Abstract
Key message
The acclimation of silver fir to the conditions of the plantation sites has greater influence on PSII thermostability and performance than does on local adaptation.
Abstract
Forest ecosystems are frequently exposed to heat stress, which adversely affects their growth, performance, and ultimately survival. The knowledge of physiological responses to heat can help mitigate the effects of climate change by facilitating the choosing of suitable reproductive material for future reforestation programs. However, the intraspecific variation of adaptive traits, including the response to heat, has not been well documented. Therefore, we attempted to determine how PSII photochemistry of different silver fir (Abies alba Mill.) provenances varies in responses to heat stress and to reveal if intraspecific variation in PSII performance and thermostability is more affected by the climate of provenance origin or the plantation site. We tested 17 fir provenances sampled at two contrasting international provenance trial sites: Hertník in Slovakia (390 m a.s.l.) and Kaprun in Austria (1100 m a.s.l.). Short-term heat stress was simulated for seven temperatures ranging from 20 to 51 °C using a water bath. The PSII thermotolerance was assessed via parameters of chlorophyll a fluorescence derived from the OKJIP transient. We observed several climatic variables of the provenances that significantly affected the photochemistry performance and thermostability of PSII, reflecting an additive hereditary basis. Almost all measured parameters varied significantly between trial plots and showed significant trial-by-provenance interactions pointing to acclimation triggered by the different conditions at the trial plots. Overall, the performance of PSII under non-stressing conditions and after heat treatment was better for provenances growing at the warmer trial site at Hertník.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Silver fir (Abies alba Mill.) is one of the most productive and ecologically valuable native European tree species. In forestry, silver fir is often studied with the respect to its significant decline that has been periodically observed in the majority of the native fir distribution range (Wiedemann 1927). This decline has been especially attributed to a high susceptibility of silver fir to NO3 and SO2 pollution. However, the levels of pollutant emissions have been significantly reduced since the 1980s, which positively affected the recovery of fir (Wolf 2003; Bošeľa et al. 2014; Büntgen et al. 2014). The positive reaction to a slight increase in temperature in late winter as well as in July is also important, indicating a potential advantage in the conditions of ongoing climate change compared to spruce, which is very sensitive to extreme weather (Zang et al. 2014; Bosela et al. 2016).
Climate change scenarios predict an increased likelihood of temperature extremes, especially heat. Currently, prolonged summer heat impacts nearly 10% of surface land, in contrast to only 1% prior to 1980 (Hansen et al. 2012). Heat significantly contributes to changes in morphological, physiological and biochemical processes, and thus affects the overall performance, growth and ultimately the survival of plants (Song et al. 2014). Heat can cause metabolic changes, which can lead to a reduction in the duration of development phases and subsequently to a reduction in light perception, resulting in the overall shortening of the life cycle. Heat induces increasing membrane fluidity and changes in the functionality of enzymes via denaturation (Wahid and Close 2007; Balogh et al. 2013). The most fundamental processes of photosynthesis are the most heat-sensitive processes in plants, as PSII components are considered highly heat sensitive (Ashraf and Harris 2013). At the thylakoid level, heat stress results in the dissociation of the Mn atoms of the oxygen-releasing complex in the reaction centers of PSII or in the separation of core and light-harvesting complexes of PSII. In addition, damage of reaction center components, e.g., major protein units D1 and D2, can occur (Pastenes and Horton 1996; De Las Rivas and Barber 1997; Yamane et al. 1998). Moreover, heat is intrinsically linked to drought, causing stomatal closure, and thus limiting plant abilities to avoid heat stress by transpiration-mediated cooling, and specific heat-associated atmospheric circulation patterns can lead to intense and prolonged effects of heat. High temperatures can therefore easily exceed the thresholds for impairment of physiological processes (Priestley and Taylor 1972; Meehl and Tebaldi 2004; Barua and Heckathorn 2006). For example, the association of heat waves with drought in the summer of 2003 is well documented. These conditions caused a reduction in gross primary productivity throughout Europe by 30%, and certain stands showed a 50% reduction in growth (Ciais et al. 2005; Bertini et al. 2011).
The mitigation of climate change impacts on forest ecosystems is therefore a pressing issue in forestry. One of the possible solutions is assisted migration, i.e., the introduction of new, non-native ecotypes and genetics variants from sites with the expected future climate to target sites. Several studies have reported the intraspecific genetic variation in responses to heat in crop plants (Havaux 1992; Yamasaki et al. 2002; Bibi et al. 2008; Brestič et al. 2010), but still little is known about trees. Due to their longevity, trees are exposed to fluctuating environmental conditions; hence, different populations have developed specific physiological mechanisms to adapt to their natural environmental conditions (Song et al. 2014). The response of tree populations to climate change may therefore differ depending on the environment conditions of the sites of origin, where the intraspecific variation in functional traits reflects different climatic selection pressures. Populations originating from warmer localities are likely to be more resistant to heat and better adapted to high temperatures than populations originating from cooler sites (Salvucci and Crafts-Brandner 2004; Ramírez-Valiente et al. 2014; Matías et al. 2016). The knowledge of the geographic and climatic patterns of adaptive trait variation is therefore essential for the selection of suitable reproductive materials (Ghouil et al. 2003; Gömöry et al. 2015; Bussotti et al. 2015). An appropriate tool for the study of intraspecific variability of such traits is provenance research. A provenance trial eliminates phenotypic differences among populations, which result from a wide range of environmental factors, by cultivating populations with different origins under equal conditions (Turesson 1925; Clausen et al. 1939).
Therefore, the goals of this study were (1) to understand how PSII of different silver fir provenances varies in responses to heat stress; (2) to identify potential geographic and climatic patterns in PSII thermostability across the range, and (3) to determine whether the photochemistry heat-stress response of fir provenances is affected by the climate of the plantation site (trial plots).
Materials and methods
Experimental material and growth conditions
Plant material was collected from two silver fir provenance trial sites belonging to the international experiment organized by IUFRO: the Slovak site was located in eastern Slovakia at Hertník (49°13′00″ N 21°16′17″ E; 390 m a.s.l.), and the Austrian site was situated near Kaprun (47°16′14″ N 12°43′43″ E; 1100 m a.s.l.). These plots are climatically contrasting: the Hertník site is situated below the fir climatic optimum, i.e., in lower altitude with warmer and drier climate, whereas the Kaprun site is located close to the upper altitudinal limit of the distribution of fir in colder and wetter region. Both sites were established in 2005 under a complete randomized block design with three replications. Seventeen and twenty-four provenances were planted at Hertník and Kaprun, respectively, covering most of the fir distribution range. Both plots were planted using five-year-old seedlings of identical batches of the respective nursery, with an initial number of 35 seedlings per provenance and block.
For the study, 17 provenances growing in both plots were selected (Table 1; Fig. 1). Samples were collected from ten individuals (two blocks, five trees per block) from each provenance. The climatic characteristics of provenance origins associated with the temperature regime were obtained from the WorldClim database at a resolution of 30 arc-seconds: T MEAN = annual mean temperature; T 59 = mean temperature during the vegetative season (May–September); BIO2 = mean diurnal temperature range [monthly mean (max temp − min temp)]; BIO3 = isothermality (BIO2/BIO7) × 10; BIO4 = temperature seasonality (standard deviation × 100); BIO5 = max temperature of warmest month; BIO7 = annual temperature range (BIO5-BIO6); BIO8 = mean temperature of wettest quarter; BIO9 = mean temperature of driest quarter; and BIO10 = mean temperature of warmest quarter. The data represent the mean values of each climatic variable from 1950 to 2000 (Hijmans et al. 2005).

(adapted from EUFORGEN 2015, http://www.euforgen.org)
Locations of the origins of tested provenances (black circles) and the trial sites at Hertník and Kaprun (stars). The gray area displays the current natural distribution of silver fir in Europe
Determination of photosynthesis and PSII performance and thermostability
Measurements occurred during the second week of June 2016. Unshaded branches from the upper third of the crown on the north side were harvested. Samples were transported to the laboratory and refrigerated overnight at 8 °C in plastic sealable bags. We used one-year-old shoots, which were enclosed in glass Erlenmeyer flasks and exposed to heat for 30 min. Heat stress was simulated using a WNE22 water bath (Memmert, Germany) for the seven water temperatures ranging from 30 to 51 °C (specifically, 30, 36, 39, 42, 45, 48 and 51 °C). The measurements were mostly repeated twice per individual and then averaged. For each temperature, a different part of the shoot of the same branch was used. Maximum air temperatures inside the flasks were measured in a separate study; these temperatures were lower than the temperature of water inside the water bath by an average of 0.5 at 30 °C, and 1.5 at 51 °C. These differences were taken into account during final data processing.
After heat treatment, needles were dark-adapted for 30 min. Then, chlorophyll a fluorescence was excited by a saturation pulse with an intensity of 3500 µmol photons m−2 s−1 and a one-second duration using a Handy PEA fluorometer (Hansatech Ltd., UK). The OJIP transients were analyzed based on the JIP test using the software Biolyzer 3.06 (Laboratory of Bioenergetics, University of Geneva, Switzerland).
The photosynthetic performance of provenances was assessed using the basic parameters of chlorophyll a fluorescence measured at 30 °C (the control measurement represented non-stressing conditions) as follows:
-
1.
F v/F m—the maximal quantum efficiency of PSII photochemistry calculated as F v/F m = (F 0 − F m)/F m, where the F 0 is the basal fluorescence of chlorophyll a measured at 50 µs, and F m represents the maximal chlorophyll a fluorescence.
-
2.
PI ABS —the absolute index of photosynthetic performance.
-
3.
RC/ABS—the number of active reaction centers per antenna (Strasser et al. 2000).
Then, the PSII thermotolerance was assessed based on the characteristics derived from the course of the OJIP transient:
-
1.
\({R_{{F_{\text{v}}}}}_{{/{F_{\text{m}}}}}\)—the ratio between the F v/F m measured at 51 °C and the control value of F v/F m measured at 30 °C. \({R_{{F_{\text{v}}}}}_{{/{F_{\text{m}}}}}\) represents the severity of heat stress, in that lower values of \({R_{{F_{\text{v}}}}}_{{/{F_{\text{m}}}}}\) reflect a higher level of heat stress and vice versa.
-
2.
T 15—the temperature threshold of PSII resistance when the F v/F m decreases by 15% with respect to the control measurement at 30 °C. For the assessment of T 15, the F v/F m values were fitted against temperatures using a third-order polynomial. T 15 was expressed as the temperature at which F v/F m reached 85% of the polynomial maximum (Froux et al. 2004).
-
3.
T C—critical temperature for PSII stability. T C was recorded as the temperature when the basal fluorescence F 0 began to sharply increase. F 0 values were correlated with temperature and were fitted by two linear regression lines. Because the substantial increase of F 0 started between 42 and 45 °C for all provenances, we used regression lines with the ranges of 30–42 and 45–51 °C. T C was expressed as the temperature where the regression lines intersected (Bigras 2000; Froux et al. 2004).
-
4.
\({T_{{W_K}}}\)—temperature at which the value of variable fluorescence at the K-step started to rapidly increase. The K-step occurred at 0.3 ms during heat treatment; higher values of W K reflect a higher degree of heat-induced damage of PSII components and vice versa. W K is calculated as \({W_K}=\frac{{{F_{0.3}} - {F_0}}}{{{F_2} - {F_0}}}\), where F 0.3 and F 2 represent the fluorescence measured at 0.3 and 2 ms, respectively, and F 0 is basal fluorescence (Lazár et al. 1999; Brestič et al. 2010). \({T_{{W_K}}}\) was assessed as T C with the difference that regression lines with the ranges of 30–39 °C and 42–48 °C were used.
Statistical analysis
Data analysis was performed using SAS 9.1.3 (SAS Institute Inc., USA) and R 3.1.2 (R Core Team, Austria). Differences among provenances were assessed by the analysis of variance ANOVA using the GLM procedure according to the model y ijkl = trial i + prov j + trial i × prov j + block k (trial i × prov j ) + ε ijkl , where the y ijkl is the measured value of the phenotype, trial i is the effect of the ith trial, provj is the effect of the jth provenance, trial i × prov j is the interaction between the trial and provenance, block k (trial i × prov j ) is the effect of the kth block and ε ijkl is the experimental error. All effects were considered fixed. For the assessment of geographic and climatic trends of photochemical performance and PSII thermostability, we used linear and quadratic regressions between the provenance means of physiological parameters and geographic coordinates and/or climatic variables of their sites of origin. The resulting probabilities were corrected by sequential Bonferroni correction. Regression curves were described using coefficient of determination (R 2), slope of the least squares line of best fit (slope) for linear regression, and the value of geographic and climatic variable, where the vertex of parabola reached the minimum or maximum (Min/Max) for quadratic regression. Moreover, the geographic patterns of the PSII thermostability parameters were displayed using ArcMap 10.2 (Esri Inc., USA).
Results
Performance of PSII under non-stressing conditions
The analysis of variance (Table 2) confirmed significant variation in PSII performance under non-stressing conditions between trial plots. The provenances showed better values, and thus a higher photochemical performance at the level of PSII under the conditions of the warmer trial plot at Hertník. The average maximal photochemical efficiency values were approximately 0.81 and 0.78 at Hertník and Kaprun, respectively. Additionally, the number of reaction centers per antenna complex was reduced by 25%, and the index of photosynthetic performance was nearly 44% less in provenances growing at the Kaprun trial plot compared to those of plots at Hertník. The value of basal fluorescence varied between trial plots, with slightly higher values in Hertník plots; however, no significant differences between the tested provenances were observed. On the other hand, a significant trial-by-provenance interaction was recorded for almost all tested parameters (except F 0), which reflects the diversity of provenance responses to the conditions of the trial plots. This was also confirmed by the geographic and climatic trends: a significant effect of several bioclimatic characteristics describing the temperature regime (BIO3, BIO4, BIO7, and BIO8) on the performance of PSII was observed in the Hertník plots, whereas no range-wide trends were identified in the Kaprun plots (Table 3). Positive effect of increasing isothermality (BIO3) on parameters of PSII performance, and effect of mean temperature of wettest quarter (BIO8) on RC/ABS remained significant even after sequential Bonferroni correction.
PSII thermostability and severity of heat stress
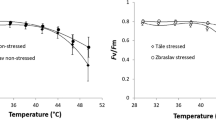
Figure 2 shows the response curves of F v/F m, F 0 and W K to increased temperature. Overall, the courses of response curves were similar for both trial plots; however, the average response curves representing trees growing in Kaprun were shifted toward less favorable values of the determined parameters compared to those in Hertník. The values of F v/F m remained stable until 42–43 °C for both trial plots but then gradually decreased. The course of F 0 was also linear until approximately 43 °C, but it increased abruptly afterward. The W K response curves had a specific pattern: W K slightly increased with increasing temperature to 39 °C, started to increase significantly up to 48 °C, and then dropped steeply. The decrease of W K was more pronounced for the Kaprun plots.
Average response curves of photochemistry parameters to temperature for the trial sites at Hertník (black) and Kaprun (gray). The response curves of maximal photochemical efficiency (Fv/F m; a), basal fluorescence (F 0; b) and K-step value (W K ; c) are displayed. The points represent the mean values per temperature and trial site; n = 170. The error bars represent the standard deviations. The points were fitted using the weighted least squares method
Based on the F v/F m, F 0 and W K response curves, we derived the parameters \({R_{{F_{\text{v}}}}}_{{/{F_{\text{m}}}}}\), T 15, T C and \({T_{{W_K}}}\) that reflect PSII thermotolerance and the severity of heat stress. Almost all parameters except \({R_{{F_{\text{v}}}}}_{{/{F_{\text{m}}}}}\) varied significantly between trial plots (Table 4). Similar to the PSII performance in non-stressing conditions, the provenances growing at the warmer trial plots at Hertník showed higher levels of PSII thermotolerance.
Because of the significant or at least marginally significant trial-by-provenance interactions, which were recorded for all parameters, we focused on the relationships between the parameters of PSII thermostability and the geographic and climatic characteristics of the provenance origins separately for each trial plot. We recognized a wide range of factors that significantly affected PSII thermostability (Table 5); six relationships maintained significant after sequential Bonferroni correction. However, there were certain differences in the response patterns of trial plots. At Hertník, PSII thermostability was negatively affected mainly by mean annual temperature and mean temperature during the vegetative season at the sites of origin. Regarding the Kaprun trial plots, positive effects of the mean diurnal range of temperature and isothermality on PSII thermostability were observed. For both trial plots, significant relationships between the T 15 and the maximal temperatures of the warmest month (BIO5) were confirmed (Fig. 3). However, the responses were not uniform: T 15 decreased linearly along the gradient of BIO5 in Hertník, whereas the response was non-linear at Kaprun, with an increase across the majority of the range.
Relationships between the parameters of temperature threshold of PSII resistance (T 15) and maximal temperature reached in the warmest month. Regressions were made separately for trial sites at Hertník (full circles) and Kaprun (empty circles). The circles present the means per provenance and trial plot; n = 20
The geographic distribution patterns of the evaluated PSII thermostability parameters were unclear (Fig. 4); moreover, the patterns were not consistent between trial plots and among parameters. In Hertník, only heat stress severity \({R_{{F_{\text{v}}}}}_{{/{F_{\text{m}}}}}\) showed a recognizable pattern with a favorable status in the Western Carpathian provenances as did severe stress in the Balkan and German provenances. Partly, such pattern could also be seen at Kaprun. However, no consistent trends could be recognized for the critical temperatures T 15, T C and \({T_{{W_K}}}\).
Geographic distribution of the parameters of PSII thermostability and severity of heat stress at trial sites at Hertník (left) and Kaprun (right). Each point represents the mean value per provenance (n = 20), and points are shaded depending on PSII thermostability and the severity of stress. Light color depicts the highest level of PSII thermostability and the lowest severity of heat stress; dark color depicts the lowest level of PSII thermostability and the highest severity of heat stress
Figure 4 indicates that populations originating from the Balkan glacial refugium, i.e., from the southeastern part of the fir distribution range (TC01, TC02, TC03, TC05 and TC06), differ in PSII thermostability. To verify this, separate ANOVAs were performed testing for differences between refugia (Table 6). Balkan populations were found to be able to withstand higher temperatures. However, after exposure to temperatures exceeding the critical temperature of damage to PSII, they showed more extensive damage to PSII components, and hence a more significant decrease in F v/F m (\({R_{{F_{\text{v}}}}}_{{/{F_{\text{m}}}}}\)). This decrease was more pronounced at the colder Kaprun site.
Discussion
The course of the OJIP transient is known to change under heat stress, which is attributed to the conformational changes of PSII related to the use of thermal energy: the basal fluorescence (F 0) sharply increases, reflecting the irreversible impairment of PSII due to the dissociation between light-harvesting complexes of PSII and the reaction centers, and maximal fluorescence (F m) decreases upon linear heating, representing a gradual closure of RCII and ultimately resulting in decreased maximum quantum yield of PSII (F v/F m ratio) (Pastenes and Horton 1996; Brestič and Živčák 2013; Kalaji et al. 2016). The F v/F m ratio of an intact plant under optimal physiological conditions is close to 0.83 across various plant species including silver fir, but this value becomes considerably reduced under the stress (Björkman and Demmig-Adams 1995). In our case, under non-stressing conditions (temperature of 30 °C), the average F v/F m reached ∼0.81 for the warmer Hertník plot and ∼0.78 at the colder Kaprun plot. Provenances at the warmer Hertník site exhibited a better vitality and performance overall under non-stressing conditions. After the heat treatment of 51 °C, we observed a decrease in F v/F m of more than 50%. This decrease, which is a proxy of the severity of heat stress, was also higher at the Hertník plot. This indicates higher heat resistance but also more severe consequences for PSII when the critical temperatures of trees planted at the warmer site are reached. At temperatures close to 50–53 °C, a 50% decrease in F v/F m was also recorded in other tree species, e.g., Pseudotsuga menziesii and Pinus ponderosa (Krause et al. 2010; Marias et al. 2017). In these species, a decrease of less than 85% of the F v/F m generally occurred at temperatures greater than 43.5 °C. Similar results were reported by Robakowski et al. (2002), who noted that a sharp decline in F v/F m in Abies alba occurred at temperatures greater than 40 °C. The temperature causing the impairment of PSII stability (T C) varies among plant species, mostly in the range of 35–49 °C (Weng and Lai 2005; Marias et al. 2017). Our results showed that the critical temperature for PSII stability of silver fir was 43.2 °C at the Hertník site and 42.7 °C at the Kaprun site, which are considerably less than those reported by Robakowski et al. (2002), who reported that the critical temperature for PSII stability in silver fir seedlings acclimated to 10 °C was close to 47 °C. Similar critical temperatures were observed for other temperate tree species (Dreyer et al. 2001). Different outcomes of our measurements may be explained by different conditions during and before sampling; our measurements were carried out at the beginning of the vegetative season, without exposure to temperatures higher than 25 °C. The mentioned studies were conducted at the peak vegetative season or after pretreatment with temperatures up to 40 °C, which suggests an earlier acclimation to higher temperatures triggered by a previous treatment by moderate heat. The history of plants can also cause these differences. The various environmental conditions and stresses can induce epigenetic control, the persistent changes in gene functions by DNA methylation, histone modifications, chromatin remodeling and small non-coding RNAs, and thus affect the individual functional status under stress (Yakovlev et al. 2010; Correia et al. 2013; Zhang et al. 2013). A wide range of morphological and physiological changes were recorded for trees, including changes of heat tolerance caused by modifications of epigenetic status (Hasbún et al. 2007; Valledor et al. 2010; Correia et al. 2013). Moreover, we simulated heat stress for 30 min per temperature; in the aforementioned studies, the heat stress was simulated by gradually increasing temperatures (1 °C per minute). Therefore, the prolonged effect of heat could cause more extensive changes and reduce the value of the critical temperature in our case (Georgieva et al. 2000; Dreyer et al. 2001; Robakowski et al. 2002).
Under heat stress, the K-step appears on the fluorescence curve at approximately 0.3 ms. A pronounced K-step occurred at a temperature close to 40 °C. Until that point, the W K values range from 0.36 to 0.44, which corresponded to the results of Brestič et al. (2010), who recorded values of W K between 0.41 and 0.46 in intact winter wheat plants. The K-step indicates irreversible heat-induced changes in PSII, either by the modification of LHC complexes or by the inhibition of electron transport from pheophytin to QA. Moreover, the K-step may be associated with the dissociation of the oxygen-evolving complex (Strasser 1997; Srivastava et al. 1997; Lazár et al. 1999). We observed a gradual increase in the K-step value with increasing temperature from 30 to 48 °C and then a subsequent drop, which was probably associated with the separation of the light-harvesting complexes of PSII from the PSII core complexes, which is also responsible for the increase in the F 0 level (Yamane et al. 1997). Similar to the other parameters, the provenances planted at Hertník showed higher \({T_{{W_K}}}\) values and thus higher PSII thermostability.
The observed significant trial-by-provenance interactions for nearly all evaluated parameters indicated that provenance responses to trial plot conditions differed, reflecting a relatively high level of plasticity of silver fir. An overall higher photochemical performance and better heat-stress response of fir trees growing at the warmer Hertník site suggest acclimation to higher temperatures of plantation sites is probably driven by a more frequent exposure to heat and more severe heat events. Increased thermostability after a previous treatment by moderate temperatures has been recorded for many plant species, e.g., black spruce (Colombo and Timmer 1992), Haig and Kennebec cultivars of potato (Havaux and Gruszecki 1993), and five oak species (three Mediterranean oaks—Q. canariensis Willd., Q. coccifera L. and Q. suber L. and two European oaks—Q. robur L. and Q. petraea Matt. Liebl.) (Daas et al. 2008). Enhanced thermotolerance can also be induced by other co-occurring stresses, such as drought (Havaux 1992; Ladjal et al. 2000), salinity (Weng and Lai 2005) and excess light intensity (Havaux 1992; Volkova et al. 2009). This acclimation is mainly attributed to the accumulation of carotenoids and the synthesis of heat shock proteins, which stabilize major membrane complexes and maintain their functionality (Marchand et al. 2005; Kotak et al. 2007).
Some parameters of PSII thermostability and PSII performance showed geographic patterns or associations with climate. Adaptive variability has previously been revealed in silver fir, i.e., intraspecific variation in growth rate (Lebourgeois 2007; Mazza et al. 2014; Gazol et al. 2015), bud burst, mortality (Hansen and Larsen 2004), and winter frost resistance (Larsen 1986).
Several parameters showed geographic patterns, but no consistent trends were observed. The PSII of southeastern provenances originating from warmer localities and from the Balkan refugium was found to withstand the highest temperatures, as indicated by the T 15 and T C values. However, after exposure to temperatures exceeding the critical temperature of damage to PSII, the decrease in F v/F m was more pronounced, especially at the colder Kaprun trial site. This probably reflects the adaptation of the Balkan provenances to a warmer climate at their sites of origin. The inter-provenance variation in PSII thermostability and overall performance of PSII points to genetic differences formed by the conditions at the sites of origin. In addition to genetic differences, the heritable variation in PSII performance and thermostability may be caused by epigenetic components. This inherited epigenetic “memory” could allow trees to retain active molecular mechanisms of stress regulation across generations, and thus better adaptation of the progeny to stress events faced by their ancestor (Correia et al. 2013). Moreover, the epigenetic inheritance of trees is still little known, e.g., Yakovlev et al. (2010) reported the temperature-dependent epigenetic memory of bud phenology in Norway spruce. Authors found out that the temperature and photoperiod during the embryogenesis significantly affect the timing of bud phenology in progeny.
Therefore, we conclude that the hereditary basis of PSII thermostability and photosynthetic performance exists, but silver fir also shows a relatively high plasticity of heat-stress response depending on conditions of the plantation sites.
Conclusions
The mitigation of climate change regarding forest ecosystems and the strengthening of their vitality by assisted migration is a key topic in current forestry. To achieve these objectives, observations of physiological processes are necessary, which allow the modeling and prediction of the reactions of trees to rapidly changing climate, and thus the identification of well-growing and adapted populations. In addition, the first motivation of provenance research was to choose tree populations with high adaptive potential, which could be used as seed sources for reforestation (Nielsen and Jørgensen 2003; König 2005). Because one of the main features of climate change is the increasing frequency of heat waves, our study focused on the assessment of PSII thermostability and performance in silver fir provenances growing at climatically contrasting trial sites. Our results showed that the association between the climatic conditions of provenance origin site and PSII thermostability and photochemical performance exists, which reflects a certain level of hereditary basis in the intraspecific variation of PSII thermostability and performance. However, silver fir showed a relatively high ability to acclimate to the current conditions of the plantation sites, which will be more important in conditions of changing climate.
Author contribution statement
DG, RL, and RK conceived the design of study; JK, ĽD, and DK planned and coordinated the experiment; AK, DK, DG, and RK collected plant material; AK and DK conducted the measurements and processed the data; DG performed the statistical analyses; and AK and DK wrote the first version of manuscript, which was discussed and approved by all the co-authors.
References
Ashraf M, Harris PJC (2013) Photosynthesis under stressful environments: an overview. Photosynthetica 51:163–190
Balogh G, Péter M, Glatz A, Gombos I, Török Z, Horváth I, Harwood JL, Vígh L (2013) Key role of lipids in heat stress management. FEBS Lett 587:1970–1980
Barua D, Heckathorn SA (2006) The interactive effects of light and temperature on heat-shock protein accumulation in Solidago altissima (Asteraceae) in the field and laboratory. Am J Bot 93:102–109
Bertini G, Amoriello T, Fabbio G, Piovosi M (2011) Forest growth and climate change: evidences from the ICP-Forests intensive monitoring in Italy. IForest 4:262
Bibi AC, Oosterhuis DM, Gonias ED (2008) Photosynthesis, quantum yield of photosystem II and membrane leakage as affected by high temperatures in cotton genotypes. J Cotton Sci 12:150–159
Bigras FJ (2000) Selection of white spruce families in the context of climate change: heat tolerance. Tree Physiol 20:1227–1234
Björkman O, Demmig-Adams B (1995) Regulation of photosynthetic light energy capture, conversion, and dissipation in leaves of higher plants. In: Schulze ED, Caldwell MM (eds) Ecophysiology of photosynthesis. Springer Berlin, Heidelberg, pp 17–47
Bosela M, Popa I, Gömöry D, Longauer R, Tobin B, Kyncl J, Kyncl T, Nechita C, Petráš R, Sidor CG, Šebeň V, Büntgen U (2016) Effects of post-glacial phylogeny and genetic diversity on the growth variability and climate sensitivity of European silver fir. J Ecol 104:716–724
Bošeľa M, Petráš R, Sitková Z, Priwitzer T, Pajtík J, Hlavatá H, Sedmák R, Tobin B (2014) Possible causes of the recent rapid increase in the radial increment of silver fir in the Western Carpathians. Environ Pollut 184:211–221
Brestič M, Živčák M (2013) PSII fluorescence techniques for measurement of drought and high temperature stress signal in crop plants: protocols and applications. In: Rout GR, Das AB (eds) Molecular stress physiology of plants. Springer, India, pp 87–131
Brestič M, Olšovská K, Pivková J (2010) Bioindication of thermotolerance of winter wheat (Triticum aestivum L.) photosynthetic apparatus. Acta Fytotechnica et Zootechnica 13:67–71
Büntgen U, Tegel W, Kaplan JO, Schaub M, Hagedorn F, Bürgi M, Brázdil R, Helle G, Carrer M, Heussner KU, Hofmann J, Kontic R, Kyncl T, Kyncl J, Camarero JJ, Tinner W, Esper J, Liebhold A (2014) Placing unprecedented recent fir growth in a European-wide and Holocene-long context. Front Ecol Environ 12:100–106
Bussotti F, Pollastrini M, Holland V, Brüggemann W (2015) Functional traits and adaptive capacity of European forests to climate change. Environ Exp Bot 111:91–113
Ciais P, Reichstein M, Viovy N, Granier A, Ogée J, Allard V, Aubinet M, Buchmann N, Bernhofer C, Carrara A, Chevallier F, Noblet ND, Friend AD, Friedlingstein P, Grünwald T, Heinesch B, Keronen P, Knohl A, Krinner G, Loustau D, Manca G, Matteucci G, Miglietta F, Ourcival JM, Papale D, Pilegaard K, Rambal S, Seufert G, Soussana JF, Sanz MJ, Schulze ED, Vesala T, Valentini R (2005) Europe-wide reduction in primary productivity caused by the heat and drought in 2003. Nature 437:529–533
Clausen J, Keck DD, Hiesey WM (1939) The concept of species based on experiment. Am J Bot 26:103–106
Colombo SJ, Timmer VR (1992) Limits of tolerance to high temperatures causing direct and indirect damage to black spruce. Tree Physiol 11:95–104
Correia B, Valledor L, Meijón M, Rodriguez JL, Dias MC, Santos C, Cañal MJ, Rodriguez R, Pinto G (2013) Is the interplay between epigenetic markers related to the acclimation of cork oak plants to high temperatures? PLoS One 8:e53543
Daas C, Montpied P, Hanchi B, Dreyer E (2008) Responses of photosynthesis to high temperatures in oak saplings assessed by chlorophyll-a fluorescence: inter-specific diversity and temperature-induced plasticity. Ann For Sci 65:305
De Las Rivas J, Barber J (1997) Structure and thermal stability of photosystem II reaction centers studied by infrared spectroscopy. BioChemistry 36:8897–8903
Dreyer E, Roux XL, Montpied P, Daudet FA, Masson F (2001) Temperature response of leaf photosynthetic capacity in seedlings from seven temperate tree species. Tree Physiol 21:223–232
Froux F, Ducrey M, Epron D, Dreyer E (2004) Seasonal variations and acclimation potential of the thermostability of photochemistry in four Mediterranean conifers. Ann For Sci 61:235–241
Gazol A, Camarero JJ, Gutiérrez E, Popa I, Andreu-Hayles L, Motta R, Nola P, Ribas M, Sangüesa-Barreda G, Urbinati C, Carrer M (2015) Distinct effects of climate warming on populations of silver fir (Abies alba) across Europe. J Biogeogr 42:1150–1162
Georgieva K, Tsonev T, Velikova V, Yordanov I (2000) Photosynthetic activity during high temperature treatment of pea plants. J Plant Physiol 157:169–176
Ghouil H, Montpied P, Epron D, Ksontini M, Hanchi B, Dreyer E (2003) Thermal optima of photosynthetic functions and thermostability of photochemistry in cork oak seedlings. Tree Physiol 23:1031–1039
Gömöry D, Longauer R, Krajmerová D (2015) Voľba lesného reprodukčného materiálu v podmienkach klimatickej zmeny [Choice of forest reproductive material under conditions of climate change]. For J 61:24–130
Hansen JK, Larsen JB (2004) European silver fir (Abies alba Mill.) provenances from Calabria, southern Italy: 15-year results from Danish provenance field trials. Eur J For R 123:127–138
Hansen J, Sato M, Ruedy R (2012) Perception of climate change. Proc Nat Acad Sci USA 109:2415–2423
Hasbún R, Valledor L, Santamaría E, Cañal MJ, Rodríguez R (2007) Dynamics of DNA methylation in chestnut trees development. Acta Hortic:563–566
Havaux M (1992) Stress tolerance of photosystem II in vivo: antagonistic effects of water, heat, and photoinhibition stresses. Plant Physiol 100:424–432
Havaux M, Gruszecki W (1993) Heat- and light-induced chlorophyll a fluorescence changes in potato leaves containing high or low levels of the carotenoid zeaxanthin: Indications of a regulatory effect of zeaxanthin on thylakoid membrane fluidity. Photochem Photobiol 58:607–614
Hijmans RJ, Cameron SE, Parra JL, Jones PG, Jarvis A (2005) Very high resolution interpolated climate surfaces for global land areas. Int J Clim 25:1965–1978
Kalaji HM, Jajoo A, Oukarroum A, Brestic M, Zivcak M, Samborska IA, Cetner MD, Łukasik I, Goltsev V, Ladle RJ (2016) Chlorophyll a fluorescence as a tool to monitor physiological status of plants under abiotic stress conditions. Acta Physiol Plant 38:102
König A (2005) Provenance research: evaluation the spatial pattern of genetic variation. In: Conservation and management of forest genetic resources in Europe. Arbora, Zvolen and IPGRI, pp 275–408
Kotak S, Larkindale J, Lee U, von Koskull-Döring P, Vierling E, Scharf KD (2007) Complexity of the heat stress response in plants. Curr Opin Plant Biol 10:310–316
Krause GH, Winter K, Krause B, Jahns P, García M, Aranda J, Virgo A (2010) High-temperature tolerance of a tropical tree, Ficus insipida: methodological reassessment and climate change considerations. Funct Plant Biol 37:890
Ladjal M, Epron D, Ducrey M (2000) Effects of drought preconditioning on thermotolerance of photosystem II and susceptibility of photosynthesis to heat stress in cedar seedlings. Tree Physiol 20:1235–1241
Larsen JB (1986) Die geographische variation der Weißtanne (Abies alba Mill.) [The geographical variation of silver fir (Abies alba Mill.)]. Wachstumsentwicklung und Frostresistenz. Forstwissenschaftliches Centralblatt 105:396–406
Lazár D, Pospíšil P, Nauš J (1999) Decrease of fluorescence intensity after the K step in chlorophyll a fluorescence induction is suppressed by electron acceptors and donors to photosystem 2. Photosynthetica 2:255–265
Lebourgeois F (2007) Climatic signal in annual growth variation of silver fir (Abies alba Mill.) and spruce (Picea abies Karst.) from the French Permanent Plot Network (RENECOFOR). Ann For Sci 64:333–343
Marchand FL, Mertens S, Kockelbergh F, Beyens L, Nijs I (2005) Performance of High Arctic tundra plants improved during but deteriorated after exposure to a simulated extreme temperature event. Glob Change Biol 11:2078–2089
Marias DE, Meinzer FC, Woodruff DR, McCulloh KA (2017) Thermotolerance and heat stress responses of Douglas-fir and ponderosa pine seedling populations from contrasting climates. Tree Physiol 37:301–315
Matías L, Gonzalez-Díaz P, Quero JL, Camarero JJ, Lloret F, Jump AS (2016) Role of geographical provenance in the response of silver fir seedlings to experimental warming and drought. Tree Physiol 36:1236–1246
Mazza G, Gallucci V, Manetti MC, Urbinati C (2014) Climate–growth relationships of silver fir (Abies alba Mill.) in marginal populations of Central Italy. Dendrochronologia 32:181–190
Meehl GA, Tebaldi C (2004) More intense, more frequent, and longer lasting heat waves in the 21st century. Science 305:994–997
Nielsen CN, Jørgensen FV (2003) Phenology and diameter increment in seedlings of European beech (Fagus sylvatica L.) as affected by different soil water contents: variation between and within provenances. For Ecol Manag 174:233–249
Pastenes C, Horton P (1996) Effect of high temperature on photosynthesis in beans (oxygen evolution and chlorophyll fluorescence). Plant Physiol 112:1245–1251
Priestley CHB, Taylor RJ (1972) On the assessment of surface heat flux and evaporation using large-scale parameters. Mon Weather Rev 100:81–92
Ramírez-Valiente JA, Valladares F, Sánchez-Gómez D, Delgado A, Aranda I (2014) Population variation and natural selection on leaf traits in cork oak throughout its distribution range. Acta Oecol 58:49–56
Robakowski P, Montpied P, Dreyer E (2002) Temperature response of photosynthesis of silver fir (Abies alba Mill.) seedlings. Ann For Sci 59:163–170
Salvucci ME, Crafts-Brandner SJ (2004) Inhibition of photosynthesis by heat stress: the activation state of Rubisco as a limiting factor in photosynthesis. Physiol Plantarum 120:179–186
Song Y, Chen Q, Ci D, Shao X, Zhang D (2014) Effects of high temperature on photosynthesis and related gene expression in poplar. BMC Plant Biol 14:111
Srivastava A, Guissé B, Greppin H, Strasser RJ (1997) Regulation of antenna structure and electron transport in Photosystem II of Pisum sativum under elevated temperature probed by the fast polyphasic chlorophyll a fluorescence transient: OKJIP. Biochim Biophys Acta Bioenerget 1320:95–106
Strasser BJ (1997) Donor side capacity of Photosystem II probed by chlorophyll a fluorescence transients. Photosynth Res 52:147–155
Strasser RJ, Srivastava A, Tsimilli-Michael M (2000) The fluorescence transient as a tool to characterize and screen photosynthetic samples. In: Yunus M, Pathre U, Mohanty P (eds) Probing photosynthesis: mechanisms, regulation and adaptation. Taylor & Francis, London, pp 445–483
Turesson G (1925) The plant species in relation to habitat and climate. Hereditas 6:147–236
Valledor L, Meijón M, Hasbún R, Jesús Cañal M, Rodríguez R (2010) Variations in DNA methylation, acetylated histone H4, and methylated histone H3 during Pinus radiata needle maturation in relation to the loss of in vitro organogenic capability. J Plant Physiol 167:351–357
Volkova L, Tausz M, Bennett LT, Dreyer E (2009) Interactive effects of high irradiance and moderate heat on photosynthesis, pigments, and tocopherol in the tree-fern Dicksonia antarctica. Funct Plant Biol 36:1046
Wahid A, Close TJ (2007) Expression of dehydrins under heat stress and their relationship with water relations of sugarcane leaves. Biol Plantarum 51:104–109
Weng JH, Lai MF (2005) Estimating heat tolerance among plant species by two chlorophyll fluorescence parameters. Photosynthetica 43:439–444
Wiedemann E (1927) Untersuchungen ueber das Tannensterben [Investigation on the dieback of silver fir]. Forstwiss Cbl:815–853
Wolf H (2003) Technical guidelines for genetic conservation and use for for silver fir (Abies alba). International Plant Genetic Resources Institute, Rome
Yakovlev IA, Fossdal CG, Johnsen Ø (2010) MicroRNAs, the epigenetic memory and climatic adaptation in Norway spruce. New Phytol 187:1154–1169
Yamane Y, Kashino Y, Koike H, Satoh K (1997) Increases in the fluorescence Fo level and reversible inhibition of Photosystem II reaction center by high-temperature treatments in higher plants. Photosynth Res 52:57–64
Yamane Y, Kashino Y, Koike H, Satoh K (1998) Effects of high temperatures on the photosynthetic systems in spinach: oxygen-evolving activities, fluorescence characteristics and the denaturation process. Photosynth Res 57:51–59
Yamasaki T, Yamakawa T, Yamane Y, Koike H, Satoh K, Katoh S (2002) Temperature acclimation of photosynthesis and related changes in photosystem II electron transport in winter wheat. Plant Physiol 128:1087–1097
Zang C, Hartl-Meier C, Dittmar C, Rothe A, Menzel A (2014) Patterns of drought tolerance in major European temperate forest trees: climatic drivers and levels of variability. Glob Change Biol 20:3767–3779
Zhang YY, Fischer M, Colot V, Bossdorf O (2013) Epigenetic variation creates potential for evolution of plant phenotypic plasticity. New Phytol 197:314–322
Acknowledgements
On behalf of all the co-authors, we would like to thank Marek Živčák for his valuable advice during the writing of manuscript and Peter Petrík, Jana Kurjaková, Lucia Hederová and Hana Húdoková for their assistance during the measurements. The study was supported by grants of the Slovak Research and Development Agency APVV-0135-12 and APVV-0744-12, and by grant of the Slovak Grant Agency for Science VEGA, 2/0034/14.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by I. Porth.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Konôpková, A., Kurjak, D., Kmeť, J. et al. Differences in photochemistry and response to heat stress between silver fir (Abies alba Mill.) provenances. Trees 32, 73–86 (2018). https://doi.org/10.1007/s00468-017-1612-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00468-017-1612-9