Abstract
Background
Contrast-induced acute kidney injury (CI-AKI) is a common pathology among adult patients, with an incidence ranging from 3–25 % depending on risk factors. Little information is available regarding CI-AKI incidence, risk factors, and prognostic impact in the pediatric population.
Methods
We performed a retrospective study of pediatric patients who underwent computed tomography (CT) scan with iodinated contrast media injection between 2005 and 2014 in five pediatric units of a university hospital. CI-AKI was defined according to Kidney Disease/Improving Global Outcomes (KDIGO) criteria.
Results
Of 346 identified patients, 233 had renal function follow-up and were included in our analyses. CI-AKI incidence was 10.3 % [95 % confidence interval (CI) 6.4–14.2 %]. CI-AKI was associated with 30-day unfavorable outcome before (45.8 % vs. 19.7 %, P = 0.007) and after [odds ratio (OR) 3.6; 95 % CI 1.4–9.5] adjustment for confounders. No independent risk factors of CI-AKI were identified.
Conclusions
CI-AKI incidence was as high as 10.3 % following intravenous contrast media administration in the pediatric setting. As reported among adults, CI-AKI was associated with unfavorable outcome after adjustment for confounders. Although additional studies are needed in the pediatric setting, our data suggest that physicians should maintain a high degree of suspicion toward this complication among pediatric patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Acute kidney injury (AKI) develops in up to 60 % of critically ill patients and is associated with high morbidity and mortality rates and an increased risk of subsequent chronic kidney disease (CKD) [1–3]. Despite improvements in dialysis technology and supportive care, AKI-associated morbidity and mortality rates have not changed for several decades [4]. Several recent studies suggest that AKI itself may influence patient outcomes independently of the nature and severity of the underlying disease and metabolic consequences of renal dysfunction [5–7].
Contrast medium use is a major risk factor for AKI, and the increasing application of contrast media for diagnostic and interventional procedures has made contrast-induced acute kidney injury (CI-AKI) the third most common cause of hospital-acquired AKI [8, 9]. CI-AKI is most commonly defined as serum creatinine (SCr) elevation of 0.5 mg/dl (44 μmol/L) or of 25 % within 48 h after exposure to contrast medium [10]. However, the relevance of this definition is controversial following reports of delayed SCr elevation and differences between AKI definitions and the specific CI-AKI definition [11, 12]. Thus, the recent Kidney Disease/Improving Global Outcomes (KDIGO) guidelines suggested their definition be used to define AKI even in this specific context [13].
CI-AKI incidence varies widely among studies depending on definitions used, study population, and baseline risk factors. CI-AKI may affect as little as 3 % of low-risk patients [14] and as many as 25 % of patients with risk factors, such as pre-existing renal dysfunction, diabetes, advanced age, or concurrent nephrotoxic drug exposure [15]. Only limited data report CI-AKI incidences in specific patient populations, including in the pediatric setting. A recent study reported a CI-AKI incidence ranging from 5 to 10 % in a population of 128 pediatric patients [16].
The primary objective of this study was to assess CI-AKI incidence among pediatric patients in a university hospital. Secondary objectives were to assess the risk factors and prognostic impact of CI-AKI in this setting.
Methods
Patients
We performed an observational retrospective cohort study in the following five units of the Saint-Etienne University Hospital: Pediatrics, Pediatric Oncology, Pediatric Surgery, Pediatric Intensive Care Unit, and Pediatric Emergency Department. The study involved pediatric patients (age <16 years) admitted between January 2005 and September 2014 who underwent computed tomography (CT) scan with iodinated contrast media injection. Patients without renal function assessment either before or following contrast media injection were secondarily excluded from the main analysis. In case of multiple CT scans with contrast media infusion or multiple hospitalizations, only the first was considered.
Definitions
In accordance with the KDIGO classification system, AKI was defined as a SCr increase of ≥26.4 μmol/L within 48 h or of ≥150 % from baseline presumed to have occurred within the prior 7 days or as urine output of <0.5 ml/kg/h for ≥6 h (i.e., oliguria) [13, 17]. Baseline renal function was based on the baseline Cr and estimated glomerular filtration rate (eGFR; determined using the revised 2009 version of Schwartz’s formula [18]) at the time of contrast media injection; any degree of AKI within 48 h was considered contrast-associated nephropathy. Urine output was defined as shift urine output observed or estimated according to weight of diapers in infants. Foley catheter was uncommonly used in the studied patients, except in intensive care unit (ICU) patients. Iodinated contrast media received during the previous 72 h either in another hospital or in prior ambulatory care was recorded. The American College of Chest Physicians/Society of Critical Care Medicine consensus conference definitions were used to evaluate sepsis, severe sepsis, or septic shock [19]. Hemorrhage was defined as any active hemorrhage requiring red blood cell transfusion. Hypovolemia at admission was diagnosed based on clinical judgment at the time of CT scan. An unfavorable outcome was defined as a need for hospital readmission within 30 days, need for ICU admission within 30 days, or 30-day mortality.
Protocol
Pediatric patients who underwent CT scan with contrast media injection were identified from the electronic hospital registry. The characteristics reported in Table 1 were extracted from the charts of these patients. We also recorded the reason for the scan, type and volume of contrast media used, unit of hospitalization, and outcome variables.
Statistical analysis
Results are reported as median and interquartile range (IQR) or as numbers and percentages. Categorical variables were compared using the chi-square test or Fisher’s exact test, as appropriate. Continuous variables were compared using the nonparametric Wilcoxon test. To identify variables significantly associated with CI-AKI and with unfavorable outcome, we performed conditional forward logistic regression analyses. The model included clinically relevant variables and variables yielding a P value of ≤0.20 in bivariate analysis, the latter being maintained in the final model. Variables are reported as estimated odds ratios (ORs) with their 95 % confidence intervals (95 % CIs). Co-linearity and interactions were tested. The Hosmer-Lemeshow test was used to check goodness of fit of the logistic regression. All tests were two sided, and P <0.05 was considered to indicate statistical significance. Statistical tests were performed using the SPSS 13 software package (IBM, Armonk, NY, USA).
Results
Study population
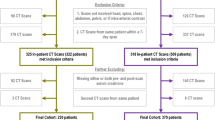
We identified 346 pediatric patients who underwent CT scan with contrast media injection (Fig. 1). Of these patients, 233 were followed for renal function and were therefore included in this study. Median patient age was of 5.9 years (IQR 1.9–13.7 years), and 142 patients (60.9 %) were boys. Table 1 presents the main characteristics of the included patients.
Few patients had important comorbidities, including eight patients with pre-existing CKD, four with chronic heart dysfunction, three with history of hypertension, and two with history of glomerular disease without renal dysfunction. No patient was diagnosed with diabetes. The most common reason for performing a CT scan was a surgical condition (118 patients; 50.6 %). According to the usual indications of CT scan in our institution, every scan was done within 48 h following admission. The median SCr level at the time of CT scan was 38 μmol/L (IQR 28–55 μmol/L) and median baseline eGFR 107 ml/min/1.73 m2 (IQR 89–132 ml/min/1.73 m2). All CT scans were performed using iso- or hypo-osmolar contrast media. Median contrast media volume was 1.9 ml/kg (IQR 1.6–2.1 ml/kg). At the time of CT scan, 57 patients (16.6 %) had received nephrotoxic drugs within the previous 72 h. No patient developed an anaphylactic reaction or required renal replacement therapy (RRT) at time of inclusion or of contrast media infusion.
Contrast-induced acute kidney injury
Among the included patients, 24 (10.3 %; 95 % CI 6.4–14.2 %) developed CI-AKI, of whom nine (38 %) had no previous history of CKD and no other identified risk factors of AKI (concomitant nephrotoxic agents or hypovolemia). Volume of infused contrast media was similar in patients without [1.9 ml/kg (IQR) 1.6-2.1] and with CI-AKI (1.9 ml/kg [IQR 1.7-2.1]; P = 0.67). Of these patients, 15 (63 %) showed elevated SCr. The remaining nine were classified as having AKI based on oliguria and two patients having both criteria. Maximum AKI severity was stage 1 in seven patients (29 %), stage 2 in six (25 %), and stage 3 in 11 (46 %). A single patient with CI-AKI required RRT. Logistic regression analysis found no factors independently associated with CI-AKI (Table 2). Factors retained in the final model, as well as OR trends, were unchanged in subgroup analyses evaluating patients reaching AKI criteria separately according to oliguria or SCr elevation.
Outcome
Unfavorable outcome was reported for 41 patients without (19.7 %) and 11 patients with (45.8 %; P = 0.007) CI-AKI. The rate of hospital readmission within 30 days was 25 % among patients with vs. 8.6 % among patients without CI-AKI (P = 0.02). Hospital mortality rate was 16.7 % among patients with vs. 6.7 % among those without CI-AKI (P = 0.09) (Fig. 2). After adjustment for confounders, unfavorable outcome was independently associated with severe sepsis or septic shock at the time of CT scan (OR 3.5; 95 % CI 1.5–7.8), younger age (OR per year 0.89; 95 % CI 0.84–0.95), and contrast-associated nephropathy (OR 3.6; 95 % CI 1.4–9.5). Lastly, unfavorable outcome in patients with CI-AKI according to SCr and urine output criteria are reported in supplementary appendix (Figure S1).
Discussion
To the best of our knowledge, this is the largest study to assess CI-AKI incidence in a pediatric setting. Using the KDIGO definition, which was recently validated in children [17], we found a CI-AKI rate similar to previously reported rates in adult patients [14, 15]. Also in line with previous studies, our findings suggest that CI-AKI is associated with poorer outcome, including a higher rehospitalization rate and a trend toward a higher 30-day mortality rate. Our analyses identified no independent risk factors for AKI in the pediatric setting.
Most studies have assessed CI-AKI incidence following cardiac catheterization, and thus CI-AKI incidence remains poorly evaluated in specific settings, such as among pediatric and ICU patients. Moreover, CI-AKI incidence varies widely across studies depending on definitions used, study population, and baseline risk factors. The recent KDIGO guidelines underline the need for further epidemiological studies in specific settings to improve available CI-AKI knowledge [12]. A recent study assessed the renal safety of two iso-osmolar contrast agents in 128 children and found CI-AKI incidences ranging from 5–10 % depending on the contrast media [11], similar to the results of our study. Interestingly, AKI risk in the pediatric population would be expected to be lower, since this patient group shows a relative lack of usual risk factors and always receives contrast media administration IV—two conditions that are usually associated with a lower CI-AKI rate [20, 21]. Interestingly, although pre-existing CKD trended to be associated with higher rate of CI-AKI, patients developing CI-AKI trended also to have higher baseline eGFR and lower baseline creatinine. This finding may reflect an augmented renal clearance previously described in sepsis and trauma patients, especially if critically ill [22, 23]. Additional studies are needed to further explore whether augmented renal clearance is a surrogate for patient severity and whether it may help identify patients at high risk of CI-AKI.
Also in accordance with previous findings, we found that CI-AKI was associated with a poorer 30-day outcome, including an increased rehospitalization rate and a trend toward higher mortality. Several studies have indicated an independent association between AKI and morbidity, hospital mortality, and long-term outcome in adult [1, 6, 24] and pediatric [25–29] settings. Similarly, CI-AKI was reportedly related to rates of higher morbidity, mortality, and long-term risk of adverse events [30–32]. Although the 30-day risk of death was not significantly higher in the CI-AKI group in our study, the heterogeneous population evaluated, the low mortality rate (7.7 %), and the wide CI (OR 2.8; 95 % CI 0.6–10.0) suggest a potential lack of statistical power to detect such an effect.
Several limitations of our study should be addressed. First, the population was heterogeneous, which may have limited the precision of our results and our ability to detect risk factors. Wide variations among evaluated patients should be expected with regard to CI-AKI risk and incidence and the potential consequences of this complication. However, this pilot study was performed with the aim of assessing global CI-AKI incidence within a pediatric population. Although our results remain to be confirmed, they clearly show an important risk of CI-AKI in the pediatric setting despite the lack of usual risk factors and the IV route of contrast media administration in this population.
To allow adequate assessment and comparability of our results, we used the KDIGO definition, which is validated both in the pediatric setting [17, 27, 28, 33] and for assessing renal consequences of contrast media administration [31]. However, this definition is highly sensitive, which may partly explain the high incidence of CI-AKI in our study [34]. Nevertheless, the higher rate of poor outcome in the group with CI-AKI suggests that this definition was adequate for estimating the risk of subsequent complications following renal insult. Additionally, the use of this definition even in a specific context, such as CI-AKI, is recommended by KDIGO guidelines [13].
Additionally, this study was not designed to assess accountability of contrast media on the observed CI-AKI. Previous studies suggest the specific toxic effect of nonionic, low-osmolar iodinated contrast medium to be minimal, especially in patients with multiple causes of renal insult [35]. These findings are to be tempered by the known pathophysiological effects of contrast media in experimental models [36]. In those models, contrast media are associated with renal hypoperfusion, direct tubular damage, and formation of reactive oxygen species that may cause further tissue damage [36]. Future studies in specific settings are required to more clearly assess the specific influence of contrast media in initiating or expanding subclinical renal insult.
The retrospective design of our study precluded adequate assessment of adherence to preventive measures or of the influence of these measures in preventing CI-AKI in this population. Hence, in the participating units, no dedicated protocol was available regarding prevention of CI-AKI. It must be noted, however, that this lack of protocol dedicated to prevention of CI-AKI was a result of the lack of information regarding the incidence of this complication in the pediatric setting. Also, our results may prompt implementation of preventive measures in this specific population. Moreover, urine output was infrequently assessed using a Foley catheter. Hence, both shift estimate in urine output and estimation of diaper weight maybe inaccurate, which might have led to erroneous estimation of urinary output [37, 38], and some patients may have been misclassified as a consequence. Additionally, we were unable to assess AKI duration in this setting, as several patients had unrecognized AKI and were discharged before renal dysfunction recovery. The retrospective design was also associated with a high dropout rate related to the absence of renal assessment following contrast media injection. One third of children exposed to contrast media had no renal function assessment or follow-up. It is not known how these excluded patients might have influenced the CI-AKI incidence in the studied population. However, this excluded population showed a 10 % rate of 30-day complications, compared with rates of 20 % in the patients without and 46 % in patients with CI-AKI (P <0.0001). This suggests that CI-AKI incidence may have been lower among the excluded patients and therefore in the exposed population. However, in the worst-case scenario, and assuming that none of these patients developed CI-AKI, this translates into a 6.4 % incidence of CI-AKI (95 % CI 4.3-9.6), which remains higher than expected in a population of patients usually considered at low risk of CI-AKI.
Despite its limitations, our study results suggest a 10.3 % incidence of CI-AKI according to the KDIGO definition (95 % CI 6.4–14.2 %) and suggests that CI-AKI is independently associated with poor short-term outcome. Thus, despite the limited prevalence of risk factors in this population and the IV administration of contrast media, pediatric patients remain exposed to CI-AKI. Additional studies are needed to confirm our results, assess risk factors, and investigate the preventive measures provided in this specific population. However, our findings suggest that physicians should maintain a high degree of suspicion toward this complication among pediatric patients. Additionally, our results may help in the design of future studies in this field by providing an estimation of CI-AKI incidence in this specific population.
References
Joannidis M, Metnitz B, Bauer P, Schusterschitz N, Moreno R, Druml W, Metnitz PGH (2009) Acute kidney injury in critically ill patients classified by AKIN versus RIFLE using the SAPS 3 database. Intensive Care Med 35:1692–1702
Nisula S, Kaukonen KM, Vaara ST, Korhonen AM, Poukkanen M, Karlsson S, Haapio M, Inkinen O, Parviainen I, Suojaranta-Ylinen R, Laurila JJ, Tenhunen J, Reinikainen M, Ala-Kokko T, Ruokonen E, Kuitunen A, Pettilä V, FINNAKI Study Group (2013) Incidence, risk factors and 90-day mortality of patients with acute kidney injury in Finnish intensive care units: the FINNAKI study. Intensive Care Med 39:420–428
Stads S, Fortrie G, van Bommel J, Zietse R, Betjes MGH (2013) Impaired kidney function at hospital discharge and long-term renal and overall survival in patients who received CRRT. Clin J Am Soc Nephrol 8:1284–1291
Hoste EAJ, Schurgers M (2008) Epidemiology of acute kidney injury: How big is the problem? Crit Care Med 36:S146–S151
Li X, Hassoun HT, Santora R, Rabb H (2009) Organ crosstalk: the role of the kidney. Curr Opin Crit Care 15:481–487
Metnitz PGH, Krenn CG, Steltzer H, Lang T, Ploder J, Lenz K, Le Gall J-R, Druml W (2002) Effect of acute renal failure requiring renal replacement therapy on outcome in critically ill patients. Crit Care Med 30:2051–2058
Liu M, Liang Y, Chigurupati S, Lathia JD, Pletnikov M, Sun Z, Crow M, Ross CA, Mattson MP, Rabb H (2008) Acute kidney injury leads to inflammation and functional changes in the brain. J Am Soc Nephrol 19:1360–1370
Rich MW, Crecelius CA (1990) Incidence, risk factors, and clinical course of acute renal insufficiency after cardiac catheterization in patients 70 years of age or older. A prospective study. Arch Intern Med 150:1237–1242
Nash K, Hafeez A, Hou S (2002) Hospital-acquired renal insufficiency. Am J Kidney Dis 39:930–936
Goldenberg I, Matetzky S (2005) Nephropathy induced by contrast media: pathogenesis, risk factors and preventive strategies. CMAJ 172:1461–1471
Waikar SS, Bonventre JV (2009) Creatinine kinetics and the definition of acute kidney injury. J Am Soc Nephrol 20:672–679
Khwaja A (2012) KDIGO clinical practice guidelines for acute kidney injury. Nephron Clin Pract 120:179–184
Kellum JA, Lameire N, KDIGO AKI Guideline Work Group (2013) Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (Part 1). Crit Care 17:204
Rihal CS, Textor SC, Grill DE, Berger PB, Ting HH, Best PJ, Singh M, Bell MR, Barsness GW, Mathew V, Garratt KN, Holmes DR Jr (2002) Incidence and prognostic importance of acute renal failure after percutaneous coronary intervention. Circulation 105:2259–2264
Rudnick MR, Goldfarb S, Tumlin J (2008) Contrast-induced nephropathy: is the picture any clearer? Clin J Am Soc Nephrol 3:261–262
Zo’o M, Hoermann M, Balassy C, Brunelle F, Azoulay R, Pariente D, Panuel M, Le Dosseur P (2011) Renal safety in pediatric imaging: randomized, double-blind phase IV clinical trial of iobitridol 300 versus iodixanol 270 in multidetector CT. Pediatr Radiol 41:1393–1400
Selewski DT, Cornell TT, Heung M, Troost JP, Ehrmann BJ, Lombel RM, Blatt NB, Luckritz K, Hieber S, Gajarski R, Kershaw DB, Shanley TP, Gipson DS (2014) Validation of the KDIGO acute kidney injury criteria in a pediatric critical care population. Intensive Care Med 40:1481–1488
Schwartz GJ, Work DF (2009) Measurement and estimation of GFR in children and adolescents. Clin J Am Soc Nephrol 4:1832–1843
Levy MM, Fink MP, Marshall JC, Abraham E, Angus D, Cook D, Cohen J, Opal SM, Vincent J-L, Ramsay G (2003) 2001 SCCM/ESICM/ACCP/ATS/SIS international sepsis definitions conference. Intensive Care Med 29:530–538
Moore RD, Steinberg EP, Powe NR, Brinker JA, Fishman EK, Graziano S, Gopalan R (1992) Nephrotoxicity of high-osmolality versus low-osmolality contrast media: randomized clinical trial. Radiology 182:649–655
D’Elia JA, Gleason RE, Alday M, Malarick C, Godley K, Warram J, Kaldany A, Weinrauch LA (1982) Nephrotoxicity from angiographic contrast material. A prospective study. Am J Med 72:719–725
Lautrette A, Phan T-N, Ouchchane L, Aithssain A, Tixier V, Heng A-E, Souweine B (2012) High creatinine clearance in critically ill patients with community-acquired acute infectious meningitis. BMC Nephrol 13:124
De Cock PAJG, Standing JF, Barker CIS, de Jaeger A, Dhont E, Carlier M, Verstraete AG, Delanghe JR, Robays H, De Paepe P (2015) Augmented renal clearance implies a need for increased Amoxicillin-Clavulanic acid dosing in critically Ill children. Antimicrob Agents Chemother 59:7027–7035
Linder A, Fjell C, Levin A, Walley KR, Russell JA, Boyd JH (2014) Small acute increases in serum creatinine are associated with decreased long-term survival in the critically ill. Am J Respir Crit Care Med 189:1075–1081
Greenberg JH, Coca S, Parikh CR (2014) Long-term risk of chronic kidney disease and mortality in children after acute kidney injury: a systematic review. BMC Nephrol 15:184
Schneider J, Khemani R, Grushkin C, Bart R (2010) Serum creatinine as stratified in the RIFLE score for acute kidney injury is associated with mortality and length of stay for children in the pediatric intensive care unit. Crit Care Med 38:933–939
Alkandari O, Eddington KA, Hyder A, Gauvin F, Ducruet T, Gottesman R, Phan V, Zappitelli M (2011) Acute kidney injury is an independent risk factor for pediatric intensive care unit mortality, longer length of stay and prolonged mechanical ventilation in critically ill children: a two-center retrospective cohort study. Crit Care 15:R146
Mammen C, Al Abbas A, Skippen P, Nadel H, Levine D, Collet JP, Matsell DG (2012) Long-term risk of CKD in children surviving episodes of acute kidney injury in the intensive care unit: a prospective cohort study. Am J Kidney Dis 59:523–530
Akcan-Arikan A, Zappitelli M, Loftis LL, Washburn KK, Jefferson LS, Goldstein SL (2007) Modified RIFLE criteria in critically ill children with acute kidney injury. Kidney Int 71:1028–1035
Rudnick M, Feldman H (2008) Contrast-induced nephropathy: what are the true clinical consequences? Clin J Am Soc Nephrol 3:263–272
Lakhal K, Ehrmann S, Chaari A, Laissy J-P, Régnier B, Wolff M, Pajot O (2011) Acute kidney injury network definition of contrast-induced nephropathy in the critically ill: incidence and outcome. J Crit Care 26:593–599
Solomon RJ, Mehran R, Natarajan MK, Doucet S, Katholi RE, Staniloae CS, Sharma SK, Labinaz M, Gelormini JL, Barrett BJ (2009) Contrast-induced nephropathy and long-term adverse events: cause and effect? Clin J Am Soc Nephrol 4:1162–1169
Sutherland SM, Byrnes JJ, Kothari M, Longhurst CA, Dutta S, Garcia P, Goldstein SL (2015) AKI in hospitalized children: comparing the pRIFLE, AKIN, and KDIGO definitions. Clin J Am Soc Nephrol 10:554–561
Chousterman BG, Bouadma L, Moutereau S, Loric S, Alvarez-Gonzalez A, Mekontso-Dessap A, Laissy JP, Rahmouni A, Katsahian S, Brochard L, Schortgen F (2013) Prevention of contrast-induced nephropathy by N-acetylcysteine in critically ill patients: different definitions, different results. J Crit Care 28:701–709
Ehrmann S, Badin J, Savath L, Pajot O, Garot D, Pham T, Capdevila X, Perrotin D, Lakhal K (2013) Acute kidney injury in the critically ill: is iodinated contrast medium really harmful? Crit Care Med 41:1017–1026
Vandenberghe W, De Corte W, Hoste EAJ (2014) Contrast-associated AKI in the critically ill: relevant or irrelevant? Curr Opin Crit Care 20:596–605
Gouyon JB, Sonveau N, d’Athis P, Chaillot B (1994) Accuracy of urine output measurement with regular disposable nappies. Pediatr Nephrol 8:88–90
Amey M, Butchard N, Hanson L, Kinross D, Mannion M, Parsons J, Wright IMR (2008) Cautionary tales from the neonatal intensive care unit: diapers may mislead urinary output estimation in extremely low birthweight infants. Pediatr Crit Care Med 9:76–79
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
This study was approved by the institutional review board of the French Society for Intensive Care Medicine (SRLF-CE-14-24). The need for informed consent was waived in accordance with French law. However, patients and family members were informed via a family leaflet and the hospital website’s family information pages that retrospective studies might be performed using patients’ charts.
Conflicts of interest
None.
Financial support
This study was supported by a grant from the French Intensive Care Society (Société de Réanimation de Langue Française).
Electronic supplementary material
Below is the link to the electronic supplementary material.
Figure S1
Contrast-induced acute kidney injury (CI-AKI) according to serum creatinine (CI-AKI creat) or urine output (CI-AKI UO) AKI criteria and unfavorable outcome at day 30 (DOC 40 kb)
Rights and permissions
About this article
Cite this article
Cantais, A., Hammouda, Z., Mory, O. et al. Incidence of contrast-induced acute kidney injury in a pediatric setting: a cohort study. Pediatr Nephrol 31, 1355–1362 (2016). https://doi.org/10.1007/s00467-016-3313-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-016-3313-9