Abstract
Background and aims
Biliary drainage is vital in managing malignant biliary obstruction (MBO). Suprapapillary stenting has emerged as a viable alternative to transpapillary stenting and is performed using inside plastic (iPS) or metal stents (iMS). This meta-analysis aims to evaluate the outcomes of suprapapillary stent placement for MBO.
Methods
The Embase, PubMed, and Web of Science databases were systematically searched to include all studies published before September 31, 2023, that reported on the outcomes of suprapapillary stents placed for MBO. Using the random-effect model, the pooled, weight-adjusted event rate estimate for the clinical outcomes was calculated with 95% confidence intervals (CIs).
Results
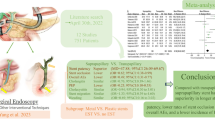
Twenty-eight studies were included, with a total of 1401 patients. The pooled clinical success rate was 98.9%. A subgroup analysis yielded non-significant differences between the iPS and iMS groups (99.3% vs. 98.6%, respectively; P = 0.44). The pooled incidence rate of adverse events (AE) with suprapapillary stents was 9.5%. In a subgroup analysis, the incidence of AEs with iPS was 10.7% compared to 9% in the iMS group without a statistical difference (P = 0.32). The most common adverse event was cholangitis (2.2%), followed by pancreatitis (1.1%), cholecystitis (0.5%), and bleeding (0.12%).
Conclusion
When technically feasible, suprapapillary stenting for MBO is a viable endoscopic option with a high clinical success rate and acceptable adverse event rates. Both iPS and iMS exhibit similar efficacy.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Malignant biliary obstruction (MBO) can manifest with significant clinical challenges due to its poor prognosis and the potential for a detrimental impact on patients’ quality of life. It is characterized by the narrowing or blockage of the bile ducts, leading to jaundice, pruritus, fatigue, and sometimes cholangitis [1]. In recent years, there has been an increasing incidence of MBO, with estimates suggesting approximately 20% of subclinical jaundice cases attributed to malignant bile duct obstruction [2].
Whether the intent of intervention is palliative (in non-operable or surgically unfit cases) or as a bridge to resection (in specific scenarios, such as cholangitis, planned neoadjuvant therapies, and delayed surgical resection), biliary drainage plays a key role in the management of MBO. It is generally advised to drain at least 50% of the viable non-atrophic liver volume, often requiring more than one stent placement [3].
Stent selection is a critical consideration in managing MBO endoscopically. Optimal stenting provides prolonged and effective drainage with low risk of cholangitis, stent migration and stent occlusion. A variety of stent types and sizes are available with different features and various benefits and disadvantages [4]. In unresectable malignant hilar strictures, the American Society of Gastrointestinal Endoscopy (ASGE) 2021 guidelines suggested the use of metal stents (MS) in patients with short life expectancy (< 3 months), or when avoiding reintervention is desired, whereas plastic stent (PS) use was suggested when the diagnosis of the stricture is yet to be confirmed or in cases where the optimal drainage method is not determined yet [3].
While the transpapillary (across-the-papilla) stenting is viewed as the conventional intervention, suprapapillary (above-the-papilla) has emerged as a reasonable alternative method that can, at least theoretically, provide longer patency time and less reflux of bacteria since the stent is placed entirely within the biliary tree, which allows to maintain the physiological function of the sphincter of Oddi. It is worth noting that the current guidelines on MBO do not address suprapapillary versus transpapillary stent placement. A meta-analysis by Kovacs et al. included 12 studies and showed the superiority of suprapapillary stents in contrast to transpapillary stents in regard to patency time [5]. Notably, single stent placement is more likely in transpapillary stents, whereas suprapapillary stent cases usually require two or more stents in over 50% of instances [6, 7].
Similar to the transpapillary approach, in suprapapillary approach both plastic and metal stents are available. Inside plastic stents are modified by the manufacturer or the endoscopist to be equipped with a thread which traverses the major papilla to allow removal. For metal stents, in most cases, uncovered stents are more commonly used, and they come with various characteristics including size, material, shape, length and diameter. Fully or partially covered metal stents are also used in some cases.
In a recent randomized controlled trial, Kanno et al. showed no major differences in efficacy and safety between suprapapillary plastic and uncovered metal stents in the management of malignant hilar strictures, although the population size was relatively small (N = 87) [8]. In this meta-analysis, we aim to comprehensively review the efficacy and safety of suprapapillary stents in the management of MBO with a focus on comparing outcomes betweeen plastic and metal stents. To the best of our knowledge, this is the first meta-analysis on this matter.
Methods
Search strategy and study eligibility
Two independent reviewers (S.A. and M.M) conducted a systematic search of studies published before September 30, 2023, reporting outcomes of suprapapillary biliary stents placed for MBO. We systematically searched the online MEDLINE, Embase and Scopus databases using key words in different combinations: (MBO or malignant biliary stricture or malignant bile duct obstruction or cholangiocarcinoma or gallbladder neoplasms) and (metal stent or plastic stent or endoprosthesis) and (intraductal or inside or suprapapillary). In addition, according to the Preferred Reporting Items for Systematic Reviews and Meta-analyses (PRISMA), we screened the reference lists of the articles and corresponded with study investigators [9]. No language restrictions were applied, as long as study outcomes were reported in the text. A third reviewer (O.T.) resolved any disagreement.
Study inclusion and exclusion
We used the following criteria in this analysis: prospective or retrospective studies which evaluated patients with MBO (patient population) who underwent endoscopic retrograde biliary stent placement entirely above the sphincter of Oddi (intervention) where clinical safety and efficacy were reported (outcomes). Studies on single stent type (PS or MS) or comparison of those were included.
The study was excluded if: (1) it involved benign biliary strictures; (2) the location of the stents was completely or partially across the major papilla; (3) it was a case report, case series with less than 10 sample sizes, animal study, editorial, meta-analysis, or review article. Studies without relevant clinical data on adverse events (AEs) were excluded.
Data extraction and quality assessment
All relevant data were extracted according to predefined table independently by (S.A. and M.M.). The following parameters were extracted: first author, year of publication, country, study design, patient demographics, technical success, functional success, and outcomes of interest. Using the Newcastle–Ottawa Scale, the methodological quality of the included cohort studies was assessed independently by two investigators (SA and OT). In a case of discrepancy, a third independent individual (MM) was consulted.
Definitions of outcomes
The endpoint outcomes include stent patency period, stent occlusion, and overall AEs. The American Society for Gastrointestinal Endoscopy lexicon for grading of severity of procedural AEs with endoscopy was used to define major AEs [10]. Clinical success was defined as a reduction in total serum bilirubin level by more than 50% at 2–4 weeks. Recurrent biliary obstruction was considered in cases of stent migration, occlusion, or tumor ingrowth within the stent.
The primary goals of this study were to evaluate the clinical success and adverse event rates of suprapapillary (or inside) stents. The secondary goals were to compare the clinical success and adverse event rates between supra-papillary plastic stents (iPS) and metal stents (iMS) and to assess the patency period of both type of stents.
Data synthesis and statistical analysis
We used R, version 3.2.3 (R Project for Statistical Computing), with Meta and Metaprop packages for all analyses. Using the Freeman-Turkey Double Arcsine Transformation (FTT) method, the pooled, weight-adjusted event rate estimate for the clinical outcomes in each group was calculated using Metaprop package. Between-study heterogeneity was assessed using the Cochrane Q-statistic (I2), which represents the percentage of total between-study variation that cannot be attributed solely to chance. Between-study heterogeneity was rated as low if 25% < I2 ≤ 50%, moderate if 50% < I2 ≤ 75%, and high if I2 > 75%. A leave-1-out meta-analysis was performed to assess the influence of the outcome by excluding each study and identifying influential studies that may contribute to heterogeneity. A subgroup analysis was performed based on the type of stent used (metal vs. plastic). Statistical tests were 2-sided and used a significance threshold of P < 0.05. The assessment of publication bias was investigated by evaluation of funnel plot asymmetry and sensitivity analysis.
Results
Literature search and study characteristics
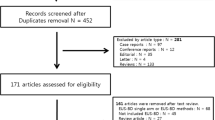
A total of 1776 unique records were identified according to the above search strategy. Finally, 28 studies with a total of 1401 patients were included in the study. PRISMA flowchart illustrates our selection process as shown in Fig. 1. Table 1 shows the baseline characteristics of the included studies and their quality analysis. Most studies were from Asia, were retrospective and were from single centers. Among the included studies, 17 were of good, 9 were of fair, and 2 were of poor quality (Table 2). Table 3 shows a detailed description of stent types that were used. Most common location of stricture in the included studies was the biliary hilum. Additionally, more than 50% of cases required 2 or more stents.
Technical and clinical success
A total of 24 studies with 1004 patients showed a pooled clinical success rate of 98.9% (95% confidence interval (CI) 98.1–99.8; I2 = 46%). A subgroup analysis yielded non-significant difference between the iPS and iMS groups (99.3% [95% CI 98–100%] vs. 98.6% [95% CI 97.4–99.8%], respectively; P = 0.44) (Fig. 2). Technical success rate was high at 99.9% ([95% CI 99.2–100%], I2 = 0%).
Overall adverse events
Overall, a total of 28 studies (1401 patients) reported on the total number of AEs related to suprapapillary stents. The pooled incidence rate of AEs with suprapapillary stents was 9.5% (95% CI 8–11%; I2 = 75%) with substantial heterogeneity. In a subgroup analysis, the incidence of AEs with iPS was 10.7% [95% CI 9–12%] compared to 9% in iMS group [95% CI 8–12%] with no significant statistical difference (P = 0.32) (Fig. 3).
Individual adverse events
The most common adverse event was cholangitis (2.2%), followed by pancreatitis (1.1%), cholecystitis (0.5%), and bleeding (0.12%) Table 4.
-
(i) Cholangitis
Twenty-six studies reported the incidence rate of cholangitis. The pooled incidence of cholangitis was 2.2% ([95% CI 1.3–3.1%]; I2 = 69%). In a subgroup analysis there was no difference between iPS and iMS groups (P = 0.69).
-
(ii) Pancreatitis
The incidence of pancreatitis has been reported in 26 studies. The pooled incidence of pancreatitis after suprapapillary stent was 1.16% ([95% CI 0.44–1.87%]; I2 = 44%). In a subgroup analysis, the incidence of pancreatitis was significantly higher in the iPS group than in the iMS group (3.88% [95% CI 1.95–5.81%] vs. 0.73% [95% CI 0–1.50%]; P < 0.01).
-
(iii) Cholecystitis
Twenty-six studies reported the incidence of cholecystitis in the iPS and iMS groups. The overall incidence of cholecystitis was 0.55% [95% CI 0–1.15%]; I2 = 6%. There was no difference in subgroup analysis of iPS and iMS groups (P = 0.69).
-
(iv) Bleeding
Twenty-eight studies reported the incidence of bleeding. The overall bleeding incidence rate was 0.12% [95% CI 0–0.63%]; I2 = 0%. When iPS group was compared to iMS group, there was no significant difference (P = 0.91).
-
(v) Recurrent biliary obstruction and reintervention
The pooled incidence of RBO was 36.4% [95% CI 33.7–39.1%]. There was significant difference of RBO incidence between the iPS and iMS groups (45% [95% CI 40.2.9–49.7%] vs. 32.3% [95% CI 29.0–35.6%]; P < 0.01). Seventeen studies reported the rate of re-intervention after suprapapillary stent placement with a pooled rate of 33.6% [95% CI 30.1–37.2%].
Patency time
A total of 25 studies reported the median patency time in days of the suprapapillary stents with a pooled median duration of 188.8 days [95% CI 106.1–260.9%]. There was no statistically significant difference between iPS and iMS (172.5 vs. 200.2 days; mean difference = 27.7 days; 95% CI [− 33.9, 89.4%], P = 0.38).
Assessment of publication bias and sensitivity analysis
A funnel plot of studies that reported on efficacy and safety of suprapapillary stents in MBO is presented in Fig. 4. The influence of a single study on the overall meta-analysis estimate was investigated by omitting one study at a time. The omission of any study resulted in no significant difference, indicating that our results were statistically reliable.
Discussion
To the best of our knowledge, this represents the first comprehensive meta-analysis of suprapapillary stents in MBO, discussing its efficacy and safety. Our meta-analysis showed a high clinical success rate of 98.9% for suprapapillary stenting and a pooled median stent patency of around 189 days. Due to the lack of removability of uncovered metal stents, and since re-interventions on occluded metal stents are often cumbersome, inside plastic stents have gained traction mostly in Asian countries due to the ease of exchangeability. Results from our analysis showed no difference of the clinical success between plastic and metal stents (99.3%vs. 98.6%, respectively) [12].
Our findings not only emphasize the efficacy of suprapapillary stents but also indicate their relative safety with an overall incidence of adverse events of 9.5%, which is consistent with Kanno et al.’s recent multicenter randomized trial [37]. Cholangitis was the most reported adverse event without difference between metal and plastic stents. Suprapapillary stenting allows for the preservation of an intact sphincter of Oddi, which acts as a physiological barrier to the reflux of bacteria and duodenal contents, potentially lowering the likelihood of developing cholangitis [38, 39]. However, not a minor proportion of cases underwent endoscopic sphincterotomy to facilitate suprapapillary stent placement, hence compromising the protective function of the sphincter of Oddi and lowering the risk reduction of cholangitis [40].
Pancreatitis is another important adverse event with higher rates in iPS compared to iMS. The existing literature indicates a higher occurrence of pancreatitis with transpapillary stents [41], possibly due to the compression of the pancreatic duct opening, leading to outflow obstruction [12]. This is also likely a key factor in the increased pancreatitis risk associated with iPS [42, 43]. Plastic stents are more prone to occlusion due to their smaller diameter and material properties, which can further exacerbate pancreatic duct obstruction and inflammation [43, 44]. Additionally, the relatively rigid nature of plastic stents may cause greater mechanical irritation to the pancreatic duct compared to more flexible metal stents. Rare complications associated with suprapapillary stenting included cholecystitis and bleeding, with no significant variation between plastic and metal subgroups. These rates were comparable to those observed with transpapillary stenting [45]. However, as bleeding has been often observed to stem from sphincterotomy [46], suprapapillary stenting decreases the need for this intervention, and could potentially reduce the incidence of this complication [14].
Our analysis showed RBO rate of 36.4%, mirroring evidence from previous literature [47]. The higher RBO incidence in iPS 45% versus 32.3% in iMS isn’t surprising given the difference in stent caliber. The higher RBO incidence of iPS emphasizes the delicate balance between the advantages of iPS removability and the increased vulnerability to occlusion. In comparison, RBO was reported to be up to 27% in transpapillary stents [48]. Our analysis also revealed a pooled median patency duration of around 189 days with numerically, but not statistically significant, longer patency of the iMS than the iPS. Thus, in cases of limited life expectancy < 3–6 months, iMS likely represents a better option given the longer patency and lower rates of re-interventions. Additionally, previous metanalysis showed that suprapapillary biliary stents have longer patency times compared to transpapillary stents [5].
While traditionally the transpapillary approach has been used in malignant bile duct occlusion, emerging evidence has been in support of suprapapillary stenting [6, 12, 30]. A recent meta-analysis comparing suprapapillary to transpapillary stenting suggested better outcomes, longer patency time and fewer adverse events with the suprapapillary approach [5]. However, multiple factors limit the widespread utilization of the iPS due to lack of robust randomized data, lack of experience and lack of easy accessibility in the Western countries [49]. Most data on suprapapillary stents come from Eastern countries which may necessitate larger prospective studies that are generated in various patient populations in Western countries for the sake of generalizability of this approach. It is important to highlight the fact that our study isn’t designed to compare the suprapapillary stenting to the standard transpapillary approach, albeit the comparable reported clinical success rates and low complication rates of the inside stenting.
A significant strength of our study is that it encompassed 28 studies involving a total of 1401 patients, making it the most comprehensive analysis that reports on the efficacy and safety of suprapapillary stents. However, our study also has certain limitations. Among the studies analyzed, only three are RCTs, with the remainder being retrospective. This distribution could introduce selection bias. Nevertheless, there was no significant publication bias based on the indicators we employed. Additionally, only two of the studies in our analysis offered a direct comparison between plastic and metal stents. Other inherent limitations were the minor and major variations in study designs including procedural techniques, number of stents, stent models, inclusion, and exclusion criteria. Additionally, we were not able to conduct analysis for overall survival. In most studies, multiple stents are required in each case (whether PS or MS) including, for metal stents, stent-in-stent, side-by-side, or both. Lastly, suprapapillary stenting is primarily practiced and studied in Asia. The results of our meta-analysis aren’t quite generalizable to the practice in the Western world.
In conclusion, suprapapillary stents have proven to be a viable effective option in managing biliary obstructions, demonstrating high clinical and technical success rates coupled with acceptable rate of adverse events. Both plastic and metal stents exhibit similarly high success rates, but the potential complications associated with each differ. Whether suprapapillary or transpapillary, when considering the type of stent to use, it remains most reasonable to take an individualized approach considering tumor location, respectability, patient values, life expectancy, and local expertise. Larger RCTs are needed to draw definitive comparisons between these two stent types and between suprapapillary and transpapillary stents.
References
Fernandez YVM, Arvanitakis M (2019) Early diagnosis and management of malignant distal biliary obstruction: a review on current recommendations and guidelines. Clin Exp Gastroenterol 12:415–432
Siegel R et al (2014) Cancer statistics, 2014. CA Cancer J Clin 64(1):9–29
Qumseya BJ et al (2021) ASGE guideline on the role of endoscopy in the management of malignant hilar obstruction. Gastrointest Endosc 94(2):222-234.e22
Elmunzer BJ et al (2023) ACG clinical guideline: diagnosis and management of biliary strictures. Am J Gastroenterol 118(3):405–426
Kovács N et al (2023) Suprapapillary biliary stents have longer patency times than transpapillary stents-a systematic review and meta-analysis. J Clin Med 12(3):898
Kubota K et al (2016) Stent placement above the sphincter of Oddi permits implementation of neoadjuvant chemotherapy in patients with initially unresectable Klatskin tumor. Endosc Int Open 4(4):E427–E433
Kanno Y et al (2020) Inside plastic stents versus metal stents for treating unresectable malignant perihilar biliary obstructions: a retrospective comparative study. Clin Endosc 53(6):735–742
Kanno Y et al (2023) Suprapapillary placement of plastic versus metal stents for malignant biliary hilar obstructions: a multicenter, randomized trial. Gastrointest Endosc 98(2):211–221.e3
Page MJ et al (2021) The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ 372:n71
Cotton PB et al (2010) A lexicon for endoscopic adverse events: report of an ASGE workshop. Gastrointest Endosc 71(3):446–454
Brijbassie A, Yeaton P, Maranki J (2011) Intraductal vs. transpapillary FCSEMS placement- does it make a difference?: 181. AJG 106:S71
Cosgrove N et al (2017) A comparison of bilateral side-by-side metal stents deployed above and across the sphincter of Oddi in the management of malignant hilar biliary obstruction. J Clin Gastroenterol 51(6):528–533
Douhara A, Ogawa H, Ozutsumi T et al (2016) Inside plastic stent is useful and safety for malignant biliary stenosis including Bismuth IV. J Gastroenterol Hepatol (Australia) 31:343. https://doi.org/10.1111/jgh.13540
Inatomi O et al (2013) Threaded biliary inside stents are a safe and effective therapeutic option in cases of malignant hilar obstruction. BMC Gastroenterol 13:31
Inoue T et al (2020) Intraductal placement of a fully covered metal stent with a long string for distal malignant biliary obstruction without endoscopic sphincterotomy: prospective multi-center feasibility study. Dig Endosc 32(6):949–956
Ishigaki K et al (2020) Retrospective comparative study of side-by-side and stent-in-stent metal stent placement for hilar malignant biliary obstruction. Dig Dis Sci 65(12):3710–3718
Ishiwatari H et al (2013) Newly designed plastic stent for endoscopic placement above the sphincter of Oddi in patients with malignant hilar biliary obstruction. Dig Endosc 25(Suppl 2):94–99
Kaneko T et al (2014) Efficacy of plastic stent placement inside bile ducts for the treatment of unresectable malignant hilar obstruction (with videos). J Hepatobiliary Pancreat Sci 21(5):349–355
Kanno Y et al (2020) Endoscopic inside stent versus metal stents for treating unresectable malignant perihilar biliary obstructions: a retrospective comparative study. Clin Endosc 53(6):735–742
Kobayashi N et al (2015) Endoscopic inside stent placement is suitable as a bridging treatment for preoperative biliary tract cancer. BMC Gastroenterol 15(1):8
Kogure H et al (2021) A prospective multicenter study of “inside stents” for biliary stricture: multicenter evolving inside stent registry (MEISteR). J Clin Med 10(13):2936
Koiwai A et al (2022) Self-expandable metal stents have longer patency and less cholangitis than inside stents in malignant perihilar biliary obstruction. JGH Open 6(5):317–323
Koizumi M et al (2020) Endoscopic stent placement above the sphincter of Oddi for biliary strictures after living donor liver transplantation. BMC Gastroenterol 20(1):92
Kurita A et al (2013) Endoscopic stent placement above the intact sphincter of Oddi for biliary strictures after living donor liver transplantation. J Gastroenterol 48(9):1097–1104
Kurita A et al (2022) Stent placement above the sphincter of Oddi is a useful option for patients with inoperable malignant hilar biliary obstruction. Surg Endosc 36(5):2869–2878
Mori H et al (2022) Comparison of an inside stent and a fully covered self-expandable metallic stent as preoperative biliary drainage for patients with resectable perihilar cholangiocarcinoma. Can J Gastroenterol Hepatol 2022:3005210
Mukai T et al (2013) Metallic stents are more efficacious than plastic stents in unresectable malignant hilar biliary strictures: a randomized controlled trial. J Hepatobiliary Pancreat Sci 20(2):214–222
Cho JN et al (2013) 287 prospective randomized trial comparing covered metal stent placed above and across the sphincter of Oddi in malignant biliary obstruction. Gastrointest Endosc 77(5, Supplement):AB139-AB140
Pedersen FM, Lassen AT, Schaffalitzky de Muckadell OB (1998) Randomized trial of stent placed above and across the sphincter of Oddi in malignant bile duct obstruction. Gastrointest Endosc 48(6):574–579
Shin J et al (2020) Comparison of the clinical outcomes of suprapapillary and transpapillary stent insertion in unresectable cholangiocarcinoma with biliary obstruction. Dig Dis Sci 65(4):1231–1238
Takada R et al (2020) Self-expandable metallic stent placement above the papilla without endoscopic sphincterotomy in patients with distal malignant biliary obstruction. Endosc Int Open 8(6):E753–E760
Taniguchi Y (2020) Su1453 intraductal vs. transpillary placement of fully covered metal stent for malignant biliary strictures. Gastrointest Endosc 91(6:Supplement):AB354
Uchida N et al (2005) Estimation of the stent placement above the intact sphincter of Oddi against malignant bile duct obstruction. J Gastroenterol 40(3):291–296
Yamada M et al (2023) Outcomes of intraductal placement of covered metal stents for unresectable distal malignant biliary obstruction. J Clin Med 12(5):2001
Yan X et al (2018) Suspended over length biliary stents versus conventional plastic biliary stents for the treatment of biliary stricture: a retrospective single-center study. Medicine (Baltimore) 97(47):e13312
Han SY et al (2023) Efficacy analysis of suprapapillary versus transpapillary self-expandable metal stents according to the level of obstruction in malignant extrahepatic biliary obstruction. Gut Liver 17(5):806–813
Okamoto T et al (2006) Placement of a metallic stent across the main duodenal papilla may predispose to cholangitis. Gastrointest Endosc 63(6):792–796
Weickert U et al (2001) Why do bilioduodenal plastic stents become occluded? A clinical and pathological investigation on 100 consecutive patients. Endoscopy 33(9):786–790
van Berkel AM et al (2005) Mechanisms of biliary stent clogging: confocal laser scanning and scanning electron microscopy. Endoscopy 37(8):729–734
Zhou H et al (2012) Endoscopic sphincterotomy associated cholangitis in patients receiving proximal biliary self-expanding metal stents. Hepatobiliary Pancreat Dis Int 11(6):643–649
Jo JH, Park BH (2015) Suprapapillary versus transpapillary stent placement for malignant biliary obstruction: which is better? J Vasc Interv Radiol 26(4):573–582
Song J, Deng J, Wen F (2023) Risk factors associated with acute pancreatitis after percutaneous biliary intervention: we do not know nearly enough. Gastroenterol Res Pract 2023:9563074
Mullen JT et al (2005) Pancreaticoduodenectomy after placement of endobiliary metal stents. J Gastrointest Surg 9(8):1094–1104
Wasan SM et al (2005) Use of expandable metallic biliary stents in resectable pancreatic cancer. Am J Gastroenterol 100(9):2056–2061
Jandura DM, Puli SR (2021) Efficacy and safety of endoscopic transpapillary gallbladder drainage in acute cholecystitis: an updated meta-analysis. World J Gastrointest Endosc 13(8):345–355
Dumonceau JM et al (2018) Endoscopic biliary stenting: indications, choice of stents, and results: European Society of Gastrointestinal Endoscopy (ESGE) clinical guideline-updated October 2017. Endoscopy 50(9):910–930
Zhang JX et al (2018) Differences in efficacy of uncovered self-expandable metal stent in relation to placement in the management of malignant distal biliary obstruction. Saudi J Gastroenterol 24(2):82–86
Zu QQ et al (2019) Percutaneous transpapillary biliary stent placement for distal malignant biliary obstruction: outcomes and survival analysis. Turk J Gastroenterol 30(8):714–721
Zilberstein NF, Mehta NA (2023) Suprapapillary stenting for malignant hilar obstruction: is it feasible in the West? Gastrointest Endosc 98(2):222–224
Funding
Saqr Alsakarneh, Mahmoud Y Madi, Fouad Jaber, Kamal Hassan, Yassine Kilani, Omar Al Ta’ani, Dushyant Singh Dahiya, Amir H. Sohail, Laith Numan, Mohammad Bilal, and Wissam Kiwan have no conflicts of interest or financial ties to disclose.
Author information
Authors and Affiliations
Contributions
(A = Study Design, B = Data collection, C = Statistical analysis, D = Data interpretation, E = Manuscript preparation, F = Literature search, G = Manuscript review). Saqr Alsakarneh: ABCDEFG, Mahmoud Y Madi: BDEFG, Kamal Hassan: BDE, Fouad Jaber: BEF, Yassine Kilani: BDE, Omar Al Ta’ani: BCD, Dushyant Dahiya: BCD, Amir H Sohail: BCD, Laith Numan: EF, Mohammad Bilal: AEFG, Wissam Kiwan: AEFG.
Corresponding author
Ethics declarations
Disclosures
Saqr Alsakarneh, Mahmoud Y. Madi, Fouad Jaber, Kamal Hassan, Yassine Kilani, Omar Al Ta’ani, Dushyant Singh Dahiya, Amir H. Sohail, Laith Numan, Mohammad Bilal, Wissam Kiwan have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alsakarneh, S., Madi, M.Y., Jaber, F. et al. Safety and efficacy of biliary suprapapillary metal and plastic stents in malignant biliary obstruction: a systematic review and meta-analysis. Surg Endosc 38, 4186–4197 (2024). https://doi.org/10.1007/s00464-024-11025-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-024-11025-0