Abstract
Background
The laparoscopic cholecystectomy has allowed the detection of an increasing number of incidental gallbladder cancers (IGBC). Although laparoscopy is employed in the management of a variety of abdominal tumors, its use in gallbladder cancer is reduced and controversial. This study analyzes the role of laparoscopy in gallbladder cancer with the focus in IGBC.
Method
We evaluated our prospective series of 51 patients with an IGBC who were treated by laparoscopy between 2006 and 2016 at the Clinica Alemana in Santiago, Chile.
Results
The series comprised 7 men and 44 women. Age ranged from 43 to 76 years (mean age 60). Regarding wall involvement, 29 patients had a T2 tumor, which was the most common. 8 and 14 patients had T1b and T3 tumors, respectively. Of the patients, 17 underwent only laparoscopic exploration. This was due to the presence tumor dissemination not being observed in the preoperative staging. 10 patients had to be converted to complete the resection, whereas 24 patients were laparoscopically resected. The quality of the resected material was not different between those who were converted and those who were treated by laparoscopy. In the laparoscopic group, the average number of harvested lymph nodes was 7.9, not statistically different from the converted group. The mean of hospital stay in the laparoscopic group (4.3 days) was significantly lower than the converted group.
Conclusions
Laparoscopy has been shown to be a safe and feasible method for the management of IGBC. This method not only allows for a complete exploration, identifying a previously unseen residual tumor, but also makes it possible to accomplish the same oncology objectives as the open procedure. Therefore, laparoscopy should be considered a valid alternative in the management of IGBC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Gallbladder cancer is associated with a dismal prognosis except in early tumors, most of which are cases of incidental gallbladder cancer (IGBC) [1, 2]. For the management of this particular patient group, reoperation to stage and resect any residual tumor is advised [3,4,5,6].
The possibility of wall perforation and the risk of tumor dissemination during gallbladder manipulation have limited the use of laparoscopy in gallbladder cancer patients [7, 8]. However, a larger number of patients are currently detected after a cholecystectomy for a presumed benign disease, and they are sent for definitive treatment once the pathology diagnosis of gallbladder cancer has been made. This would allow resection by laparoscopy without risk of dissemination.
On the other hand, for cancers such as gastric and colorectal, laparoscopic surgery has shown outcomes similar to open surgery and is a validated method [9,10,11].
In this light, we wanted to analyze our protocol for the IGBC management through laparoscopy with emphasis on its feasibility, intraoperative findings, and preliminary results.
Materials and methods
A prospective laparoscopic management protocol for patients with gallbladder cancer has been underway at the Department of Surgery at the Clinica Alemana in Santiago, Chile since 2006. Patient data are collected in a prospective database and include demographic data, histopathology of the initial cholecystectomy, surgery details, histopathology of the resected specimen, morbidity, mortality, and follow-up.
Tumors were classified according to the TNM classification of the American Joint Committee on Cancer and lymph node location according to the General Rules for Surgical and Pathological Studies on Cancer of the biliary tract [12, 13].
The indication for a laparoscopic approach in patients with gallbladder cancer was the presence of an IGBC. To consider a patient eligible for the laparoscopic approach, the tumor had to be confined to the gallbladder or to adjacent areas susceptible to resection in a formal lymphadenectomy of the hepatic pedicle and a wedge resection of the gallbladder bed. Each patient provided informed consent for the laparoscopic procedure.
Most patients underwent a cholecystectomy in centers other than the Clinica Alemana and were referred for assessment and possible resection in our clinic.
As part of the preoperative evaluation, the cholecystectomy operatory chart was reviewed and contact was made with the surgeon who performed the cholecystectomy to obtain information about the surgical details. Patients were interviewed and examined by the main author, and their pathology, treatment, and prognosis explained to them. Patients were free to accept or decline a second operation and the laparoscopic approach.
In addition to the physical exam, common blood tests and a helical CT were performed as a standard staging method. Inoperability was defined by the presence of metastatic disease or local infiltration of the main vascular structures (portal vein and hepatic artery).
Postoperative complications within 90 days after surgery were classified using Clavien–Dindo classification [14]. Postoperative mortality was defined as death within 90 days after surgery.
Data were analyzed using the Mann–Whitney–Wilcoxon, Chi-squared, or Fischer’s exact tests where appropriate.
Surgical technique
Five ports are used in a smile-shaped configuration. After abdominal exploration including laparoscopic ultrasound to exclude peritoneal dissemination and liver metastasis, we mobilize the hepatic flexure of the colon, retracting the hepatic flexure down. We free the duodenal arch, exposing the plane between the kidney and the second portion of the duodenum. The plane between the aorta and the vena cava is dissected and lymph nodes are removed for frozen section. Involvement at this level determines the end of the operation. The procedure continues with the lymphadenectomy of the hepatic pedicle. The area of the hepatic pedicle can show varying degrees of inflammation, making it difficult to recognize the structures. Recognition of the common bile duct is normally the most challenging part of the procedure.
Remnant cystic duct evaluation is also important; involvement of this structure determines whether to convert the procedure and perform the resection of the common bile duct. A circumferential dissection of the hepatic pedicle is completed. The extent of the lymphadenectomy included, in addition to the lymph nodes of the hepatic pedicle, nodes located along the hepatic artery and retropancreatic region. Assessment of the choledochoduodenal lymph node is extremely important because this is where the tumor will spread to the lymph nodes beyond the regional area.
After dissecting the hepatic pedicle, we begin with the resection of the gallbladder bed. A wedge of segments IVb and V is marked using a hook cautery. To transect the liver, different devices are employed. We mainly prefer to use both ultrasonic shears and a bipolar. Resection of port sites is rarely performed as per the surgeon’s preference.
Results
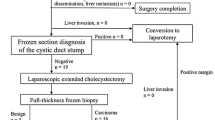
Between 2006 and 2016, 51 patients underwent exploratory laparoscopy for an IGBC (Fig. 1). Of these patients, 7 were male and 44 were female, ranging in age from 43 to 76 years (mean age 60, SD 7.08). The level of wall involvement of these patients is shown in Table 1, with T2 patients being the most common group.
Of the patients, 17 underwent only laparoscopic exploration. Causes for non-resection were peritoneal dissemination in eight patients, distant lymph node invasion in five, extensive local infiltration in two, and one patient had a minor basal coagulopathy that limited pursuit of the resection and one had a renal vein tear that occurred during the laparoscopy that required an open repair (Table 2).
Ten patients had to be converted after beginning the laparoscopic resection. Of these, four had a severe inflammatory process surrounding the hepatic pedicle, three persistent bleeding from the liver transection and one each of a suspicious bile leak from the porta hepatis area, explored after converting, a positive cystic duct stump that required common bile duct resection, and an involved retropancreatic lymph node that we converted for resecting. This last patient was treated at the beginning of our experience.
Finally, 24 patients underwent a complete laparoscopic resection of the gallbladder bed and a lymphadenectomy of the hepatic pedicle.
Table 3 shows the relationship between the T stage and the resection rate. All patients with a T1b tumor could be resected; of the 29 T2 patients, 19 (65%) could undergo resection. (P = 0.07 with T1b). On the other hand, the resection rate in T3 patients was 50%, being statistically significant different when compared with T1b (0.02). Finally, the difference in the resectability rate between T3 and T2 was not statistically significant.
Concerning intraoperative complications, among the patients who underwent the procedure totally laparoscopically, one patient had a small perforation of the left biliary duct that was repaired laparoscopically. No blood transfusion was required in this patient group. By contrast, among those who had to be converted, five patients required intraoperative blood transfusions.
Table 4 illustrates the postoperative complications according to the Clavien–Dindo Classification in both groups: Among those who underwent a totally laparoscopic resection, there were two bile leaks associated with abdominal collections originating on the raw surface of the liver resection. One of these patients was laparoscopically drained, while the other patient was managed percutaneously. Another patient, who developed fever and a suspicious case of an abdominal collection, was treated only with antibiotics. Finally, one patient developed a pulmonary atelectasis and another a pulmonary embolism that were successfully treated.
Postoperative findings
Among the 34 patients who underwent resection, none had liver involvement, while 9 had at least one lymph node involved. The number of dissected lymph nodes ranged from 3 to 16 in the laparoscopic series (mean 7.9), while in the converted group it ranged from 4 to 12 (mean 8.7) (P > 0.05) .
Lymph node status and its relation to the T stage are shown in Table 5. Among the eight patients with a T1b tumor who underwent lymph node dissection, only one (12.5%) had lymph node involvement. In patients with T2 and T3 tumors, the percentage of lymph node involvement was 17.2 and 21.4, respectively.
Length of hospital stay ranged from 2 to 18 days (mean 4.3, SD 3.1) in the laparoscopic group, whereas the stay ranged from 4 to 11 days (mean 6.3, SD 2.3) (P = 0.02) in the patients who underwent resection after converting.
Discussion
Since the emergence of the laparoscopic cholecystectomy, diagnosis of early types and incidental tumors has increased worldwide [4, 5].
Laparoscopy has also enabled a change in the way many digestive tumors are managed. Colon cancer was the first digestive tumor where laparoscopic management showed oncological results similar to the open approach, being the first step in the spread of laparoscopic management of different abdominal tumors [9,10,11]. In IGBC, laparoscopy was initially used only for staging. Our results also support the value of laparoscopy in assessing patients prior to resection. In one-third of our patients, laparoscopic exploration enabled detection of unobserved dissemination during the staging prior to the resection.
In 2010, our group and also Gumbs reported the first cases of laparoscopic management of gallbladder cancer [15, 16]. Gumbs provided a case report of a preoperatively suspected gallbladder cancer, while our series only included patients with IGBC. Since those reports, other authors have published their experiences with both incidental and preoperatively suspected tumors [17,18,19]. Piccolo just published a review of this issue, highlighting the feasibility of the laparoscopic approach and the comparable results in terms of the quality of the resected specimen with the traditional approach [20]. Our series confirms the above in a larger number of patients from a single center.
With regard to liver resections, recommendations have varied from limited to extended resections [21]. The size of the resection will mainly depend on the depth of wall invasion and the side of the tumor in the gallbladder [22]. Since our series focuses on IGBC and early tumors, non-anatomical wedge resections of the gallbladder bed are commonly preferred over larger resections, with such resections being safer and more feasible via laparoscopy. Among our patients, surgical complications due to bleeding and inflammation were the main causes for conversion. Although in none of the patients did bleeding lead to hemodynamic instability, the difficulty in controlling this complication indicated conversion to an open procedure. Inflammation of the hepatic pedicle secondary to the previous cholecystectomy was also associated with conversion. This was a common finding in our patients that made the proper identification of the pedicle structures more difficult.
Lymphadenectomy is the other key factor in the management of these patients. In addition to its value for staging, lymph node invasion represents an independent prognosis-related factor. As other authors have highlighted, our results shows that the number and location of resected lymph nodes are comparable to those obtained via the open approach [18, 20].
Concerning length of hospitalization, differences between groups were statistically significant. Patients with a longer stay in each group were due to the management of perioperative complications.
The main limitation of this study is that it is an observational study without a randomized control group. Moreover, the effect of this approach on survival compared with the open approach is another drawback. Nevertheless, this study represents one of the largest series of IGC treated by laparoscopy in a single center.
From the results, we may conclude that the laparoscopic management of IGBC is a feasible procedure that is not only useful for staging patients undergoing reoperation, but also represents a valid alternative for the treatment. Once the respective learning curve is surpassed, this approach should become the standard for managing patients with IGBC.
References
Stinton LM, Shaffer EA (2012) Epidemiology of gallbladder disease: cholelithiasis and cancer. Gut Liver 6:172–187
Wistuba II, Gaazdar AF (2004) Gallbladder cancer: lessons from a rare tumor. Nat Rev Cancer 4:695–706
De Aretxabala X, Roa I, Burgos L, Losada H, Roa JC, Mora J, Hepp J, Leon J, Maluenda F (2006) Gallbladder cancer: an analysis of a series of 139 patients with invasion restricted to the subserosal layer. J Gastrointest Surg 10:186–192
Tantia O, Jain M, Khanna S, Sen B (2009) Incidental carcinoma of the gallbladder during laparoscopic cholecystectomy for symptomatic gallstone disease. Surg Endosc 23:2021–20126
Hueman MT, Vollmer CM Jr, Pawlik TM (2009) Evolving treatment strategies for gallbladder cancer. Ann Surg Oncol 16:2101–2115
Barlett DL, Fong Y, Fortner JG, Brennan MF, Blumgart LH (1996) Long term results after resection for gallbladder cancer. Implications for staging and management Ann Surg 224:639–646
Lee JM, Kim BW, Kim WH, Wang HJ, Kim MW (2011) Clinical implications of bile spillage in patients undergoing cholecystectomy for gallbladder cancer. Am Surg 7:697–701
Ahmad J, Mayne AI, Loughrey MB, Kelly P, Taylor M (2014) Spilled gallstones during laparoscopic cholecystectomy. Ann R Coll Surg Engl 96:18–20
Honda M, Hiki N, Kinoshita T, Yabusaki H, Abe T, Nunobe S, Terada M, Matsuki A, Sunagawa H, Aizawa M, Healy MA, Iwasaki M, Furukawa TA (2016) Long term outcomes of laparoscopic versus open surgery for clinical stage I gastric cancer: the LOC-1 Study. Ann Surg 264:214–222
Jiang JB, Jiang K, Dai Y, Wang RX, Wu WZ, Wang JJ, Xie FB, Li XM (2015) Laparoscopic versus open surgery for mid-low rectal cancer: a systematic review and meta-analysis on short and long term outcomes. J Gastrointest Surg 19:1497–1512
Deijen CL, Vasmel JE, de lange-deKlerk ES, Cuesta MA, Coene PLO. Lange JF, Meijerink WJHJ., Jakimowicz JJ, Jeekel J, Kazemier G, Janssen IM, Pahlman L, Haglind E, Bojer HJ, Color (Colon cancer laparoscopic or open Resection) study group (2017) Ten year outcomes of randomised trial of laparoscopic versus open surgery for colon cancer. Surg Endosc 31:2607–2615
Kondo S, Takada T, Miyazaki M, Miyakawa S, Tsukada K, Nagino M, Furuse J, Saito H, Tsuyuguchi T, Yamamoto M, Kayahara M, Kimura F, Yoshitomi H, Nozawa S, Yoshoda M, Wada K, Hirano S, Amano H, Miura F (2008) Japanese Association of Biliary Surgery, Japanese Society of Clinical Oncology Guidelines for the management of biliary tract and ampullary carcinoma: surgical treatment. J Hepatobiliary Pancreat Surg 15:41–54
Edge SB, Compton CC (2010) The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol 17:1471–1474
Dindo DN, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240:205–213
de Aretxabala X, Leon J, Hepp J, Maluenda F, Roa I (2010) Gallbladder cancer: role of laparoscopy in the management of potentially resectable tumors. Surg Endosc 24:2192–2196
Gumbs AA, Hofmann JP (2010) Laparoscopic completion radical cholecystectomy for T2 gallbladder cancer. Surg Endosc 24:3221–3322
Agarwal AK, Javed A, Kalayarasan R, Sakhuja P (2015) Minimally invasive versus the conventional open surgical approach of a radical cholecystectomy for gallbladder cancer, a retrospective comparative study. HPB 17:536–541
Itano O, Oshima G. Minagawa T. Shinoda M, Kitago M, Abe Y, Hibi T, Yagi H, Ikoma N, Aiko S, Kawaida M, Musugi Y, Kameyama K, Sakamoto M, Kitagawa Y (2015) Novel strategy for treatment of T2 gallbladder carcinoma. Surg Endosc 29:3600–3607
Palanisamy S, Patel N, Sabnis S, Palanisamy N, Vijay A, Palanivelu P, Parthasarthi R, PChinnusamy (2016) Laparoscopic radical cholecystectomy for suspected gallbladder carcinoma: thinking beyond convention. Surg Endosc 30:2442–2448
Piccolo G, Piozzi GN (2017) Laparoscopic radical cholecystectomy for primary or incidental early gallbladder cancer. The new rules governing the treatment of gallbladder cancer. Gastroenterol Res Pract. https://doi.org/10.1155/2017/8570502
Qadan M, Kingham P (2016) Technical aspects of gallbladder cancer surgery. Surg Clin N Am 96:229–245
Shindoh J, de Aretxabala X, Aloia TA, Roa JC, Roa I, Zimmitti G, JAvle M. Conrad C, Maru DM, Aoki T, Vigano L, Ribero D, Kokudo N, Capusotti L, Vauthey JN (2015) Tumor location is a strong predictor of tumor progression and survival in T2 gallbladder cancer. Ann Surg 261:733–739
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Xabier de Aretxabala, Federico Oppliger, Nicolas Solano, Guillermo Rencoret, Marcelo Vivanco, Daniel Carvajal, Juan Hepp, and Ivan Roa have no conflicts of interest or financial ties to disclose.
Rights and permissions
About this article
Cite this article
de Aretxabala, X., Oppliger, F., Solano, N. et al. Laparoscopic management of incidental gallbladder cancer. Surg Endosc 32, 4251–4255 (2018). https://doi.org/10.1007/s00464-018-6173-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-018-6173-5