Abstract
Ureteropelvic junction obstruction (UPJO) is the most common obstructive uropathy and its optimal management remains controversial. However, there is a current trend towards non-surgical management. We aimed to determine the effects of the non-surgical management in children with unilateral UPJO. For a systematic review, we searched MEDLINE, EMBASE, CENTRAL, clinical trials registries, and selected conference proceedings for eligible studies. Any type of study reporting the outcomes renal function, secondary surgical intervention, drainage pattern or hydronephrosis of non-surgical management in children with unilateral UPJO was included. Data from 20 studies were extracted and evaluated by two independent authors. The pooled prevalence was 21% for split renal function deterioration, 27.9% for secondary surgical intervention, 3.2% for progressive hydronephrosis, and 82.2% for improved drainage pattern. Not all patients with surgical intervention regained split renal function from enrolment. Renal imaging methods did not strongly correlate with each other. Many studies had to be excluded because of a lack of detection of an obstruction or mixed populations with bilateral UPJO or other uropathies. The variable definitions of UPJO, different criteria for surgical intervention, incongruity of management protocols, and the imprecise reporting of outcomes were limiting factors in the comparability of the results, leading to heterogeneity in meta-analyses. Although the available evidence cannot recommend or refute the current non-surgical management, the systematic review clarifies aspects of the ongoing controversy by providing realistic estimates for non-surgical management in children with unilateral UPJO. Additionally, it reveals unclear potential risks, particularly for long-term outcomes, which were rarely reported.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Unilateral ureteropelvic junction obstruction (UPJO) is defined as impeded urine outflow from the renal pelvis to the ureter, which may result in progressive damage to the kidney [1]. With a varying incidence of approximately 1 per 750–2,000, unilateral UPJO represents the most common obstructive uropathy [2]. Despite its epidemiological significance, the management of children with unilateral UPJO is still controversially debated and practice varies widely [3–5]. The current diagnostic approach with renal imaging cannot reliably determine if patients have a significant obstruction and are at risk for permanent kidney damage [6–9]. Therefore, it is difficult to time intervention and to track potential progression of split renal function deterioration [10–13]. The management of unilateral UPJO has undergone a paradigm shift in the recent decades [5, 14, 15]. The observation of deteriorating split renal function seemed indicative for early surgical intervention [16, 17]. Animal models supported the surgical approach, however, the total obstruction by ligature of the ureter does not reflect the situation in humans [18–22]. It has become apparent that patients may be treated safely by an approach with close monitoring and serial imaging [23, 24]. Therefore, non-surgical management has become the favoured approach for most asymptomatic patients without diminished split renal function [4, 25, 26]. A recently published systematic review, however, emphasised the need to evaluate the outcome of the non-surgical management of patients with unilateral UPJO without the restriction of randomised controlled trials [27]. Therefore, the purpose of this systematic review was to determine the pooled prevalence of the effects of non-surgical management of these patients for the outcomes split renal function, secondary surgical intervention, hydronephrosis, and drainage pattern.
Materials and methods
All methods used for this systematic review were specified in advance and documented in a protocol available in the International Prospective Register of Systematic Reviews (PROSPERO; http://www.crd.york.ac.uk/PROSPERO) under registration number CRD42016034013. The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement (http://www.prisma-statement.org/) and items of the Cochrane Handbook for Systematic Reviews of Interventions were used for reporting.
Data sources and search strategies
We developed a search strategy to identify all studies reporting on outcomes of the non-surgical management of children younger than 18 years of age with unilateral UPJO (Table 1). We searched the electronic databases CENTRAL (Cochrane Central Register of Controlled Trials, Issue 12 2015), MEDLINE and EMBASE (from their inception to 21 December 2015) without language restriction. In addition, we searched the following clinical trials registries for ongoing or recently completed trials using keywords (“hydronephrosis,” “obstruction,” “junction,” “ureteric-pelvic,” “ureteropelvic,” “pelvi-ureteric,” “ureteral obstruction,” “pyeloplasty”): www.clinicaltrials.gov; www.controlled-trials.com; www.trialscentral.org; apps.who.int/trialsearch; www.drks.de; www.anzctr.org.au. Conference proceedings listed in Supplementary Material 1 were screened using the same keywords. The reference lists of relevant articles and reviews were also searched for trials and publications.
Study selection
We included all types of studies reporting any of the following outcomes: split renal function, secondary surgical intervention (children initially managed non-surgically, but later requiring surgery owing to worsening of the condition), hydronephrosis, and drainage pattern. Renal scintigraphy (with or without the administration of diuretics) or any other appropriate cross-sectional imaging method, such as magnetic resonance imaging, was required for the diagnosis of unilateral UPJO. For the purposes of this review we consistently refer to the term “obstruction” if any urinary outflow impairment is detected, compared with the contralateral kidney. Therefore, the detection of obstruction is not equivalent to an inevitable indication for surgical intervention. Studies were excluded if one of the following criteria was present: diminished/absent contralateral kidney function, acute clinical symptoms or distinct hydronephrosis leading to immediate surgery, nephrostomy, stenting, or crossing vessels. Case reports or case series including ≤ 3 patients were excluded.
If studies also included data on patients who did not meet the inclusion criteria, or if additional information was needed for decision-making, study authors were contacted to retrieve required data. Eligibility of study inclusion was determined independently by two review authors, and disagreements were resolved by a third author.
Data extraction
Data extraction was done independently by two authors using a standardised data extraction form. If a study reported on separate subpopulations (e.g., with different group characteristics), these were extracted separately. If more than one publication on a given study existed, reports were merged to form an exhaustive, but nonredundant data set with the most recent information available. Information required from the original authors was requested by written correspondence and subsequently included in the review. Any disagreement between two independent reviewers was resolved by a third author.
Quality assessment
The risk of bias assessment at the study level was defined according to the evidence-based medicine criteria and the Cochrane ACROBAT-NRSI (https://sites.google.com/site/riskofbiastool), with adaptations to the objective of this review [28–30]. The assessment was carried out for all the studies included by two independent reviewers using a standard assessment form (Supplementary Material 2) and summarised for each study for the developed categories to ensure comparability and integrative analysis.
Data synthesis and analysis
The main outcomes were measured as prevalences, calculated from the number of individuals and the total size of the population, with accompanying 95% confidence intervals (CI). Outcomes were grouped for the population with or without secondary surgical intervention, and for the combination of both. The given numbers for patients with and without secondary surgical intervention did not have to add up to the total number of patients, as not all studies report on both subpopulations equally. The inconsistency of effects (heterogeneity of variance across studies) was addressed by inspection of forest plots and calculation of Q and I2 for each outcome per group with a random effects model [31, 32]. I2 values of more than 25, 50, and 75% correspond to low, medium, and high levels of heterogeneity. Meta-analysis was performed using the random-effects model, thereby adjusting for a variation of the effect between studies. To include studies with a prevalence of 0.0 and 100.0%, a frequency increment of 0.00001 was added to these values. If pooling was not possible, we provided descriptive results of studies. All analyses were carried out using R 3.1.2 and the additional package meta 4.3.2 [33, 34]. We performed sensitivity analyses to explore the influence of the following factors on pooled effect size:
-
1.
Large/small studies (defined as ≤ 100 patients or ≤ 20 patients)
-
2.
Studies not specifying a split renal function > 40% at time of enrolment
-
3.
Studies defining less than 50% of the population as having a half-time drainage of > 20 min
-
4.
Studies whose participants do not have a hydronephrosis of SFU grade ≥ 3
-
5.
Studies that do not report on children < 1 year of age
-
6.
Studies in which most or all participants were followed up for at least 2 years
A multi-variable meta-regression was performed to determine the influence of the following predictors on the prevalence of secondary surgery: age at presentation (all participants < 1 year/ other), country of study origin (Anglosphere/not Anglosphere), split renal function at presentation (all patients ≥ 40%/other), drainage pattern at presentation (at least 50% of all patients with a drainage half-time > 20 min/other) and hydronephrosis at presentation (all patients with SFU grade ≥ 3/other). Publication bias was visualised with funnel plots [35].
Results
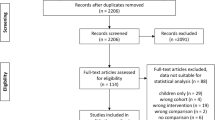
The initial search yielded 53,266 records, none of them from clinical trials registries. After removal of duplicates, 43,080 titles or abstracts were screened and 42,818 were found to be irrelevant to the review. After full-text assessment, another 242 records were excluded with reason (Supplementary Material 3), among them 16 records with missing/unclear information on study population or outcomes for which authors did not provide details upon request, and two records whose study population was later described in more detail [36, 37]. The remaining 20 records [24, 26, 38–55] were included in this review (Fig. 1) and are characterised in Supplementary Material 4.
Study flow diagram according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement [77]
Study characteristics
The studies included were mostly monocentric and initiated between 1980 and 1995, predominantly in the Anglosphere. Most of them were published in the 1990s and all but two were observational, either case series or cohort studies [24, 51]. Many studies had to be excluded owing to reporting of mixed populations (uni- and bilateral UPJO) or missing functional renal imaging.
Across all studies, the selection criteria for non-surgical management were often not reported. Approximately 50% of all children in the studies included showed a renal function ≥ 40% at enrolment. Although the diagnostic methods used were comparable, the interpretation was not consistent, especially regarding the evaluation of the drainage pattern. All but three studies did not provide a precise definition of obstruction [41, 49, 50]. Follow-up times were not specified at all or were not reported in a consistent manner across studies, as mean, median or a range [38, 44, 46–48]. Patients were followed up from 2.5 months to 19.6 years. Only 3 studies reported an observational period of more than 2 years for all patients [26, 45, 55]; an additional 5 studies had follow-up times of at least 1 year for each patient [39, 40, 42, 43, 49].
Quality assessment
We summarised the customised quality assessment in a risk of bias for each study and across all studies (Figs. 2, 3). For almost all studies, “measurement of interventions” and “measurement of outcomes” had to be classified as “high risk.” Approximately one third of the studies were biased because of the selection of the reported results. Missing data were apparent in less than 50% of all studies. The two randomised controlled trials included [24, 51] did not show an overall superior quality for the assessed risk of bias categories compared with the observational studies (Fig. 3).
Split renal function deterioration
Split renal function was the most common reported outcome of the studies included, usually only in the descriptive manner “number of patients with improvement, stabilisation or deterioration”. Only few studies reported mean values at enrolment and at the last follow-up for the entire population [39, 45, 48, 51]. The pooled prevalence of the total population with worsened split renal function (Fig. 4) showed significant heterogeneity (p < 0.0001), high variance across studies (I2 = 96.6), and amounted to 21.0% (95% CI = 4.7–37.4%) for 367 patients. The subgroups with and without secondary surgery (89 and 39 patients respectively) revealed a higher prevalence for the secondary surgical subgroup (28.4%, 95% CI = 0.0–66.7%, I2 = 91.6%) than for the solely non-surgical group (18.9%, 95% CI = 0–59.1%, I2 = 98.5%). Studies showed an increased pooled prevalence of 26.7% (95% CI = 3.4–49.9%, 295 patients) for patients with a split renal function of ≥ 40% at enrolment and a pooled prevalence of 23.1% (95% CI = 12.7–33.5%) for all patients < 1 year of age at enrolment, both with similar heterogeneity and inconsistency.
Pooled prevalence of split renal function deterioration in all children with initial non-surgical management (total), and the subgroups of children with and without secondary surgery. Each study is marked according to the information available for split renal function at enrolment. A all patients ≥ 40%, B additionally patients with < 40% included, C unknown
Secondary surgery
The prevalence of children with a secondary surgical intervention was reported in 18 studies with 1,076 patients and showed statistically significant heterogeneity (p < 0.0001) and true variance between studies (I2 = 97.7%) (Fig. 5). Prevalences ranged from 0% [47] to 73.4% [46], with a pooled prevalence of 27.9% (95% CI = 17.7–38.2%). Even when inclusion criteria for split renal function at enrolment were restricted to patients with values ≥ 40%, the prevalence of patients with secondary surgical intervention was highly variable. The pooled prevalence was 28.3% (95% CI = 18.1–38.5%, 485 patients) with significant heterogeneity (p < 0.0001) and high inconsistency (I2 = 96.3%). Studies exclusively reporting on children < 1 year yielded a lower pooled prevalence of 23.1% (95% CI = 12.7–33.5%, 905 patients); the heterogeneity was significant (p < 0.0001) and the true variance between studies remained high (I2 = 97.5%). Removal of large (≥100 patients) and small (≤20 patients) studies had no effect on the pooled prevalence (29.8%, 95% CI = 12.5–47.1%, 424 patients), with retained significant heterogeneity (p < 0.0001) and considerable inconsistency (I2 = 97.1%). If only studies with follow-up times of more than 2 years for most or all patients were considered (18.6%, 95% CI = 3.5–33.7%, 203 patients), heterogeneity remained significant (p < 0.0001) and variance among studies was high (I2 = 93.4%).
Pooled prevalence of secondary surgery. Data visualised as described in Fig. 3
We aimed to model the secondary surgery prevalence using meta-regression. Significant portions of variability were explained by the predictors age at presentation (negative correlation with age < 1 year) and study country (higher rates outside of the Anglosphere). Overall, residual variance could be reduced to 73.5% through regression analysis. A stratification of studies on children < 1 year revealed a lower pooled secondary surgery prevalence if all patients presented with a split renal function ≥ 40% (7.1%, 95% CI = 1.2–12.9%, 235 patients), compared with studies that also included patients presenting with a split renal function < 40% (27.2%, 95% CI = 0–54.5%, 463 patients).
The criteria for secondary surgical intervention were usually not precisely defined in the 18 studies available. In all but 2 studies [43, 50] the criterion “deterioration of split renal function” was reported, but only 5 studies [24, 39, 42, 48, 52] specified the deterioration as a decrease of 5 or 10%. The criteria progressive hydronephrosis, drainage pattern, or clinical associated symptoms were used less frequently, and only defined for hydronephrosis in one study [39].
Hydronephrosis deterioration
A progression of hydronephrosis was reported in 4 studies (173 patients), 2 of which used a grading system (Society for Fetal Urology, SFU) [26, 55]. The pooled prevalence of hydronephrosis deterioration (Fig. 6) was not heterogeneous (p = 0.1436) and showed moderate variance across studies (I2 = 44.6%). It amounted to 3.2% (95% CI = 0–8.1%) for a total of 173 patients. The separate analysis of patients with and without secondary surgical intervention showed the same trend, without heterogeneity (p = 0.5647 and p = 0.5104) or inconsistency (I2 = 0%), and a pooled prevalence of 0% (95% CI = 0.0–0.0%) each.
Pooled prevalence of hydronephrosis deterioration. Each study is marked according to the information available for urinary tract dilatation at enrolment. A all patients with SFU grade 3 or 4, B additionally patients with lower SFU grades, C unknown. Data visualised as described in Fig. 4
Drainage pattern improvement
For the outcome “drainage pattern” we used improvement as the criterion owing to difficulties in the evaluation of an already progressively obstructed drainage pattern. Only 2 studies defined the outcome, either as a change to a non-obstructed state or as a decrease to 15–25 min drainage half-time [45, 50]. The outcome was reported for a total of 112 patients (Fig. 7) and amounted to a pooled prevalence of 82.2% (95% CI = 69.2–95.2%), with significant heterogeneity and moderate inconsistency (I2 = 64.7%). For the 76 children with split renal function ≥ 40% at enrolment, the prevalence was almost identical (83.1%, 95% CI = 64.3–100%, p = 0.1794, I2 = 41.8%). Subgroup analysis showed no variation in this outcome for patients with secondary surgery (n = 55, pooled prevalence: 100%, 95% CI = 100.0–100.0%, p = 1, I2 = 0%), but significant heterogeneity (p = 0.0005) and inconsistency (I2 = 83%) for the solely non-surgical patients (n = 193, pooled prevalence 47.6%, 95% CI = 26.9–68.4%).
Pooled prevalence of drainage pattern improvement. Each study is marked according to the information available for the drainage pattern at enrolment. A at least 50% of study patients with drainage half-time ≥ 20 min, B additionally patients with shorter drainage half-times, C unknown. Data visualised as described in Fig. 4
Discussion
To the best of our knowledge we present the first systematic review assessing the non-surgical management of unilateral UPJO in children. Previous systematic reviews and meta-analyses have summarised the sonographic outcome of patients with prenatal hydronephrosis [56, 57]. Knowledge concerning the aetiology of hydronephrosis, however, is indispensable for treatment in the daily clinical routine. Our systematic review focuses on patients with unilateral UPJO to determine if there is sufficient evidence supporting the current trend towards non-surgical management and to highlight areas of ambiguity and inconsistency that need further clarification.
The applied literature search was extensive and broad enough to detect any current evidence of unilateral UPJO in children. During study selection, several important aspects in the evaluation of the literature attracted attention. At first, the distinction between a dilated upper urinary tract that is obstructed and a dilated but unobstructed system is of fundamental importance [26, 58, 59]. Nevertheless, a considerable number of publications report cohorts of patients as having unilateral UPJO based on the sonographic detection of hydronephrosis, without presenting any evidence of impaired urinary outflow (Supplementary Material 3). Furthermore, the therapy decision for upper urinary tract dilatation is considerably biased depending on whether the obstruction involves one or both kidneys [60, 61]. Yet, a significant number of publications report on a mixed population with uni- and bilateral UPJO (Supplementary Material 3). Renal imaging in bilateral cases of UPJO cannot reliably assess split renal function, and therefore the threshold for clinical decision-making towards surgical intervention is often lower than in patients with unilateral UPJO. Studies limited by these criteria may lead to an over- or underestimation of the effects of non-surgical management in children with unilateral UPJO and, therefore, needed to be excluded [23, 62–70].
We were able to include 1,083 patients, for whom outcomes were seldom reported as quantitative measures, but rather as qualitative statements, thereby limiting the informative value of changes seen in split renal function and revealing the need for the establishment of precise and constant definitions of improvement and deterioration.
Even though deterioration of split renal function plays a crucial role in clinical decision-making, this outcome was reported for only one third of all study patients and mostly for children with good renal function at enrolment (≥40%). We showed that 21% of patients seem to have a risk for split renal function deterioration during the study period, contrasting the largest study included with only 3.6% [41]. On average, the secondary surgical group comprised a considerable number of children, who had impaired function at the last follow-up and did not regain split renal function values comparable with the measurement at enrolment. This finding challenges the hypothesis that surgical treatment might alleviate the progression of split renal function deterioration [15, 53].
For patient counselling it is necessary to assess the risk for secondary surgical intervention. The pooled prevalence for patients with ≥ 40% split renal function (28.3%) was comparable with previous findings [24, 51]. Interestingly, the pooled prevalence did not change if all children, independently of initial renal function, were included (27.9%). In addition, if only small children (<1 year of age) were taken into account, the rate remained similar at approximately 25%, revealing that older children with clinical symptoms or already progressed renal function deterioration did not lead to bias.
Meta-regression elucidated a significant influence of two parameters (study country and age at presentation) on the secondary surgery rate, yet did not greatly reduce overall variance among studies. The effect of the study country could be explained by different customs in the diagnostic and therapeutic management of UPJO. Studies including very young children (<1 year of age) with a split renal function < 40% also have an increased secondary surgery prevalence compared with those presenting with split renal function > 40% only, reflecting the hypothesis that initially decreased renal function is associated with a more severe renal drainage impairment [39, 52, 53].
Despite the fact that ultrasound is the preferred screening tool for detecting a progressive grade of hydronephrosis, this outcome was rarely reported; yet, it was the outcome least affected by heterogeneity, even though different systems of classification were used for its assessment. Only 3.2% of all patients showed an increase in hydronephrosis. Our low prevalence seems surprising, as it did not correlate with the much higher prevalence seen for split renal function deterioration and the rate of secondary surgery. This finding raises the question whether ultrasound screening is an adequate tool in the detection of UPJO progression by using a customary grading system, or if it poses the risk of underestimating the pathology of non-surgical management downward.
The drainage pattern was the least often reported outcome (112 patients) and its profound variability in prevalences reflects the challenges associated with the interpretation of this imaging technique. We saw a pooled prevalence of 82.2% for improved drainage (irrespective of split renal function at enrolment), which implies that on average only 17.8% of all patients had an unchanged or increased obstruction during follow-up. This represents a much smaller rate than would have been expected by the deterioration rates seen in split renal function. Surprisingly, the subgroup of non-operated children had an almost 50% chance of drainage improvement without intervention during follow-up.
The studies included in this systematic review show a lack of proper reporting, which may reflect pre-2001 Consolidated Standards of Reporting Trials (CONSORT) conditions (http://www.consort-statement.org/), but can have a large impact on study results. Heterogeneity of meta-analyses was found to be substantial for almost all outcomes, even though the selection of patients in the individual studies was predominantly adequate (Figs. 2, 3). Reasons for heterogeneity are likely found in the observed high risk of bias in the measurement of interventions and outcomes of the studies included. Meta-regression and sensitivity analysis did not alleviate the underlying problem.
The variable definitions of unilateral UPJO and the lack of standardised criteria in the use and assessment of renal imaging hampers not only the interpretation, but also the comparability of results. Incongruity of management protocols may challenge the risk assessment of the non-surgical management of the pre-specified outcomes. Furthermore, the criteria for surgical intervention remained unclear in most of the studies.
In the short-term follow-up, approximately 4 out of 5 children with unilateral UPJO may be treated safely with non-surgical management; yet, most studies had a follow-up period that was too short to evaluate the long-term outcome. For patients with secondary surgical intervention, neither progressive split renal function deterioration nor improvement could be observed.
Conclusion
In summary, the major concerns demonstrated in the review are the variable definitions of unilateral UPJO, the indistinct interpretation of the diagnostic methods applied and the inconsistencies and inaccuracies in the reporting of methods and outcomes [6, 25, 71, 72]. Against this background, the evidence for effective non-surgical management in children with unilateral UPJO needs to be critically assessed, especially given that non-surgical UPJO management entails long-term monitoring throughout childhood, accompanied by serial functional imaging with radiation exposure, anaesthesia and/or analgo-sedation, which could potentially lead to serious adverse effects [73–76]. Currently, recommendations cannot be made in favour of or against the non-surgical treatment of UPJO in children based on the studies available to date. Nevertheless, even though we could not resolve the ongoing controversy, we were able to gain further insight by providing realistic estimates of non-surgical management of unilateral UPJO. The currently favoured non-surgical management is based on data of poor quality with incongruent types of decision trees for clinical practice within the studies included, and emphasises the need for a high-quality clinical trial that overcomes the reported limitations of previous studies, and provides not only sufficient data for patient risk stratification, but also for long-term outcome. A collaboration between pediatric nephrologists and urologists with common pre-defined therapy criteria is needed for the implementation of such a study.
References
Koff SA (2008) Requirements for accurately diagnosing chronic partial upper urinary tract obstruction in children with hydronephrosis. Pediatr Radiol 38 [Suppl 1]:S41–S48
Woodward M, Frank D (2002) Postnatal management of antenatal hydronephrosis. BJU Int 89(2):149–156
Braga LH, Ruzhynsky V, Pemberton J, Farrokhyar F, Demaria J, Lorenzo AJ (2014) Evaluating practice patterns in postnatal management of antenatal hydronephrosis: a national survey of Canadian pediatric urologists and nephrologists. Urology 83(4):909–914
Williams B, Tareen B, Resnick MI (2007) Pathophysiology and treatment of ureteropelvic junction obstruction. Curr Urol Rep 8(2):111–117
Ingraham SE, McHugh KM (2011) Current perspectives on congenital obstructive nephropathy. Pediatr Nephrol 26(9):1453–1461
Nguyen HT, Herndon CDA, Cooper C, Gatti J, Kirsch A, Kokorowski P, Lee R, Perez-Brayf-Ield M, Metcalfe P, Yerkes E, Cendron M, Campbell JB (2010) The Society for Fetal Urology consensus statement on the evaluation and management of antenatal hydronephrosis. J Pediatr Urol 6(3):212–231
Piepsz A (2011) Antenatal detection of pelviureteric junction stenosis: main controversies. Semin Nucl Med 41(1):11–19
Riccabona M, Avni FE, Blickman JG, Dacher JN, Darge K, Lobo ML, Willi U (2009) Imaging recommendations in paediatric uroradiology. minutes of the ESPR uroradiology task force session on childhood obstructive uropathy, high-grade fetal hydronephrosis, childhood haematuria, and urolithiasis in childhood. ESPR Annual Congress, Edinburgh, UK, June 2008. Pediatr Radiol 39(8):891–898
Tondeur M, De Palma D, Roca I, Piepsz A, Ham H (2008) Inter-observer reproducibility in reporting on renal drainage in children with hydronephrosis: a large collaborative study. Eur J Nucl Med Mol Imaging 35(3):644–654
Csaicsich D, Greenbaum LA, Aufricht C (2004) Upper urinary tract: when is obstruction obstruction? Curr Opin Urol 14(4):213–217
Malki M, Linton KD, Mackinnon R, Hall J (2012) Conservative management of pelvi-ureteric junction obstruction (PUJO): is it appropriate and if so what duration of follow-up is needed? BJU Int 110(3):446–448
Shokeir AA, Nijman RJ (2000) Antenatal hydronephrosis: changing concepts in diagnosis and subsequent management. BJU Int 85(8):987–994
Zhang PL, Peters CA, Rosen S (2000) Ureteropelvic junction obstruction: morphological and clinical studies. Pediatr Nephrol 14(8-9):820–826
Josephson S (2002) Antenatally detected, unilateral dilatation of the renal pelvis: a critical review. 2. postnatal non-operative treatment—long-term hazards, urgent research. Scand J Urol Nephrol 36(4):251–259
Josephson S (2002) Antenatally detected, unilateral dilatation of the renal pelvis: a critical review. 1. postnatal non-operative treatment 20 years on—is it safe? Scand J Urol Nephrol 36(4):243–250
Bernstein GT, Mandell J, Lebowitz RL, Bauer SB, Colodny AH, Retik AB (1988) Ureteropelvic junction obstruction in the neonate. J Urol 140(5 Pt 2):1216–1221
King LR, Coughlin PW, Bloch EC, Bowie JD, Ansong K, Hanna MK (1984) The case for immediate pyeloplasty in the neonate with ureteropelvic junction obstruction. J Urol 132(4):725–728
Chevalier RL, Forbes MS, Thornhill BA (2009) Ureteral obstruction as a model of renal interstitial fibrosis and obstructive nephropathy. Kidney Int 75(11):1145–1152
Chevalier RL, Thornhill BA, Forbes MS, Kiley SC (2010) Mechanisms of renal injury and progression of renal disease in congenital obstructive nephropathy. Pediatr Nephrol 25(4):687–697
Chevalier RL, Thornhill BA, Wolstenholme JT, Kim A (1999) Unilateral ureteral obstruction in early development alters renal growth: dependence on the duration of obstruction. J Urol 161(1):309–313
Shi Y, Pedersen M, Li C, Wen JG, Thomsen K, Stodkilde-Jorgensen H, Jorgensen TM, Knepper MA, Nielsen S, Djurhuus JC, Frokiaer J (2004) Early release of neonatal ureteral obstruction preserves renal function. Am J Physiol Renal Physiol 286(6):F1087–F1099
Thornhill BA, Burt LE, Chen C, Forbes MS, Chevalier RL (2005) Variable chronic partial ureteral obstruction in the neonatal rat: a new model of ureteropelvic junction obstruction. Kidney Int 67(1):42–52
Heinlen JE, Manatt CS, Bright BC, Kropp BP, Campbell JB, Frimberger D (2009) Operative versus nonoperative management of ureteropelvic junction obstruction in children. Urology 73(3):521–525, discussion 525
Dhillon HK (1998) Prenatally diagnosed hydronephrosis: the Great Ormond Street experience. Br J Urol 81 [Suppl 2]:39–44
Nguyen HT, Benson CB, Bromley B, Campbell JB, Chow J, Coleman B, Cooper C, Crino J, Darge K, Herndon CD, Odibo AO, Somers MJ, Stein DR (2014) Multidisciplinary consensus on the classification of prenatal and postnatal urinary tract dilation (UTD classification system). J Pediatr Urol 10(6):982–998
Ulman I, Jayanthi VR, Koff SA (2000) The long-term followup of newborns with severe unilateral hydronephrosis initially treated nonoperatively. J Urol 164(3 Pt 2):1101–1105
Weitz M, Portz S, Laube GF, Meerpohl JJ, Bassler D (2016) Surgery versus non-surgical management for unilateral ureteric-pelvic junction obstruction in newborns and infants less than two years of age. Cochrane Database Syst Rev 7:CD010716
Grimes DA, Schulz KF (2002) Cohort studies: marching towards outcomes. Lancet 359(9303):341–345
Laupacis A, Wells G, Richardson WS, Tugwell P (1994) Users’ guides to the medical literature. V. how to use an article about prognosis. evidence-based medicine working group. JAMA 272(3):234–237
Mulder RL, van Dalen EC, Van den Hof M, Bresters D, Koot BG, Castellino SM, Loke Y, Leclercq E, Post PN, Caron HN, Postma A, Kremer LC (2011) Hepatic late adverse effects after antineoplastic treatment for childhood cancer. Cochrane Database Syst Rev 7:CD008205
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560
Higgins JPT, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21(11):1539–1558
R Core Team (2014) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, https://www.R-project.org
Schwarzer G (2015) meta: General package for meta-analysis. http://CRAN.R-project.org/package=meta
Egger M, Davey Smith G, Schneider M, Minder C (1997) Bias in meta-analysis detected by a simple, graphical test. BMJ 315(7109):629–634
Ransley PG, Dhillon HK, Gordon I, Duffy PG, Dillon MJ, Barratt TM (1990) The postnatal management of hydronephrosis diagnosed by prenatal ultrasound. J Urol 144(2 Pt 2):584–587; discussion 593-584
Gordon I, Dhillon HK, Gatanash H, Peters AM (1991) Antenatal diagnosis of pelvic hydronephrosis - assessment of renal-function and drainage as a guide to management. J Nucl Med 32(9):1649–1654
Cartwright PC, Duckett JW, Keating MA, Snyder HM 3rd, Escala J, Blyth B, Heyman S (1992) Managing apparent ureteropelvic junction obstruction in the newborn. J Urol 148(4):1224–1228
Chertin B, Pollack A, Koulikov D, Rabinowitz R, Hain D, Hadas-Halpren I, Farkas A (2006) Conservative treatment of ureteropelvic junction obstruction in children with antenatal diagnosis of hydronephrosis: lessons learned after 16 years of follow-up. Eur Urol 49(4):734–738
Duong HP, Piepsz A, Collier F, Khelif K, Christophe C, Cassart M, Janssen F, Hall M, Ismaili K (2013) Predicting the clinical outcome of antenatally detected unilateral pelviureteric junction stenosis. Urology 82(3):691–696
Freedman ER, Rickwood AM (1994) Prenatally diagnosed pelviureteric junction obstruction: a benign condition? J Pediatr Surg 29(6):769–772
Gordon I, Dhillon HK, Peters AM (1991) Antenatal diagnosis of renal pelvic dilatation--the natural history of conservative management. Pediatr Radiol 21(4):272–273
Gruenewald SM, Cohen RC, Antico VF, Farlow DC, Cass DT (1990) Diagnosis and treatment of antenatal uropathies. J Paediatr Child Health 26(3):142–147
Homsy YL, Williot P, Danais S (1986) Transitional neonatal hydronephrosis: fact or fantasy? J Urol 136(1 Pt 2):339–341
Jain V, Agarwala S, Bhatnagar V, Gupta AK, Kumar R, Bal CS (2012) Long term outcome of management of antenatally diagnosed pelvi-ureteric junction obstruction. Indian J Pediatr 79(6):769–773
Kaselas C, Papouis G, Grigoriadis G, Klokkaris A, Kaselas V (2007) Pattern of renal function deterioration as a predictive factor of unilateral ureteropelvic junction obstruction treatment. Eur Urol 51(2):551–555
Koff SA, Campbell K (1992) Nonoperative management of unilateral neonatal hydronephrosis. J Urol 148(2 Pt 2):525–531
Koff SA, Campbell KD (1994) The nonoperative management of unilateral neonatal hydronephrosis: natural history of poorly functioning kidneys. J Urol 152(2 Pt 2):593–595
Madden NP, Thomas DF, Gordon AC, Arthur RJ, Irving HC, Smith SE (1991) Antenatally detected pelviureteric junction obstruction. Is non-operation safe? Br J Urol 68(3):305–310
Nonomura K, Yamashita T, Kanagawa K, Itoh K, Koyanagi T (1994) Management and outcome of antenatally diagnosed hydronephrosis. Int J Urol 1(2):121–128
Palmer LS, Maizels M, Cartwright PC, Fernbach SK, Conway JJ (1998) Surgery versus observation for managing obstructive grade 3 to 4 unilateral hydronephrosis: a report from the Society for Fetal Urology. J Urol 159(1):222–228
Shokeir AA, El-Sherbiny MT, Gad HM, Dawaba M, Hafez AT, Taha MA, Saida M, Ghaly AM (2005) Postnatal unilateral pelviureteral junction obstruction: impact of pyeloplasty and conservative management on renal function. Urology 65(5):980–985; discussion 985
Subramaniam R, Kouriefs C, Dickson AP (1999) Antenatally detected pelvi-ureteric junction obstruction: concerns about conservative management. BJU Int 84(3):335–338
Thorup J, Lenz K, Rabol A, Passalides A, Nielsen OH (1996) Follow-up of prenatally diagnosed unilateral hydronephrosis. Pediatr Surg Int 11(1):18–21
Ylinen E, Ala-Houhala M, Wikstrom S (2004) Outcome of patients with antenatally detected pelviureteric junction obstruction. Pediatr Nephrol 19(8):880–887
Sidhu G, Beyene J, Rosenblum ND (2006) Outcome of isolated antenatal hydronephrosis: a systematic review and meta-analysis. Pediatr Nephrol 21(2):218–224
Lee RS, Cendron M, Kinnamon DD, Nguyen HT (2006) Antenatal hydronephrosis as a predictor of postnatal outcome: a meta-analysis. Pediatrics 118(2):586–593
Parkhouse HF, Barratt TM (1988) Investigation of the dilated urinary tract. Pediatr Nephrol 2(1):43–47
Piepsz A, Gordon I, Brock J 3rd, Koff S (2009) Round table on the management of renal pelvic dilatation in children. J Pediatr Urol 5(6):437–444
Onen A, Jayanthi VR, Koff SA (2002) Long-term followup of prenatally detected severe bilateral newborn hydronephrosis initially managed nonoperatively. J Urol 168(3):1118–1120
Josephson S, Dhillon HK, Ransley PG (1993) Post-natal management of antenatally detected, bilateral hydronephrosis. Urol Int 51(2):79–84
Buisson P, Ricard J, Boudailliez B, Canarelli JP (2003) Change in management of ureteropelvic junction obstruction. Arch Pediatr 10(3):215–220
Csaicsich D, Muller T, Arbeiter K, Balzar E, Thereska L, Aufricht C (2004) Management of congenital hydronephrosis with ureteropelvic junction obstruction: the Vienna-AKH experience 1986-2001. Wien Klin Wochenschr 116(21–22):725–729
Ismail A, Elkholy A, Zaghmout O, Alkadhi A, Elnaggar O, Khairat A, Elhassanat H, Mosleh A, Hamad B, Elzomer J, Elkaabi A (2006) Postnatal management of antenatally diagnosed ureteropelvic junction obstruction. J Pediatr Urol 2(3):163–168
Karnak I, Woo LL, Shah SN, Sirajuddin A, Ross JH (2009) Results of a practical protocol for management of prenatally detected hydronephrosis due to ureteropelvic junction obstruction. Pediatr Surg Int 25(1):61–67
Ocheke IE, Antwi S, Gajjar P, McCulloch MI, Nourse P (2014) Pelvi-ureteric junction obstruction at Red Cross Children’s Hospital, Cape Town: a six year review. Arab J Nephrol Transplant 7(1):33–36
Onen A (2007) An alternative grading system to refine the criteria for severity of hydronephrosis and optimal treatment guidelines in neonates with primary UPJ-type hydronephrosis. J Pediatr Urol 3(3):200–205
Onen A (2007) Treatment and outcome of prenatally detected newborn hydronephrosis. J Pediatr Urol 3(6):469–476
Rickwood AM, Godiwalla SY (1997) The natural history of pelvi-ureteric junction obstruction in children presenting clinically with the complaint. Br J Urol 80(5):793–796
Ross SS, Kardos S, Krill A, Bourland J, Sprague B, Majd M, Pohl HG, Gibbons MD, Belman AB, Rushton HG (2011) Observation of infants with SFU grades 3-4 hydronephrosis: worsening drainage with serial diuresis renography indicates surgical intervention and helps prevent loss of renal function. J Pediatr Urol 7(3):266–271
Fernbach SK, Maizels M, Conway JJ (1993) Ultrasound grading of hydronephrosis: introduction to the system used by the Society for Fetal Urology. Pediatr Radiol 23(6):478–480
Conway J, Maizels M (1992) The well tempered diuretic renogram—a standard method to examine the asymptomatic neonate with hydronephrosis or hydroureteronephrosis. J Nucl Med 33(11):2047–2051
Fazel R, Krumholz HM, Wang Y, Ross JS, Chen J, Ting HH, Shah ND, Nasir K, Einstein AJ, Nallamothu BK (2009) Exposure to low-dose ionizing radiation from medical imaging procedures. N Engl J Med 361(9):849–857
Karnak I, Woo LL, Shah SN, Sirajuddin A, Kay R, Ross JH (2008) Prenatally detected ureteropelvic junction obstruction: clinical features and associated urologic abnormalities. Pediatr Surg Int 24(4):395–402
McCann ME, Soriano SG (2012) General anesthetics in pediatric anesthesia: influences on the developing brain. Curr Drug Targets 13(7):944–951
Stratton KL, Pope JC, Adams MC, Brock JW 3rd, Thomas JC (2010) Implications of ionizing radiation in the pediatric urology patient. J Urol 183(6):2137–2142
Moher D, Liberati A, Tetzlaff J, Altman DG, Group P (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6(7):e1000097
Acknowledgements
We would like to thank Gail Higgins, the Trials Search Co-ordinator of the Cochrane Renal Group and Christina Walter, the Search Co-ordinator at the University Hospital Tübingen (Germany) for developing and running the search strategy used in this review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Weitz, M., Schmidt, M. & Laube, G. Primary non-surgical management of unilateral ureteropelvic junction obstruction in children: a systematic review. Pediatr Nephrol 32, 2203–2213 (2017). https://doi.org/10.1007/s00467-016-3566-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-016-3566-3