Abstract
Purpose
This study collected data on the use of ferric carboxymaltose (FCM) in a cancer patient population in France to evaluate the feasibility and the conditions of use of FCM in routine clinical practice beyond the limiting criteria of clinical trials.
Methods
This observational, prospective study of patients with a solid tumour or a haematological malignancy who have received treatment with FCM after 01 July 2011 evaluated data about the circumstances of iron administration, concomitant medication and laboratory tests in the period from 3 months prior to the first FCM administration (baseline) until 3 months post-baseline.
Results
Data from 367 FCM-treated patients were analysed. FCM was mainly given as a single dose at baseline (69.2 %) and without additional erythropoiesis-stimulating agent (ESA, 64.3 %). The median total iron dose was 1000 mg per patient. Median haemoglobin (Hb) levels of FCM-treated patients improved from 10.3 g/dL (interquartile range 9.5, 11.1 g/dL) at baseline to 11.8 g/dL (11.1, 13.0 g/dL) until the end of the 3-month observational period. Patients treated with FCM alone or additional ESA achieved similar median Hb increase (1.3 [0.4, 2.1] g/dL and 1.4 [0.4, 2.5] g/dL, respectively). Patients with baseline Hb up to 11.0 g/dL and serum ferritin up to 500 ng/mL and beyond achieved stable median Hb levels ≥11.0 g/dL without signs of iron overload. No severe or serious adverse reaction and no hypersensitivity reactions were reported.
Conclusions
The results of this observational study confirm the effectiveness and tolerability of FCM when given in clinical routine practice alone or in combination with an ESA.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Guidelines and experts on the treatment of cancer-related anaemia strongly recommend the reduction or prevention of blood transfusions and a restricted use of erythropoiesis-stimulating agents (ESAs) at the lowest required dose [1–4]. Controlled clinical studies in anaemic cancer patients have shown that intravenous (i.v.) iron in combination with ESAs effectively improves haemoglobin (Hb) levels compared to no or oral iron supplementation [2]. Additional controlled studies and an observational study in Germany suggest that also patients without concomitant ESA can benefit from i.v. iron [5–9].
Both anaemia (Hb <12 g/dL) and iron deficiency (ID, transferrin saturation [TSAT] <20 %) are frequent complications in patients with solid tumours (45.9 and 33.0 %, respectively) or haematologic malignancies (35.4 and 33.9 %, respectively) [10]. Anaemia is associated with impaired quality of life (QoL) and may be related to impaired response to cancer treatment and reduced overall survival [11, 12]. ID can be associated with impaired physical function and fatigue symptoms [10, 13–15]. If left undiagnosed or untreated, ID can lead to anaemia [1].
In routine practice, iron status is assessed only at low frequency [16] although assessment of haematopoiesis and iron metabolism in patients eligible for ESA therapy identified 21 % of patients to be absolute iron-deficient, defined by elevated ferritin index, and 29 % of patients to have absolute and/or functional iron deficiency, defined by elevated ferritin and/or diminished Hb content of reticulocytes, before the start of anaemia treatment [17]. Iron therapy, if given at all, is mainly based on oral iron despite that clinical studies have shown that oral iron had limited effect in ESA-treated cancer patients [18, 19]. Stable iron complexes can be administered at doses up to 20 mg iron/kg body weight with infusion times varying between 15 min and 6 h [20]. Ferric carboxymaltose (FCM; Ferinject®; Vifor Pharma, Switzerland) is a stable iron carbohydrate complex for i.v. iron administration, which provides controlled release of iron into the metabolic pathway and a positive benefit/risk profile for the treatment of ID and iron deficiency anaemia (IDA) in many acute and chronic conditions [20].
The study reported here aimed to collect data on the use (e.g. indication, dose and frequency of administration, and combination with other anaemia therapies) of FCM in an overall oncological patient population in France to evaluate the feasibility and the conditions of use of FCM in routine clinical practice beyond the limiting inclusion and exclusion criteria of clinical trials.
Methods
Study design and population
The study was designed as an observational, prospective study in 45 active centres in France. Eligible cancer patients had to be at least 18 years of age, diagnosed with a solid tumour or a haematological malignancy, provide informed consent and receive treatment with FCM after 01 July 2011. The prospective enrolment period was from 1 July 2011 to January 2013. Patients who have been enrolled in a parallel study on the use of iron sucrose and red blood cell (RBC) transfusions in 2010 were excluded from enrolment. Patients receiving an investigational anaemia therapy or being actively enrolled in another investigational anaemia study or who had not completed such a study by at least 30 days before baseline were excluded from enrolment. The study was conducted in line with the International Conference on Harmonisation Good Clinical Practice (ICH-GCP, CPMP/ICH/135/95) and the Declaration of Helsinki and in compliance with all applicable local and national guidelines and regulations.
Patient management and data collection
The frequency of study visits as well as the performed diagnostic tests and treatments was left to the investigator’s discretion according to their routine practice. The first day of FCM treatment was defined as baseline, and the observational period was scheduled for 3 months.
Anonymised data from patient records were collected by the investigator or a clinical research assistant via paper case report forms. At baseline, data on patient demographics, cancer type and treatments, reasons for initiation of iron treatment, and laboratory tests (Hb, mean corpuscular volume [MCV], mean corpuscular haemoglobin concentration [MCHC], mean corpuscular haemoglobin [MCH], serum iron, serum ferritin, serum transferrin, TSAT, soluble transferrin receptor [sTfrR], white cell count, platelets, C-reactive protein [CRP], aspartate aminotransferase [AST], alanine aminotransferase [ALT], creatinine, total bilirubin, calcium, phosphate) were recorded when available. Furthermore, all available data on laboratory tests and concomitant medications in the period from 3 months prior (M−3) to the first FCM administration until 3 months post-baseline (M+3) were recorded. All observed adverse and serious adverse drug reactions (related or possibly drug-related events), pregnancies and special events such as medication abuse, misuse, overdose and medication errors that occurred during the observational period were recorded.
The primary objective was to describe the indication and administered dose of FCM, the frequency, route and duration of FCM administration and the location of administration (hospital, private practice, patient’s home).
Data analysis
Categorical data are summarised by number and percentage; continuous data are presented as mean and median values and interquartile range (Q1, Q3 [25 %, 75 % percentile]) or full range (minimum–maximum). Missing values were not imputed. The statistical analysis was performed by the contract research organisation ICTA PM (Fontaine-les-Dijon, France). Statistical tests for differences comprised the one-sided Wilcoxon signed rank-test for paired samples and the two-sided Wilcoxon-Man-Whitney test with t approximation for non-Gaussian variables.
In order to assess the actual treatment effect of FCM, all Hb laboratory data and iron status data of patients who received a RBC transfusion after or more than 1 month prior to the first FCM dose were censored from analysis during 1 month after the RBC transfusion. Patients who have received a RBC transfusion within 1 month prior to the first FCM dose were excluded from the population. Hb and iron status data were also censored from analysis from the time patients have received i.v. iron sucrose.
Additional analysis was performed without any censoring, leading to the same conclusions as analysis of censored data.
Collected data were subjected to data queries to ensure data quality and consistency of the database. A quality control of the data entry (30 randomly selected patient records) confirmed that the accepted 1 ‰ rate of inconsistencies was respected.
The necessary sample size was estimated to 405 patients based on a required precision of at least 5 % for the expected proportions and a usual alpha risk level of 5 % (i.e. 95 % CI). Furthermore, a 5 % lost to follow-up or non-evaluable patients over the 3-month follow-up period was anticipated (based on an approximately 10 % reported in the ECAS study after 6 months) [21].
Results
Patient characteristics
Of 413 enrolled cancer patients that were included in the study set, data from 367 patients were analysed (Fig. 1).
Median age of patients was 64 years (21–90 years); 44 % were male (Table 1). The majority of patients (96 %) have presented with solid tumours; main malignancies were gastrointestinal tumours (38 %) and breast cancer (24 %). Others had tumours at multiple or unspecified sites (2.2 %) or haematological malignancy (2 %). Metastatic disease was reported for 55 %, and 68 % have received ongoing cytotoxic chemotherapy at baseline. The median creatinine level was 68.1 μmol/L with 27.0 % of patients having an abnormal creatinine level.
Of the 367 analysed patients, 180 (49.0 %) have completed the study at the theoretical month 3 post-baseline visit (M+3). Among the other 187 patients, 130 (35.4 %) did not reach the theoretical 90-day observational period (but were considered by their physician as having completed the study), 37 (10.1 %) died (all deaths considered as unrelated to the study drugs) and 15 (4.1 %) were lost to follow-up. No patient withdrew consent, 4 (1.1 %) stated other reasons for early termination (Fig. 1).
Overall, 39 (10.6 %) deaths were recorded during the observational period from baseline to M+3. Most deaths were due to disease progression (89.7 %), and one (2.6 %) due to general physical health deterioration, pneumonia and digestive haemorrhage, respectively. In one case, reason of death was missing; all deaths were considered unrelated to the study drug.
Circumstances of iron administration and RBC transfusions
Most patients (69.2 %) received their FCM treatment as a single dose at baseline (Fig. 2a, b). Only a minority of patients had a treatment duration ≥1 month (Fig. 2b). Median total iron dosage was 1000 mg (250–5000 mg). FCM was almost exclusively administered via i.v. infusion (99.1 vs. 0.9 % via bolus injection), and most administrations were given via a central vein (75.9 vs. 24.6 % via a peripheral vein). Based on physician assessment, 57 % of patients were iron-deficient at baseline. Of those, 44.8 % had iron deficiency without anaemia and 55.2 % had iron deficiency anaemia.
FCM treatment was given without any additional ESA in 64.3 %. The monthly cumulative median ESA dose increased from 100,000 IU (68,571, 132,142 IU) at baseline to 132,857 IU (76,190, 147,619 IU) at M+3 (p = 0.007 M+3 vs. baseline). During the period from M−3 to M+3, RBC transfusions were given to 19.3 % of patients (Table 2). A RBC transfusion was given to 6.8 % before baseline and 15.3 % after baseline. Four patients (1.1 %) received iron sucrose and 15 (4.1 %) received oral iron after the initial FCM administration.
Iron administration was almost exclusively performed by nurses (99.7 %). The majority of patients (64.9 %) have received the iron treatment at home; the rate of iron administrations in private practice was negligible (1.9 %).
Effectiveness of FCM and Hb evolution over time
Mean Hb levels steadily improved after the first iron dose (Fig. 3a). FCM-treated patients achieved a median Hb of 11.1 g/dL (10.0, 12.0 g/dL) 1 month post-baseline, and median Hb levels remained in the range of 11.0–12.0 g/dL until the end of the 3-month follow-up period. In FCM-treated patients with or without additional ESA, median Hb levels improved by 1.6 and 1.3 g/dL, respectively, during the 3-month observational period after the first FCM dose, and both subgroups reached levels >11.0 g/dL (Fig. 3b). Notably, patients who have been treated with additional ESA had lower median Hb levels at baseline than those who have received FCM alone (9.9 vs. 10.7 g/dL; p = 0.007).
Stratification of FCM-treated patients by baseline Hb and baseline ferritin showed that median Hb levels improved in all subgroups (Fig. 3c, d). For patients with baseline Hb >11 g/dL who maintained stable Hb levels, the median Hb level did not exceed normal levels during the follow-up period.
Censoring data of FCM-treated patients who had received a RBC transfusions or iron sucrose treatment did not significantly influence the calculated median Hb levels at baseline and the end of the observational period (uncensored: Hbbaseline 10.3 [9.5, 11.1], HbM+3 11.7 [10.8,12.8] g/dL; censored: Hbbaseline 10.3 [9.5, 11.1], HbM+3 11.8 [11.1, 13.0] g/dL; p < 0.001 for HbM+3 vs. Hbbaseline).
Serum ferritin and TSAT evolution over time
Baseline serum ferritin and TSAT were assessed in 54.0 and 53.7 % of patients, respectively. Among the tested patients, mean serum ferritin was elevated during the entire observational period (baseline, 245 [89, 601] ng/mL; M+1, 782 [503, 1152] ng/mL; M+2, 685 [406, 1072] ng/mL; M+3, 514 [283, 952] ng/mL; p < 0.001 for all time points vs. baseline). A baseline TSAT <20 % was detected in 90.8 % of patients and a TSAT >50 % in 1 %. TSAT improved from 13.0 [9.2, 16.9] % at baseline to 22.1 [15.0, 28.4] % at M+1 and remained in this normal range over the entire observation period (Fig. 4).
C-reactive protein
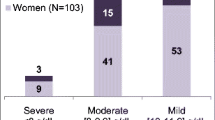
Baseline CRP was assessed in 29.4 % of patients. Among the tested patients, 70.4 % had elevated baseline CRP levels. Of note, patients with strong underlying inflammation (CRP >49 mg/L) responded well to FCM treatment and achieved a median Hb of 11.6 g/dL (10.8, 13.1 g/dL) at the end of the study. Notably, this subgroup had lower Hb levels at baseline (9.7 [9.0, 10.4] g/dL) and during the 3-month period before treatment compared to patients in the categories with baseline CRP <7.5 mg/L (baseline Hb 11.9 [10.8, 12.9] g/dL) and baseline CRP 7.5–49 mg/L (baseline Hb 10.7 [9.7, 11.2] g/dL), respectively.
Tolerability and adverse reactions
Neither a severe nor serious adverse reaction as well as no adverse reaction leading to death was reported (Table 3). Adverse reactions were experienced by 12.1 % (in 8.7 % due to FCM and 3.4 % due to iron sucrose). The most common FCM-related adverse reaction was hypophosphataemia (6.1 %). Median blood phosphate levels transiently decreased from 1.0 mmol/L (0.9, 1.2 mmol/L) at baseline to 0.9 mmol/L (0.7, 1.1 mmol/L) after 1 month and recovered to 1.0 mmol/L (0.8, 1.2 mmol/L) and 1.0 mmol/L (0.9, 1.2 mmol/L) after 2 and 3 months, respectively. No hypersensitivity reaction was reported in this study.
Discussion
This observational study in cancer patients documents routine practice conditions of i.v. iron treatment with ferric carboxymaltose (FCM). In order to assess the actual effect of the FCM treatment, several data were censored, once a RBC transfusion or iron sucrose was received. Globally, results were not different between analyses of censored and non-censored data.
FCM was most commonly given at home, reducing the need for hospital visits, and has been mainly administered as a single dose at baseline (median total iron dose per patient 1000 mg). After the conduct of this study, the European Medicines Agency published the results of their review of the safety and exposure data of all i.v. iron products which concluded that all i.v. iron products have a positive benefit-risk balance but nevertheless mandated measures intending to maximise their safe use. Accordingly, oncology patients who are routinely treated in day hospitals in France now also receive their i.v. iron treatment in this outpatient setting.
Interestingly, the majority of FCM-treated patients have received their iron treatment without additional ESA. Whether this is a general trend towards treatment practice in line with guidelines and recommendations to minimise ESA doses [1–4] or if this is related to the higher single iron dose that can be administered with FCM compared to iron sucrose cannot be concluded from the available data. Similarly and also in line with long-standing recommendations to prevent or minimise RBC transfusions, a relatively low proportion (approximately one fifth) of patients have received a transfusion.
Overall, FCM treatment improved median Hb and TSAT to levels ≥11 g/dL and >20 % within 1 month after initial treatment. Hb improvement was independent of baseline Hb, and median Hb levels remained stable between 11 and 12 g/dL. In spite of its effectiveness in improving Hb levels, FCM treatment did not increase mean Hb beyond recommended levels which can be the case with ESAs and requires careful Hb monitoring and ESA dose adjustment to ensure that Hb levels do not exceed the target level [1].
In line with controlled studies that showed the efficacy of i.v. iron when given without an additional ESA, also patients who received FCM alone (i.e. without additional ESA) responded well to the iron treatment and achieved a median Hb above 11 g/dL. In fact, the increase in median Hb from baseline to the end of the 3-month observational period was very similar in patients that received an ESA or not (+1.6 and +1.3 g/dL, respectively). However, these subgroups (with or without ESA) have to be considered as two different populations since ESA-treated patients had lower baseline Hb levels. Notably, both subgroups had similar Hb levels 3 months before the first FCM treatment; however, patients who received an ESA had already a strong decrease in Hb levels between months 2 and 1 prior to the first FCM treatment. Earlier initiation of iron treatment with FCM in this subgroup might have avoided the need for additional and costly treatment with an ESA.
The rather low baseline TSAT values are in line with the requirement of a confirmed diagnosis of iron deficiency before treatment with FCM. Notably, 3 months before treatment (M−3), median TSAT in this study population was close to values in an unselected cancer patient population [10]. However, a substantial decrease in median TSAT was already observed 1 month before the start of iron treatment, suggesting that a considerable proportion of patients in this study may have benefited from earlier initiation of i.v. iron (FCM) treatment. Baseline Hb levels and Hb evolution over time were independent of baseline TSAT levels (data not shown).
The high prevalence of elevated CRP levels among patients tested for CRP confirms that cancer is often associated with chronic inflammation. Since utilisation of iron stores is impaired by inflammation-induced cytokines and hepcidin [22, 23], a large proportion of these patients might have been in functional iron deficiency. Since serum ferritin is also an acute phase protein, elevated serum ferritin levels in patients with cancer or other chronic diseases may be more indicative of an underlying inflammation than normal or even elevated iron stores. Also, the observation that patients with baseline serum ferritin levels up to 500 ng/mL responded well to FCM shows that these patients were functionally iron-deficient and did not have iron overload or haemochromatosis. Interestingly, patients with very high baseline ferritin levels (≥500 ng/mL) had the lowest median Hb at baseline and responded to iron treatment by achieving a median Hb level >11.0 g/dL after 2 months. By this observation and the finding that also patients with elevated CRP levels achieved Hb response to FCM, this study confirms the effectiveness of FCM in patients with active or chronic inflammation. This is a key advantage of FCM and other i.v. iron products compared to oral iron which is ineffective in patients with inflammation and functional iron deficiency [1]. The considerable rate of patients with abnormal creatinine levels suggests a substantial prevalence of renal conditions, a typical confounder of anaemia, in this patient population.
Furthermore, the study confirms that FCM, given at a median total dosage of 1000 mg iron, is well tolerated. Neither a hypersensitivity reaction nor drug-related serious adverse events were reported in this study. Hypophosphataemia, the most common adverse reaction that was related to FCM, comprises a transient, clinically asymptomatic reaction that generally resolves spontaneously without treatment and has been already described by earlier studies [20]. Other adverse reactions have been reported with low frequency. The mortality and completion rate are in line with other populations of cancer patients with metastatic disease (55 %).
The actual study protocol also included a retrospective group for the collection of data on the use of iron sucrose and RBC transfusions before the introduction of FCM in France. Since the prospective group of FCM-treated patients (reported here) and the retrospective group treated with iron sucrose and/or RBC transfusions were not comparable due to (expected) differences in demographic data and disease characteristics, only results of the prospective FCM group were included in this manuscript.
In conclusion, the results of this observational study confirm the effectiveness and tolerability of FCM when given in clinical routine practice either alone or in combination with an ESA. The good effectiveness and safety results in FCM-treated patients are in line with an observational study performed in Germany [8] that showed the effectiveness of FCM with or without an additional ESA in a large cancer patient population and confirmed former results of clinical studies showing benefits of i.v. iron alone as anaemia therapy [5–7]. Earlier initiation of parenteral iron therapy, particularly with a compound such as FCM that can be given as a single dose of up to 1000 mg iron, may prevent or reduce the need for use of ESAs or rescue treatment with RBC transfusions.
References
Aapro M, Osterborg A, Gascon P, Ludwig H, Beguin Y (2012) Prevalence and management of cancer-related anaemia, iron deficiency and the specific role of intravenous iron. Ann Oncol 23:1954–1962
Aapro MS, Link H (2008) September 2007 update on EORTC guidelines and anemia management with erythropoiesis-stimulating agents. Oncologist 13(Suppl 3):33–36
National Comprehensive Cancer Network Inc (2014) NCCN Practice Guidelines in Oncology; Cancer and Chemotherapy-Induced Anemia - Version 2.2015. http://www.nccn.org/professionals/physician_gls/f_guidelines.asp#supportive. Accessed 28 October 2014
Rizzo JD, Brouwers M, Hurley P, Seidenfeld J, Arcasoy MO, Spivak JL, Bennett CL, Bohlius J, Evanchuk D, Goode MJ, Jakubowski AA, Regan DH, Somerfield MR (2010) American Society of Hematology/American Society of Clinical Oncology clinical practice guideline update on the use of epoetin and darbepoetin in adult patients with cancer. Blood 116:4045–4059
Athibovonsuk P, Manchana T, Sirisabya N (2013) Prevention of blood transfusion with intravenous iron in gynecologic cancer patients receiving platinum-based chemotherapy. Gynecol Oncol 131:679–682
Dangsuwan P, Manchana T (2010) Blood transfusion reduction with intravenous iron in gynecologic cancer patients receiving chemotherapy. Gynecol Oncol 116:522–525
Kim YT, Kim SW, Yoon BS, Cho HJ, Nahm EJ, Kim SH, Kim JH, Kim JW (2007) Effect of intravenously administered iron sucrose on the prevention of anemia in the cervical cancer patients treated with concurrent chemoradiotherapy. Gynecol Oncol 105:199–204
Steinmetz T, Tschechne B, Harlin O, Klement B, Franzem M, Wamhoff J, Tesch H, Rohrberg R, Marschner N (2013) Clinical experience with ferric carboxymaltose in the treatment of cancer- and chemotherapy-associated anaemia. Ann Oncol 24:475–482
Hedenus M, Karlsson T, Ludwig H, Felder M, Roubert B, Birgegard G (2013) Intravenous Ferric Carboxymaltose As Sole Anemia Therapy In Patients With Lymphoid Malignancies, Chemotherapy-Induced Anemia and Functional Iron Deficiency. Blood 122:3439
Ludwig H, Muldur E, Endler G, Hubl W (2013) Prevalence of iron deficiency across different tumors and its association with poor performance status, disease status and anemia. Ann Oncol 24:1886–1892
Cella D, Kallich J, McDermott A, Xu X (2004) The longitudinal relationship of hemoglobin, fatigue and quality of life in anemic cancer patients: results from five randomized clinical trials. Ann Oncol 15:979–986
Hudis CA, Van BS, Chang J, Muenstedt K (2004) rHuEPO and treatment outcomes: the clinical experience. Oncologist 9(Suppl 5):55–69
Brownlie T, Utermohlen V, Hinton PS, Haas JD (2004) Tissue iron deficiency without anemia impairs adaptation in endurance capacity after aerobic training in previously untrained women. Am J Clin Nutr 79:437–443
Favrat B, Balck K, Breymann C, Hedenus M, Keller T, Mezzacasa A, Gasche C (2014) Evaluation of a Single Dose of Ferric Carboxymaltose in Fatigued, Iron-Deficient Women - PREFER a Randomized, Placebo-Controlled Study. PLoS One 9, e94217
Krayenbuehl PA, Battegay E, Breymann C, Furrer J, Schulthess G (2011) Intravenous iron for the treatment of fatigue in nonanemic, premenopausal women with low serum ferritin concentration. Blood 118:3222–3227
Ludwig H, Aapro M, Bokemeyer C, Glaspy J, Hedenus M, Littlewood TJ, Osterborg A, Rzychon B, Mitchell D, Beguin Y (2014) A European patient record study on diagnosis and treatment of chemotherapy-induced anaemia. Support Care Cancer 22:2197–2206
Steinmetz HT, Tsamaloukas A, Schmitz S, Wiegand J, Rohrberg R, Eggert J, Breuer F, Tessen HW, Eustermann H, Thomas L (2010) A new concept for the differential diagnosis and therapy of anaemia in cancer patients. Support Care Cancer 19:261–269
Bastit L, Vandebroek A, Altintas S, Gaede B, Pinter T, Suto TS, Mossman TW, Smith KE, Vansteenkiste JF (2008) Randomized, multicenter, controlled trial comparing the efficacy and safety of darbepoetin alpha administered every 3 weeks with or without intravenous iron in patients with chemotherapy-induced anemia. J Clin Oncol 26:1611–1618
Henry DH, Dahl NV, Auerbach M, Tchekmedyian S, Laufman LR (2007) Intravenous ferric gluconate significantly improves response to epoetin alfa versus oral iron or no iron in anemic patients with cancer receiving chemotherapy. Oncologist 12:231–242
Munoz M, Martin-Montanez E (2012) Ferric carboxymaltose for the treatment of iron-deficiency anemia. [corrected]. Expert Opin Pharmacother 13:907–921
Ludwig H, Van BS, Barrett-Lee P, Birgegard G, Bokemeyer C, Gascon P, Kosmidis P, Krzakowski M, Nortier J, Olmi P, Schneider M, Schrijvers D (2004) The European Cancer Anaemia Survey (ECAS): a large, multinational, prospective survey defining the prevalence, incidence, and treatment of anaemia in cancer patients. Eur J Cancer 40:2293–2306
Hentze MW, Muckenthaler MU, Galy B, Camaschella C (2010) Two to tango: regulation of Mammalian iron metabolism. Cell 142:24–38
Weiss G, Goodnough LT (2005) Anemia of chronic disease. N Engl J Med 352:1011–1023
Acknowledgments
Preparation and conduct of the study have been sponsored by Vifor France SA. Data Management and biostatistical analyses were performed by ICTA PM, France, and funded by Vifor France SA. Medical writing support in the preparation of the manuscript was provided by Walter Fürst, SFL Regulatory Affairs & Scientific Communication, Switzerland, and funded by Vifor Pharma Ltd., Switzerland. The manuscript was reviewed and commented by Beate Rzychon (Vifor Pharma Ltd., Glattbrugg, Switzerland). The authors would like to thank the local investigators who actively participated in this study: Grégory Dendleux, Centre Hospitalier Pierre Oudot; Gérard Dine, Saïd Brahimi, Audrey Echegasd and Nadia Ali Amar, Centre Hospitalier Troyes; Jean-Loup Mouysset, Clinique Rambot-Provencale; Abderraouf Radji, Centre Frederic Joliot; Laurent Zelek, Kader Chouahnia, Gaetan Des Guetz, Marie-Christine Pailler and Thierry Bouillet, Hopital Avicenne; Pierre Bory, Centre Hospitalier Bastia Cedex; Mihaela Achille, Emmanuel Achille and France Campos, Institut De Cancerologie Prive De L'orangerie; Hamah Lamallem, Hopital Americain De Paris; Faress Husseini, Hopital Louis Pasteur; Claude Boiron, Anne Donnadieu, Centre Rene Huguenin; Bertrand Mennecier, Nouvel Hopital Civil; Jean-Philippe Wagner, Centre De Radiothérapie De Dunkerque; Jérôme Dauba, Hopital Layne; Isabelle Gabelle, Chu De Grenoble; Joel Castelli, Pascale Revole and Pascal Bourlet, Centre Hospitalier De Castelluccio; Younes El Masmoudi, Clinique Des Murlins; Delphine Borchiellini and Axel Leysalle, Centre Antoine Lacassagne; Dominique Jaubert, Nathalie Bonichon-Lamichhane, Hortense Laharie and Christophe Debelleix, Clinique Tivoli; Alain Zannetti and Elouen Boughalem, Centre Hospitalier Cholet Cedex; Louis-Marie Dourthe, Clinique Sainte Anne; Anne Floquet, Dominique Bechade and Marie Sire, Institut Bergonie; Isabelle Ray-Coquard, Centre Leon Berard; Jean-Pierre Crumbach, Centre Hospitalier Freyming Merlebach Cedex; Philippe Agape, Tawfiq Henni, Tahar Touahri and Jean-Francois Paitel, Centre Hospitalier Felix Guyon - Allée Des Topazes; Valérie Moulin, Aurélie Ferru, Patrick Bouchaert, Chu La Miletrie; Dominique Spaeth, Fabien Brocard and Célia Becuwe, Centre D'oncologie De Gentilly; Fabienne Watelle, Centre Hospitalier Dr Schaffner; Nadine Paillot, Caroline Cuvier, Marjorie Lallom and Sylvie Giacchotti, Hopital De Mercy; Sandrine Richard and Julie Gachet, Hopital Europeen Georges Pompidou; Marielle Guillet, Jean-Christophe Souquet and Lionel Wander, Hopital Croix Rousse; Marc Espie, Florence Coussy, Sylvie Giacchetti, Caroline Cuvier and Marjorie Lallom, Hopital Saint Louis; Karima Yakendji Bekredda, Polyclinique Sainte Marguerite; Laurent Bastit, Clinique Pasteur; Alain Saad, Selva David and Tony Nakry, Centre Hospitalier Beziers Cedex; Elisabeth Perez and Patricia Zunic, Groupe Hospitalier Sud Reunion; Jean-Philippe Metges, Institut De Cancérologie Et D'hématologie - Hôpital Morvan; Hugues Bourgeois, Clinique Victor Hugo; Philippe Laplaige, Polyclinique De Blois; Nathalie Denizon, Centre Hospitalier Le Mans Cedex 9; Michaël Finaud, Djamal Hadjadj Aoul, Velardocchio, Clinique Vert Coteau; Alain Ducolone and Elisabeth Quoix, Nouvel Hopital Civil; Jean-Pierre Lotz, Hopital Tenon; Anne Mercier-Blas and Xavier Artignan, Centre Hospitalier Prive Saint Gregoire; Jean-Louis Lacaze and Audrey Eche, Institut Claudius Regaud; and Olivier Romano, Hopital Prive La Louviere.
Conflict of interest
Alain Toledano, Elisabeth Luporsi, Kamel Laribi, Jérôme Barrière, Philippe Huot-Marchand and Roland Bugat reported no conflict of interest. Florian Scotté is consultant to Vifor Pharma. Jean François Morere received funding from Vifor Pharma. Ladan Duvillié and Victor Hugo Concas are employees of Vifor Pharma France. The authors are fully responsible for content and editorial decisions for this paper. All authors had access to the primary study data. All authors reviewed the paper and approved the final version.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Toledano, A., Luporsi, E., Morere, J.F. et al. Clinical use of ferric carboxymaltose in patients with solid tumours or haematological malignancies in France. Support Care Cancer 24, 67–75 (2016). https://doi.org/10.1007/s00520-015-2728-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-015-2728-3