care 18(2):141–147
Graf J, Stengel A (2021) Psychological burden and psycho-oncological interventions for patients with hepatobiliary cancers-a systematic review. Front Psychol 12:662777
Shun SC, Lai YH, Jing TT et al (2005) Fatigue patterns and correlates in male liver cancer patients receiving transcatheter hepatic arterial chemoembolization. Support Care Cancer 13(5):311–317
Huang J, Song P, Hang K et al (2021) Sleep deprivation disturbs immune surveillance and promotes the progression of hepatocellular carcinoma. Front Immunol 12:727959
Peng JK, Hepgul N, Higginson IJ, Gao W (2019) Symptom prevalence and quality of life of patients with end-stage liver disease: a systematic review and meta-analysis. Palliat Med 33(1):24–36
Cao W, Li J, Hu C et al (2013) Symptom clusters and symptom interference of HCC patients undergoing TACE: a cross-sectional study in China. Support Care Cancer 21(2):475–483
Saran U, Humar B, Kolly P, Dufour JF (2016) Hepatocellular carcinoma and lifestyles. J Hepatol 64(1):203–214
Yang JD, Hainaut P, Gores GJ, Amadou A, Plymoth A, Roberts LR (2019) A global view of hepatocellular carcinoma: trends, risk, prevention and management. Nat Rev Gastroenterol Hepatol 16(10):589–604
Li Y, Schoufour J, Wang DD et al (2020) Healthy lifestyle and life expectancy free of cancer, cardiovascular disease, and type 2 diabetes: prospective cohort study. BMJ 368:l6669
Bortolato B, Hyphantis TN, Valpione S et al (2017) Depression in cancer: the many biobehavioral pathways driving tumor progression. Cancer Treat Rev 52:58–70
St-Onge MP, Grandner MA, Brown D et al (2016) Sleep duration and quality: impact on lifestyle behaviors and cardiometabolic health: a scientific statement from the American Heart Association. Circulation 134(18):e367–e386
Kang D, Shim S, Cho J, Lim HK (2020) Systematic review of studies assessing the health-related quality of life of hepatocellular carcinoma patients from 2009 to 2018. Korean J Radiol 21(6):633–646
Chiu CC, Lee KT, Wang JJ, Sun DP, Lee HH, Shi HY (2018) Health-related quality of life before and after surgical resection of hepatocellular carcinoma: a prospective study. Asian Pac J Cancer Prev 19(1):65–72
Lei JY, Yan LN, Wang WT, Zhu JQ, Li DJ (2016) Health-related quality of life and psychological distress in patients with early-stage hepatocellular carcinoma after hepatic resection or transplantation. Transplant Proc 48(6):2107–2111
Feldbrugge L, Langenscheidt A, Krenzien F et al (2021) Health-related quality of life and mental health after surgical treatment of hepatocellular carcinoma in the era of minimal-invasive surgery: resection versus transplantation. Healthcare (Basel) 9(6):1-11
He Q, Jiang JJ, Jiang YX, Wang WT, Yang L, Liver SG (2018) Health-related quality of life comparisons after radical therapy for early-stage hepatocellular carcinoma. Transplant Proc 50(5):1470–1474
Vere CC, Streba CT, Streba LM, Ionescu AG, Sima F (2009) Psychosocial stress and liver disease status. World J Gastroenterol: WJG 15(24):2980
Steel J, Carney M, Carr B, Baum A (2004) The role of psychosocial factors in the progression of hepatocellular carcinoma. Med Hypotheses 62(1):86–94
Rossetto A, De Re V, Steffan A et al (2019) Carcinogenesis and metastasis in liver: cell physiological basis. Cancers 11(11):1731
Antoni MH, Moreno PI, Penedo FJ (2023) Stress management interventions to facilitate psychological and physiological adaptation and optimal health outcomes in cancer patients and survivors. Annu Rev Psychol 74:423–455
Demark-Wahnefried W, Schmitz KH, Alfano CM et al (2018) Weight management and physical activity throughout the cancer care continuum. CA Cancer J Clin 68(1):64–89
Iyengar NM, Jones LW (2019) Development of exercise as interception therapy for cancer: a review. JAMA Oncol 5(11):1620–1627
Land SR, Toll BA, Moinpour CM et al (2016) Research priorities, measures, and recommendations for assessment of tobacco use in clinical cancer research. Clin Cancer Res 22(8):1907–1913
Funding
This work was supported by the National Natural Science Foundation of China (81960308).
Author information
Authors and Affiliations
Contributions
X.-M. Y conceived the study; all authors participated in the acquisition of the data; F.-R. L and F.-J. Z performed follow-up the data; R.-R.H, F.-J. Z and F.-R. L analyzed data; R.-R.H is a statistician and he checked the statistics. F.-J. Z and X.-M. Y drafted and revised the manuscript; all authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhao, FJ., Huo, RR., Li, FR. et al. Associations of tumor-related psychiatric symptoms and healthy behaviors with dynamic quality of life after hepatocellular carcinoma hepatectomy. Support Care Cancer 32, 589 (2024). https://doi.org/10.1007/s00520-024-08790-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00520-024-08790-y