Abstract
The Tibetan Plateau holds some of the world’s highest undisturbed natural treelines and timberlines. Such extreme environments constitute potentially valuable monitoring sites of the effects of climate warming on high-elevation forests. Here, we analyze a network of 21 Smith fir forests situated in the Sygera Mountains, southeastern Tibetan Plateau, using tree-ring width (TRW) and basal area increment (BAI) chronologies. Sampled sites encompassed a wide elevation gradient, from 3600 to 4400 m, including some treeline sites and diverse aspects and tree ages. In comparison with TRW series, BAI series better capture the long-term warming signal. Previous November and current April and summer temperatures are the dominant climatic factors controlling Smith fir radial growth. The mean inter-series correlations of TRW increased upwards, but the forest limit presented the highest potential to reconstruct past temperature variability. Moreover, the growth responses of young trees were less stable than those of trees older than 100 years. Climate warming is accelerating radial growth of Smith fir forest subjected to mesic conditions. Collectively, these findings confirm that the effects of site elevation and tree age should be considered when quantifying climate-growth relationships. The type of tree-ring data (BAI vs. TRW) is also relevant since BAI indices seem to be a better climatic proxy of low-frequency temperature signals than TRW indices. Therefore, site (e.g., elevation) and tree (e.g., age) features should be considered to properly evaluate the effects of climate warming on growth of high-elevation forests.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Ongoing climate warming is expected to modify the growth and distribution of tree species, especially at mountain regions where warmer temperatures are expected to enhance tree growth (Albright and Peterson 2013). However, topographical features, especially elevation and slope aspect, create topoclimates in mountain regions which vary significantly over small geographical distances (Holst et al. 2005; Barry 2008). As a result, trees located at different topographical situations respond differently to climate (Bunn et al. 2005; Liang et al. 2006; Leal et al. 2007; Wang et al. 2015). Therefore, these topographical features should be explicitly considered to understand how forest growth reacts to climate in mountainous regions.
Evaluations of climate-growth relationships based on dendroclimatology generally assume that these associations are age independent once the biological growth trend has been removed (Fritts 2001). However, several studies have shown that trees respond to climate depending on their age (Szeicz and MacDonald 1994; Carrer and Urbinati 2004; Rozas et al. 2009; Li et al. 2013). For instance, old trees usually present greater growth sensitivity to climatic constraints than young trees (Yu et al. 2008; Linares et al. 2013). Such age dependence of tree growth responsiveness to climate could be explained by physiological changes (alterations in photosynthesis rates, changes in carbon or nutrient allocation to sinks or growing meristems, loss of hydraulic conductivity, etc.) the trees experience as they age and enlarge (Bond 2000; Ryan et al. 2006). Given this aforementioned background, tree-ring based studies should explicitly consider how topographical features modulate the age dependence of tree growth responses to climate warming in high-elevation forests (e.g., Primicia et al. 2015).
Some of the world’s highest natural treelines and timberlines are located in the southeastern Tibetan Plateau (hereafter abbreviated as SE TP) (Miehe et al. 2007). In this region, most treelines and high-elevation forests are dominated by Smith fir (Abies georgei var. smithii (Viguie & Gaussen) W. C. Cheng & L. K. Fu) which is distributed across a wide elevation range extending from 3600 to 4400 m asl (Wang et al. 2012). Several studies have shown that Smith fir radial growth is enhanced by warmer summer temperatures (Liang et al. 2009, 2010), but neither topography nor age has been considered as relevant factors modulating the growth responsiveness to climate warming.
In this study, a tree-ring network of 21 sampling sites was developed across an 800-m elevation transect located in the SE TP. Mean indexed tree-ring width (TRW) and basal area increment (BAI) site chronologies were built at each locality to quantify the relative importance of topography and age as modulators of tree growth responses to climate. We hypothesize that the radial growth of Smith fir will respond differently to climate across sites differing in topography (elevation, aspect) and among tree age classes.
The specific objectives of this study are the following: (1) to explore the dominant climatic factors limiting radial growth of Smith fir; (2) to assess the effects of elevation and slope aspect on climate-growth relationships; (3) to determine how tree age affects climate-growth relationships; and (4) to quantify long-term forest growth using basal area increment (BAI), since this is a proxy of changes in aboveground biomass (Babst et al. 2014). Understanding the topographical mediation and the age dependence of growth responses to climate is a fundamental task to evaluate the responses of mountain forests to global warming. Further, this information will, in turn, be crucial for adequately reconstructing past climate variability, especially low-frequency signals, considering local site factors and the age of sampled trees to obtain more reliable climatic proxies.
Material and methods
Study area
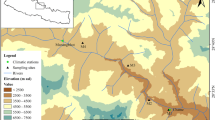
The study area is located in the Sygera Mountains (latitude 29°10′–30°15′N, longitude 93°12′–95°35′E), in the SE TP. This area is characterized by a temperate semi-humid climate. The South Asian monsoon reaches the study area along the Yarlung Zangbo River, resulting in abundant summer monsoon rainfall. Records from the meteorological station in Nyingchi (29°34′N, 93°28′E, 3000 m asl, located 30 km southwest from the study area) show a mean annual precipitation of 672 mm for the 1960–2014 period, with 72 % of annual rainfall recorded during the monsoon season (June to September). July (mean temperature of 15.8 °C) and January (0.6 °C) are the warmest and coldest months of the year, respectively. Records from an automatic weather station located at a Blackseed Juniper (Juniperus saltuaria Rehd. & E.H. Wilson) forest limit (29°39′N, 95°43′E, 4390 m asl) on an east-facing slope show that the annual total precipitation was 871 mm during the 2007–2009 period, whereas the mean air temperatures for the coldest (January) and warmest months (July) were −7.1 and 7.9 °C, respectively (Liu et al. 2011).
Smith fir is a shade-tolerant conifer species, spanning an 800-m wide elevation range in the study area, from 3600 to 4400 m asl. The Smith fir forests are most abundant from 3800 to 4000 m, and its upper forest limit is located from 4250 to 4400 m, depending upon site aspect (Liang et al. 2010; Wang et al. 2012).
Tree-ring sampling and chronology development
A total of 21 sampling sites, with mean slope angles of 10°–15°, showing different elevations, slope aspects, and tree ages, were selected during the 2006–2010 period. In total, 480 increment cores were collected at 1.3 m from 415 healthy Smith fir trees using increment borers. The tree-ring samples were processed following standard dendrochronological methods by air drying and sanding the cores (Cook and Kairiukstis 1990). After a rigorous visual cross-dating of the tree-ring cores, ring widths were measured by using a Lintab 5 system with a resolution of 0.01 mm, and then the quality of the cross-dating was checked by using the COFECHA program (Holmes 1983). No missing rings were found. The tree-ring measurements were standardized to remove the biological growth trend, as well as any other low-frequency variation due to stand dynamics, by fitting a 100-year cubic smoothing spline to preserve inter-annual to multi-decadal variability using the ARSTAN program (Cook 1985). Then, the resulting standard TRW indices of each tree were averaged for each site using a biweight robust mean and no autoregressive modeling was performed. Therefore, we used the standard site chronologies in further analyses. The age of trees with pith was determined by counting the number of rings. In cases of cores without pith, the distance to the pith was estimated by fitting a template of concentric circles with known radii to the curve of the innermost rings (Duncan 1989). The number of growth rings in the missing radius was obtained by dividing the estimated length of the missing radius by the mean ring width obtained from the 20 innermost growth rings. Cores whose innermost rings were not close enough to the pith or did not curve were not used in the analyses of age effects.
The TRW data were converted into BAI assuming a circular shape of the stem and using the following formula:
where R is the radius of the tree and j is the year of tree-ring formation.
We carried out a conservative detrending method. First, BAI was converted to normalized BAI (NBAI), a dimensionless variable, to make comparable the among-tree variation in growth. Then, an age-related trend of NBAI series was computed (cambial age was considered at coring height). Finally, BAI indices were obtained as follows:
where BAI index ij stands for the value calculated for the year j in the tree i, observed NBAI ij is the normalized BAI value of the year j in the tree i; NBAI j is the computed age-related normalized BAI value for the year j. We obtained BAI chronologies for 19 sites since we excluded sites DYS-3840 and SW-4340 where the estimated distances to innermost rings were too long in some trees to adequately estimate BAI.
Several descriptive statistics commonly used in dendroclimatology were adopted to quantify their common signal and to assess their sensitivity to climatic fluctuations (Fritts 2001). These statistics included the mean sensitivity (MS) to describe the year-to-year variation in each growth index, the first-order serial autocorrelation (AC) to indicate the relationship with previous growth, the Expressed Population Signal (EPS) to establish a criterion for selecting a common period where chronologies would be replicated enough (Wigley et al. 1984), the mean correlation calculated over all trees (Rbar), the signal-to-noise ratio (SNR), and the variance explained by the first principle component (PC1) to estimate the common growth variations shared among trees of the same site (Fritts 2001).
Analyses along the elevation gradients of the tree growth chronologies
Correlations (Pearson coefficients) and principal component analysis (PCA) were used to characterize the relationships among the 21 TRW and the 19 BAI chronologies over the common 1901–2006 period, in which all chronologies presented EPS > 0.80. Correlations were calculated between TRW and BAI site chronologies, and the PCA was based on the covariance matrix calculated among those chronologies. In the PCA, only the components that explained at least 5 % of the variation, usually the first three principal components (PC1, PC2, and PC3) were retained (Jolliffe 2002).
Age-specific analyses of growth data chronologies
To analyze how tree age affects climate-growth associations, we selected trees located above 4100 m asl, which presented more variability in year-to-year growth indices. In total, 155 trees were studied and they were grouped into six 50-year age classes (1–50 years, 51–100 years, 101–150 years, 151–200 years, 201–250 years, >250 years). For the youngest class (age ≤ 50 years), the descriptive statistics were calculated for the 1970–2006 period; while for the other five age classes, their common period was 1961–2006.
Climate-growth relationships
The influence of climatic factors on the annual growth of Smith fir trees was investigated by correlating TRW and BAI indexed series with monthly mean, minimum, and maximum temperatures and total precipitation over the 1961–2006 period. Climate data were obtained from the Nyingchi meteorological station. Considering the lagged effects of climatic variables on Smith fir growth, the window of analyses included the data from July of the previous growth year to September of the current year (Liang et al. 2010).
Results
Characteristics of the TRW and BAI datasets and chronologies
Table 1 lists the statistical characteristics of the 21 TRW and the 19 BAI chronologies. The mean TRW was 0.85 mm (range 0.54–1.19 mm) and the mean BAI was 816 mm2 (range 385–1629 mm2). Due to the cool and moist climatic conditions of the study area, the year-to-year variability of TRW chronologies (average MS is 0.11 ± 0.01) was relatively small. In the case of TRW chronologies, their relatively high values of mean Rbar (0.27 ± 0.08), SNR (7.50 ± 4.61) and EPS (0.86 ± 0.08) indicated that the TRW series contained strong common signals, probably reflecting similar climatic influences. In the case of AC, their high values (TRW, mean AC was 0.65 ± 0.06; BAI, mean AC was 0.74 ± 0.11) indicate that growth was significantly influenced by previous-year growth.
Elevation-specific growth responses to climate
As elevation increased, TRW and BAI decreased, while the Rbar, SNR, and EPS of TRW chronologies increased significantly (p < 0.05; Table 1). These statistics indicate that high-elevation Smith fir forests record more common growth signals, probably mediated by climatic influences, than low-elevation forests.
The first principal component (PC1) yielded higher loadings on the high-elevation sites either for TRW or for BAI chronologies (Fig. 1), and the PC1s of both TRW and BAI chronologies were significantly correlated with site elevation (TRW, r = 0.78, p < 0.001; BAI, r = 0.49, p < 0.05). The high percentage of TRW or BAI variation retained by the PC1 (49–52 %) confirmed that Smith fir growth data reflected a common regional climatic signal. Moreover, the PC2 loadings differentiated TRW chronologies and BAI chronologies into two groups separated by an elevation threshold located at about 4000 m (Fig. 1). The lack of significant differences in the statistical characteristics of TRW and BAI chronologies when grouped by site aspect was also particularly remarkable.
Loadings for the first three principal components (PC1, PC2, and PC3) derived from relating the 21 standard Smith fir tree-ring width (TRW) chronologies (a) or the 19 Smith fir basal area increment (BAI) chronologies (b) considering data for the 1901–2006 period. The percentage of variation of each principal component is indicated between parentheses
In the case of both TRW and BAI chronologies, most of these series along the elevation gradient were significant correlated (p < 0.01) (Fig. 2). Furthermore, the adjacent sites and also the high-elevation sites presented the highest correlations. In the case of BAI chronologies, the inadequate sample sizes for some sites (Table 1) may result in the non-significant correlations (Fig. 2).
Correlation matrix calculated by relating the 21 standard tree-ring width (TRW) and 19 basal area increment (BAI) chronologies considering the 1901–2006 period. Filled and empty circles indicate positive and negative correlations, respectively. The shaded gray background indicates significance at the 0.05 level. Numbers located below and above the diagonal correspond to TRW and BAI chronologies, respectively
Age-specific growth responses to climate
Growth patterns were similar among the six considered age classes regarding TRW and BAI chronologies (Fig. 3). In fact, TRW and BAI series of the different age classes were significantly correlated with each other, except for the youngest age class. The correlation coefficients reached maximum values for the two older age classes (151–200 and 201–250 years) (Table 2). Furthermore, mean TRW decreased as trees aged, while MS and AC increased with tree age and stabilized when trees were older than 100 years (Table 3). All these results indicate that Smith fir trees older than 100 years tend to present much more stable and coherent growth responses to climate than younger trees. It should be also noted that the differences in sample depth, shorter common period of the youngest age class, and the fact that most samples of this class came from one site (DYP-4400) may also explain these differences. Notably, although TRW and BAI chronologies displayed similar year-to-year fluctuation, BAI indices showed an increasing trend during the past 250 years, with some decadal peaks during warm periods (e.g., 1950s, 1990s, and 2000s), showing a high potential to record low-frequency climatic signals such as long-term rises in temperature (Fig. 3).
Topographic and age-related influences on growth responses to climate
The correlation analyses suggest that irrespective of slope, elevation, and tree age, the radial growth of Smith fir is limited by similar climate factors (Figs. 4 and 5). Both TRW and BAI chronologies were positively correlated with mean, minimum, and maximum temperatures during the previous November and with April minimum temperatures and summer (June to August) temperatures of the year of tree-ring formation (Figs. 4 and 5). However, precipitation was less related to growth as compared with temperatures excepting current June precipitation which showed negative associations with TRW and BAI chronologies.
Correlations obtained by relating the standard tree-ring width (TRW) chronologies (a) or mean basal area increment (BAI) chronologies (b) with monthly climate variables considering four different elevations. Climate variables go from the July of the previous year to current September of the year of tree-ring formation. The dotted and dashed horizontal lines indicate the 95 and 99 % confidence limits, respectively
Legend is as in Fig. 4 but considering six 50-year age classes
It is worthwhile to note that as elevation increased, the relationship between tree growth and climate became more stable and reinforced, especially above 4000 m. At high elevations, not only prior November temperature but also previous October temperature was positively related to TRW and BAI data. Moreover, the positive relationships of current August temperature and TRW or BAI data strengthened at low elevations (Fig. 4). Overall, trees younger than 51 years presented weaker and less regular TRW and BAI responses to climate than trees older than 100 years (Fig. 5).
Discussion
Here we show how site features and tree age modulate Smith fir’s growth responses to temperature and precipitation at mountain forests in the TP. The presented research emphasizes that local topographical conditions and tree age should explicitly be considered to adequately assess how high-elevation forests respond to climate warming in terms of radial growth and productivity. Furthermore, the building of suitable tree-ring proxies of past temperature variability in mountain areas should take into account the local topography of the sampled sites and the tree ages whenever possible.
Comparing TRW and BAI as temperature proxies
In the context of global climate warming, recovering low-frequency climatic signals from tree-ring data has attracted the attention of dendroclimatologists (Esper et al. 2003). TRW is one of the most important proxies to reconstruct past climate variability (Fritts 2001). However, tree-ring data are usually detrended and standardized to remove long-term growth trends due to age and size ontogenetic changes and to maximize the common climatic signal between trees (Cook and Kairiukstis 1990). On the other hand, BAI is considered to be a more meaningful indicator of growth than TRW because it removes variation in growth attributable to increasing stem circumference but keeps relevant information related to the tree ontogeny as the juvenile (rising) and mature (stable) phases (Biondi and Qeadan 2008; Liu et al. 2013; Camarero et al. 2015). However, BAI determination assumes a circular shape of the stem which could not always be assumed in mountain forests where trees usually grow on steep slopes and form eccentric stem shapes.
In our study, both TRW and BAI series captured past temperature variation, but the centennial-scale warming trend was only detected in BAI series. To further assess the validity of our chronologies and the differences between TRW and BAI series, we compared them with tree-ring (regional curve standardization method) based summer temperature reconstructions for the Linzhi-Bomi region located in the SE TP (Zhu et al. 2011). Significant correlations were found between the Linzhi-Bomi temperature reconstruction and the TRW (r = 0.30, p < 0.001) and BAI series (r = 0.78, p < 0.001) for the 1750–2002 period. Considering a 20-year moving average, the correlations improved for BAI series (r = 0.96, p < 0.001) while decreased in the case of TRW series (r = 0.17, p < 0.05). The mentioned long-term growth trend has also been pointed out in other studies carried out in the TP and elsewhere (Liu et al. 2007; Wilson et al. 2007; Liu et al. 2009). This evidence suggests that BAI is a better climatic proxy to record low-frequency temperature signals than TRW.
Features of Smith fir TRW chronologies along the elevation gradient
The semi-humid climate in the study area is favorable for tree growth, which explains the high TRW values and their relative low year-to-year variability (Table 1). As elevation increased, TRW decreased significantly (p < 0.001), while the MS and Rbar of TRW standard chronologies increased, which had been found in other studies developed at mountain area (Splechtna et al. 2000; Wang et al. 2005). This finding is probably explained by the increasingly harsher environmental conditions trees face as elevation increases, chiefly low temperatures and a short growing season (Körner 2012). Furthermore, the fact that the statistical quality of the Smith fir chronologies increases upwards as we approach the forest limit situated at ca. 4400 m indicates that chronologies developed at this ecotone have the highest potential to explore past temperature variability in the TP. This differs from what other studies have found along elevation gradients in nearby mountains subjected to semi-arid conditions (Gou et al. 2005; Wang et al. 2005; Chen et al. 2012). Such discrepancy is mainly due to the different macroclimate conditions for the study areas and suggests that different precipitation regimes could influence how tree growth responds to temperature gradients in mountain regions and indirectly impact local treeline formation (Liu et al. 2006; Yang et al. 2013; Liang et al. 2014; González-de Andrés et al. 2015).
Despite the differences listed above, the radial growth of Smith fir is affected by common regional climatic signals as a positive influence of warmer summer conditions on TRW. This similarity of growth variations along elevation gradient agrees with previous studies carried out in SE Tibet (Liang et al. 2010; Li et al. 2012). However, our findings do not concur with studies carried out along an elevation gradient from 3200 to 4200 m situated in the central Hengduan Mountains (Fan et al. 2009), which found that winter temperature was the most consistent constrain of growth at high to middle elevation, whereas tree growth was limited by spring moisture availability at low elevations. The observed climate-growth relationships for Smith fir are probably due to the relatively high elevation of the studied forests, the humid climate conditions, and the short growing season of wood formation observed in such high-elevation sites from late May to mid-August (according to Li et al. 2013). We expect similar temperature-driven responses in forests from humid and temperate mountain areas.
Age-specific characteristics of Smith fir growth
At high-elevation sites, low summer temperatures constrain Smith fir radial growth, which justifies the wide use of TRW as a proxy to reconstruct past temperature variation in the TP (Liang et al. 2009; Li et al. 2013). However, those studies assumed that climate-growth relationships were mostly age independent once the biological growth trend has been removed. This study found that tree radial climate-growth relationships are more stable when trees are older than 100 years. This may be because growth of older trees is more limited by temperatures. As trees age and enlarge and their height increases, the length of water transportation through the stem increases, and thus the rate of water transportation, nutrient use, and photosynthesis decrease (Yoder et al. 1994; Bond 2000; Ryan et al. 2006). However, the loss of hydraulic efficiency in old and tall trees cannot well explain our results, considering that tree height did not change greatly as a function of tree age (e.g., mean heights of trees with ages of 100 and 200 years are 6.8 ± 1.5 m and 10.1 ± 1.6 m, respectively). We propose that the age dependence of growth processes at high-elevation forests such as xylogenesis could better explain the different climate-growth associations observed across Smith fir forests (Rossi et al. 2008). For instance, studies on xylem formation at 3850 m in the study area indicate that young (43 years) and old trees (162 years) responded to changes of the minimum air temperature, but young trees presented an earlier onset of xylogenesis, a longer growing season, and a higher growth rate (Li et al. 2013). Thus, age-dependent growth responses to climate can give important clues to predict how differently young and old trees will react to climate warming. According to our results, Smith firs older than 100 years would allow maximizing the climate signals recovered from TRW and BAI series to reconstruct past temperatures.
However, tree age is significantly and positively related to the tree diameter at breast height (R 2 ranges from 0.69 to 0.95, p < 0.001) for the study sites and species along the altitudinal gradients (Liang et al. 2011; Wang et al. 2012). Unlike other studies (Mencuccini et al. 2005; De Luis et al. 2009; Rozas et al. 2009), our data cannot separately evaluate the effects of tree age and size on tree growth.
Growth responses to temperature at high-elevation forests
The existence of alpine forest limits and treelines is generally considered to be primarily related to summer heat deficiency (Körner 2012). Our study also found that summer temperature was the main limiting factor of Smith fir growth, independent of site elevation, slope aspect and tree age. Similar findings have also been reported for other tree species at the upper timberlines on the SE (Liang et al. 2009, 2010; Zhu et al. 2011) and NE TP (Liang et al. 2006), as well as other areas (Zhang et al. 2012; Dang et al. 2013). This finding can be explained by the direct effects of temperature on cambium activity and radial growth. On one hand, low temperature during the growing season could limit the cambium activity in early spring (Deslauriers et al. 2003; Schmitt et al. 2004) and could also cause spring frost injuries of needles and shoots or even damage cambial tissues (Gurskaya and Shiyatov 2006). On the other hand, higher summer temperatures favor radial growth by increasing cell production rates (Wieser and Tausz 2007).
The positive relationship observed between radial tree growth and the prior late fall (October to November) temperature was also observed in other mountain forests from the TP (Fan et al. 2009; Liang et al. 2006, 2009) and elsewhere (Tardif et al. 2003; Oberhuber 2004). During the late fall and early winter, the protecting snow cover has not been formed and the frost resistance of tree buds and fine roots is still low (Owens and Singh 1982). Thus, extremely low night temperatures may damage recently formed buds and fine roots, reduce the activity of mycorrhizal fungi, and damage the photosynthetic ability of high-elevation trees (Kozlowski and Pallardy 1997; Weih and Karlsson 2002). Furthermore, winter temperatures indirectly influence the depth and duration of soil snow packs, which largely determine the onset and length of the growing season in the following spring (Vaganov et al. 1999).
Monthly mean minimum temperatures during April had a positive correlation with Smith fir radial growth, suggesting that the higher spring temperatures would advance the onset of growing season by increasing the melting of snow cover and by warming soil and stem meristems (Li et al. 2013). However, in some rain-shadow areas, the maximum temperatures in the early growing season negatively affected tree growth which could be related to an increased moisture stress (Bräuning 1999; Čufar et al. 2008; Zhang et al. 2015).
Conclusions
Based on a network of Smith fir TRW and BAI chronologies distributed along a wide elevation gradient, we explored how topography and tree age influence radial growth responses to climate on the Tibetan Plateau. Previous late fall and current spring and summer temperatures drive Smith fir growth. Site elevation and tree age modulate these responses. The stands located above 4000 m show the highest potential to reconstruct past temperature variability. The responses of trees older than 100 years to climate are much more stable and stronger than those in young trees. According to the presented results, it is better to sample trees older than 100 years and growing in sites higher than 4000 m if the objective is to reconstruct past temperature variability. Finally, BAI better captured the regional and long-term climate warming than TRW.
References
Albright WL, Peterson DL (2013) Tree growth and climate in the Pacific northwest, North America: a broad-scale analysis of changing growth environments. J Biogeogr 40:2119–2133
Babst F, Bouriaud O, Alexander R, Trouet V, Frank D (2014) Toward consistent measurements of carbon accumulation: A multi-site assessment of biomass and basal area increment across Europe. Dendrochronologia 32:153–161
Barry RG (2008) Mountain weather and climate. Cambridge University Press, Cambridge
Biondi F, Qeadan F (2008) A theory-driven approach to tree-ring standardization: defining the biological trend from expected basal area increment. Tree-Ring Res 64:81–96
Bond BJ (2000) Age-related changes in photosynthesis of woody plants. Trends Plant Sci 5:349–353
Bräuning A (1999) Dendroclimatological potential of drought-sensitive tree stands in southern Tibet for the reconstruction of monsoonal activity. IAWA J 20:325–338
Bunn AG, Waggoner LA, Graumlich LJ (2005) Topographic mediation of growth in high elevation foxtail pine (Pinus balfouriana Grev. et Balf.) forests in the Sierra Nevada, USA. Glob Ecol Biogeogr 14:103–114
Camarero JJ, Gazol A, Galván JD, Sangüesa-Barreda G, Gutiérrez E (2015) Disparate effects of global-change drivers on mountain conifer forests: warming-induced growth enhancement in young trees vs. CO2 fertilization in old trees from wet sites. Glob Chang Biol 21:738–749
Carrer M, Urbinati C (2004) Age-dependent tree-ring growth responses to climate in Larix decidua and Pinus cembra. Ecology 85:730–740
Chen F, Yuan Y, Wen W, Yu S, Fan Z, Zhang R, Zhang T, Shang H (2012) Tree-ring-based reconstruction of precipitation in the Changling Mountains, China, since A.D.1691. Int J Biometeorol 56:765–774
Cook ER (1985) A time-series analysis approach to tree ring standardization. University of Arizona, Tucson, Dissertation
Cook ER, Kairiukstis LA (1990) Methods of dendrochronology: applications in the environmental sciences. Kluwer, Dordrecht
Čufar K, De Luis M, Eckstein D, Kajfež-Bogataj L (2008) Reconstructing dry and wet summers in SE Slovenia from oak tree-ring series. Int J Biometeorol 52:607–615
Dang H, Zhang Y, Zhang K, Jiang M, Zhang Q (2013) Climate-growth relationships of subalpine fir (Abies fargesii) across the altitudinal range in the Shennongjia Mountains, central China. Clim Chang 117:903–917
De Luis M, Novak K, Čufar K, Raventós J (2009) Size mediated climate-growth relationships in Pinus halepensis and Pinus pinea. Trees-Struct Funct 23:1065–1073
Deslauriers A, Morin H, Begin Y (2003) Cellular phenology of annual ring formation of Abies balsamea in the Quebec boreal forest (Canada). Can J for Res 33:190–200
Duncan R (1989) An evaluation of errors in tree age estimates based on increment cores in kahikatea (Dacrycarpus dacrydioides). New Zeal Natural Sci 16:1–37
Esper J, Cook ER, Krusic PJ, Peters K, Schweingruber FH (2003) Tests of the RCS method for preserving low-frequency variability in long tree-ring chronologies. Tree-Ring Res 59:81–98
Fan Z, Bräuning A, Cao K, Zhu S (2009) Growth-climate responses of high-elevation conifers in the central Hengduan Mountains, southwestern China. Forest Ecol Manag 258:306–313
Fritts HC (2001) Tree rings and climate. Blackburn Press, Caldwell
González-de Andrés E, Camarero JJ, Büntgen U (2015) Complex climate constraints of upper treeline formation in the Pyrenees. Trees-Struct Funct 29:941–952
Gou X, Chen F, Yang M, Li J, Peng J, Jin L (2005) Climatic response of thick leaf spruce (Picea crassifolia) tree-ring width at different elevations over Qilian Mountains, northwestern China. J Arid Environ 61:513–524
Gurskaya M, Shiyatov S (2006) Distribution of frost injuries in the wood of conifers. Russ J Ecol 37:7–12
Holmes RL (1983) Computer-assisted quality control in tree-ring dating and measurement. Tree-Ring Bull 43:69–78
Holst T, Rost J, Mayer H (2005) Net radiation balance for two forested slopes on opposite slides of a valley. Int J Biometeorol 49:275–284
Jolliffe IT (2002) Principal component analysis. Springer, New York
Körner C (2012) Alpine treelines. functional ecology of the global high elevation tree limits. Springer, Basel
Kozlowski TT, Pallardy SG (1997) Growth control in Woody Plants. Academic, San Diego
Leal S, Melvin TM, Grabner M, Wimmer R, Briffa KR (2007) Tree-ring growth variability in the Austrian Alps: the influence of site, altitude, tree species and climate. Boreas 36:426–440
Li Z, Liu G, Fu B, Hu C, Luo S, Liu X, He F (2012) Anomalous temperature-growth response of Abies faxoniana to sustained freezing stress along elevational gradients in China’s Western Sichuan Province. Trees-Struct Funct 26:1373–1388
Li X, Liang E, Gričar J, Prislan P, Rossi S, Čufar K (2013) Age dependence of xylogenesis and its climatic sensitivity in Smith fir on the south-eastern Tibetan Plateau. Tree Physiol 33:48–56
Liang E, Shao X, Eckstein D, Huang L, Liu X (2006) Topography-and species-dependent growth responses of Sabina przewalskii and Picea crassifolia to climate on the northeast Tibetan Plateau. Forest Ecol Manag 236:268–277
Liang E, Shao X, Xu Y (2009) Tree-ring evidence of recent abnormal warming on the southeast Tibetan Plateau. Theor Appl Climatol 98:9–18
Liang E, Wang Y, Xu Y, Liu B, Shao X (2010) Growth variation in Abies georgei var. smithii along altitudinal gradients in the Sygera Mountains, southeastern Tibetan Plateau. Trees-Struct Funct 24:363–373
Liang E, Wang Y, Eckstein D, Luo T (2011) Little change in the fir tree-line position on the Southeastern Tibetan Plateau after 200 years of warming. New Phytol 190:760–769
Liang E, Dawadi B, Pederson N, Eckstein D (2014) Is the growth of birch at the upper timberline in the Himalayas limited by moisture or by temperature? Ecology 95:2453–2465
Linares JC, Taïqui L, Sangüesa-Barreda G, Seco JI, Camarero JJ (2013) Age-related drought sensitivity of Atlas cedar (Cedrus atlantica) in the Moroccan Middle Atlas forests. Dendrochronologia 31:88–96
Liu L, Shao X, Liang E (2006) Climate signals from tree ring chronologies of the upper and lower treelines in the Dulan region of the Northeastern Qinghai-Tibetan Plateau. J Integr Plant Biol 48:278–285
Liu X, Shao X, Zhao L, Qin D, Chen T, Ren J (2007) Dendroclimatic temperature record derived from tree-ring width and stable carbon isotope chronologies in the Middle Qilian Mountains, China. Arct Antarct Alp Res 39:651–657
Liu Y, An Z, Linderholm HW, Chen D, Song H, Cai Q, Sun J, Tian H (2009) Annual temperatures during the last 2485 years in the mid-eastern Tibetan Plateau inferred from tree rings. Sci China Ser D 52:348–359
Liu B, Liang E, Zhu L (2011) Microclimatic conditions for Juniperus saltuaria treeline in the Sygera Mountains, Southeastern Tibetan Plateau. Mt Res Dev 31:45–53
Liu H, Williams AP, Allen CD, Guo D, Wu X, Anenkhonov OA, Liang E, Sandanov DV, Yin Y, Qi Z, Badmaeva NK (2013) Rapid warming accelerates tree growth decline in semi-arid forests of Inner Asia. Glob Chang Biol 19:2500–2510
Mencuccini M, Martínez-Vilalta J, Vanderklein D, Hamid HA, Korakaki E, Lee S, Michiels B (2005) Size-mediated ageing reduces vigour in trees. Ecol Lett 8:1183–1190
Miehe G, Miehe S, Vogel J, Co S, La D (2007) Highest treeline in the northern hemisphere found in southern Tibet. Mt Res Dev 27:169–173
Oberhuber W (2004) Influence of climate on radial growth of Pinus cembra within the alpine forest limit ecotone. Tree Physiol 24:291–301
Owens JN, Singh H (1982) Vegetative bud development and the time and method of cone initiation in subalpine fir (Abies lasiocarpa). Can J Bot 60:2249–2262
Primicia I, Camarero JJ, Janda P, Čada V, Morrissey RC, Trotsiuk V, Bače R, Teodosiu M, Svoboda M (2015) Age, competition, disturbance and elevation effects on tree and stand growth response of primary Picea abies forest to climate. For Ecol Manag 354:77–86
Rossi S, Deslauriers A, Anfodillo T, Carrer M (2008) Age-dependent xylogenesis in timberline conifers. New Phytol 177:199–208
Rozas V, DeSoto L, Olano JM (2009) Sex-specific, age-dependent sensitivity of tree-ring growth to climate in the dioecious tree Juniperus thurifera. New Phytol 182:687–697
Ryan MG, Phillips N, Bond BJ (2006) The hydraulic limitation hypothesis revisited. Plant Cell Environ 29:367–381
Schmitt U, Jalkanen R, Eckstein D (2004) Cambium dynamics of Pinus sylvestris and Betula spp. in the northern boreal forest in Finland. Silva Fenn 38:167–178
Splechtna BE, Dobrys J, Klinka K (2000) Tree-ring characteristics of subalpine fir (Abies lasiocarpa (Hook.) Nutt.) in relation to elevation and climatic fluctuations. Ann for Sci 57:89–100
Szeicz JM, MacDonald GM (1994) Age dependent tree ring growth responses of subarctic white spruce to climate. Can J For Res 24:120–132
Tardif J, Camarero JJ, Ribas M, Gutiérrez E (2003) Spatiotemporal variability in tree ring growth in the Central Pyrenees: climatic and site influences. Ecol Monogr 73:241–257
Vaganov EA, Hughes MK, Kirdyanov AV, Schweingruber FH, Silkin PP (1999) Influence of snowfall and melt timing on tree growth in subarctic Eurasia. Nature 400:149–151
Wang T, Ren H, Ma K (2005) Climatic signals in tree ring of Picea schrenkiana along an altitudinal gradient in the central Tianshan Mountains, northwestern China. Trees-Struct Funct 19:736–742
Wang Y, Čufar K, Eckstein D, Liang E (2012) Variation of maximum tree height and annual shoot growth of Smith fir at various elevations in the Sygera Mountains, southeastern Tibetan Plateau. PLoS one 7:e31725. doi:10.1371/journal.pone.0031725
Wang H, Ge Q, Dai J, Tao Z (2015) Geographical pattern in first bloom variablity and its relation to temperature sensitivity in the USA and China. Int J Biometeorol 59:961–969
Weih M, Karlsson PS (2002) Low winter soil temperature affects summertime nutrient uptake capacity and growth rate of mountain birch seedlings in the subarctic, Swedish lapland. Arct Antarct Alp Res 34:434–439
Wieser G, Tausz M (2007) Trees at their upper limit: tree life limitation at the Alpine Forest limit. Springer, Dordrecht
Wigley T, Briffa KR, Jones PD (1984) On the average value of correlated time series, with applications in dendroclimatology and hydrometeorolog. J Clim Appl Meteorol 23:201–213
Wilson R, D’Arrigo R, Buckley B, Büntgen U, Esper J, Frank D, Luckman B, Payette S, Vose R, Youngblut D (2007) A matter of divergence: tracking recent warming at hemispheric scales using tree ring data. J Geophys Res 112:D17103. doi:10.1029/2006JD008318
Yang B, He M, Melvin TM, Zhao Y, Briffa KR (2013) Climate control on tree growth at the upper and lower treelines: a case study in the Qilian Mountains, Tibetan Plateau. PLoS one 8:e69065
Yoder B, Ryan M, Waring R, Schoettle A, Kaufmann M (1994) Evidence of reduced photosynthetic rates in old trees. For Sci 40:513–527
Yu G, Liu Y, Wang X, Ma K (2008) Age-dependent tree-ring growth responses to climate in Qilian juniper (Sabina przewalskii Kom.). Trees-Struct Funct 22:197–204
Zhang W, Jiang Y, Dong M, Kang M, Yang H (2012) Relationship between the radial growth of Picea meyeri and climate along elevations of the Luyashan Mountain in North-Central China. Forest Ecol Manag 265:142–149
Zhang Q, Evans MN, Lyu L (2015) Moisture dipole over the Tibetan Plateau during the past five and a half centuries. Nat Commun 6:8062
Zhu H, Shao X, Yin Z, Xu P, Xu Y, Tian H (2011) August temperature variability in the southeastern Tibetan Plateau since AD 1385 inferred from tree rings. Palaeogeogr Palaeocl 305:84–92
Acknowledgments
This work was supported by the National Natural Science Foundation of China (41130529, 41525001), the National Basic Research Program of China (2012FY111400), and Action Plan for West Development of the Chinese Academy of Science (KZCX2-XB3-08-02). We appreciate the great support for our fieldwork by the Southeast Tibet Station for Alpine Environment, Observation and Research, Chinese Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Liu, B., Wang, Y., Zhu, H. et al. Topography and age mediate the growth responses of Smith fir to climate warming in the southeastern Tibetan Plateau. Int J Biometeorol 60, 1577–1587 (2016). https://doi.org/10.1007/s00484-016-1148-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00484-016-1148-5