Abstract
Tyrosol and hydroxytyrosol, by-products of olive oil production, are valuable substrates for enzymatic transglycosylation that can provide products with pharmaceutical potential. Phenylethanoid fructosides are produced from sucrose and phenylethanoids by the catalytic action of β-fructofuranosidases. This work dealt with the potential of the most abundant β-fructofuranosidase, baker's yeast invertase, for this bioconversion. The effects of sucrose and phenylethanoid concentrations were investigated with a focus on the selectivity of phenylethanoid transfructosylation and fructoside yields. For this purpose, initial rate and progress curve experiments were carried out for the initial (hydroxy)tyrosol and sucrose concentrations of 0.072–0.3 M and 1–2 M, respectively. Reaction courses exhibited either a maximum or plateau of fructoside yield in the range of about 10–18%. The addition of deep eutectic solvents was applied in the concentration range from 5 to 70% (v/v) to investigate the possibility of shifting the reaction equilibrium towards fructoside synthesis.
Graphical abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The significant antioxidant activity of tyrosol and hydroxytyrosol appears to have a potential in the prevention of various diseases associated with oxidative stress. Among various age-related diseases, pharmacological studies indicate their ability to increase central nervous system activity, cardiovascular and cognitive functions [1, 2]; especially, hydroxytyrosol has been proposed as a promising anticancer agent [3].
The naturally accessible source of these phenyletanoids are olives and their industrial by-products such as olive oil mill wastewater [4]. Glycosylated forms of phenyletanoids have improved solubility, stability, bioavailability, and even, in some cases, pharmacological activity [5, 6]. The coupling of various glycones also provides glycosides with improved properties for their use as functional food supplements.
Most papers deal with the glycosylation of tyrosol simply due to its much lower cost than that of hydroxytyrosol. Protocols for the enzymatic synthesis of tyrosol β-D-glucopyranoside (salidroside), a laboriously isolated natural glycophenolic, have been published by several authors [7–10]. Salidroside is known to be a major carrier of the therapeutic effect in Rhodiola sp. [11]. Also, a variety of new-to-nature tyrosol glycoside derivatives have been prepared. Synthetic procedures have been developed for the preparation of β-D-galactopyranoside [8, 12, 13] β-D-fructofuranoside [14, 15] and rutinoside [16] tyrosol derivatives.
Since hydroxytyrosol β-D-fructofuranoside (hydroxytyrosol fructoside, HT-Fru) is one of the most effective antioxidants, the search for new and improved processes for its synthesis is of high interest [6]. Enzymatic fructosylation is a useful option for this biosynthesis. In general, different fructosyltransferases (EC 2.4.1.9) and β-fructofuranosidases (EC 3.2.1.26) can be used for this purpose [17]. Fructosyltransferases have a more dominant transfructosylating activity, while the main catalytic mechanism of β-fructofuranosidases comprises sucrose hydrolysis into its individual components, fructose, and glucose. Depending on their microbial origin, a high transfructosylating ability of β-fructofuranosidases towards various fructosyl acceptors was observed [18]. The nature of these enzymes plays an important role in their acceptor specificity.
The selection of a suitable biocatalyst for the desired process is performed on a case-by-case basis. Efficient enzymatic synthesis of tyrosol β-D-fructofuranoside (tyrosol fructoside, Tyr-Fru) has been reported using an industrial β-galactosidase preparation with a high β-fructofuranosidase side activity [14]. Hollá et al. [15] pointed out the differences in the ability to transfer fructosyl moieties to tyrosol among 32 industrial enzyme preparations investigated mainly originating from filamentous fungi. The most efficient preparation, which produced fructooligosaccharides as the dominant product, originated from an Aspergillus niger strain. The Tyr-Fru yield reached a maximum of 6%. In contrast, our latest work concluded that enzymes with high fructosyltransferase activity towards sucrose appear to be less likely to succeed in tyrosol transfructosylation [13]. Higher yields, 27.6%, were obtained for yeast enzyme preparations. The highest Tyr-Fru yield was achieved with a Kluyveromyces lactis preparation possessing a β-fructofuranosidase side activity.
A drawback of industrial preparations with the β-fructofuranosidase side activity is that this side activity can be significantly reduced or eliminated if the preparations are replaced by an innovated product by the manufacturer. Therefore, it is worth considering preparations that have β-fructofuranosidase as the main enzyme component. The best-known enzyme of this kind is β-fructofuranosidase from Saccharomyces cerevisiae, also called invertase. It is widely used in the food and beverage industries to produce invert sugar by sucrose hydrolysis. Baker’s yeast invertase exhibits low oligosaccharide synthesis rates in aqueous sucrose solutions [19–23]. It, however, performs much better if other substrates are used as fructosyl moiety acceptors. Its transfructosylation capacity was investigated for various aliphatic and aromatic alcohols a long time ago [24, 25]. Later, the effect of various reaction parameters such as sucrose and alcohol concentration, enzyme loading, pH, and the temperature was studied [26–28].
The course of reactions catalysed by glycosidases can be manipulated using high acceptor concentrations or cosolvent addition that lowers hydrolysis [29]. Among others, deep eutectic solvents (DESs) represent an interesting, new type of cosolvents. DESs are formed by the interaction of a hydrogen bond donor (HBD; such as a quaternary ammonium salt) and a hydrogen bond acceptor (HBA; such as sugar, polyol, amine, or carboxylic acid) in a eutectic molar ratio at which the mixture has a liquid state at ambient temperature. The range of low-cost precursors, ease of synthesis, sustainability, non-volatility, non-flammability, and low toxicity are advantages that lead scientists to implement these solvents in various processes as a more environmentally friendly option compared to organic solvents or ionic liquids [30, 31].
Uhoraningoga et al. [32] reported an improved synthesis of alkyl glucosides by reverse hydrolysis in DES containing 40% (v/v) choline chloride and glycerol [32]. In another study, β-glucosidase from almonds was implemented for alkyl glucoside synthesis in a two-in-one DES system, in which the substrate is part of DES. However, the yield achieved was relatively low [33]. Factors that influence the activity and stability of enzymes in the DES systems include the type of HBA and HBD, their molar ratio, and concentration in the reaction medium [34, 35]. However, from the data obtained so far, no general conclusion was obtained for the prediction of enzyme behaviour in the DES environment.
This work deals with the performance of an industrial enzyme preparation of β-fructofuranosidase from Saccharomyces cerevisiae, Invertase (Novozymes), in the synthesis of tyrosol and hydroxytyrosol fructosides from sucrose. The effect of sucrose and phenylethanoid concentrations on the enzyme acceptor specificity was investigated using initial rate measurements. Progress curve measurements provided the effect of these concentrations on the yields of phenylethanoid fructosides and fructooligosaccharide by-products. The potential of DES addition to influence the thermodynamics of the reaction network was studied for 12 different DESs. They were screened by evaluating enzyme activity and tyrosol transfructosylation efficacy.
Materials and methods
Materials
The chemical reagents purchased from Sigma-Aldrich Productions GmbH (Steinheim, Germany) were tyrosol, sucrose, glucose, and fructose. Buffer components and methanol in purity pro analysis were purchased from Centralchem (Bratislava, Slovakia). Hydroxytyrosol was purchased from Biosynth (Compton, Berkshire, UK). The enzymatic assay kit Bio-La-Test Glu 500 was obtained from Erba Lachema s.r.o. (Brno, Czech Republic). Acetonitrile and methanol for HPLC were purchased from Merck (Darmstadt, Germany). Tyrosol β-D-fructofuranoside, used as an HPLC calibration standard, was prepared following the procedure described by Karnišová Potocká et al. [14]. Carbohydrate standards including 1-kestose and nystose were purchased from Wako Pure Chemical (Osaka, Japan). Choline chloride (99%) and betaine were purchased from Carbosynth (Compton, Berkshire, UK). Choline acetate was obtained from Abcr GmbH (Karlsruhe, Germany).
Enzyme
Invertase, an industrial enzymatic preparation of β-fructofuranosidase from Saccharomyces cerevisiae, was supplied by Novozymes A/S (Bagsvared, Denmark). The specific activity of Invertase, determined as described below in the part “β-Fructofuranosidase activity assay”, was 245 U/mg (per solid weight).
Synthesis of deep eutectic solvents
Components of DESs, hydrogen bond acceptors, choline chloride (ChCl), choline acetate (ChAc), or betaine (Bet) were mixed with hydrogen bond donors such as urea, glycerol (Gly) and ethylene glycol (EtGly) in specific molar ratios given in Table 1. The components were stirred and heated up in a water bath up to 80 °C, until a clear homogeneous liquid was formed. DES–buffer solutions were prepared by dissolving a DES in sodium acetate buffer to the required volume fraction.
β-Fructofuranosidase activity assay
β-Fructofuranosidase activity was determined using a 146 mM sucrose solution in 0.1 M acetate buffer with pH 6. The concentration of the enzyme stock solution was 2.6 mg/mL. The reaction was started by adding 100 µL of 20 times diluted enzyme stock solution in the identical buffer to 1 mL of the substrate solution preheated to 40 °C. Samples (100 µL) were taken at regular intervals and the reaction was stopped by the addition of 100% methanol (300 µL). The release of glucose was measured through a glucose oxidase colorimetric assay. 10 µL of the sample was mixed with 1000 µL of the Bio-La-Test Glu 500 solution under the specified conditions and concentration of the released glucose was determined by spectrophotometric measurement at 500 nm. One unit was defined as the amount of enzyme that produces 1 µmol of glucose per minute under the test conditions. The assay was carried out in triplicate. The activity of the stock solution was 638 ± 25 U/mL (mean ± standard deviation).
Activity assay using a 96-well microtiter plate
The effect of DES type on β-fructofuranosidase activity was evaluated using a glucose assay kit adapted to a 96-well microtiter plate. The screening was based on the intensity of the colour response, which is directly proportional to the activity of the enzyme. Invertase was dissolved in sodium acetate buffer with pH 5 at a concentration of 0.13 mg/mL. 250 µL of 146 mM sucrose solution in a mixture of the identical buffer and the relevant DES were mixed with 25 µL of enzyme at 40 °C for 40 min. The reaction was terminated by transferring 20 µL of the sample to a new 96-well plate with 360 µL of 100% methanol. 10 µL of this mixture was then added to 370 µL of the Bio-La-Test Glu 500 solution.
Enzymatic production of tyrosol fructoside and hydroxytyrosol fructoside
The transfructosylation of tyrosol and hydroxytyrosol in the absence of DES was carried out in a reaction mixture (4.5 mL) consisting of sucrose and either tyrosol or hydroxytyrosol in 0.1 M sodium acetate buffer with pH 6. The concentration intervals examined for these substrates were 1.0 to 2.0 M for sucrose, 0.072 M to 0.3 M for tyrosol, and 0.072 M to 0.13 M for hydroxytyrosol, respectively. The reactions were carried out at 50 °C (for tyrosol) or 40 °C (for hydroxytyrosol) with the enzyme concentration adjusted to 3.5 U/mL. Both phenylethanoids are sufficiently stable at these temperatures [36, 37]. In the presence of DES in the reaction mixture, the process conditions were slightly modified. The transfructosylation reaction was carried out at pH 5 and 1.5 M sucrose and 0.072 M tyrosol. Aliquots (100 µL) were collected at the appropriate time intervals and diluted with 400 µL of 100% methanol to inactivate the enzyme. The tyrosol and hydroxytyrosol fructoside yields were calculated as the ratio of the molar concentration of the particular product to the initial concentration of the respective acceptor.
Initial reaction rates of transfructosylation reactions and sucrose hydrolysis under reaction conditions
Initial reaction rates for the hydrolytic activity and transferase activity were determined with respect to both sucrose and tyrosol. Samples were taken from the reaction mixture at regular intervals of 20 min to obtain at least six experimental points. The reaction conditions, substrate concentrations, and sample treatment were identical to those described above. The rate of fructose formation was used to represent the rate of sucrose hydrolysis. The time course of the concentration of 6-kestose and tyrosol fructoside was monitored to determine the transfer rates to sucrose and tyrosol, respectively. Initial reaction rates were calculated from the slope of the linear region of a product formation curve over time. The molar concentrations of 6-kestose, tyrosol fructoside, and fructose were quantified by HPLC analysis described below.
HPLC analyses
HPLC analyses were performed using an Agilent 1260 system equipped with an Asahipak NH2P-50 4E column (250 × 4.6 mm, 5 µm; Shodex, Japan) and an NH2P-50G 4A guard column (4.6 × 10 mm, 5 µm; Shodex, Japan) with a flow rate of 1.0 mL/min and a temperature maintained at 35 °C. Tyrosol and tyrosol glycosides were detected by a diode array detector (DAD) at 275 nm. The HPLC system was coupled to an Agilent 1110 refractive index (RI) detector operated at 35 °C, used for the determination of saccharides. The composition of the eluent was fixed at 68% acetonitrile/water (v/v). Samples withdrawn from the reaction medium were diluted 45-fold to avoid precipitation in further analysis steps. They were mixed with acetonitrile to match its concentration in the mobile phase. All compounds were quantified by the external standard method on the basis of peak area. Due to the absence of a 6-kestose standard, it was identified as the peak with the retention time of 10.1 min. The molar refractivity of 1-kestose was used for its quantification. The formation of a trisaccharide was also verified by the mass balance of saccharide moieties. Hydroxytyrosol and hydroxytyrosol fructoside were analysed on a Zorbax Eclipse C8 column (Merck, Germany) and detected by a DAD detector at 275 nm. The column temperature was maintained at 35 °C. The samples were diluted with redistilled water to adjust their concentration to 30% v/v. Analytes were eluted with 30% methanol/water (v/v) at a flow rate of 1.0 mL/min.
Results
Effect of tyrosol and sucrose concentrations on the reaction network
Tyr-Fru formation is accompanied by other parallel processes in the reaction system such as sucrose hydrolysis and transfructosylation and secondary hydrolysis of the synthesized products. Tyrosol thus competes as an acceptor of fructosyl residues with water and sucrose. The reaction network is shown in Fig. 1. As mentioned above, the extent of individual reactions depends on the reaction conditions.
Therefore, in addition to Tyr-Fru, monosaccharides, glucose and fructose and fructooligosaccharides are formed during the reaction. A typical composition of the reaction mixture is illustrated in the supplementary material (Fig. S1). Trisaccharide, 6-kestose, was the main product among the synthesized oligosaccharides. At later reaction times (after 2–4 h), very small peaks corresponding to 1-kestose and an uncharacterized disaccharide were observed (Fig. S2). According to the previously studied profile of oligosaccharides produced by Saccharomyces cerevisiae β-fructofuranosidase the unidentified peak may belong to inulobiose or 6-β-fructofuranosylglucose [38]. The amount of 6-kestose formed was used to determine the initial reaction rate of fructosyl transfer to sucrose.
The initial rate method was applied to investigate the effect of sucrose and tyrosol concentrations on the reaction network. Sucrose concentrations (from 1 to 2 M) were chosen to promote transfer reactions and suppress sucrose hydrolysis. The range of investigated concentrations of tyrosol, from 0.072 M to 0.3 M, was limited by its solubility limit. Figure 2A–C presents three measured initial rates of parallel reactions. The initial rates of sucrose hydrolysis (Fig. 2A), tyrosol transfructosylation (Fig. 2B), and sucrose transfructosylation (Fig. 2C) are presented as a function of sucrose concentration with the tyrosol concentration as a parameter. These three rates were used to calculate the ratio of the total transfructosylation rate to the sucrose hydrolysis rate presented in Fig. 2D.
Effect of sucrose and tyrosol concentration on the reaction network at pH 6.0 and 50 °C investigated using the initial rate method. A Sucrose hydrolysis rate, B tyrosol transfructosylation rate, C sucrose transfructosylation rate, and D ratio of total (sucrose plus tyrosol) transfructosylation rate to sucrose hydrolysis rate. The symbols represent different tyrosol concentrations: 0.072 M (blue square), 0.181 M (orange diamond), and 0.3 M (grey triangle) (colour figure online)
The first elementary step of the reaction mechanism is the release of glucose from sucrose accompanied by the formation of an activated enzyme complex with the fructosyl moiety. The rate of this first step is essentially rate determining for the subsequent parallel reactions of the activated complex with fructosyl acceptors. It is well known that the kinetics of the enzymatic hydrolysis of sucrose by Saccharomyces cerevisiae β-fructofuranosidase complies with the substrate-inhibition equation [39, 40]. The reason for the deviation from the classic Michaelis–Menten kinetics is attributed mainly to the thermodynamic nonideality of concentrated sucrose solutions caused by sucrose aggregation [41]. The unfavourable influence of high sucrose concentrations is apparent for both sucrose hydrolysis (Fig. 2A) and tyrosol transfructosylation (Fig. 2B) rates. This trend is independent of tyrosol concentration.
Figure 2A, B demonstrates that the rates of these reactions were approximately twice higher for 1 M sucrose than for 2 M sucrose, except for the sucrose hydrolysis rate for the highest tyrosol concentration of 0.3 M, where the effect of a high sucrose concentration was somewhat weaker. Figure 2B clearly shows the role of tyrosol as the second substrate competing for fructosyl moieties. The rate of tyrosol transfructosylation increased with its concentration to a degree that did not depend much on the sucrose concentration. On the contrary, the rate of sucrose hydrolysis decreased with the concentration of tyrosol in an analogous manner. For 0.3 M tyrosol, the tyrosol transfructosylation rate was approximately 50–60% of the corresponding sucrose hydrolysis rate, while it was only 10–15% for the lowest tyrosol concentration.
Figure 2C shows that the sucrose transfructosylation rates were much lower than the tyrosol transfructosylation rates. It means that sucrose was a poorer acceptor of fructosyl moieties than tyrosol. At the highest tyrosol concentration (0.3 M), tyrosol transfructosylation rates were approximately 15 times higher than sucrose transfructosylation rates. These initial reaction rates were nearly equal at the lowest tyrosol concentration (0.072 M). The sucrose transfructosylation rates were up to 10 times higher at 0.072 M tyrosol compared to 0.3 M tyrosol. In contrast to the trends presented in Fig. 2A, B, sucrose concentration had a minimal effect on the sucrose transfructosylation rate. An explanation is that the inhibitory effect of high sucrose concentrations is counteracted by its function as the fructosyl moiety acceptor. This hypothesis is compatible with the conclusions of the studies focused on the kinetics of sucrose transfructosylation [19, 42]. In the sucrose concentration range of 1–2 M, the substrate only slightly inhibited the formation of 6-kestose.
The ratio of transfructosylation and hydrolytic activities is a useful parameter that characterizes the selectivity of β-fructofuranosidases. The total transfructosylation rate was obtained as a sum of the tyrosol and sucrose transfructosylation rates presented in Fig. 2B, C. It was related to the sucrose hydrolysis rate (Fig. 2A) and is plotted in Fig. 2D. It is evident from this figure that this ratio was only minimally affected by the sucrose concentration, and it increased with the tyrosol concentration. These conclusions are analogous to the ones explained above for tyrosol fructosylation. It is easily comprehensible, since tyrosol transfructosylation is the dominant transfructosylation reaction.
Influence of reaction conditions on the course of tyrosol fructoside production
Previous studies on the effect of pH on the hydrolytic activity of Saccharomyces cerevisiae β-fructofuranosidase indicated only a slight change in reaction rate with pH in the region close to the optimal pH of 4–6 [43, 44]. However, according to our present results, pH affected the rate of tyrosol transglycosylation. Tyrosol fructoside yield was approximately 15% higher at pH 6 compared to the yield obtained at pH 4 (data not shown here).
The competition of water, saccharides, and tyrosol reacting with fructosyl moieties released from sucrose at the reaction start was discussed in the previous section. But the overall picture of the competition changes during the progress of the reaction. The synthesis of tyrosol fructoside is kinetically controlled; therefore, the transfructosylation production process should be stopped before the hydrolysis of the product prevails over the rate of its synthesis. Therefore, it was reasonable to study the effect of sucrose and tyrosol concentrations on the time course of tyrosol fructoside production (Fig. 3). The same combinations of initial tyrosol and sucrose concentrations were used as in the experiments presented in the previous section.
Progress curves of yields of tyrosol fructoside obtained by a transfructosylation reaction catalysed by β-fructofuranosidase from Saccharomyces cerevisiae at pH 6.0 and 50 °C and at different initial concentrations of tyrosol and sucrose. A 0.072 M tyrosol, B 0.181 M tyrosol, and C 0.300 M tyrosol. The symbols correspond to the following initial sucrose concentrations: 1.0 M (blue square), 1.5 M (orange diamond), 1.75 M (grey triangle), and 2.0 M (yellow circle)
The results obtained are somewhat surprising when considering the conclusions observed for the initial rate measurements. The effect of tyrosol concentration on the yield of tyrosol fructoside (Fig. 3) is completely opposite to that of the initial transfructosylation rate (Fig. 2). The highest yield of 16–18% was achieved for the lowest concentration of 0.072 M tyrosol (Fig. 3A), while it was only 12–13% for the highest concentration of 0.3 M (Fig. 3C). For the medium tyrosol concentration of 0.181 M, the yield was 14–15.5%. Figure 3 also shows that the process dynamics were fastest at the lowest tyrosol concentration. Furthermore, the progress curves have different shapes for the chosen total reaction time of 8 h.
A sharp maximum of Tyr-Fru yield was observed at the lowest tyrosol concentration of 0.072 M (Fig. 3A). It means that the rate of reverse Tyr-Fru hydrolysis overcame the rate of its formation by transfructosylation. The Tyr-Fru maximum was most pronounced at the lowest sucrose concentration of 1.0 M. Figure 3A shows that the increase in the initial sucrose concentration slowed the backward hydrolysis of tyrosol fructoside. Consequently, somewhat higher yields of Tyr-Fru were achieved. Higher sucrose concentrations also caused a gradual increase in the time in which the maximum yield was obtained. The highest yields were reached for sucrose concentrations of 1.5–1.75 M after 2–3 h of reaction time.
Figure 3B, C demonstrates that backward hydrolysis of Tyr-Fru was also suppressed by higher tyrosol concentrations. We suppose that this was caused by lower water activity at higher both tyrosol and sucrose concentrations. All but one progress curve asymptotically approached a yield value without exhibiting a maximum. The only exception was the experiment for 0.181 M tyrosol and 1.5 M sucrose. Here, the yield reached a maximum in 3–4 h and decreased slightly afterwards.
Although the decrease in maximal Tyr-Fru yield with the initial concentration of tyrosol represents important information about the course of reactions, the influence of high tyrosol concentration cannot be negatively interpreted from the process point of view. Figure 4 demonstrates the advantage of a higher tyrosol concentration. The Tyr-Fru concentration in the final product increased almost proportionally with the initial tyrosol concentration. It reached maximal values close to 40 mM for 0.3 M tyrosol. As follows from Figs. 3 and 4, the effect of the sucrose concentration was weak at this tyrosol concentration. A higher Tyr-Fru concentration is advantageous for downstream processing, whereas a somewhat lower yield does not essentially change the requirements for tyrosol recycling. However, it is necessary to mention that a higher concentration of tyrosol prolongs the time to reach the maximum amount of tyrosol fructoside.
Final sucrose conversion and optimal tyrosol fructoside concentration obtained by a transfructosylation reaction catalysed by β-fructofuranosidase from Saccharomyces cerevisiae at pH 6.0 and 50 °C and at different initial concentrations of tyrosol and sucrose. A 1 M sucrose, B 1.5 M sucrose, C 1.75 M, and D 2 M sucrose. The white bars represent the percentage of converted sucrose after 8 h of reaction time. The black bars represent the molar concentration of tyrosol fructoside after 4 h for 0.072 M tyrosol and after 8 h for 0.181 M and 0.300 M tyrosol
Figure 4 also shows that the rate of sucrose conversion decreased significantly with increasing sucrose and tyrosol concentrations. For example, Fig. 4D shows that, at an initial sucrose concentration of 2 M, sucrose conversion after 8 h of reaction was reduced from about 46% in the absence of tyrosol to 18% at 0.3 M tyrosol. The molar concentration of released fructose decreased in about the same proportion (data not shown). Hydrolytic activity of the enzyme was thus significantly suppressed by tyrosol. Consequently, tyrosol transfructosylation was more pronounced. The shift in the transfructosylation selectivity with tyrosol concentration is demonstrated by 3–4 times higher optimal Tyr-Fru concentrations at the highest initial tyrosol concentration in Fig. 4. This relative increase of the optimal Tyr-Fru concentrations is about twice higher than the increase in the ratio of initial transfructosylation and hydrolysis rates.
Figure 2 shows that the transfer of fructosyl residues to sucrose was slower compared to the transfer to tyrosol and this behaviour was exacerbated at higher tyrosol concentrations. It also resulted in low 6-kestose yields shown in the supplementary material (Fig. S3). These findings indicate a weak enzyme affinity towards the sucrose acceptor. The maximum molar yield of 6-kestose (6.7%, 0.0514 M) was obtained at concentrations of 1.5 M sucrose and 0.072 M tyrosol.
It should be emphasized that the 6-kestose and Tyr-Fru yields refer to very different initial concentrations of sucrose and tyrosol, respectively. For that reason, the final molar concentrations of 6-kestose were in most cases higher than those of Tyr-Fru. Only, the application of a 0.30 M concentration of the phenylethanoid acceptor suppressed fructosyl transfer to sucrose in such a measure that the Tyr-Fru molar concentrations became higher. The sucrose transformation process into 6-kestose was independent of the overall kinetically controlled mechanism. There was no indication of the onset of 6-kestose hydrolysis during the reaction timeframe.
Production of tyrosol β-fructoside in deep eutectic solvent solutions
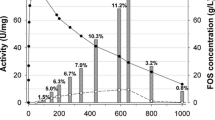
The type of solvent system is crucial in biocatalytic reactions. In terms of improving the Tyr-Fru yield, a potential shift of the reaction equilibrium towards glycoside synthesis by the addition of DES was investigated. Following the existing information on the behaviour of glycosidases in these solvents presented in the Introduction, 12 convenient DESs were selected for the investigation in this work. The DESs used and their molar compositions are listed in Table 1. A wide range of DES concentrations in the reaction mixture, from 5 to 70% v/v, was used. To rapidly examine the effect of both the type and the concentration of DES on the β-fructofuranosidase activity, a qualitative activity assay was designed using a 96-well microtiter plate. Figure 5 presents the results for 12 types of DES and 8 different concentrations of DES when the coloration intensity is directly proportional to enzyme activity.
Screening of β-fructofuranosidase activity in DES solutions by glucose assay kit after 40 min of reaction. DESs: 1—choline chloride:glycerol 1:2, 2—choline chloride:ethylene glycol 1:2, 3 –choline chloride:urea 1:1, 4—choline chloride:urea 1:2, 5—choline acetate:glycerol 1:2, 6—choline acetate:ethylene glycol 1:2, 7—choline acetate:urea 1:2, 8—betaine:glycerol 1:2, 9—betaine: ethylene glycol, 10—betaine:urea:glycerol 1:2:3, 11—betaine:ethylene glycol:glycerol 1:2:2, 12—urea:glycerol 1:9, Ref.—reference sample not containing DES
Figure 5 does not show any enhancement of enzyme activity by adding DES. Only for low concentrations of DES 11, the enzyme activity appeared to be close to that in a DES-free medium. On the contrary, the lighter coloration of most samples compared to the reference (three wells in the upper right corner in Fig. 5) implies the negative effect of these cosolvents on the formation of glucose, which represents a combined effect of hydrolytic and transfructosylation reactions. The assay showed that DES 5 and DES 7 based on choline acetate gave the worst results in this assay. At 30% and higher concentrations of these eutectics, no enzyme activity was observed under the given test conditions. There were relatively low differences among the performance of other DESs tested.
DES 2, DES 3, DES 6, DES 9 and DES 11 were selected for a subsequent set of experiments as the best-performing solvents and DES 7 as the worst-performing solvent. Tyrosol fructoside (Tyr-Fru) production was monitored at DES concentrations of 5% (v/v) to 40% (v/v). In the presence of 40% DES, the transfructosylation of tyrosol was completely stopped in all cases. Therefore, the results of transfructosylation in DES solution systems are presented in Fig. 6 with 20% DES as an upper limit. (Full progress curves are given in the supplementary material, Fig. S4 and Fig. S5). The Tyr-Fru yields are plotted as relative values with the corresponding yield in DES-free conditions as reference.
Figure 6 documents that Tyr-Fru synthesis was suppressed by DESs when there was not much difference between the DESs identified as the best and the worst in the screening experiments. All DESs caused a significant decline in Tyr-Fru yield with the DES concentration. The lowest tested concentration of DES 11, 5% (v/v), resulted in a 26% reduction in the Tyr-Fru yield. When the DES content was 10% (v/v), the Tyr-Fru yields dropped by 33–55% and in 20% DES (v/v) the yield was typically only 30–35% compared to the DES-free conditions. Even worse results were obtained for DES 2 consisting of ChCl and EtGly, where the Tyr-Fru yield decreased by 80%. The reported negative effect of DES on tyrosol transfructosylation was much greater compared to the previously examined β-fructofuranosidase from Kluyveromyces lactis [13].
Figure 6 offers several interesting comparisons. For example, there was a notable difference between Tyr-Fru yields in ChCl:EtGly (DES 2) and ChCl:Urea (DES 3). Despite the same hydrogen bond acceptor (ChCl), the Tyr-Fru yield was 14% higher in DES 3 although its hydrogen bond donor, urea, is known for its denaturing effects. An explanation can be found in the presence of ethylene glycol as a more reactive fructosyl acceptor than tyrosol. In aqueous solutions, the DES components are no longer linked by hydrogen bonds [46]. In this case, the DES components based on polyols (glycerol, ethylene glycol) become available in reaction media for fructosylation. Their competition for fructosyl residues can be enhanced by their high concentration in the reaction medium.
The same pattern was also observed for another hydrogen acceptor, choline acetate (ChAc). The Tyr-Fru yield was better in case of DES 7 (urea hydrogen donor) than DES 6 (ethylene glycol hydrogen donor). Choline acetate and urea (DES 7) represented the best combination of hydrogen acceptor and donor for Tyr-Fru production among all tested DESs. This is quite surprising since DES 7 was identified as the worst environment in the first set of screening experiments (Fig. 5). The product progress curves shown in the supplementary material (Fig. S4 and Fig. S5) confirm that both observations are correct. Although the initial Tyr-Fru production rate was the lowest for DES 7, the final Tyr-Fru yield was the highest. On the contrary, the highest initial rates were obtained for betaine-based DESs (DES 9 and DES 11), but the yields were among the poorest. Since all these DESs were shown to stabilize β-fructofuranosidase against denaturation [13], the lower product yields in DESs were the effect of reaction inhibition and interference of the DES components with the Tyr-Fru synthesis.
Production of hydroxytyrosol fructoside
Due to an additional hydroxy group, hydroxytyrosol (HT) has remarkably attractive biochemical properties that increase its market demand [6, 45]. Therefore, after successful transfructosylation of tyrosol by β-fructofuranosidase from Saccharomyces cerevisiae, this enzyme was applied for the production of hydroxytyrosol fructoside (HT-Fru).
Due to the higher sensitivity of HT to elevated temperatures, the reaction temperature was reduced to 40 °C. HT-Fru formation was examined in 1 M and 1.5 M sucrose with an initial HT concentration of 0.072 M (11 g/L) and 0.130 M (20 g/L). The 1 M concentration was selected based on the HT transfructosylation reaction conditions used in the previous studies [14, 47] and 1.5 M sucrose is our optimized concentration for Tyr-Fru formation using baker's yeast β-fructofuranosidase. The course of HT-Fru synthesis is shown in Fig. 7.
Progress curves of yields of hydroxytyrosol fructoside obtained by a transfructosylation reaction catalysed by β-fructofuranosidase from Saccharomyces cerevisiae at pH 6.0 and 40 °C. A 0.072 M hydroxytyrosol and B 0.130 M hydroxytyrosol. The symbols represent different initial concentrations of sucrose (1 and 1.5 M, respectively)
Similarly to Tyr-Fru formation, higher sucrose concentrations suppressed the secondary hydrolysis of HT-Fru and increased the product molar yield (Fig. 7). The highest HT-Fru yield, 16.4%, was obtained for 1.5 M sucrose and 0.072 M HT. Likewise as for Tyr-Fru, the higher concentration of the alcohol acceptor increased the product concentration, but decreased its yield. The transfructosylation rate and the overall yield of fructoside were slightly lower for the reaction with HT compared to tyrosol.
The synthesis of HT-Fru by Saccharomyces cerevisiae β-fructofuranosidase reported in this work is quite attractive compared to the results published so far. Although Karnišová Potocká et al. achieved a higher molar yield (around 33%), the enzyme preparation used in their work is no longer on the market [14]. Compared to HT fructosylation reported by Míguez et al., who reached approximately 23% molar yield of HT-Fru with a genetically modified enzyme [47], the advantage of the synthesis described here is that it uses one of the most available, inexpensive, and easily produced enzymes.
Conclusions
This work demonstrates that baker's yeast β-fructofuranosidase is a very suitable biocatalyst for the synthesis of fructosides of tyrosol and hydroxytyrosol. Higher initial sucrose concentrations (above 1.5 M) were found to be beneficial for the biocatalyst transfructosylation selectivity. Higher phenylethanoid concentrations (0.3 M) also suppressed the secondary product hydrolysis, although they slowed all reactions. Inhibition of all reaction rates was also observed when deep eutectic solvents were added to the substrates solutions. An expected positive effect of the shift of reaction equilibrium towards transglycosylation reactions was thus not achieved in this way.
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Ma GP, Zheng Q, Xu MB et al (2018) Rhodiola rosea L. improves learning and memory function: preclinical evidence and possible mechanisms. Front Pharmacol 9:1415. https://doi.org/10.3389/fphar.2018.01415
Karković Marković A, Torić J, Barbarić M, Jakobušić Brala C (2019) Hydroxytyrosol, tyrosol and derivatives and their potential effects on human health. Molecules 24:1–39. https://doi.org/10.3390/molecules24102001
Emma MR, Augello G, di Stefano V et al (2021) Potential uses of olive oil secoiridoids for the prevention and treatment of cancer: a narrative review of preclinical studies. Int J Mol Sci 22:1–22. https://doi.org/10.3390/ijms22031234
Pedan V, Popp M, Rohn S et al (2019) Characterization of phenolic compounds and their contribution to sensory properties of olive oil. Molecules. https://doi.org/10.3390/molecules24112041
Horvathova E, Mastihubova M, Karnisova Potocka E et al (2019) Comparative study of relationship between structure of phenylethanoid glycopyranosides and their activities using cell-free assays and human cells cultured in vitro. Toxicol in Vitro. https://doi.org/10.1016/j.tiv.2019.104646
Kis P, Horváthová E, Gálová E et al (2021) Synthesis of tyrosol and hydroxytyrosol glycofuranosides and their biochemical and biological activities in cell-free and cellular assays. Molecules. https://doi.org/10.3390/molecules26247607
Akita H, Kawahara E, Kishida M, Kato K (2006) Synthesis of naturally occurring β-d-glucopyranoside based on enzymatic β-glycosidation. J Mol Catal B Enzym 40:8–15. https://doi.org/10.1016/j.molcatb.2006.01.031
Potocká E, Mastihubová M, Mastihuba V (2015) Enzymatic synthesis of tyrosol glycosides. J Mol Catal B Enzym 113:23–28. https://doi.org/10.1016/j.molcatb.2014.12.017
Wang F, Huang D, Ma Y et al (2019) Preparation of salidroside with n -butyl β -D-glucoside as the glycone donor via a two-step enzymatic synthesis catalyzed by immobilized β -glucosidase from bitter almonds. Biocatal Biotransform 37:246–260. https://doi.org/10.1080/10242422.2018.1549236
Bi Y, Wang Z, Mao Y et al (2012) Ionic liquid effects on the activity of β-glycosidase for the synthesis of salidroside in co-solvent systems. Chin J Catal 33:1161–1165. https://doi.org/10.1016/S1872-2067(11)60395-1
Panossian A, Wikman G, Sarris J (2010) Rosenroot (Rhodiola rosea): Traditional use, chemical composition, pharmacology and clinical efficacy. Phytomedicine 17:481–493. https://doi.org/10.1016/j.phymed.2010.02.002
Qi T, Gu G, Xu L et al (2017) Efficient synthesis of tyrosol galactosides by the β-galactosidase from Enterobacter cloacae B5. Appl Microbiol Biotechnol 101:4995–5003. https://doi.org/10.1007/s00253-017-8249-x
Hollá V, Hill R, Antošová M, Polakovič M (2021) Design of immobilized biocatalyst and optimal conditions for tyrosol β-galactoside production. Bioprocess Biosyst Eng 44:93–101. https://doi.org/10.1007/s00449-020-02425-2
Karnišová Potocká E, Mastihubová M, Mastihuba V (2019) Enzymatic synthesis of tyrosol and hydroxytyrosol β-d-fructofuranosides. Biocatal Biotransform 37:18–24. https://doi.org/10.1080/10242422.2017.1423060
Hollá V, Antošová M, Karkeszová K et al (2019) Screening of commercial enzymes for transfructosylation of tyrosol: effect of process conditions and reaction network. Biotechnol J. https://doi.org/10.1002/biot.201800571
Karnišová Potocká E, Mastihubová M, Mastihuba V (2021) Transrutinosylation of tyrosol by flower buds of Sophora japonica. Food Chem. https://doi.org/10.1016/j.foodchem.2020.127674
Toledo LET, García DM, Cruz EP, et al (2018) Fructosyltransferases and invertases: useful enzymes in the food and feed industries. In: Kuddus M (ed) Enzymes in food biotechnology: production, applications, and future prospects. Elsevier, pp 451–469
Antošová M, Polakovič M (2001) Fructosyltransferases: the enzymes catalyzing production. Chem Pap 55:350–358
Straathof AJJ, Kieboom APG, van Bekkum H (1986) Invertase-catalysed fructosyl transfer in concentrated solutions of sucrose. Carbohydr Res 146:154–159. https://doi.org/10.1016/0008-6215(86)85033-9
Vega-Paulino RJ, Zúniga-Hansen ME (2012) Potential application of commercial enzyme preparations for industrial production of short-chain fructooligosaccharides. J Mol Catal B Enzym 76:44–51. https://doi.org/10.1016/j.molcatb.2011.12.007
Marín-Navarro J, Talens-Perales D, Polaina J (2015) One-pot production of fructooligosaccharides by a Saccharomyces cerevisiae strain expressing an engineered invertase. Appl Microbiol Biotechnol 99:2549–2555. https://doi.org/10.1007/s00253-014-6312-4
Ganaie MA, Gupta US, Kango N (2013) Screening of biocatalysts for transformation of sucrose to fructooligosaccharides. J Mol Catal B Enzym 97:12–17. https://doi.org/10.1016/j.molcatb.2013.07.008
Lafraya Á, Sanz-Aparicio J, Polaina J, Marín-Navarro J (2011) Fructo-oligosaccharide synthesis by mutant versions of Saccharomyces cerevisiae invertase. Appl Environ Microbiol 77:6148–6157. https://doi.org/10.1128/AEM.05032-11
Breuer HJ, Bacon JSD (1957) Sucrose formation by Taka-diastase: action of the enzyme on methyl β-fructofuranoside and raffinose. Biochem J 66:462–468. https://doi.org/10.1042/bj0660462
Baseer A, Shall S (1971) The enzymic synthesis of fructosides using purified external β-fructosidase from Saccharomyces cerevisiae. Int J Biochem 2:503–506. https://doi.org/10.1016/0020-711X(71)90019-X
Straathof AJJ, Vrijenhoef JP, Sprangers EPAT et al (1988) Enzymic formation of β-D-fructofuranosides from sucrose: activity and selectivity of invertase in mixtures of water and alcohol. J Carbohydr Chem 7:223–238. https://doi.org/10.1080/07328308808058916
Selisko B, Ulbrich R, Schellenberger A, Müller U (1990) Invertase-catalyzed reactions in alcoholic solutions. Biotechnol Bioeng 35:1006–1010. https://doi.org/10.1002/bit.260351008
Rodríguez M, Gómez A, González F et al (1996) Selectivity of methyl-fructoside synthesis with β-fructofuranosidase. Appl Biochem Biotechnol 59:167–175. https://doi.org/10.1007/BF02787818
Abdul Manas NH, Md. Illias R, Mahadi NM (2018) Strategy in manipulating transglycosylation activity of glycosyl hydrolase for oligosaccharide production. Crit Rev Biotechnol 38:272–293. https://doi.org/10.1080/07388551.2017.1339664
Panić M, Cvjetko Bubalo M, Radojčić Redovniković I (2021) Designing a biocatalytic process involving deep eutectic solvents. J Chem Technol Biotechnol 96:14–30. https://doi.org/10.1002/jctb.6545
Kist JA, Zhao H, Mitchell-Koch KR, Baker GA (2021) The study and application of biomolecules in deep eutectic solvents. J Mater Chem B 9:536–566. https://doi.org/10.1039/d0tb01656j
Uhoraningoga A, Kinsella GK, Henehan GT, Ryan BJ (2021) β-glucosidase from Streptomyces griseus: ester hydrolysis and alkyl glucoside synthesis in the presence of Deep Eutectic Solvents. Curr Opin Green Sustain Chem. https://doi.org/10.1016/j.crgsc.2021.100129
Delavault A, Grüninger J, Kapp D et al (2022) Enzymatic synthesis of alkyl glucosides by β-glucosidases in a 2-in-1 deep eutectic solvent system. Chem Ing Tech 94:417–426. https://doi.org/10.1002/cite.202100150
Cheng QB, Zhang LW (2017) Highly efficient enzymatic preparation of daidzein in deep eutectic solvents. Molecules. https://doi.org/10.3390/molecules22010186
Hoppe J, Drozd R, Byzia E, Smiglak M (2019) Deep eutectic solvents based on choline cation—physicochemical properties and influence on enzymatic reaction with β-galactosidase. Int J Biol Macromol 136:296–304. https://doi.org/10.1016/j.ijbiomac.2019.06.027
Attya M, Benabdelkamel H, Perri E, Russo A, Sindona G (2010) Effects of conventional heating on the stability of major olive oil phenolic compounds by tandem mass spectrometry and isotope dilution assay. Molecules 15:8734–8746. https://doi.org/10.3390/molecules15128734
Zafra-Gómez A, Luzón-Toro B, Capel-Cuevas S, Carlos-Morales J (2011) Stability of hydroxytyrosol in aqueous solutions at different concentration, temperature and with different ionic content: a study using UPLC-MS. Food Nutr Sci 2:1114–1120. https://doi.org/10.4236/fns.2011.210149
Farine S, Versluis C, Bonnici PJ et al (2001) Application of high performance anion exchange chromatography to study invertase-catalysed hydrolysis of sucrose and formation of intermediate fructan products. Appl Microbiol Biotechnol 55:55–60. https://doi.org/10.1007/s002530000493
Bowski L, Saini R, Ryu DY, Vieth WR (1971) Kinetic modeling of the hydrolysis of sucrose by invertase. Biotechnol Bioeng 13:641–656. https://doi.org/10.1002/bit.260130505
Nelson JM, Schubert MP (1928) Water concentration and the rate of hydrolysis of sucrose by invertase. J Am Chem Soc 50:2189–2193. https://doi.org/10.1021/ja01395a017
Shearwin KE, Winzor DJ (1988) Substrate as a source of thermodynamic nonideality in enzyme kinetic studies: invertase-catalyzed hydrolysis of sucrose. Arch Biochem Biophys 260:532–539. https://doi.org/10.1016/0003-9861(88)90478-X
Antošová M, Illeová V, Vandáková M et al (2008) Chromatographic separation and kinetic properties of fructosyltransferase from Aureobasidium pullulans. J Biotechnol 135:58–63. https://doi.org/10.1016/j.jbiotec.2008.02.016
Polakovič M, Báleš V, Dluhý M, Štefuca V (1993) Bioprocess engineering optimization of a packed bed bioreactor with immobilized cells using experimental design. Bioprocess Eng 9:225–230. https://doi.org/10.1007/BF00369406
Valerio SG, Alves JS, Klein MP et al (2013) High operational stability of invertase from Saccharomyces cerevisiae immobilized on chitosan nanoparticles. Carbohydr Polym 92:462–468. https://doi.org/10.1016/j.carbpol.2012.09.001
Méndez-Líter JA, Tundidor I, Nieto-Domínguez M et al (2019) Transglycosylation products generated by Talaromyces amestolkiae GH3 β-glucosidases: effect of hydroxytyrosol, vanillin and its glucosides on breast cancer cells. Microb Cell Fact 18:1–12. https://doi.org/10.1186/s12934-019-1147-4
Gabriele F, Chiarini M, Germani R et al (2019) Effect of water addition on choline chloride/glycol deep eutectic solvents: characterization of their structural and physicochemical properties. J Mol Liq. https://doi.org/10.1016/j.molliq.2019.111301
Míguez N, Ramírez-Escudero M, Gimeno-Pérez M et al (2018) Fructosylation of hydroxytyrosol by the β-fructofuranosidase from Xanthophyllomyces dendrorhous: insights into the molecular basis of the enzyme specificity. ChemCatChem 10:4878–4887. https://doi.org/10.1002/cctc.201801171
Acknowledgements
This work was supported by grants from the Slovak Research and Development Agency (Grant No. APVV-18-0188) and the Slovak Grant Agency for Science (Grant No. VEGA 1/0515/22).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethics approval
No ethical issues are related to the conducted research.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Karkeszová, K., Antošová, M., Potocká, E.K. et al. Medium engineering of phenylethanoid transfructosylation catalysed by yeast β-fructofuranosidase. Bioprocess Biosyst Eng 46, 237–249 (2023). https://doi.org/10.1007/s00449-022-02828-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-022-02828-3