Abstract
Purpose
Lung cancer treatment can lead to negative health consequences. We analyzed the effects of curative-intent lung cancer treatment on functional exercise capacity (EC) and patient-reported outcomes (PROs).
Methods
We performed a prospective, observational cohort study of consecutive patients with stage I–IIIA lung cancer undergoing curative-intent therapy and assessed functional EC (primary outcome, six-minute walk distance (6MWD)), cancer-specific quality of life (QoL) (secondary outcome, European Organization for Research and Treatment of Cancer QoL Questionnaire Core 30 (EORTC-QLQ-C30) summary score), and exploratory outcomes including dyspnea (University of California San Diego Shortness of Breath Questionnaire (UCSD SOBQ)) and fatigue Brief Fatigue Inventory (BFI)) symptoms before and at 1 to 3 months post-treatment. We analyzed the time effect of treatment on outcomes using multivariable generalized estimating equations.
Results
In 35 enrolled participants, treatment was associated with a clinically meaningful and borderline-significant decline in functional EC ((mean change, 95% CI) 6MWD = − 25.4 m (− 55.3, + 4.47), p = 0.10), clinically meaningful and statistically significant higher dyspnea (UCSD SOBQ = + 13.1 (+ 5.7, + 20.6), p = 0.001) and fatigue (BFI = + 10.0 (+ 2.9, + 17.0), p = 0.006), but no clinically meaningful or statistically significant change in cancer-specific QoL (EORTC-QLQ-C30 summary score = − 3.4 (− 9.8, + 3.0), p = 0.30).
Conclusions
Among the first prospective analysis of the effect of curative-intent lung cancer treatment on functional EC and PROs, we observed worsening dyspnea and fatigue, and possibly a decline in functional EC but not cancer-specific QoL at 1 to 3 months post-treatment. Interventions to reduce treatment-related morbidities and improve lung cancer survivorship may need to focus on reducing dyspnea, fatigue, and/or improving functional EC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Approximately 35% of patients with non-small cell lung cancer (NSCLC) are diagnosed with stage I–IIIA disease [1, 2] and eligible to undergo curative-intent therapy through a combination of lung cancer resection surgery, definitive radiation, or concurrent chemoradiation. The number of earlier stage lung cancer is expected to increase [3] given the findings of the US National Lung Screening Trial [4], and many professional societies [5,6,7,8,9] endorsing lung cancer screening with low-dose computed tomography in high-risk individuals. Immediately following curative-intent therapy, lung cancer patients are at risk for worsening health due to the toxicities and side effects of treatment. Depending on the extent of resection, a loss of 10–15% of lung function is expected at 3–6 months following lung cancer resection surgery and may persist at 1 year [10]. In addition, perioperative pulmonary [11] and cardiopulmonary [12] complications occur in 15% and 35% of patients, respectively, and can lead to negative health consequences beyond the perioperative period. In those undergoing definitive radiation, 5–15% will develop radiation pneumonitis [13] and worsening respiratory symptoms. Patients undergoing chemotherapy including adjuvant therapy can experience neutropenia, cardiac ischemia, heart failure (HF), neuropathy, and worsening fatigue. Also, lung cancer patients have major comorbidities including chronic obstructive pulmonary disease (COPD, present in approximately 50% of patients) due to tobacco exposure and heart failure (approximately 13%) [14], the health effects of which can be exacerbated by lung cancer treatment.
The identification and quantification of peri-treatment changes in health may identify important decrements which can be prevented and/or minimized to improve lung cancer morbidity and mortality. In addition, peri-treatment efforts to optimize cardiopulmonary function and reduce symptom burden may improve lung cancer survivors’ quality of life (QoL) and survival. In this project, we assessed the changes in health as reflected by functional exercise testing and patient-reported outcomes (PROs). We hypothesized that curative-intent therapy of stage I–IIIA lung cancer is associated with decrements in health as reflected by functional exercise capacity (EC) and cancer-specific QoL.
Methods
Study overview
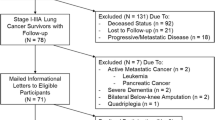
We performed a prospective, observational cohort study of patients undergoing curative-intent therapy for lung cancer. We identified eligible patients from a weekly list of consecutive cases presented at the VA San Diego Healthcare System (VASDHS) chest tumor board (CTB). We included adult lung cancer patients with clinical stage I–IIIA disease who are recommended by the CTB to undergo lung cancer resection surgery, definitive radio-ablation, or concurrent chemoradiation as the primary mode of the treatment. We excluded patients undergoing concurrent systemic therapy for other cancers or those physically unable to perform functional EC evaluation (e.g., quadriplegia or amputees) (Fig. 1).
Between August 2016 and March 2018, we mailed informational letters to eligible patients after CTB management plans were communicated to patients and followed up with a telephone call approximately 1 week later to gauge their interest. Patients who were interested and willing to participate in our study were scheduled in-person visits, during which all were provided written informed consent prior to study procedures. Outcome assessments were performed between August 2016 and May 2018, both before and at 1 to 3 months after completion of therapy. We followed the STROBE guideline recommendations to report our findings [15]. The VASDHS Institutional Review Board approved this protocol (#H150158).
Covariates
We collected baseline clinical characteristics and potential confounders important in lung cancer and cardiopulmonary health and QoL, including age, sex, body mass index, tobacco exposure, comorbidities (including COPD, HF, and psychiatric illness), lung function, and echocardiographic findings where available; lung cancer–related information included histologic subtype, clinical stage, and primary treatment modality. All covariates were abstracted from the electronic health records and verified by a board-certified physician with subspecialty training in pulmonology (DH).
Functional EC and PRO assessments
Our primary endpoint was functional EC as assessed by the six-minute walk test (6MWT) distance (6MWD). We chose the 6MWT [16] based on practical considerations of availability, ease of performance for testing, and the likelihood that daily activities of living are performed at submaximal exercise intensity. In lung cancer survivors, the 6MWD has concurrent validity against cardiopulmonary fitness [17], discriminant validity compared to age-, sex-, height-, and weight-matched adults [18], predictive validity for cancer-specific QoL [18], responsiveness to treatment [19], and a defined minimal clinically important difference (MCID) for interpretation (22–42 m, or a change of 9.5%) [19]. We performed the 6MWT according to the standard protocol at the VASDHS which follows the American Thoracic Society recommendations [20].
Our secondary endpoint was a validated composite score of cancer-specific QoL, assessed by the European Organization for Research and Treatment of Cancer QoL Questionnaire Core 30 (EORTC-QLQ-C30) summary score [21]. We chose the EORTC-QLQ-C30 [22] based on availability, inclusion of core domains of QoL and other subdomains relevant to lung cancer (e.g., dyspnea, fatigue, pain), and a validated summary score [21] to avoid multiple testing of individual health domains. We also performed exploratory PRO assessments for lung cancer-specific symptoms, generic health, sleep quality, dyspnea, fatigue, and anxiety/depression using the EORTC-QLQ-Lung Cancer Module 13 (LC13) [23], EuroQoL-5 Dimensions/visual analogue scale (EQ-5D/VAS) [24], Pittsburgh Sleep Quality Index (PSQI) [25], University California San Diego Shortness of Breath Questionnaire (UCSD SOBQ) [26], Brief Fatigue Inventory (BFI) [27], and Hospital Anxiety and Depression Scale (HADS) [28] questionnaires, respectively. We used separate PRO questionnaires and not the EORTC subscales to assess dyspnea and fatigue because these two symptoms are assessed by only one and three questions, respectively, on the EORTC-QLQ-C30 [22], and three and zero questions, respectively, on the LC13 [23]. We administered all questionnaires in-person when possible and on printed forms without modifications; all questionnaires were scored as per their respective instruction manuals.
We interpreted results using the MCIDs (for the respective questionnaires) where available, 0.06 points (EQ-5D US-index [29]), 7 points (EQ-5D VAS [29]), 3 points (PSQI [30]) 5 points (UCSD SOBQ [31]), 7 points (BFI [27]), and 1.5 points (HADS subscales [32]). Since the MCID for the EORTC-QLQ-C30 summary score is not yet established, we calculated a range of MCID using 0.2 to 0.5 standard deviations of the mean [33] using the data from a previous study [18] of lung cancer survivors following curative intent therapy, 3.6 to 9.0 points.
Sample size
We calculated sample sizes assuming a significance level of p < 0.05 in two-tailed tests, and 80% power to detect a difference in outcomes. For our primary endpoint, we calculated that a sample size of 29 participants will be needed, using a MCID in the 6MWD of 40 m [34] and standard deviation (SD) of 74 m as reported by previous literature [19]. For our secondary endpoint, we calculated a sample size of 30 participants based on a decline of 9 EORTC-QLQ-C30 summary score points following surgical lung cancer treatment [35] and SD of 17 as reported by previous literature [21]. These calculations were performed using PS Power and Sample Size Calculations software, version 3.0.
Statistical analyses
We summarized descriptive statistics as appropriate. All outcomes were recorded and analyzed as continuous variables. To examine the distribution of outcome variables, we visually inspected all histograms and used skewness and kurtosis distribution statistics of ± 2 to define normal distribution [36]. We interpreted the 6MWD using the reference equations in healthy adults [37] and PROs using reference values where available [38]. We used the paired sample t tests and multivariable generalized estimating equations (GEE) models to assess and analyze the effects of time and/or treatment on outcomes. We chose GEE models as they generally provide better model fits compared with linear mixed effects models for studies with a relatively large sample size (N > 30) and few follow-up assessments [39]. To identify potential confounders, we used univariable (UVA) and multivariable (MVA) linear regression analyses to assess the relationship between baseline characteristics and the outcome of interest. We performed MVAs using stepwise backward selection modeling including all baseline characteristics with p < 0.20. We used model R-squared and adjusted R-squared values to gauge model fitting, and defined overfitting as a difference of ≥ 20% in adjusted and unadjusted R-squared [40]; those in the final MVA models were selected to enter multivariable GEE models. We further selected for covariates included in the final GEE models using stepwise backwards selection and p value cutoff < 0.20. To investigate the effect of treatment on outcomes, the effect of time (pre-/post-treatment) was forced into the model regardless of statistical significance. We also performed a pre-specified subgroup analysis of stage I lung cancer patients to compare the effects of surgery vs definitive radio-ablation on outcomes. We used beta coefficients (β) and 95% CIs to describe effect size and defined statistical significance as p < 0.05 in two-tailed tests. All data were analyzed using IBM® SPSS® Statistics software version 24.0.
Results
Participants
We screened 55 stage I–IIIA lung cancer patients, mailed recruitment letters to 50 eligible, and had a final enrollment of 35 participants (Fig. 1); their baseline characteristics are summarized in Table 1. Most had a tobacco exposure history (32 participants, 91%), COPD (25, 71%), and stage I disease (29, 83%). There were no significant differences in baseline clinical characteristics for participants compared with non-participants except for a higher proportion of non-participants having stage II–IIIA disease (E-Table 1).
Baseline functional EC and PRO assessments
Participants’ baseline functional EC was low (mean 6MWD = 370 m (69% predicted) and impaired in 24 participants (69%) (Table 2)). Cancer-specific QoL was also reduced (mean = 72.0 points on scale range 0–100). Approximately, half of the participants reported abnormal physical function, pain, insomnia, appetite loss, or dyspnea on the EORTC-QLQ-C30/LC-13 (Table 2) questionnaire (defined as raw scores < mean reference value for functional scales and raw scores > mean reference value for symptom scales [38]). Baseline exploratory outcome assessments are summarized in Table 2.
Curative-intent treatment
All but two participants underwent either surgical resection, definitive radio-ablative therapy, or concurrent chemoradiation for treatment. Of the 18 (51%) participants who underwent surgical resection, all but two received lobectomy; one underwent pneumonectomy due a central tumor location, and another underwent wedge resection due to poor lung function and planned stereotactic body radiotherapy (SBRT) for a synchronous primary lung cancer (follow-up outcome assessments were obtained after wedge resection in this participant); no participant received adjuvant chemo- or radiotherapy. Among the 12 (34%) participants who underwent definitive radio-ablative therapy, all but two received SBRT; one received cryoablation due to a history of pneumonitis following radiotherapy for a previous primary lung cancer, and another received radiofrequency ablation (RFA) and SBRT for synchronous primary lung cancers (follow-up outcome assessments were obtained following completion of SBRT and RFA). Of the 5 (14%) participants undergoing concurrent chemoradiation, two received tri-modality therapy (follow-up assessments were performed at 1 to 3 months following completion lobectomy in these participants). No participant with stage I disease received adjuvant (chemo- or radio-) therapy. We provided in-depth treatment-associated morbidities in our online data supplements.
Completion of follow-up assessments
Following treatment, 28 (80%) of the 35 participants completed the follow-up 6MWT and 31 (89%) completed the PRO questionnaires. Two participants had transportation challenges and declined the follow-up 6MWT but completed PRO questionnaires remotely (one via mail and another via telephone). Three participants did not have regular follow-up clinic visits and/or transportation challenges and, therefore, did not complete either 6MWT or PRO re-assessments within the 1–3-month post-treatment period. One participant suffered medical complications following treatment and died during the follow-up period.
Effect of treatment on outcomes
Following curative-intent therapy, there was a possibly clinically meaningful (and statistically non-significant) decrease in the primary outcome functional EC (mean change (95% CI) 6MWD = − 25.5 m (− 58.4, + 7.3), p = 0.12)) (Fig. 2a (i), as well as possibly clinically meaningful (and statistically non-significant) decrease in the secondary outcome cancer-specific QoL (EORTC-QLQ-C30 summary score = − 3.89 (− 10.9, + 3.08), p = 0.26)) (Fig. 2a (ii) as assessed by the paired sample t tests. Exploratory outcome assessments showed that dyspnea (mean change (95% CI) UCSD SOBQ = + 12.9 (+ 4.77, + 21.0), p = 0.003) (Fig. 2a (iii) and fatigue (BFI = + 10.4 points (+ 2.87, + 17.9), p = 0.008) (Fig. 2a (iv) scores were clinically higher/worse following treatment and no clinically meaningful changes in other exploratory outcomes listed in Table 2.
a Changes in primary, secondary, and significant exploratory outcomes associated with curative-intent lung cancer treatment. i Functional EC (primary outcome); complete follow-up data in 28 participants; mean difference (post-/pre-treatment) = − 25.5 m (95% CI – 58.4, + 7.29), p = 0.12. ii Cancer-specific QoL (secondary outcome); complete follow-up data in 31 participants; mean difference (post-/pre-treatment) = − 3.89 points (95% CI – 10.9, + 3.08), p = 0.26. iii Dyspnea (UCSD SOBQ, significant exploratory outcome) complete data in 30 participants; mean difference (post-/pre-treatment) = + 12.9 points (95% CI + 4.77, + 21.0), p = 0.003. iv Fatigue (BFI, significant exploratory outcome); complete data in 31 participants; mean difference (post-/pre-treatment) = + 10.4 points (95% CI + 2.87, + 17.9), p = 0.008. b Changes in primary, secondary, and significant exploratory outcomes for stage I lung cancer stratified by treatment. i Functional EC (primary outcome); complete response and follow-up in 13 participants for surgical resection and 11 for definitive radio-ablation; no significant between-treatment effect (p = 0.77). ii Cancer-specific QoL (secondary outcome); complete response and follow-up in 14 participants for surgical resection and 12 for definitive radio-ablation; significant between-treatment effect (p = 0.04). iii Dyspnea (UCSD SOBQ, exploratory outcome); complete response and follow-up in 14 participants for surgical resection and 12 for definitive radio-ablation; no significant between-treatment effect (p = 0.77). iv Fatigue (BFI, exploratory outcome); complete response and follow-up in 14 participants for surgical resection and 12 for definitive radio-ablation; no significant between-treatment effect (p = 0.45). Horizontal lines inside the boxes represent the median values, ends of boxes represent upper and lower quartiles, and whiskers represent highest and lowest observations. BFI, brief fatigue inventory; EC, exercise capacity; QoL, quality of life; UCSD SOBQ, University of California San Diego Shortness of Breath Questionnaire
Results of UVAs and MVAs to identify baseline clinical characteristics associated with the endpoints are shown in E-Tables 2–5. In multivariable GEEs adjusting for all confounders associated with the outcomes, the effect of time (pre-/post-treatment) was associated with possibly clinically meaningful decrements in functional EC (mean 6MWD change − 25.4 m, p = 0.096) (Table 3) and no clinically meaningful decrease in cancer-specific QoL (mean EORTC-QLQ-C30 summary score change − 3.39 points, p = 0.30) (Table 4); dyspnea (mean UCSD SOBQ increase 13.1 points, p = 0.001) (Table 5) and fatigue (mean BFI increase 9.97 points, p = 0.006) (Table 6) symptoms were clinically higher/worse following treatment.
In a pre-specified subgroup analysis of stage I patients (N = 29) to compare the effects of surgical resection (n = 17, 59%) vs definitive radio-ablation (n = 12, 41%) on outcomes (Fig. 2b), surgical treatment resulted in a clinically meaningful higher decrement in the secondary outcome cancer-specific QoL following treatment (mean change − 15.1 points (− 0.83, − 29.4), p = 0.04) (Table 7), but no clinically meaningful differences in the primary outcome functional EC (Table 8) or exploratory outcomes dyspnea or fatigue as assessed by the UCSD SOBQ (Table 9) or BFI (Table 10), respectively. The effects of time (pre-/post-treatment) for stage I patients are also shown in Tables 7, 8, 9, and 10.
Discussion
In a prospective, observational cohort study of stage I–IIIA lung cancer patients undergoing curative-intent therapy, we observed (1) a possibly clinically meaningful decline in functional EC, the primary outcome; (2) no clinically meaningful change in the secondary outcome, cancer-specific QoL; and (3) clinically meaningful worsening of exploratory outcomes dyspnea and fatigue symptoms at 1 to 3 months following treatment completion.
Much of the attention on the effects of curative-intent lung cancer therapy focuses on physiological (i.e., lung function and maximal/peak EC) and clinical (i.e., perioperative morbidity and mortality) outcomes [41]. Currently, there is a lack of clinical emphasis on patient-centered outcomes (e.g., functional EC and cancer-specific QoL) which may be more important than survival for some patients [42]. As such, in its most recent clinical guideline for follow-up and surveillance after curative-intent therapy of lung cancer, the American College of Chest Physicians called for additional research to clarify which curative-intent treatment modalities affect QoL the most and to identify patients who are at the most risk for impairments after treatment [43].
The 6MWD is an important patient-centered and functional outcome associated with perioperative complications [44] and survival [45] in patients with early stage lung cancer [16]. To the best of our knowledge, only two previous studies (both by Granger and colleagues [34, 46]) examined the effects of stage I–III lung cancer treatment on functional EC. In both studies, the authors reported clinically and statistically significant declines in the 6MWD following lung cancer resection surgery. While our study did not detect a statistically significant change, the 25-m reduction is likely clinically significant as suggested by another analysis reporting 6MWD changes of 22–42 m as the MCID in the lung cancer population [19]. Our study also provides complementary information to a study by Granger and colleagues which reported a 43-m reduction in 6MWD in a cohort of 40 stage I–IIIB lung cancer survivors at 10 weeks following diagnosis. In contrast to their study, we excluded patients with stage IIIB disease and those undergoing palliative therapy or sequential chemoradiation, thereby targeting a different patient population (i.e., those undergoing curative-intent therapy). Moreover, whereas some of the follow-up assessments in the study by Granger and colleagues were performed before or during the course of treatment [34]; all our follow-up assessments were performed after completion of curative-intent therapy, providing additional insights into their post-treatment course.
While lacking long-term follow-up, our study adds to existing literature [47,48,49,50,51,52,53,54] on the effects of curative-intent lung cancer treatment on PROs and QoL. Among the largest studies to date, Brunelli and colleagues [52] reported that in 156 consecutive patients undergoing lung cancer resection surgery, the physical composite scale in the generic QoL was significantly reduced at 1 month but completely recovered at 3 months, and the mental composite scale remained unchanged. In the same study, the authors also found poor correlation (coefficients < 0.2) between these generic health measures and FEV1, DLCO, and EC as assessed by the height reached on the stair-climbing test [52]. In contrast, in a study with 2-year follow-up, Ilonen and colleagues [53] observed that in 53 patients undergoing lung cancer resection surgery, the generic QoL was decreased compared with preoperative values at 3, 12, and 24 months following surgery. They also found no correlation between preoperative FEV1 or DLCO and QoL at any of the follow-up assessment time points [53]. Similarly, in a prospective cohort study of 131 lung cancer patients undergoing lobectomy or bilobectomy, Schulte and colleagues [54] found that most health domains, including physical function, pain, and dyspnea, were significantly impaired after surgery and remained so for up to 24 months following treatment.
While these contrasting findings may partly be due to differences in baseline characteristics of included patients and surgical techniques used (open thoracotomy vs video-/robotic-assisted thoracoscopy), standard physiological outcome assessments including pulmonary function testing do not appear to adequately capture all the effects of curative-intent lung cancer treatment on health. Also, these studies suggest that results may vary depending on the PRO or QoL instruments used, possibly due to a lack of a validated questionnaire for lung cancer patients undergoing curative-intent therapy, variations in psychometric properties between instruments including sensitivity to change, or the availability of a composite score to avoid multiple testing and minimize chance bias. To the best of our knowledge, our study is among the first to examine the effects of curative-intent lung cancer therapy on cancer-specific QoL using a validated, composite, summary score [21], suggested to be more sensitive to change compared with traditional QoL scores [35].
In contrast to previous studies that used a cross-sectional design [18, 55,56,57], our study is among the first to use the UCSD SOBQ and BFI questionnaires to prospectively assess changes in dyspnea and fatigue, respectively, in patients undergoing curative-intent therapy. While the EORTC-QLQ-C30 [22]/LC13 [23] questionnaires are commonly used cancer-/lung cancer–specific PRO instruments, dyspnea and fatigue—two important and commonly abnormal symptoms in lung cancer patients—are assessed by only one and three questions, respectively, in the 30-question EORTC-QLQ-C30, and only three and zero questions, respectively, in the 13-question EORTC-QLQ-LC13. While there are ongoing efforts to create a novel EORTC-QLQ-LC29 instrument with a summary score [58] to more accurately capture lung cancer-specific health, we used separate PRO questionnaires with more questions specifically on these two important symptoms (24 items in the UCSD SOBQ and 9 in the BFI). In this small sample, we detected clinically meaningful and statistically significant increases/worsening in dyspnea and fatigue symptoms following treatment.
Curative-intent therapy of stage I–IIIA lung cancer is heterogenous and uses a combination of treatment modalities and varies according to stage and clinical assessment of fitness to tolerate treatment. To this end, we performed a pre-specified subgroup analysis of stage I patients to compare the effects of surgical vs radio-ablative therapy. Similar to the entire cohort, our subgroup analysis showed a possibly clinically meaningful decline in functional EC and clinically meaningful higher/worsening of dyspnea and fatigue associated with stage I lung cancer treatment. Compared with radio-ablation, surgical resection may lead to greater decrement in cancer-specific QoL at 1 to 3 months following treatment. While the sample sizes are small and selected, these findings are similar to a recent exploratory analysis of a RCT involving 22 stage IA NSCLC patients to investigate the effects of surgical resection vs SBRT on global QoL [59]. To the best of our knowledge, our subgroup analysis is among the first to prospectively examine the effects of surgical vs definitive radio-ablative therapy in stage I lung cancer patients using a validated, composite, cancer-specific QoL score. As the number of early stage lung cancer survivors increases due to advances in screening and treatment techniques, these findings have implications in future studies involving the shared decision-making, treatment selection, and/or post-treatment care for these patients.
Many adult cancer survivors can experience reduced QoL as the result of physical impairments which can go undetected and untreated and result in disability [60]. In lung cancer patients, systematic reviews suggest that preoperative exercise training improves cardiopulmonary fitness and may reduce surgical complications [61], while postoperative training may improve exercise capacity and QoL [62]. While these findings support the utility of exercise to improve lung cancer–related outcomes, these studies can be affected by volunteer and selection bias and inadequate sample size [61, 62]. Our exploratory PRO assessments suggest that decreasing symptom burden due to dyspnea and fatigue (e.g., through optimizing medical therapy for cardiopulmonary disease) may be important to improve exercise, function, and/or QoL in these patients (E-Fig. 1).
Our study has limitations. First, the small sample size may not be adequately powered to detect statistically significant differences in the primary or secondary outcomes and pre-disposes our multivariable models to overfitting. Second, the range of 1 to 3 months for follow-up assessments may lead to additional variations in the outcomes measured and, therefore, diminished statistical power. Third, the absence of long-term follow-up assessments limits our ability to draw conclusions on the effect of time on outcomes including exploratory variables following treatment. For instance, it is possible that some of the worsening in dyspnea and fatigue may improve spontaneously after the 3-month follow-up period. Finally, our findings may have limited generalizability due to it being a single-institutional study involving a predominantly white male veteran patient population with a significant tobacco exposure and higher than expected prevalence of comorbidities, including coronary artery disease and COPD [14].
The strengths of our study include pre-specified primary, secondary, and exploratory outcomes to minimize chance bias. In addition, all baseline and most follow-up functional EC and PRO assessments were performed in-person by one observer (DH), maximizing the completeness and accuracy of the data collected and minimizing inter-observer variability. Equally important, we had a high completion rate (at least 80%) on all outcomes measured, maximizing the validity of our findings. In addition, unlike many of the published studies to date, we used multivariable GEE analyses to adjust for baseline characteristics including lung function associated with the outcomes enhancing our conclusions. Finally, we provided detailed descriptions of important clinical events following curative-intent lung cancer treatment and interpreted outcome changes using MCIDs, facilitating translation to the clinical setting.
We conclude that in a prospective observational cohort study of lung cancer patients undergoing curative-intent therapy, there were clinically meaningful and statistically significant worsening of dyspnea and fatigue symptoms, possible decreases in functional EC, but no significant change in cancer-specific QoL at 1 to 3 months following treatment. In stage I lung cancer patients, surgical treatment may lead to a greater decrement in cancer-specific QoL compared with definitive radio-ablative therapy. These results provide a proof-of-concept on the information provided by physio-psychological assessments in this patient population and may facilitate future studies to reduce symptom burden, and/or improve functional EC and QoL.
Abbreviations
- 6MWD:
-
Six-minute walk distance
- 6MWT:
-
Six-minute walk test
- AECOPD:
-
Acute exacerbation of COPD
- BFI:
-
Brief Fatigue Inventory
- CTB:
-
Chest tumor board
- DLCO :
-
Diffusion capacity of the lung for carbon monoxide
- EC:
-
Exercise capacity
- EORTC-QLQ-C30/LC13:
-
European Organization for Research and Treatment of Cancer QoL Questionnaire Core 30/Lung Cancer Module 13
- EQ-5D/VAS:
-
EuroQoL-5 Dimensions/visual analogue scale
- FACT-G/L:
-
Functional Assessment of Cancer Therapy – General/Lung
- GEE:
-
Generalized estimating equations
- HADS:
-
Hospital Anxiety and Depression Scale
- HF:
-
Heart failure
- LCS:
-
Lung cancer screening
- MCID:
-
Minimal clinically important difference
- MVA:
-
Multivariable linear regression analysis
- NSCLC:
-
Non-small cell lung cancer
- PRO:
-
Patient-reported outcome
- PSQI:
-
Pittsburgh Sleep Quality Index
- QoL:
-
Quality of life
- RCT:
-
Randomized clinical trial
- RFA:
-
Radiofrequency ablation
- SBRT:
-
Stereotactic body radiotherapy
- TLC:
-
Total lung capacity
- UCSD SOBQ:
-
University California San Diego Shortness of Breath Questionnaire
- US:
-
United States
- UVA:
-
Univariable linear regression analysis
- VASDHS:
-
VA San Diego Healthcare System
References
Dinan MA, Curtis LH, Carpenter WR, Biddle AK, Abernethy AP, Patz EF Jr, Schulman KA, Weinberger M (2012) Stage migration, selection bias, and survival associated with the adoption of positron emission tomography among medicare beneficiaries with non-small-cell lung cancer, 1998-2003. J Clin Oncol 30:2725–2730
Siegel RL, Miller KD, Jemal A (2019) Cancer statistics, 2019. CA Cancer J Clin 69:7–34
Wang Z, Hu Y, Wang Y, Han W, Wang L, Xue F, Sui X, Song W, Shi R, Jiang J (2016) Can CT screening give rise to a beneficial stage shift in lung cancer patients? Systematic review and meta-analysis. PLoS One 11:e0164416
National Lung Screening Trial Research Team, Aberle DR, Adams AM, Berg CD, Black WC, Clapp JD, Fagerstrom RM, Gareen IF, Gatsonis C, Marcus PM, Sicks JD (2011) Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 365:395–409
Jaklitsch MT, Jacobson FL, Austin JH, Field JK, Jett JR, Keshavjee S, MacMahon H, Mulshine JL, Munden RF, Salgia R, Strauss GM, Swanson SJ, Travis WD, Sugarbaker DJ (2012) The American Association for Thoracic Surgery guidelines for lung cancer screening using low-dose computed tomography scans for lung cancer survivors and other high-risk groups. J Thorac Cardiovasc Surg 144:33–38
Mazzone PJ, Silvestri GA, Patel S, Kanne JP, Kinsinger LS, Wiener RS, Soo Hoo G, Detterbeck FC (2018) Screening for lung cancer: CHEST guideline and expert panel report. Chest 153:954–985
Moyer VA, U.S. Preventive Services Task Force (2014) Screening for lung cancer: U.S. preventive services task force recommendation statement. Ann Intern Med 160:330–338
Smith RA, Andrews K, Brooks D, DeSantis CE, Fedewa SA, Lortet-Tieulent J, Manassaram-Baptiste D, Brawley OW, Wender RC (2016) Cancer screening in the United States, 2016: a review of current American Cancer Society guidelines and current issues in cancer screening. CA Cancer J Clin 66:96–114
Wood DE, Kazerooni EA, Baum SL, Eapen GA, Ettinger DS, Hou L, Jackman DM, Klippenstein D, Kumar R, Lackner RP, Leard LE, Lennes IT, Leung ANC, Makani SS, Massion PP, Mazzone P, Merritt RE, Meyers BF, Midthun DE, Pipavath S, Pratt C, Reddy C, Reid ME, Rotter AJ, Sachs PB, Schabath MB, Schiebler ML, Tong BC, Travis WD, Wei B, Yang SC, Gregory KM, Hughes M (2018) Lung Cancer Screening, Version 3.2018, NCCN clinical practice guidelines in oncology. J Natl Compr Cancer Netw 16:412–441
Wang JS, Abboud RT, Wang LM (2006) Effect of lung resection on exercise capacity and on carbon monoxide diffusing capacity during exercise. Chest 129:863–872
Agostini P, Cieslik H, Rathinam S, Bishay E, Kalkat MS, Rajesh PB, Steyn RS, Singh S, Naidu B (2010) Postoperative pulmonary complications following thoracic surgery: are there any modifiable risk factors? Thorax 65:815–818
Ha D, Choi H, Zell K, Raymond DP, Stephans K, Wang XF, Videtic G, McCarthy K, Minai OA, Mazzone PJ (2014) Association of impaired heart rate recovery with cardiopulmonary complications after lung cancer resection surgery. J Thorac Cardiovasc Surg 149:1168–1173
Carver JR, Shapiro CL, Ng A, Jacobs L, Schwartz C, Virgo KS, Hagerty KL, Somerfield MR, Vaughn DJ, ASCO Cancer Survivorship Expert Panel (2007) American Society of Clinical Oncology clinical evidence review on the ongoing care of adult cancer survivors: cardiac and pulmonary late effects. J Clin Oncol 25:3991–4008
Islam KM, Jiang X, Anggondowati T, Lin G, Ganti AK (2015) Comorbidity and survival in lung cancer patients. Cancer Epidemiol Biomark Prev 24:1079–1085
von Elm E, Altman DG, Egger M, Pocock SJ, Gotzsche PC, Vandenbroucke JP, STROBE Initiative (2007) The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. Ann Intern Med 147:573–577
Ha D, Mazzone PJ, Ries AL, Malhotra A, Fuster M (2016) The utility of exercise testing in patients with lung cancer. J Thorac Oncol 11:1397–1410
Schmidt K, Vogt L, Thiel C, Jager E, Banzer W (2013) Validity of the six-minute walk test in cancer patients. Int J Sports Med 34:631–636
Ha D, Ries AL, Mazzone PJ, Lippman SM, Fuster MM (2018) Exercise capacity and cancer-specific quality of life following curative intent treatment of stage I-IIIA lung cancer. Support Care Cancer 26:2459–2469
Granger CL, Holland AE, Gordon IR, Denehy L (2015) Minimal important difference of the 6-minute walk distance in lung cancer. Chron Respir Dis 12:146–154
ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories (2002) ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med 166:111–117
Giesinger JM, Kieffer JM, Fayers PM, Groenvold M, Petersen MA, Scott NW, Sprangers MA, Velikova G, Aaronson NK, EORTC Quality of Life Group (2016) Replication and validation of higher order models demonstrated that a summary score for the EORTC QLQ-C30 is robust. J Clin Epidemiol 69:79–88
Aaronson NK, Ahmedzai S, Bergman B, Bullinger M, Cull A, Duez NJ, Filiberti A, Flechtner H, Fleishman SB, de Haes JC (1993) The European Organization for Research and Treatment of Cancer QLQ-C30: a quality-of-life instrument for use in international clinical trials in oncology. J Natl Cancer Inst 85:365–376
Bergman B, Aaronson NK, Ahmedzai S, Kaasa S, Sullivan M (1994) The EORTC QLQ-LC13: a modular supplement to the EORTC core quality of life questionnaire (QLQ-C30) for use in lung cancer clinical trials. EORTC study group on quality of life. Eur J Cancer 30A:635–642
Shaw JW, Johnson JA, Coons SJ (2005) US valuation of the EQ-5D health states: development and testing of the D1 valuation model. Med Care 43:203–220
Buysse DJ, Reynolds CF 3rd, Monk TH, Berman SR, Kupfer DJ (1989) The Pittsburgh Sleep Quality Index: a new instrument for psychiatric practice and research. Psychiatry Res 28:193–213
Eakin EG, Resnikoff PM, Prewitt LM, Ries AL, Kaplan RM (1998) Validation of a new dyspnea measure: the UCSD Shortness of Breath Questionnaire. University of California, San Diego. Chest 113:619–624
Mendoza TR, Wang XS, Cleeland CS, Morrissey M, Johnson BA, Wendt JK, Huber SL (1999) The rapid assessment of fatigue severity in cancer patients: use of the brief fatigue inventory. Cancer 85:1186–1196
Zigmond AS, Snaith RP (1983) The hospital anxiety and depression scale. Acta Psychiatr Scand 67:361–370
Pickard AS, Neary MP, Cella D (2007) Estimation of minimally important differences in EQ-5D utility and VAS scores in cancer. Health Qual Life Outcomes 5:70
Hughes CM, McCullough CA, Bradbury I, Boyde C, Hume D, Yuan J, Quinn F, McDonough SM (2009) Acupuncture and reflexology for insomnia: a feasibility study. Acupunct Med 27:163–168
Ries AL (2005) Minimally clinically important difference for the UCSD shortness of breath questionnaire, Borg scale, and visual analog scale. COPD 2:105–110
Puhan MA, Frey M, Buchi S, Schunemann HJ (2008) The minimal important difference of the hospital anxiety and depression scale in patients with chronic obstructive pulmonary disease. Health Qual Life Outcomes 6:46-7525-6-46
Musoro JZ, Coens C, Fiteni F, Katarzyna P, Cardoso F, Russell NS, King MT, Cocks K, Sprangers MA, Groenvold M, Velikova G, Flechtner H, Bottomley A, EORTC Breast and Quality of Life Groups (2019) minimally important differences for interpreting EORTC QLQ-C30 scores in patients with advanced breast cancer. jncics 3
Granger CL, McDonald CF, Irving L, Clark RA, Gough K, Murnane A, Mileshkin L, Krishnasamy M, Denehy L (2014) Low physical activity levels and functional decline in individuals with lung cancer. Lung Cancer 83:292–299
Pompili C, Koller M, Velikova G, Franks K, Absolom K, Callister M, Robson J, Imperatori A, Brunelli A (2018) EORTC QLQ-C30 summary score reliably detects changes in QoL three months after anatomic lung resection for non-small cell lung Cancer (NSCLC). Lung Cancer 123:149–154
Pallant J (2013) SPSS survival manual: a step by step guide to data analysis using IBM SPSS. Open University Press
Enright PL, Sherrill DL (1998) Reference equations for the six-minute walk in healthy adults. Am J Respir Crit Care Med 158:1384–1387
Scott NW, Fayers PM, Aaronson NK, Bottomley A, de Graeff A, Groenvold M, Gundy C, Koller M, Peterson MA, Sprangers MAG (2008) EORTC QLQ-C30 Reference Values. EORTC Quality of Life Group
Zeger SL, Liang KY, Albert PS (1988) Models for longitudinal data: a generalized estimating equation approach. Biometrics 44:1049–1060
Babyak MA (2004) What you see may not be what you get: a brief, nontechnical introduction to overfitting in regression-type models. Psychosom Med 66:411–421
Brunelli A, Kim AW, Berger KI, Addrizzo-Harris DJ (2013) Physiologic evaluation of the patient with lung cancer being considered for resectional surgery: diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 143:e166S–e190S
Gralla RJ, Hollen PJ, Msaouel P, Davis BV, Petersen J (2014) An evidence-based determination of issues affecting quality of life and patient-reported outcomes in lung cancer: results of a survey of 660 patients. J Thorac Oncol 9:1243–1248
Colt HG, Murgu SD, Korst RJ, Slatore CG, Unger M, Quadrelli S (2013) Follow-up and surveillance of the patient with lung cancer after curative-intent therapy: diagnosis and management of lung cancer, 3rd ed: American College of Chest Physicians evidence-based clinical practice guidelines. Chest 143:e437S–e454S
Hattori K, Matsuda T, Takagi Y, Nagaya M, Inoue T, Nishida Y, Hasegawa Y, Kawaguchi K, Fukui T, Ozeki N, Yokoi K, Ito S (2018) Preoperative six-minute walk distance is associated with pneumonia after lung resection. Interact Cardiovasc Thorac Surg 26:277–283
Hamada K, Irie M, Fujino Y, Hyodo M, Hanagiri T (2019) Prognostic value of preoperative exercise capacity in patients undergoing thoracoscopic lobectomy for non-small cell lung cancer. Lung Cancer 128:47–52
Granger CL, Parry SM, Edbrooke L, Denehy L (2016) Deterioration in physical activity and function differs according to treatment type in non-small cell lung cancer - future directions for physiotherapy management. Physiotherapy 102:256–263
Balduyck B, Hendriks J, Lauwers P, Van Schil P (2007) Quality of life evolution after lung cancer surgery: a prospective study in 100 patients. Lung Cancer 56:423–431
Bezjak A, Lee CW, Ding K, Brundage M, Winton T, Graham B, Whitehead M, Johnson DH, Livingston RB, Seymour L, Shepherd FA (2008) Quality-of-life outcomes for adjuvant chemotherapy in early-stage non-small-cell lung cancer: results from a randomized trial, JBR.10. J Clin Oncol 26:5052–5059
Handy JR Jr, Asaph JW, Skokan L, Reed CE, Koh S, Brooks G, Douville EC, Tsen AC, Ott GY, Silvestri GA (2002) What happens to patients undergoing lung cancer surgery? Outcomes and quality of life before and after surgery. Chest 122:21–30
Schulte T, Schniewind B, Dohrmann P, Kuchler T, Kurdow R (2009) The extent of lung parenchyma resection significantly impacts long-term quality of life in patients with non-small cell lung cancer. Chest 135:322–329
Sloan JA, Cheville AL, Liu H, Novotny PJ, Wampfler JA, Garces YI, Clark MM, Yang P (2016) Impact of self-reported physical activity and health promotion behaviors on lung cancer survivorship. Health Qual Life Outcomes 14:66-016-0461-3
Brunelli A, Socci L, Refai M, Salati M, Xiume F, Sabbatini A (2007) Quality of life before and after major lung resection for lung cancer: a prospective follow-up analysis. Ann Thorac Surg 84:410–416
Ilonen IK, Rasanen JV, Knuuttila A, Sihvo EI, Sintonen H, Sovijarvi AR, Salo JA (2010) Quality of life following lobectomy or bilobectomy for non-small cell lung cancer, a two-year prospective follow-up study. Lung Cancer 70:347–351
Schulte T, Schniewind B, Walter J, Dohrmann P, Kuchler T, Kurdow R (2010) Age-related impairment of quality of life after lung resection for non-small cell lung cancer. Lung Cancer 68:115–120
Feinstein MB, Krebs P, Coups EJ, Park BJ, Steingart RM, Burkhalter J, Logue A, Ostroff JS (2010) Current dyspnea among long-term survivors of early-stage non-small cell lung cancer. J Thorac Oncol 5:1221–1226
Iyer S, Roughley A, Rider A, Taylor-Stokes G (2014) The symptom burden of non-small cell lung cancer in the USA: a real-world cross-sectional study. Support Care Cancer 22:181–187
Lowery AE, Krebs P, Coups EJ, Feinstein MB, Burkhalter JE, Park BJ, Ostroff JS (2014) Impact of symptom burden in post-surgical non-small cell lung cancer survivors. Support Care Cancer 22:173–180
Koller M, Hjermstad MJ, Tomaszewski KA, Tomaszewska IM, Hornslien K, Harle A, Arraras JI, Morag O, Pompili C, Ioannidis G, Georgiou M, Navarra C, Chie WC, Johnson CD, Himpel A, Schulz C, Bohrer T, Janssens A, Kulis D, Bottomley A, EORTC Quality of Life Group, EORTC Lung Cancer Group, European Society of Thoracic Surgeons (2017) An international study to revise the EORTC questionnaire for assessing quality of life in lung cancer patients. Ann Oncol 28:2874–2881
Louie AV, van Werkhoven E, Chen H, Smit EF, Paul MA, Widder J, Groen HJ, van den Borne BE, De Jaeger K, Slotman BJ, Senan S (2015) Patient reported outcomes following stereotactic ablative radiotherapy or surgery for stage IA non-small-cell lung cancer: results from the ROSEL multicenter randomized trial. Radiother Oncol 117:44–48
Silver JK, Baima J, Mayer RS (2013) Impairment-driven cancer rehabilitation: an essential component of quality care and survivorship. CA Cancer J Clin 63:295–317
Cavalheri V, Granger C (2017) Preoperative exercise training for patients with non-small cell lung cancer. Cochrane Database Syst Rev 6:CD012020
Driessen EJ, Peeters ME, Bongers BC, Maas HA, Bootsma GP, van Meeteren NL, Janssen-Heijnen ML (2017) Effects of prehabilitation and rehabilitation including a home-based component on physical fitness, adherence, treatment tolerance, and recovery in patients with non-small cell lung cancer: a systematic review. Crit Rev Oncol Hematol 114:63–76
Acknowledgments
We would like to thank Dr. Florin Vaida and Anya Umlauf for the assistance in the statistical analyses for this study, the Section of Pulmonary and Critical Care Medicine at the VA San Diego Healthcare System (VASDHS), notably Dr. Judd Landsberg and Dr. Philippe Montgrain for their relentless support, Svetlana Sheinkman for maintaining an ongoing list of lung cancer patients, Christine Miller for the assistance with patient recruitment and enrollment, and the staff in the Pulmonary Function Laboratory for the assistance with patient scheduling and six-minute walk test assessments. We would also like to give tremendous thanks to all the veteran lung cancer survivors who volunteered and donated their time for this study.
Funding
We would like to thank the Division of Pulmonary, Critical Care, and Sleep Medicine and the Clinical and Translational Research Institute (CTRI) Clinical Research Enhancement through Supplemental Training (CREST)/Master of Advanced Study (MAS) in Clinical Research Program at the University of California San Diego (UCSD) for the financial and scholarship support. This work was supported in part by the National Institutes of Health (L30CA208950 and 1T32HL134632-01), the American Cancer Society (PF-17-020-01-CPPB), and Veterans Affairs (VA Merit I01BX003688). The content is solely the responsibility of the authors and does not necessarily represent the official views of the funding agencies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical conduct of research
This study was conducted at the VA San Diego Healthcare System. All human investigations were performed after approval by the VA San Diego Healthcare System institutional review board (protocol # H150158) and in accord with an assurance filed with and approved by the US Department of Health and Human Services and with the 1964 Helsinki Declaration and its later amendments.
Informed consent
Written informed consent was obtained from each participant included in this study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The contents of this manuscript were previously made available in a pre-print publication (https://doi.org/10.1101/508234), revised, and updated in this current version.
Electronic supplementary material
ESM 1
(DOCX 162 kb)
Rights and permissions
About this article
Cite this article
Ha, D., Ries, A.L., Lippman, S.M. et al. Effects of curative-intent lung cancer therapy on functional exercise capacity and patient-reported outcomes. Support Care Cancer 28, 4707–4720 (2020). https://doi.org/10.1007/s00520-020-05294-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-020-05294-3