Abstract
Purpose
The aim of this study was to investigate the feasibility of tongue strength measures (TSMs) and the influence of bulb location, sex, and self-perceived pain and mucositis in head and neck cancer (HNC) patients during chemoradiotherapy (CRT).
Methods
Twenty-six newly diagnosed HNC patients treated with CRT performed anterior and posterior maximal isometric tongue pressures by means of the Iowa Oral Performance Instrument (IOPI). The Oral Mucositis Weekly Questionnaire (OMWQ) and a Visual Analogue Scale (VAS) for pain during swallowing were completed weekly from baseline to 1 week post CRT.
Results
Feasibility of TSMs during CRT declines significantly from 96 to 100% at baseline to 46% after 6 weeks of CRT. But post-hoc analyses reveal only significant differences in feasibility between baseline and measurements after 4 weeks of treatment. No effect of gender or bulb location was established, but feasibility is influenced by pain and mucositis.
Conclusions
Feasibility of TSMs declines during CRT and is influenced by mucositis and pain. For the majority of subjects, TSMs were feasible within the first 4 weeks, which provides a window of scientific and clinical opportunities in this patient population.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
HNC is one of the most distressing cancers with a major impact on quality of life (QoL) [1]. The improvement of tumor response, loco-regional control and survival by using organ-sparing (chemo)radiotherapy (CRT) in the last decade, is unfortunately not reflected in a function-sparing outcome [2,3,4,5]. Sequelae of CRT such as pain, oedema, xerostomia, and ongoing fibrosis negatively impact mouth opening, chewing, speech, and swallowing [6, 7].
Dysphagia is a common, and one of the most serious and disabling complications associated with CRT in HNC patients, but underreported [8]. CRT-associated collateral damage to healthy tissues involved in the oropharyngeal swallow is often inevitable. The acute radiation effects and ever-continuing radiation-induced fibrosis ultimately result in muscular disuse or atrophy, contributing to the decline in swallowing function [6, 8, 9]. This results primarily in difficulties with adequate and safe transportation of food and/or liquids from the mouth to the pharynx and subsequently into the esophagus, which can lead to residue and aspiration [10]. Secondary complications of dysphagia can include prolonged meal duration, malnutrition, feeding tube dependency, hospitalization for treatment of pain or weight loss, aspiration pneumonia, and increased mortality [8, 11]. All these aspects, directly or indirectly, can negatively impact a person’s QoL [12,13,14].
Acute dysphagia (defined as problems during ongoing CRT or shortly afterwards) has often been considered of lesser importance by clinicians due to its transient nature. However, a pioneering study demonstrates muscle deterioration even shortly after completion of CRT caused by reduced strength, atrophy, and fatty infiltration [15]. Due to this insight and the notion that radiotherapy(RT)-induced fibrosis syndrome is a never-ending, progressive process, the importance of prophylactic exercises is increasingly acknowledged. Data indicating that prophylactic swallowing therapy may prevent or limit long-term swallowing CRT-induced swallowing disorders are accumulating [14, 16,17,18,19,20,21]. Due to a lack of sufficient knowledge on functional and physiological changes during CRT, a consensus on therapy content is missing [22].
One important underlying mechanism of dysphagia in HNC patients following CRT is reduced tongue strength (TS) due to the described muscular disuse and/or muscle atrophy [23, 24]. TS is the main driving force for food propulsion [10]. Besides bolus propulsion, insufficient TS is associated with aspiration and endangers adequate oral nutrition [25,26,27]. This pivotal role of TS merits a more profound knowledge and forms the basis of our research question.
Measuring tongue strength in HNC patients during and following their treatment will raise the knowledge and insight in the described process of muscle deterioration. In the long run, profound knowledge on the evolution of tongue strength is necessary to develop therapeutic interventions to prevent and rehabilitate oropharyngeal dysphagia. Although measuring TS during CRT has both a clinical and scientific value, the feasibility of these measures has not yet been documented.
To measure TS, intra-oral electrodes often fixed to a hard shield plate or, an air-filled balloon such as the Iowa Oral Performance Instrument IOPI [28] are used to register the tongue-palate pressures generated by the patient. This implies surface contact between mucosa of the tongue and hard palate and the electrode or plastic bulb, respectively. Extensive normative data collection in healthy adults has verified the feasibility of measuring TS by the IOPI [29,30,31,32]. However, CRT-treated HNC patients must deal with oral mucositis as an acute side effect, which can cause pain and soreness in the mouth and throat [33, 34]. Therefore, TSMs by means of a device during CRT have been regarded controversial and consequently have not been investigated.
The main aim of this study was to investigate the feasibility of TSMs in CRT-treated HNC patients at baseline, weekly during CRT and 1 week after completion of the treatment. Secondary aims are examining the influence of anterior or posterior bulb location, sex, and self-perceived effects of pain and mucositis on feasibility. Our research hypotheses are:
-
(1)
For the majority of the HNC patients, it is feasible to measure tongue strength during CRT, although feasibility will decline during the treatment.
-
(2)
There will be a significant difference between the feasibility of anterior and posterior TSMs because the anterior bulb position will be more easy to tolerate by the subjects.
-
(3)
There is no significant gender effect in feasibility.
-
(4)
There is a significant impact of perceived pain and mucositis on feasibility.
Methods
Material and procedure
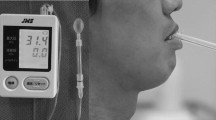
The Iowa Oral Performance Instrument (IOPI [28], Fig. 1) with an air-filled bulb was used to perform TSMs. This instrument is the international standard method for research and use in clinical practice [1, 11, 17, 23,24,25, 29, 31, 32, 34, 35]. For anterior TSMs, the distal end of the air filled balloon was placed right behind the upper incisors; for posterior TSMs, the tip of the balloon was positioned at the transition between the soft and hard palate (Fig. 2). Participants were instructed to generate maximal isometric tongue-palate pressures (MIP), pushing the tongue as hard as possible against the tongue bulb for 3 s. These motivated trials using verbal encouragement were repeated 3 times anteriorly and 3 times posteriorly with 10-s breaks between consecutive measurements. The examiner visually assessed correct strip placement between each trial.
Data collection
TSMs were performed prior to CRT (baseline, BL), after 1, 2, 3, 4, 5, and 6 weeks of CRT (CRT1, CRT2, CRT3, CRT4, CRT5, CRT6), and 1 week post CRT (post CRT). Feasibility was expressed as the percentage of participants able to perform 3 consecutive anterior or posterior TSMs. TSMs were only classified as feasible if the subject felt unrestricted and able to produce maximal tongue-palate pressures.
The Oral Mucositis Weekly Questionnaire-Head and Neck Cancer or OMWQ-HN [36] was used to investigate self-perceived effects of mucositis. The OMWQ-HN is a validated patient reported outcome (PROM) questionnaire that measures the symptoms of mucositis, including mouth and throat soreness and their impact on patient’s well-being and function. It consists of 6 questions with a maximum score of 61; the higher the score, the higher the impact of mucositis on well-being and function. Subjects also completed a 100 mm visual analogue scale (VAS)—ranging from “no pain at all at swallowing” (0) to “swallowing is extremely painful” (100).
Data analysis
Statistical analysis was performed using SPSS software (IBM Corp. Released 2012. IBM SPSS Statistics for Windows, Version 21.0. Armonk, NY). Descriptive analyses were used to calculate the overall feasibility and the feasibility at each investigated point in time. The effects of time on feasibility of TSMs was investigated by Cochrane’s Q test, supplemented with McNemar tests with Holm-Bonferoni correction as post-hoc analysis [37]. The effect of gender and bulb location was determined by chi-square (χ2) analyses. The effect of pain during swallowing and self-perceived mucositis was investigated by comparing global results on VAS and OMWQ between the group of feasible TSMs (group 1) and the group of unfeasible TSMs (group 2).
Ethical committee
This trial was not registered, but this study was approved by the Ethical Committee of the Antwerp University Hospital, Belgium (B300201318333). All subjects agreed voluntarily to participate in this study and signed an informed consent.
Results
Participants
This paper presents data of 26 subjects collected at the Antwerp University Hospital in the context of the Cancer Plan Project KPC29_033 on swallowing between August 2012 and April 2015. During this period, all patients with a new diagnosis of a primary squamous cell carcinoma in the head-neck region, including oral cavity, oropharynx, hypopharynx, and/or larynx, and meeting the inclusion/exclusion criteria were invited to participate in the Cancer Plan study. Inclusion criteria were the presence of sufficient cognitive and language abilities and CRT as unique scheduled treatment. A history of prior carcinoma and/or cancer surgery or CRT in the head and neck region and presence of metastasis were essential exclusion criteria. The Cancer Plan Project is a multicentric collaboration between different Belgian treatment centers, but for this specific research topic, only patients from the coordinating center were included. Table 1 provides detailed information on age, gender, tumor size, and treatment of each subject. From all oropharynx-tumors, there were 8 tonsil cancers and 9 base of tongue cancers (1 was not specified in the patient file).
Feasibility of tongue strength measures
We used an alpha level of .05 for all statistical tests. Figure 3 illustrates the evolution of TSMs feasibility during CRT. This evolution is highly significant (p < .001) with an almost linear decline of feasibility during CRT. Figure 3 shows TSMs to be feasible in the majority of participants until CRT5, followed by a drop in feasibility. Post-hoc analyses showed statistically significant differences in feasibility of anterior TSMs between baseline and CRT4 (p = .040), CRT5 (p = .024), CRT6 (p = .000), and post CRT (p = .048). For posterior TSMs, statistically significant differences were found between baseline and CRT4 (p = .016), CRT5 (p = .010), CRT6 (p = .000), and post CRT (p = .006), with an additional significant difference between BL and CRT1 (p = .048). There were no other significant effects between baseline and other moments, nor between 2 consecutive moments in time. The effect of bulb location was not significant (χ2 = 0,001; p = .980).
Effect of gender on feasibility
No gender effect was found for either anterior or posterior TSMs (χ2 = 0,715; p = .398 and χ2 = 0,893; p = .345).
Effect of mucositis related symptoms on feasibility
Figures 4 and 5 respectively illustrate the distribution of the OMWQ and VAS values of group 1 (feasible TSMs) and group 2 (unfeasible TSMs). The difference between both groups is highly significant for the OMWQ score (t(128) = 3.154, p = .002 for ANT TSMs and t(128) = 3.570, p = .001 for POST TSMs), as well as for the pain during swallowing (t(153) = 3.497, p = .001 for ANT TSMs and t(153) = 3.611, p < .001 for POST TSMs). The scores of group 2 are higher for both variables, indicating a higher self-perceived presence and impact of mucositis. Nonetheless, Figs. 4 and 5 show a substantial overlap between the values of both groups.
Discussion
As discussed in the introduction, TSMs during CRT have a high clinical and scientific relevance. Since TSMs and/or tongue strengthening exercises (TSE) involve direct contact between the oral mucosa and the measuring/therapy device, the feasibility of these activities has been questioned.
The results of this study show a significant decline in feasibility from 96 to 100% anteriorly and posteriorly, respectively, at baseline to 46% for both locations after 6 weeks of CRT. Post-hoc analyses reveal significant decrease in feasibility from 4 weeks of treatment on. A slight but remarkable decrease in feasibility is situated between baseline and the first week of CRT. A conceivable explanation is fear or stress arising with starting the treatment.
No significant effect of gender or bulb location was found, but feasibility is clearly influenced by (self-perceived) mucositis and pain. The latter stresses the presumable importance of pain management in this population [22, 29]. Adequate pain management is not only necessary for preservation of swallowing function and eating during CRT [38] but it also creates opportunities for prophylactic swallowing interventions.
However, the substantial overlap in scores for mucositis and pain between the feasible and non-feasible group also shows that the relationship between pain and mucositis is not straight forward. This implies that those factors are insufficient to guide clinicians whether to continue TSE or not. Based on our observations during the study, we hypothesize that variables like intrinsic motivation and personality traits are influencing factors. These variables were not documented in this study but should be subject of further research.
Our study is the first to demonstrate the feasibility of TSMs with a device which implicates surface contact with the—often painful—tissue of the tongue and palate during CRT.
The main limitation of this pilot study is the relative small number of subjects and the monocentric design. The limited sample size is largely explained by the strict inclusion/exclusion criteria, as well as lack of patients’ motivation. Patients in our study group with tumors in the oral cavity are standard treated with surgery and therefore not included in this study. We assume that these patients would have more burden from their mucositis on the surfaces in contact with the bulb and thereby experience more pain which may lead to a lower feasibility.
In summary, the high feasibility during the first 4–5 weeks of treatment creates opportunities to collect data about evolution of TS during (chemo)radiotherapy. In addition, it provides support for the use of prophylactic tongue strengthening exercise regimens during CRT. This opens a window of opportunities to expand our knowledge on the acute physiological impact of CRT, as well as the feasibility of TSE in a prophylactic swallowing setting. The results of this study cannot be generalized to other HNC-patients, like patients treated with surgery as primary treatment modality. The data about TSMs obtained during this research will create interesting insights in the evolution from TS during this treatment, but these data will be published in an upcoming manuscript.
References
Lazarus CL, Husaini H, Falciglia M, DeLacure RC, Branski D, Kraus D, Lee N, Ho M, Ganz C, Sith B, Sanfilippo N (2014) Effect s of exercise on swallowing and tongue strength in patients with oral and oropharyngeal cancer treated with primary radiotherapy with or without chemotherapy. Int J Oral Maxillofac Surg 43(5):523–530. doi:10.1016/j.ijom.2013.10.023
Pignon JP, le Maîre A, Maillard E, Bourhis J, MACH-NC Collaborative Group (2009) Meta-analysis of chemotherapy in head and neck cancer (MACH-NC) : an update on 93 randomised trials and 17,346 patients. Radiother Oncol 92(1):4–14. doi:10.1016/j.radonc.2009.04.014
Lefebvre JL, Chevalier D, Luboinski B, Kirkpatrick A, Collette L, Sahmoud T (1996) Larynx preservation in pyriform sinus cancer: preliminary results of a European Organization for Research and Treatment of Cancer phase III trial. EORTC Head and Neck Cancer Cooperative Group. J Natl Cancer Inst 88:890–899
Forastiere AA, Goepfert H, Maor M, Pajak TF, Weber R, Morrison W, Glisson B, Trotti A, Ridge JA, Chao C, Peters G, Lee DJ, Leaf A, Ensley J, Cooper J (2003) Concurrent chemotherapy and radiotherapy for organ preservation in advanced laryngeal cancer. New Engl J Med 349:2091–2098
Maurer J, Hipp M, Schäfer C, Kölbl O (2010) Dysphagia. Impact on quality of life after radio(chemo)therapy of head and neck cancer. Strahlenther Onkol 11:744–749. doi:10.1007/s00066-011-2275-x
Raber-Durlacher JE, Brennan MT, Verdonck-de Leeuw IM, Gibson RJ, Eilers JG, Waltimo T, Bots CP, Michelet M, Sollecito TP, Rouleau TS, Sewnaik A, Bensadoun R-J, Fliedner MC, Silverman S Jr, Spijkervet FKL, Dysphagia Section, Oral Care Study Group, Multinational Association of Supportive Care in Cancer (MASCC)/International Society of Oral Oncology (ISOO) (2010) Swallowing dysfunction in cancer patients. Support Care Cancer 20:433–443. doi:10.1007/s00520-011-1342-2
Langendijk JA, Doornaert P, Verdonck-de Leeuw IM, Leemans CR, Aaronson NK, Slotman BJ (2008) Impact of late treatment-related toxicity on quality of life among patients with head and neck cancer treated with radiotherapy. J Clin Oncol 26(22):3770–3776. doi:10.1200/JCO.2007.14.6647
Szcesniak MM, Maclean J, Zhang T, Graham PH, Cook IJ (2014) Persistant dysphagia after head and neck radiotherapy: a common and under-reported complication with significant effect on non-cancer-related mortality. Clin Oncol 26:697–703. doi:10.1016/j.clon.2014.08.009
Tedla M, Valach M, Carrau RL, Varga I, Profant M, Mráz P, Weismann P (2012) Impact of radiotherapy on laryngeal intrinsic muscles. Eur Arch of Otorhinolaryngol 169(3):953–658. doi:10.1007/s00405-011-1686-8
Hutcheson KA, Lewin JS, Barringer DA, Lisec A, Gunn B, Moore MWS, Holsinger FC (2012) Late dysphagia after radiotherapy-based treatment of head and neck cancer. Cancer 118(23):5796–5799. doi:10.1002/cncr.27631
De Bodt M, Guns C, D’Hondt M, Vanderwegen J, Van Nuffelen G (2015) Dysfagie. Handboek voor de klinische praktijk. Garant, Antwerpen
List MA, Siston A, Haraf D (1999) Quality of life and performance in advanced head and neck cancer patients on concomitant chemoradiotherapy: a prospective examination. J Clin Oncol 17:1020–1028
Nund RL, Scarinci NA, Cartmill B, Ward EC, Kuipers P, Porceddu SV (2014) Application of the international classification of functioning, disability and health (ICF) to people with dysphagia following non-surgical head and neck cancer management. Dysphagia 29(6):692–793. doi:10.1007/s00455-014-9563-4
Murphy BA, Deng J (2015) Advances in supportive care for late effects of head and neck cancer. Journal of clinical oncology 33(29):3314–3321. doi:10.1200/JCO.2015.61.3836
King SN, Dunlap NE, Tennant PA, Pitts T (2016) Pathophysiology of radiation-induced dysphagia in head and neck cancer. Dysphagia 31(3):339–351. doi:10.1007/s00455-016-9710-1
Rosenthal D, Lewin J, Eisbruch A (2006) Prevention and treatment of dysphagia and aspiration after chemoradiation for head and neck cancer. J Clin Oncol 24:2636–2643
Virani A, Kunduk M, Fink DS (2015) Effects of 2 different swallowing exercise regimens during organ-preservation therapies for head and neck cancers on swallowing function. Head Neck 37(2):162–170. doi:10.1002/hed.23570
Kulbersh BD, Rosenthal EL, McGrew BM, Duncan RD, McColloch NL, Carroll WR, Magnuson JS (2006) Pretreatment, preoperative swallowing exercises may improve dysphagia quality of life. Laryngoscope 116(6):883–886. doi:10.1097/01.mlg.0000217278.96901.fc
Duarte VM, Chhetri DK, Liu YF, Erman AA, Wang MB (2013) Swallow preservation exercises during chemoradiation therapy maintains swallow function. Otolaryngol Head Neck Surg 149(6):878–884. doi:10.1177/0194599813502310
Van der Molen L, Van Rossum MA, Burkhead LM, Smeele LE, Rasch CRN, Hilgers FJM (2011) A randomized preventive rehabilitation trial in advanced head and neck cancer patients treated wih chemoradiotherapy: feasibility, compliance, and short-term effects. Dysphagia 26:155–170. doi:10.1007/s00455-010-9288-y
Carnaby-Mann G, Crary MA, Schmalfuss I, Amdur R (2012) "Pharyngocise": randomized controlled trial of preventative exercises to maintain muscle structure and swallowing function during head-and-neck chemoradiotherapy. Int J Radiat Oncol Biol Phys 83(1):210–219. doi:10.1016/j.ijrobp.2011.06.1954
Schindler A, Denaro N, Russi EG, Pizzorni N, Bossi P, Merlotti A, Spadola Bissetti M, Numico G, Gava A, Orlandi E, Caspiani O, Buglione M, Alterio D, Bacigalupo A, De Sanctis V, Pavanato G, Ripamonti C, Merlano MC, Licitra L, Sanquineti G, Langendijk JA, Murphy B (2015) Dysphagia in head and neck cancer patients treated with radiotherapy and systemic therapies: literature review and consensus. Crit Rev Oncol Hematol 96(2):372–384. doi:10.1016/j.critrevonc.2015.06.005
Lazarus CL, Logemann JA, Pauloski BR (2007) Effects of radiotherapy with or without chemotherapy on tongue strength and swallowing in patients with oral cancer. Head Neck 29:632–637
Lazarus CL, Logemann JA, Pauloski BR (2000) Swallowing and tongue function following treatment for oral and oropharyngeal cancer. J Speech Lang Hear Res 43:1011–1023
Butler SG, Stuart A, Leng X, Wilhelm E, Rees C, Williamson J, Kritchevsky SB (2011) The relationship of aspiration status with tongue and handgrip strength in healthy older adults. J Gerontol A Biol Sci Med Sci 66(4):452–458. doi:10.1093/gerona/glq234
Ku PK, Yuen EH, Cheung DM, Chan BY, Ahuja A, Leung SF, Tong MC, van Hasselt A (2007) Early swallowing problems in a cohort of patients with nasopharyngeal carcinoma: symptomatology and videofluoroscopic findings. Laryngoscope 117(1):142–146
Smith RV, Kotz T, Beitler JJ, Wadler S (2000) Long-term swallowing problems after organ preservation therapy with concomitant radiation therapy and intravenous hydroxyuera: initial results. Arch Otolaryngol Head Neck Surg 126(3):384–389
Iowa Oral Performance Instrument. http://www.iopimedical.com Accessed 14th April 2016.
Vanderwegen J, Guns C, Van Nuffelen G, Elen R, De Bodt M (2013) The influence of age, sex, bulb position, visual feedback, and the order of testing on maximum anterior and posterior tongue strength and endurance in healthy Belgian adults. Dysphagia 2:159–166. doi:10.1007/s00455-012-9425-x
Oh DH, Park JS, Jo YM, Chang M (2016) Differences in maximal isometric tongue strength and endurance of healthy young vs. older adults. J Phys Ther Sci 3:854–856. doi:10.1589/jpts.28.854
Mendes AE, Nascimento L, Mansur LL, Callegaro D, Jacob Filho W (2015) Tongue forces and handgrip strength in normal individuals: association with swallowing. Clinics 70(1):41–45. doi:10.6061/clinics/2015(01)08
Todd JT, Lintzenich CR, Butler SG (2013) Isometric and swallowing tongue strength in healthy adults. Laryngoscope 123(10):2469–2473. doi:10.1002/lary.23852
De Sanctis V, Bossi P, Sanguineti G, Trippa F, Ferrari D, Bacigalupo A, Ripamonti CI, Buglione M, Pergolizzi S, Langendijk JA, Murphy B, Raber-Durlacher J, Russi EG, Lalla RV (2016) Mucostis in head and neck cancer patients treated with radiotherapy and systemtic therapies: literature review and consensus statements. Crit Rev Oncol Hematol 100:147–166. doi:10.1016/j.critrevonc.2016.01.010
Mercadante S, Ajelli F, Adile C, Ferrera P, Valle A, Fusco F, Caruselli A, Cartoni C, Massimo P, Masedu F, Valenti M, Porzio G (2015) Prevalence of oral mucositis, dry mouth, and dysphagia in advanced cancer patients. Support Care Cancer 23(11):3249–3255. doi:10.1007/s00520-015-2720-y
Van Nuffelen G, Van den Steen L, Vanderveken O, Specenier P, Van Laer C, Van Rompaey D, Guns C, Mariën S, Peeters M, Van de Heyning P, Vanderwegen J, De Bodt M (2015) Study protocol for a randomized controlled trial: tongue strengthening exercises in head and neck cancer patients, does exercise load matter? Trials 4(16):395. doi:10.1186/s13063-015-0889-5
Epstein JB, Beaumont JL, Gwede CK, Murphy B, Garden A, Meredith R, Le QT, Brizel D, Isitt J, Cella D (2007) Longitudinal evaluation of the oral mucositis weekly questionnaire-head and neck cancer, a patient-reported outcomes questionnaire. Cancer 109(9):1–9
Holm S (1979) A simple sequentially rejective multiple test procedure. Scand J Stat 6(2):65–70
Britton B, Mc Carter K, Baker A, Wolfenden L, Wratten C, Bauer J, Beck A, Mc Elduff P, Halpin S, Carter G (2015) Eating eating as treatment (EAT) study protocol: a stepped wedge, randomised controlled trial of health behaviour change intervention provided by dietitians to improve nutrition in patients with head and neck cancer undergoing radiotherapy. BMJ Open 5(7):e008921. doi:10.1136/bmjopen-2015-008921
Acknowledgements
This work was funded by Belgian Federal Cancer Plan (KPC29_033).
We thank all researchers from the KP-study group, the patients, their families, and caregivers.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
Mrs. Van den Steen reports grants from Belgian Federal Cancer Plan (KPC29_033) during the conduct of the study.
Rights and permissions
About this article
Cite this article
Van den Steen, L., Vanderveken, O., Vanderwegen, J. et al. Feasibility of tongue strength measurements during (chemo)radiotherapy in head and neck cancer patients. Support Care Cancer 25, 3417–3423 (2017). https://doi.org/10.1007/s00520-017-3761-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-017-3761-1