Abstract
Purpose
Breast cancer is the most frequently diagnosed cancer and leading cause of cancer death among women, representing a considerable public health burden in South Africa and other low-middle income countries. Short- and long-term complications of these treatments include shoulder morbidities such as pain, decreased range of motion, tightness, weakness, pain, numbness and lymphoedema and may be present for up to 6 years post-treatment. An understanding of baseline demographic and clinical risk factors can guide rehabilitation and management strategies for high-risk patients. The aims of this study were to quantify the burden of shoulder pain and disability in a tertiary academic hospital in Cape Town, South Africa, and identify potential risk factors for the development of shoulder morbidity.
Methods
This study was a cross-sectional analysis of the prevalence of shoulder pain and dysfunction in women attending their post-treatment annual follow-up visit for unilateral breast carcinoma.
Results
Three in four patients reported a presence of any pain or disability while only 9% experienced severe pain and disability. Multivariable ordinal logistic regression analysis identified race, side, axillary surgery, chemotherapy and age as significant predictors of pain and chemotherapy a significant predictor of disability.
Conclusion
The substantial burden of shoulder morbidity in this population represents a significant public health burden. The use of identified clinical and demographic characteristics may guide in the development of survivorship programmes incorporating surveillance and management of these high-risk patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Breast cancer is the most frequently diagnosed cancer and leading cause of cancer death among women in both low-to-middle-income countries (LMICs) and high-income countries (HICs) [1]. According to the 2011 South African National Cancer Registry, breast cancer accounts for 21% of all cancers in females [2, 3], accompanied by a mortality rate of 16% from 2012 [4], representing a considerable public health burden in South Africa [2]. In LMICs such as South Africa, many patients present with advanced disease and have a lack of access to newest treatment options that may be available in HICs [5, 6].
The medical management of breast cancer may result in short- and long-term complications, including impaired shoulder function such as pain, decreased range of motion (ROM), tightness, weakness, pain, numbness and lymphoedema [7,8,9,10]. These complications affect activities of daily living (ADL) including the ability to return to work [9]. Reduced ROM and pain are commonly described shoulder morbidities [11] that occur shortly after surgery [12] and may be present for up to 6 years post-surgery [13, 14]. Different treatment modalities are associated with varying levels of shoulder morbidity, such as a higher prevalence in those receiving axillary lymph node biopsy (ALNB) compared to sentinel node biopsy (SNB), in those receiving mastectomy compared to breast-conserving therapy (BCT), and with the use of chemotherapy and radiotherapy [8, 9, 11, 15,16,17]. Demographics associated with increased shoulder morbidity include the number of nodes removed, older age, the left side being affected and high body mass index (BMI) [13, 14, 18, 19].
The aims of this study were to quantify the burden of shoulder pain and disability in a tertiary academic hospital in Cape Town, South Africa, and identify potential risk factors for the development of shoulder morbidity.
Materials and methods
Methods
Women attending their annual check-up at the breast clinic at Groote Schuur Hospital, Cape Town, South Africa, were approached to be included in the study. Informed consent was received for all participants prior to collection of data. Personal information was de-identified and each participant was assigned a study number. Patients were reviewed according to pre-determined inclusion and exclusion criteria (see below), and upon informed consent, completed the Shoulder Pain and Disability Index (SPADI) questionnaire in the language of their choice. SPADI is a self-administered index consisting of 13 items divided into two subscales; 5 items for pain and 8 items for disability, which has shown consistency and reliability. The questionnaire is easy to complete and imposes very little burden on the patient. Those requiring assistance to complete the questionnaire were assisted by research staff fluent in the language of the participant. Additional clinical and demographic data was collected such as race (self-identified), age and side affected.
This study is a cross-sectional analysis of the prevalence of shoulder pain and disability in this sample of women. The primary objective of this study was to determine the prevalence of shoulder morbidity in patients and the secondary objective was to evaluate associations between shoulder morbidity and covariates such as treatment protocol and baseline demographics.
Inclusion and exclusion criteria
Inclusion criteria
-
1.
Women 18 years of age and older
-
2.
≥ 1 year post-surgery for unilateral carcinoma of the breast
Exclusion criteria
-
1.
Reconstructive surgery
-
2.
Current or previous history of shoulder complex trauma, surgery, pathology or dysfunction
-
3.
Current or previous history of cervical neuropathy
Statistical considerations and data analysis
Sample size
This was a convenience sample of women attending annual follow-up appointments at the breast clinic at the hospital between 2015 and 2017. The SPADI questionnaire was completed by 349 participants. Not all participants’ medical records provided the required demographic and clinical data.
Statistical analysis
Data was analysed using STATA 14.0 (College Station, Texas, USA). Demographic and clinical data were described by summary statistics, where categorical variables were reported in frequencies and percentages and numerical variables were described by the mean and standard deviation.
Pain and disability were categorised into four categories according to scores from the SPADI questionnaire where 0 depicts ‘no pain/no disability’, 1–30 ‘mild pain/mild disability’, 31–50 ‘moderate pain/moderate disability’ and > 50 ‘severe pain/severe disability’, respectively. Ordinal logistic regression was used for determining the association(s) between pain and disability scores with covariates, such as baseline demographics. Ordinal logistic regression was used due to the presence of several ordered categories of the outcomes, pain and disability. Potential confounders and interacting variables were considered in the analysis. In the univariable analysis, odds ratios (OR) and 95% confidence intervals (CIs) were calculated to summarise the effect of each covariate on pain and disability outcomes. Covariates with a p value < 0.2 in the univariate analysis were selected for consideration in the multivariable model. A backward selection procedure was used to investigate the most significant predictors of pain and disability scores in the multivariable analysis. The final model was presented by the OR and 95% CI for the OR of the predictor. A p value of < 0.05 was considered statistically significant. The proportional odds assumption was investigated for the final model.
Ethical considerations
Ethical clearance for this study was granted by the University of Cape Town Human Research Ethics Committee (approval number: 317/2017). This study was conducted in accordance with the Declaration of Helsinki (last updated in 2013) and International guidelines for Good Clinical Practice (ICH 1997).
Results
Demographic and clinical characteristics are summarised in Table 1. The vast majority of patients were of mixed ancestry. Age ranged from 34 to 84 years, with a mean of 60 years. Follow-up was on average 6.5 years since surgery, with a minimum of 2 years and maximum of 17 years. The large majority of patients underwent ALND compared to SNB (78% vs. 13%). There were no radical mastectomies, but a very high percentage of modified radical mastectomy (MRM).
A summary of the SPADI results are presented in Table 2, according to individual items and total scores for pain and disability, respectively. Overall, the prevalence of shoulder morbidity in this population was 75% with only a quarter of patients reporting no pain or disability. There was a significant difference between total SPADI score when comparing the proportion who reported mild versus the proportion who reported moderate (p < 0.001) whereas there was no significant difference between the proportion who reported moderate versus the proportion who reported severe SPADI scores (p = 0.13).
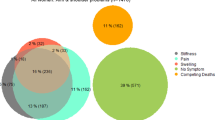
When evaluating the congruence between pain and disability, 26% had no pain and 38% had no disability, whereas 14% had severe pain and 7% severe disability. Overall, more pain is present relative to disability. Similarly, as seen in Fig. 1, there is not a complete association between all categories of pain and disability. The Pearson’s correlation coefficient between pain and disability scores was 0.82 showing a strong linear relationship between pain and disability.
As seen in the individual components of pain and disability in Table 2, 74% of patients experienced some degree of pain (similar to the overall shoulder morbidity of 75%) whereas 63% experienced some level of disability. Mild pain was experienced by 46% of patients and moderate and severe pain by 14% of patients, respectively. Mild disability was experienced by 53% of patients, moderate by 13% and severe disability by 9% of patients.
Ranking scale for severity grading:
For total pain, disability, and SPADI: none: score of zero, mild: score of 1–30, moderate: score of 31–50, severe: score > 50.
For pain and disability individual questions: none: score of zero, mild: score of 1–3, moderate: score of 3–5, severe: score > 5.
Table 3 and Table 4 summarise the results from univariable and multivariable ordinal logistic regression with respect to pain (Table 3) and disability (Table 4). The odds ratio is interpreted as the increase in odds of being in a higher pain or disability category per unit increase in the risk factor. The proportional odds assumptions were met for the final models of both pain and disability.
Utilising univariable ordinal logistic regression analysis, variables associated with pain in this population group (all p < 0.2) were race, the affected side of the primary tumour, total number of nodes affected, use of chemotherapy as an additional treatment modality, age, use of radiotherapy as an additional treatment modality and axillary surgery (Table 3). In the multivariable ordinal logistic regression analysis, race, side, axillary surgery, chemotherapy and age remained as the most significant variables associated with pain, whereas radiotherapy was no longer significant.
As seen in Table 3, Caucasian and mixed ancestry patients experienced less pain compared to Black African patients, but this OR was not statistically significant for mixed ancestry patients (Caucasian: OR 0.21; 95% CI 0.05–0.82, p = 0.024 and mixed ancestry: OR 0.54; 95% CI 0.22–1.37, p = 0.194). Those patients who had the left side affected had 3.2 times increased odds of being in worse pain (OR 3.22; 95% CI 0.97–10, p = 0.056) compared with patients who had their right side affected. For each unit increase in age, there was a corresponding 5% reduction in the odds of being in a higher pain category (OR 0.95; 95% CI 0.93–0.98, p < 0.0001). The lack of chemotherapy as an additional treatment was associated with a 61% reduction in the odds of pain (OR 0.39; 95% CI 0.18–0.83, p = 0.015). ALND was associated with a reduced odds of being in a higher pain category by 52% (OR 0.48; 95% CI 0.23–0.98, p = 0.044).
To directly test the hypothesis that the side effect may differ by race, an overall model was constructed that included the covariates described and an interaction term for race and side. Both the covariates remained significantly associated with higher pain, as did the interaction term for Caucasian when compared to the reference group of Black African (p = 0.034), but not for mixed ancestry when compared to the reference group of Black African (p = 0.156). Exploratory analysis of additional interaction terms showed no significant association with the outcome.
Covariates found to be correlated with disability in univariable ordinal logistic regression analysis included race, chemotherapy, age, radiotherapy and axillary surgery (Table 4). In the multivariable logistic regression analysis, chemotherapy was the only one that remained as a significant predictor of disability, after adjustment. The lack of exposure to chemotherapy as an additional treatment modality was associated with a 63% reduction in the odds of experiencing a worse category of disability (OR 0.37, 95% CI 0.18–0.77, p = 0.007).
Discussion
This analysis reports on a group of individuals in a LMIC state-provided facility in Cape Town, South Africa. Approximately 600 new patients and many more follow-ups are seen annually at this large, tertiary academic breast cancer clinic, from a broad range of referral centres within Cape Town. On average, patients were 6.52 years post-surgery when they were enrolled into this study, with a minimum of 2.37 years and a maximum of 17.5 years, representing long-term outcome data post-surgery for breast cancer. This is a similar finding to previous work from an author in this group from a European population group showing that pain continues to persistent 6 years after treatment [13]. This similarity exists in the presence of a large number of radical mastectomies in this patient group compared to those in the European study [13].
A total of 75% of patients experienced some level of pain and disability, with 25% experiencing none. The overall percentage was almost identical for both total SPADI score and the domain of pain scores (75% and 74%, respectively), whereas the disability score was somewhat less with 63% experiencing mild, moderate or severe disability. These figures represent a significant burden of long-term morbidity. The reduced prevalence of disability compared to pain indicates that pain may be the more significant burden of shoulder morbidity, accompanied by disability in the large majority. According to overall SPADI score, 53% of patients were mild, 13% moderate and 9% severe, respectively. The proportion of patients in each group of overall SPADI score was statistically significantly different when comparing mild versus moderate groups, but not for moderate versus severe groups, respectively. Mild pain and disability accounts for the largest group; 46% of patients in each of the domains. Differences lie between pain and disability in the moderate and severe groups, suggesting that more pain exists but not necessarily in the presence of accompanying disability.
Separate ordinal logistic regression models were constructed for pain and disability due to the differing burden each one represents, as seen from Table 2 and Fig. 1. The proportional odds assumptions were met for the final multivariable ordinal regression models of pain and disability, confirming that the ORs increase at a consistent proportion in the model. Whereas age, side affected, chemotherapy, axillary surgery and race were risk factors for pain, only the use of chemotherapy was a risk factor for disability.
Black African patients had a greater odds of being in the higher pain categories, when compared to Caucasian and mixed ancestry patients. We are aware of differences existing between racial groups indicating that Black patients are at a higher risk, although this is largely from the African-American population [20, 21]. Differences between the racial groups may exist due to cultural approaches to treatment-seeking behaviours, delays in seeking or accessing treatment and socioeconomic status (SES). The prevalence of obesity is often higher in the Black population, particularly Black women [22] and given high BMI is a known risk factor for shoulder morbidity [11], one needs to consider the presence of obesity in Black patients as a mediator of pain [20]. Mediation analysis has suggested that BMI can attenuate the association between race and disability by 40% [20]. However, as this was not measured in our study, we cannot make inferences regarding it. This is a public sector hospital and therefore patients seen are generally at a lower SES, which in addition to being of Black African race, may be associated with presenting with more advanced disease and receiving fewer treatment options [16, 20, 21]. Forty percent of patients presented with grade II tumours, although 18% had missing data for this variable. A recent study at this breast cancer facility found that 78% of women were unemployed despite having received treatment more than 2 years prior (personal communication). It is important to note that more than 70% of the patients included in the group were of mixed ancestry. This is not reflective of the South African population where the largest racial group is Black African; however, it is representative of the Cape Town region where mixed ancestry is the largest population group [23].
In this study, the mean age was 60 years, ranging from 34 to 83 years. Increasing age was statistically significantly associated with reduced pain, with an OR of 0.95 (95% CI 0.93–0.98, p < 0.0001). Older age (> 65 years) is a risk factor for reduced ROM and limitation in ADL; however, younger age is associated with reduced muscle strength [11]. This discrepancy may be due to younger patients being more sensitive to the discomfort resulting from axillary surgery or having different expectations [12]. A study with a relatively similar population age group to ours (mean age of patients was 56 years) found age to be a strong predictor of long-term shoulder–arm function [24].
Left side affected was associated with a higher risk of pain, which corroborates what has been shown previously. It is well understood that left affected side represents a higher-risk group for development of pain and disability [14]. A number of other studies have also reported increased frequency of pain on the left side [13, 18]. A functional asymmetry towards the right hemisphere for pain perception has been reported by several authors [14]. The right hemisphere is also dominant in processing emotional experience, suggesting a possible link between the right hemisphere and the emotional component of pain processing, which is supported by evidence that factors such as anxiety and anticipation of pain have been shown to stimulate affective/cognitive pain perception pathways [14]. Chemotherapy treatment was associated with a 37% increased odds of having a worse disability score and 39% of having a worse pain score. Chemotherapy has been linked to increased upper limb pain and dysfunction and lymphoedema [9, 11, 25], although has also been shown to reduce the odds of reduced ROM reduction when compared with not receiving chemotherapy [11]. Patients received chemotherapy including cyclophosphamide, doxorubicin, epirubicin and 5-fluorouracil); however, the regimen received was not analysed at patient-level. ALND was associated with reduced pain when compared to SNB. This is a surprising finding given most of the evidence suggests that SNB has more favourable outcome in terms of shoulder morbidity [8, 11]. A similar proportion of patients in both groups received hormonal treatment, radiotherapy and chemotherapy. Furthermore, more patients in the ALND groups also had left side affected (57% ALND vs. 33% SNB). Many patients had received additional treatments with chemotherapy (70%), radiotherapy (63%) and hormonal treatment (70%). Given radiotherapy result in long-term morbidities, including diminished tissue healing or increased tissue fibrosis, perhaps this provides evidence of shoulder morbidity being less related to type of axillary surgery only, but rather the additional other treatments [25].
The SPADI questionnaire was used as it is quick and easy to complete [26]. It has also been shown to have high reliability and validity [27] and is able to identify key functional limitations in breast cancer survivors, making it a useful tool for identification of patients at risk of developing shoulder pain and disability [9]. It has not been validated in a South African population; however, a study evaluated the concurrent validity, construct validity and the test-retest reliability of a Face-SPADI in South African breast cancer survivors and showed excellent concurrent validity, internal consistency and test-retest reliability (personal communication). Thus, we can be confident of the results of this study which utilised the SPADI numerical questionnaire. The SPADI questionnaire requires self-reporting of pain and disability, introducing the challenges of subjective reporting and self-perceived outcomes. However, as discussed above, SPADI has been shown to be reliable and valid and has frequently been employed in research settings for assessment of shoulder dysfunction.
There are several limitations to this study. There is a proportion of missing demographic and clinical data and the small Black African and Caucasian samples may have influenced findings. This may affect some of the statistical analysis with regards to regression in terms of sample size and confidence intervals. Although ten patients did not fully complete the SPADI questionnaire, the questionnaire makes adjustment for this in the calculation. Although internal validity is likely to be high, there is limited external generalizability. However, this study was aimed at providing data for a specific group of patients in a LMIC. No patients in this group underwent a radical mastectomy, as this is not the preferred treatment of choice. We are aware that radical mastectomy is a risk factor for shoulder morbidity so perhaps this may underrepresent the burden.
The fact that this data shows such a significant burden of shoulder morbidity 6 years after treatment highlights the importance of long-term surveillance and rehabilitation. There is a clear need for prospective surveillance programmes that can be integrated into survivorship programmes, particularly within South Africa and other LMICs [11, 28]. The ability to identify those patients at risk of long-term morbidity reduces the need for intensive rehabilitation and those associated costs [28]. This has an economic public health burden in that many of these women require to contribute to their households to feed and house their families. Oftentimes, these women work in jobs that require a high level of physical functioning, and thus, the inability to move one’s shoulder represents a significant barrier to return to work. Rehabilitation would reduce the public health burden in an already-burdened service and public health sector facility. Having key baseline data and predictors enables us to design and implement care pathways for those patients at risk. A greater focus needs to be given to survivorship strategies, surveillance and early intervention warning systems as part of an integrated cancer care model. The authors have developed an early warning system for long-term surveillance based on SPADI domains and items [28].
References
Jemal A, Bray F, Ferlay J (2011) Global Cancer statistics: 2011. CA Cancer J Clin 61:69–90. https://doi.org/10.3322/caac.20107.Available
Herbst MC (2016) Cancer Association of South Africa (CANSA) Fact Sheet on the Incidence of Cancer Among Females from 2000 to 2011
National Health Laboratory Service (2011) National Cancer Registry.; 2011. http://www.cansa.org.za/south-african-cancer-statistics/. Accessed 15 Nov 2017
International Agency for Research on Cancer (IARC). Globocan (2012) Estimated Cancer Incidence, Mortality and Prevalence Worldwide in 2012. http://globocan.iarc.fr/Pages/fact_sheets_population.aspx. Accessed February 24, 2017
Mutebi M, Edge J (2014) Stigma, survivorship and solutions: addressing the challenges of living with breast cancer in low-resource areas. South African Med J 104(5):383–385. https://doi.org/10.7196/SAMJ.8253
Marcus TS, Lunda S, Fernandez L (2013) Delayed breast cancer presentation: hospital data should inform proactive primary care. African J Prim Heal Care Fam Med 5(1):1–7. https://doi.org/10.4102/phcfm.v5i1.503
Freitas-Silva R, Conde D, de Freitas-Junior R, Martinez E (2010) Comparison of quality of life, satisfaction with surgery and shoulder-arm morbidity in breast cancer survivors submitted to breast-conserving therapy or mastectomy followed by immediate breast reconstruction. Clinics 65(8):781–787
De Groef A, Van Kampen M, Tieto E et al (2016) Arm lymphoedema and upper limb impairments in sentinel node-negative breast cancer patients: a one year follow-up study. Breast 29:102–108. https://doi.org/10.1016/j.breast.2016.07.021
Shamley D (2015) A Cross-Disciplinary Look at Shoulder Pain and Dysfunction after Treatment for Breast Cancer. Int J Cancer Clin Res 1(9)
Koehler LA, Blaes AH, Haddad TC, Hunter DW, Hirsch AT, Ludewig PM (2015) Movement, function, pain, and postoperative edema in axillary web syndrome. Phys Ther 95(10):1345–1353
Hidding JT, Beurskens CHG, Van Der Wees PJ, Van Laarhoven HWM, Nijhuis-van Der Sanden MWG (2014) Treatment related impairments in arm and shoulder in patients with breast cancer: A systematic review. PLoS One 9(5). doi:https://doi.org/10.1371/journal.pone.0096748
Fleissig A, Fallowfield LJ, Langridge CI, Johnson L, Newcombe RG, Dixon JM, Kissin M, Mansel RE (2006) Post-operative arm morbidity and quality of life. Results of the ALMANAC randomised trial comparing sentinel node biopsy with standard axillary treatment in the management of patients with early breast cancer. Breast Cancer Res Treat 95(3):279–293
Shamley D, Lascurain-aguirrebeña I, Oskrochi R (2012) Shoulder morbidity after treatment for breast cancer is bilateral and greater after mastectomy. Acta Oncol (Madr) 51(8):1045–1053. https://doi.org/10.3109/0284186X.2012.695087
Shamley D, Srinaganathan R, Oskrochi R, Lascurain-aguirrebeña I, Sugden E (2009) Three-dimensional scapulothoracic motion following treatment for breast cancer. Breast Cancer Res Treat 118(2):315–322. https://doi.org/10.1007/s10549-008-0240-x
Levangie PK, Drouin J (2009) Magnitude of late effects of breast cancer treatments on shoulder function: a systematic review. Breast Cancer Res Treat 116(1):1–15. https://doi.org/10.1007/s10549-008-0246-4
Smoot B, Paul SM, Aouizerat BE, Dunn L, Elboim C, Schmidt B, Hamolsky D, Levine JD, Abrams G, Mastick J, Topp K, Miaskowski C (2016) Predictors of altered upper extremity function during the first year after breast Cancer treatment. Am J Phys Med Rehabil 1:639–655. https://doi.org/10.1097/PHM.0000000000000455
Helms G, Ku T, Moser L, Remmel E, Kreienberg R (2009) Shoulder-arm morbidity in patients with sentinel node biopsy and complete axillary dissection - data from a prospective randomised trial. J Cancer Surg 35:696–701. https://doi.org/10.1016/j.ejso.2008.06.013
Shamley DR, Srinanaganathan R, Weatherall R, Oskrochi R, Watson M, Ostlere S, Sugden E (2007) Changes in shoulder muscle size and activity following treatment for breast cancer. Breast Cancer Res Treat 106(1):19–27. https://doi.org/10.1007/s10549-006-9466-7
Levy EW, Pfalzer LA, Danoff J, Springer BA, McGarvey C, Shieh CY, Morehead-Gee A, Gerber LH, Stout NL (2012) Predictors of functional shoulder recovery at 1 and 12 months after breast cancer surgery. Breast Cancer Res Treat 134(1):315–324. https://doi.org/10.1007/s10549-012-2061-1
Dean L, DeMichele A, LeBlanc M, Stephens-Shields A, Li S (2015) Black breast Cancer survivors experience greater upper extremity disability. Breast Cancer Res Treat 154(1):117–125. https://doi.org/10.1016/j.bbamem.2015.02.010.Cationic
Blinder VS, Griggs JJ (2013) Health disparities and the cancer survivor. Semin Oncol 40(6):796–803. https://doi.org/10.1053/j.seminoncol.2013.09.003
Hendley Y, Zhao L, Coverson DL, Din-Dzietham R, Morris A, Quyyumi AA, Gibbons GH, Vaccarino V (2011) Differences in weight perception among blacks and whites. J Women’s Heal 20(12):1805–1811. https://doi.org/10.1089/jwh.2010.2262
Race, Ethnicity and Language in South Africa | World Elections. https://welections.wordpress.com/guide-to-the-2014-south-african-election/race-ethnicity-and-language-in-south-africa/. Published 2014. Accessed September 18, 2017
Kootstra JJ, Dijkstra PU, Rietman H, de Vries J, Baas P, Geertzen JHB, Hoekstra HJ, Hoekstra-Weebers JEHM (2013) A longitudinal study of shoulder and arm morbidity in breast cancer survivors 7 years after sentinel lymph node biopsy or axillary lymph node dissection. Breast Cancer Res Treat 139(1):125–134. https://doi.org/10.1007/s10549-013-2509-y
Gosselink R, Rouffaer L, Vanhelden P, Piot W, Troosters T, Christiaens MR (2003) Recovery of upper limb function after axillary dissection. J Surg Oncol 83(4):204–211. https://doi.org/10.1002/jso.10271
Torres-Lacomba M, Sánchez-Sánchez B, Prieto-Gómez V, Pacheco-da-Costa S, Yuste-Sánchez MJ, Navarro-Brazález B, Gutiérrez-Ortega C (2015) Spanish cultural adaptation and validation of the shoulder pain and disability index, and the oxford shoulder score after breast cancer surgery. Health Qual Life Outcomes 13(1):63. https://doi.org/10.1186/s12955-015-0256-y
Roach K, Budiman-Mak E, Songsiridej N, Lertratanakul Y (1991) Development of a shoulder pain and disability index. Arthritis Care Res 4(4):143–149
Shamley D, Robb K (2015) An early warning surveillance programme for detecting upper limb deterioration after treatment for breast cancer : a novel technology supported system. BMC Cancer 15:1–4. https://doi.org/10.1186/s12885-015-1636-8
Funding
Funding for publication was via an NRF grant (RRR Grant 93541).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical clearance for this study was granted by the University of Cape Town Human Research Ethics Committee (approval number: 317/2017). This study was conducted in accordance with the Declaration of Helsinki (last updated in 2013) and International guidelines for Good Clinical Practice (ICH 1997).
Conflict of interest
The authors declare that they have no conflict of interest.
Appendix 1: SPADI Questionnaire
Appendix 1: SPADI Questionnaire
Pain Scale
How severe is your pain?
Circle the number that best describes your pain where: 0 = no pain at all and 10 = the worst pain imaginable.
1.At its worst? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
2.When lying on the involved side? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
3.Reaching for something on a high shelf? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
4.Touching the back of your neck? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
5.Pushing with the involved arm? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
Disability Scale
How much difficulty do you have?
Circle the number that best describes your experience where: 0 = no difficulty and 10 = so difficult it requires help.
1.Washing your hair? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
2.Washing your back? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
3.Putting on an undershirt or jumper? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
4.Putting on a shirt that buttons down the front? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
5.Putting on your pants? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
6.Placing an object on a high shelf? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
7.Carrying a heavy object of 10 pounds (4.5 kg) | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
8.Removing something from your back pocket? | 0 | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
Rights and permissions
About this article
Cite this article
Kramer, N., Ramjith, J. & Shamley, D. Prevalence of shoulder morbidity after treatment for breast Cancer in South Africa. Support Care Cancer 27, 2591–2598 (2019). https://doi.org/10.1007/s00520-018-4540-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-018-4540-3