Abstract
Background
Despite good short-term results and patient satisfaction with endoscopic thoracic sympathectomy (ETS), there has been much debate on the level of sympathectomy for treatment of palmar hyperhidrosis (PH) in terms of long-term clinical outcomes.
Objective
The aim of the study was to analyze the long-term recurrence and compensatory hyperhidrosis (CH) rates of ETS, comparing single-level T2 against multi-level T2–T3 ablation in single patients.
Methods
Patients who had undergone treatment for PH with unilateral T2 and contralateral T2–T3 ablation in ETS were retrospectively reviewed. They were subjected to telephone interview using standardized set of interview script and questionnaire with a scoring system similar to hyperhidrosis disease severity scale. All patients were evaluated for comparison of symptom resolution, site and severity of CH, and satisfaction rates. To compare between T2 and T2–T3, the level of sympathectomy on one side is matched to the ipsilateral recurrence of PH and CH occurrence.
Results
Twenty-two patients with a mean age of 36.5 years could be reached. The mean follow-up was 8 years (range 38–153 months). The global recurrence rate for PH is 18 %. CH was observed in 20 (91 %) patients, and trunk compensation was the most common (18/22–82 %), followed by lower limb (14/22–64 %) and axilla (10/22–45 %). Overall, 72.8 % (16) of the patients were satisfied with the operation. Among the six patients who were not satisfied, two patients reported recurrence of symptoms, while four patients experienced some form of compensation. There was no absolute difference in the severity of sweating bilaterally for patients who reported recurrence of PH. The site and severity of CH were also bilaterally symmetrical for all patients.
Conclusion
There was no difference in recurrence rates and CH between single-level (T2) and multi-level (T2–T3) ETSs in the long term.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Endoscopic thoracic sympathectomy (ETS) has become the treatment of choice for patients suffering from moderate-to-severe palmar hyperhidrosis [1–3]. Despite impressive patient satisfaction rates with ETS, there has been much debate on the level of sympathectomy for treatment of hyperhidrosis. Compensatory hyperhidrosis (CH) is the most common side effect of ETS and exerts a negative impact on patient’s postoperative quality of life [4, 5]. The relationship of CH to the extent and levels of sympathectomy has been the subject of intense debate among thoracic surgeons [6]. Recent expert consensus by Cerfelio et al. [7] has suggested that the most common risk factor cited in the literature for moderate-to-severe CH was T2 ganglion interruption [8–10]. However, the number of levels interrupted has been proven inconclusive as a risk factor.
While short-term results and postoperative complications have been reported extensively in the literature, long-term follow-ups analyzing the progression of the effected treatment and its side effects for 5 years and above are rare [11, 12] and difficult.
Therefore, the aim of this study is to analyze and compare the long-term results of two different levels of denervation, T2 and T2–T3 to verify the effectiveness of different ablations on symptoms of hyperhidrosis, as well as side effects and to determine patient satisfaction from our previous randomized controlled trial [13].
Materials and methods
Most studies on VATS for palmar hyperhidrosis often involve symmetrical, single-level (T2 only) or multi-level (T2–T3) sympathectomy bilaterally. In our study, we recruited patients who had undergone asymmetrical sympathectomy to reduce the confounding effect in terms of difference in patient factors. All patients were worked up preoperatively to exclude secondary causes of hyperhidrosis including hyperthyroidism, chronic infections, malignancy, and immunological disorders. Long-term follow-up was carried out between December 2012 and December 2013.
A total of 39 patients from 2003 to 2010 were enrolled for this study and were randomized to undergo unilateral T2 and T3 blockade with contralateral blockade of T2 only. The side undergoing ablation at levels T2 or T2–T3 was randomized by the “closed envelope” method similar to the one mentioned from Katara et al. [13].
Between 2012 and 2013, these patients were subjected to telephone interview using a standardized set of questions and interview script. Both patients and the observer (interviewer) were blinded to the levels of sympathectomy. The questions can be found in Table 1. Our questionnaire used the same definition of disease severity from hyperhidrosis disease severity scale [14] adapted from the International Hyperhidrosis Society, except that instead of using a point system, we used a modified 4-grade scale: (1) none, (2) mild, (3) moderate, and (4) severe. Each patient was analyzed as a whole for overall satisfaction, the presence of recurrence of palmar hyperhidrosis, and severity of compensatory sweating. Satisfaction was rated by patients on a scale of 1 (most satisfied) to 10 (least satisfied), and the scores were categorized into four main groups: very satisfied, satisfied, dissatisfied, and very dissatisfied. Recurrence was considered when a person experienced a return of palm sweating after it was confirmed to be dry after operation, while compensation was considered if a person experienced new sweating or worsening sweating in other parts of the body that was not present before the surgery. Recurrence and compensatory hyperhidrosis of five sites (lower limb, trunk, axillae, face, and groin) were also assessed using our 4-grade scale.
To compare T2 and T2–T3, we adopted a novel approach. The level of sympathectomy on one side was matched to the ipsilateral recurrence of palmar hyperhidrosis, the site and the severity of compensatory sweating for body parts that were not found in the midline (axilla and lower limbs specifically). The outcomes of single-level versus multi-level ablation were then compared.
Patients were informed of the purpose of the study. Domain-specific review board (DSRB) approval was granted (Reference 2012/00264). All the statistics were calculated using SPSS software version 20 (SPSS Inc., Chicago, IL, USA).
Surgical procedure
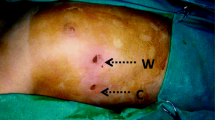
The ETS procedure was performed with the patient under general anesthesia using single-lung ventilation via a double-lumen endotracheal tube as previously described by Katara et al. [13]. A needlescopic technique was utilized. The T2 and/or T3 ganglia were identified (above the corresponding rib) and ablated using monopolar diathermy until the two ablated ends were at least 1 cm apart. A positive ablation was confirmed by a rise in temperature measured on the finger probe.
Results and analysis
Patient descriptive (Table 2)
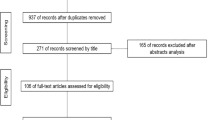
Twenty-two out of thirty-four patients (56.4 %) responded to the telephone interview, with the others lost to follow-up (Fig. 1) due to loss of contact (patients’ contact numbers were not updated, or patients left the country). The study group consisted of patients with a male-to-female ratio of 3:1 and a mean age of 36.5 years (range 14–58 years). The side for T2 or T2–T3 ablation was randomly selected with 22 (81.8 %) patients undergoing two-level ablation on the right (group A) and 4 (18.2 %) patients undergoing the procedure on the left (group B).
Operation details and short-term outcomes (Table 3)
Average operative time was 42 min. All patients had a rise of >0.5 °C in probe temperature over their palms after ablation. Four cases of postoperative pneumothorax and four cases of subcutaneous emphysema were reported, and all of them were treated conservatively with in-patient observation, requiring at most a day or two of prolonged hospital stay. No mortalities were recorded.
Long-term overall results (Table 4)
Patients were followed up for a mean of 105 months (range 38–153 months). All patients had confirmed that their palmar hyperhidrosis had resolved immediately after the operation, with both palms equally dry. Of the 22 patients, 4 (18 %) patients suffered from mild recurrence of palmar hyperhidrosis and 20 (91 %) patients experienced some form of compensatory sweating. Of these 20 patients, trunk compensatory was the most common with 18 reported cases (82 %), followed by the lower limb (14 cases—64 %) and axilla (10 cases—45 %). Overall, 59.1 % (13) of the patients were very satisfied with the operation, 13.6 % (3) merely satisfied, 13.6 % (3) dissatisfied, and 13.6 % (3) very dissatisfied. There were no other reported complications.
Long-term results based on level of ablation (T2 vs T2–T3)
Ipsilateral outcomes were compared to the level of sympathectomy performed (T2 vs T2–T3). Of the four patients who suffered from recurrence of palmar hyperhidrosis, there was no absolute difference in the severity of the sweating bilaterally. There was also no absolute difference between the site (lower limb and axilla) and severity of compensatory sweating.
Overall level of satisfaction
A total of 72.8 % of the respondents (16 out of 22) were either very satisfied or satisfied with the procedure. Among the six patients who are dissatisfied with the procedure, two (33.3 %) patients had recurrence of palmar hyperhidrosis, while four (66.6 %) patients experienced some form of compensatory sweating.
Discussion
ETS is currently recognized as the best method of treatment of palmar hyperhidrosis in terms of its efficacy and safety profile [15].
In our long-term analysis, there was no relative difference in recurrence rate between the two different levels of sympathectomy as recurrence in patients occurred symmetrically and bilaterally. Both different levels of sympathectomy brought good effects in reducing palmar hyperhidrosis equally, with immediate postoperative relief in all patients and up to 81.8 % having nonrecurrence up till 5 years postoperatively. Only two patients reported to have recurrence of symptoms of which both only had mild symptoms that did not severely affect their activities of daily living. Recurrent hyperhidrosis has been widely reported as one of the potential side effect from hyperhidrosis surgery, with incidence rate varying considerably from 0 to 65 % [11, 16]. Failure of symptom resolution can be attributed to various causes such as mistaken identification of the sympathetic level, incomplete ablation, or accessory nerve pathways [6] (Kuntz fiber). Resympathectomy has been advocated and successfully performed for immediate failures and recurrence cases [17] if the surgeon acknowledges possible anatomic variations and can overcome the problems related to pleural adhesions [18].
Compensatory sweating is the most common and distressing complication for sympathectomy. It is characterized by the appearance of excessive perspiration in regions of the body where it had not been previously observed. Different studies have had controversial results in terms of this side effect, which occurs in the literature from 3 to 98 % [19, 20]. Our study describes a higher rate of CH (91 %) than reported usual rate of around 60 % [11]. This could be attributed to the hot and humid environment of Singapore, with previous reports from countries such as Taiwan [21, 22] and Middle East countries [23] that have similar climate conditions showing high rates of CH. Besides climate, if patients are already experiencing increased sweating in regions such as the trunk, groin, or upper thighs, they are at increased risk of developing CH [7] and they should be counseled before proceeding with procedure.
The overall satisfaction rate of ETS (72.7 %) in our study was much lower than that of most studies in the literature. About two-thirds of the patients who indicated that they were dissatisfied with the procedure had complained of CH. In studies with high rates of CH, a similar level of satisfaction was seen [21, 24]. Gossot et al. [11] also reported that majority of the patients who were dissatisfied with the surgery gave severe CH as the main reason. Panhofer et al. [25] also reported that the quality of life in patients who developed severe CH was worse than before surgery. Therefore, it is important to identify the risk factors for the development of CH, so that those patients with higher risk of developing severe CH can be excluded for surgery.
Another reason that could explain the lower rates of satisfaction is the nature of the study being a long-term one. Patient satisfaction is related not only to the results (resolution of palmar sweating), but also to patient tolerance to side effects. Once the initial euphoria over the improvement in their palmar sweating dissipates, the patient’s unsatisfied perception about postoperative side effects will be amplified.
Besides compensatory hyperhidrosis, excessive dryness of the hands can also influence the degree of postoperative patient satisfaction [26, 27]. In our study, almost a quarter of patients experienced some form of overdryness of the hand. Excessive dry hands are a cause for dissatisfaction as they are inconvenient in daily life activities and require frequent use of moisturizers. Liu et al. [28] have suggested a correlation between excessively dry hands and lower degree of patient satisfaction. Although we were unable to show a statistically significant correlation in our study between dry hands and level of satisfaction due to small sample size, it is important to consider this side effect when counseling patient for operation.
In addition, it is often suggested that CH progressively disappears with time [29–32]. Our experience demonstrates that CH was still present after more than 2 years and patients did not report improvement with time. Although we did not specifically ask for the onset of CH, it was well noted in the literature [33] that most CH occurred within the first 8 weeks to a year after operation. In our study, the mean follow-up of patients with CH is about 8 years, with more than 90.2 % of the patients complaining of some form of CH still persisting. Hence, it can be admitted that the common assertion that CH vanishes with time is most likely wrong. This is in accordance with many long-term studies [11, 12] that report similar high rates of CH after long periods of follow-up.
The strength of our study lies in its long follow-up time period. In our study, we captured long-term clinical outcomes such as recurrence, CH, and patient satisfaction, which may not be accurately captured at short-term follow-up. Admittedly, our nonresponse rate was significant, despite measures to contact our patients. In addition, the unique part of our study is that we look specifically at patients who underwent asymmetrical level of thoracic sympathectomy to compare the effects of extent of sympathectomy with its long-term outcomes, as most studies compared patients who received the same level of sympathectomy bilaterally. This way of comparing reduces the heterogeneity of patient factors in affecting outcome and gives us a more accurate view of whether T2 ablation is as effective as T2–T3 ablation.
Conclusion
In conclusion, endoscopic thoracic sympathectomy is the treatment of choice for patients with primary hyperhidrosis. Our study concluded that T2 ablation in VATS for palmar hyperhidrosis was as effective as T2–T3 in terms of outcome, recurrence, and compensatory sweating. CH was an important factor in patient satisfaction, and our results suggested that a warmer climate may precipitate increased rate of severe CH. Hence, patients have to be counseled on the risks of CH, especially since our results suggest that a warmer climate may result in higher rates of CH. They must also be counseled on the other risks, such as the possibility of recurrence and hand overdryness, since these factors also have an impact on patient satisfaction. Although we had a limited number of cases and need to randomize a larger study group to substantiate our results, there seemed to be no significant difference between the extent of sympathectomy and the risks of CH or recurrence noted in our study at present.
References
Dumont P, Denoyer A, Robin P (2004) Long-term results of thoracoscopic sympathectomy for hyperhidrosis. Ann Thor Surg 78:1801–1807
Kwong KF, Hobbs JL, Cooper LB, Burrows W, Gamliel Z, Krasna MJ (2008) Stratified analysis of clinical outcomes in thoracoscopic sympathicotomy for hyperhidrosis. Ann Thorac Surg 85:390–393 (discussion 393–394)
Zacherl J, Huber ER, Imhof M, Plas EG, Herbst F, Fugger R (1998) Long-term results of 630 thoracoscopic sympathicotomies for primary hyperhidrosis: the Vienna experience. Eur J Surg 580:43–46
Ojimba TA, Cameron AE (2004) Drawbacks of endoscopic thoracic sympathectomy. Br J Surg 91:264–269
Rex LO, Drott C, Claes G, Gothberg G, Dalman P (1998) The Boras experience of endoscopic thoracic sympathicotomy for palmar, axillary, facial hyperhidrosis and facial blushing. Eur J Surg 580:23–26
Li X, Tu YR, Lin M, Lai FC, Chen JF, Dai ZJ (2008) Endoscopic thoracic sympathectomy for palmar hyperhidrosis: a randomized control trial comparing T3 and T2-4 ablation. Ann Thor Surg 85:1747–1751
Cerfolio RJ, De Campos JR, Bryant AS, Connery CP, Miller DL, DeCamp MM, McKenna RJ, Krasna MJ (2011) The Society of Thoracic Surgeons expert consensus for the surgical treatment of hyperhidrosis. Ann Thor Surg 91:1642–1648
Lai CL, Chen WJ, Liu YB, Lee YT (2001) Bradycardia and permanent pacing after bilateral thoracoscopic T2-sympathectomy for primary hyperhidrosis. Pacing Clin Electrophysiol PACE 24:524–525
Munia MA, Wolosker N, Kauffman P, de Campos JR, Puech-Leao P (2007) A randomized trial of T3-T4 versus T4 sympathectomy for isolated axillary hyperhidrosis. J Vasc Surg 45:130–133
Yang J, Tan JJ, Ye GL, Gu WQ, Wang J, Liu YG (2007) T3/T4 thoracic sympathictomy and compensatory sweating in treatment of palmar hyperhidrosis. Chin Med J 120:1574–1577
Gossot D, Galetta D, Pascal A, Debrosse D, Caliandro R, Girard P, Stern JB, Grunenwald D (2003) Long-term results of endoscopic thoracic sympathectomy for upper limb hyperhidrosis. Ann Thor Surg 75:1075–1079
Herbst F, Plas EG, Fugger R, Fritsch A (1994) Endoscopic thoracic sympathectomy for primary hyperhidrosis of the upper limbs. A critical analysis and long-term results of 480 operations. Ann Surg 220:86–90
Katara AN, Domino JP, Cheah WK, So JB, Ning C, Lomanto D (2007) Comparing T2 and T2–T3 ablation in thoracoscopic sympathectomy for palmar hyperhidrosis: a randomized control trial. Surg Endosc 21:1768–1771
Solish N, Bertucci V, Dansereau A, Hong HC-H, Lynde C, Lupin M, Smith KC, Storwick G (2007) A Comprehensive Approach to the Recognition, Diagnosis, and Severity-Based Treatment of Focal Hyperhidrosis: recommendations of the Canadian Hyperhidrosis Advisory Committee. Dermatol Surg 33:908–923
Schmidt J, Bechara FG, Altmeyer P, Zirngibl H (2006) Endoscopic thoracic sympathectomy for severe hyperhidrosis: impact of restrictive denervation on compensatory sweating. Ann Thor Surg 81:1048–1055
Yazbek G, Wolosker N, de Campos JR, Kauffman P, Ishy A, Puech-Leao P (2005) Palmar hyperhidrosis–which is the best level of denervation using video-assisted thoracoscopic sympathectomy: T2 or T3 ganglion? J Vasc Surg 42:281–285
Hsu CP, Chen CY, Hsia JY, Shai SE (1998) Resympathectomy for palmar and axillary hyperhidrosis. Br J Surg 85:1504–1505
Lin TS (2001) Video-assisted thoracoscopic “resympathicotomy” for palmar hyperhidrosis: analysis of 42 cases. Ann Thor Surg 72:895–898
Lyra Rde M, Campos JR, Kang DW, Loureiro Mde P, Furian MB, Costa MG, Coelho Mde S, Brasileira Sociedade, de Cirurgia T (2008) Guidelines for the prevention, diagnosis and treatment of compensatory hyperhidrosis. Jornal brasileiro de pneumologia: publicacao oficial da Sociedade Brasileira de Pneumologia e Tisilogia 34:967–977
Sugimura H, Spratt EH, Compeau CG, Kattail D, Shargall Y (2009) Thoracoscopic sympathetic clipping for hyperhidrosis: long-term results and reversibility. J Thorac Cardiovasc Surg 137:1370–1376 (discussion 1376–1377)
Chiou TS, Chen SC (1999) Intermediate-term results of endoscopic transaxillary T2 sympathectomy for primary palmar hyperhidrosis. Br J Surg 86:45–47
Lai YT, Yang LH, Chio CC, Chen HH (1997) Complications in patients with palmar hyperhidrosis treated with transthoracic endoscopic sympathectomy. Neurosurgery 41:110–113 (discussion 113-115)
Al Dohayan A (1999) Transaxillary thoracoscopic sympathectomy experience in a hot climate: management of the dominant hand. Surgical laparoscopy, endoscopy & percutaneous techniques 9:317–321
Chou SH, Kao EL, Lin CC, Chang YT, Huang MF (2006) The importance of classification in sympathetic surgery and a proposed mechanism for compensatory hyperhidrosis: experience with 464 cases. Surg Endosc 20:1749–1753
Panhofer P, Zacherl J, Jakesz R, Bischof G, Neumayer C (2006) Improved quality of life after sympathetic block for upper limb hyperhidrosis. Br J Surg 93:582–586
Kopelman D, Hashmonai M (2008) The Correlation Between the Method of Sympathetic Ablation for Palmar Hyperhidrosis and the Occurrence of Compensatory Hyperhidrosis: a Review. World J Surg 32:2343–2356
Milanez de Campos JR, Kauffman P, de Campos Werebe E, Andrade Filho LO, Kusniek S, Wolosker N, Biscegli Jatene F (2003) Quality of life, before and after thoracic sympathectomy: report on 378 operated patients. Ann Thor Surg 76:886–891
Liu Y, Yang J, Liu J, Yang F, Jiang G, Li J, Huang Y, Wang J (2009) Surgical treatment of primary palmar hyperhidrosis: a prospective randomized study comparing T3 and T4 sympathicotomy. Eur J Cardio Thorac Surg 35:398–402
Byrne J, Walsh TN, Hederman WP (1990) Endoscopic transthoracic electrocautery of the sympathetic chain for palmar and axillary hyperhidrosis. Br J Surg 77:1046–1049
Drott C, Gothberg G, Claes G (1995) Endoscopic transthoracic sympathectomy: an efficient and safe method for the treatment of hyperhidrosis. J Am Acad Dermatol 33:78–81
Hashmonai M, Kopelman D, Kein O, Schein M (1992) Upper thoracic sympathectomy for primary palmar hyperhidrosis: long-term follow-up. Br J Surg 79:268–271
Kopelman D, Hashmonai M, Ehrenreich M, Assalia A (1998) Thoracoscopic sympathectomy for hyperhidrosis: is there a learning curve? Surg Laparosc Endosc 8:370–375
Chiou TS (2005) Chronological changes of postsympathectomy compensatory hyperhidrosis and recurrent sweating in patients with palmar hyperhidrosis. J Neurosurg Spine 2:151–154
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
Drs. Wilson Ong, Alvin Lee, Tan Wee Boon, and Davide Lomanto have no conflicts of interest or financial ties to disclose.
Rights and permissions
About this article
Cite this article
Ong, W., Lee, A., Tan, W.B. et al. Long-term results of a randomized controlled trial of T2 versus T2–T3 ablation in endoscopic thoracic sympathectomy for palmar hyperhidrosis. Surg Endosc 30, 1219–1225 (2016). https://doi.org/10.1007/s00464-015-4335-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00464-015-4335-2