Abstract
The cellulase production by filamentous fungi Aspergillus fumigatus JCM 10253 was carried out using agro-industrial waste ragi husk as a substrate in the microbial fermentation. The effect of the process parameters such as temperature, substrate concentration, pH, and incubation process time and their interdependence was studied using response surface methodology. The optimum cellulase activities were obtained at 50 °C under the conditions with 1–2% of substrate concentration at pH 2–4 for the incubation period of 7–8 days. The maximum carboxymethyl cellulase (CMCase) and β-glucosidase activities with optimized process variables were 95.2 IU/mL and 0.174 IU/mL, respectively. The morphological characterization of fungus by scanning electron microscopy (SEM) and Fourier transform infrared spectroscopy (FTIR) revealed the presence of secondary protein structures. Furthermore, this study demonstrated that the application of ragi husk could be a promising feedstock for value-added industrial products. The thermo-acidophilic nature of isolated strain Aspergillus fumigatus JCM 10253 possessed a significant potential for higher titer of cellulase production that could be further employed for lignocellulosic bioethanol production.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Lignocellulosic materials have gained more importance as a raw material for biofuel production due to worldwide availability of low-cost renewable feedstock in abundance; fossil fuel usage causes global and greenhouse gas emission; uncertainty supply of petroleum due to high consumption and present no food production conflict. Lignocellulosic polysaccharide polymers are cross-linked with strong bonds that lead to a robust structure. Due to the highly complex structure of these natural polymers, economically feasible production of lignocellulosic bioethanol is challenging. Cellulases are a combination of three major enzyme endoglucanases (EC 3.2.1.4), exoglucanases, including cellobiohydrolases (CBHs) (EC 3.2.1.91), and β-glucosidases (EC 3.2.1.21). Cellulases are the enzymes that catalyze the hydrolysis of cellulose into glucose by attacking the glycosidic linkages [1]. Due to the complex structure of lignocellulosic biomass, the enzyme quantity required for hydrolysis is onefold higher than for starch [2]. The production cost and low yield of these enzymes are the foremost issues for industrial applications. The purpose of agro-waste as a solid substrate in the fermentation system for the production of the enzymes has many advantages due to its lower operating expenses and lower capital assets relative to other techniques [3]. Another advantage of fermentation processes allows the utilization of forestry, agriculture, and agro-industrial residues as substrates, which maintains the microbial growth and metabolism by allowing the microorganisms to grow on moist solid substrates [4, 5].
Since commercial processes require high temperature, there is a rising demand for a supply of thermotolerant enzymes mainly those which are resistant to inhibition of product and are extremely thermostable [6, 7]. Presently, thermophilic fungal enzymes have received more attention due to its industrial biotechnology relevant applications as a potential biocatalyst such as stability of their protein at elevated temperatures and oxidative ligninolytic enzymes for lignocellulosic biomass degradation at high temperatures. Thus, these enzymes have substantial industrial potential, even though under extreme operational conditions provide better yields [8].
In preliminary studies, Aspergillus fumigatus JCM 10253 was isolated from a soil sample and screened for cellulase activity by the plate assay method using carboxymethyl cellulose (CMC) agar medium with Congo red staining and β-glucosidase activity was screened by gel diffusion plate assay method. It was observed that strain JCM 10253 has shown the highest cellulolytic enzyme producer among other isolates [9]. In the present study, ragi (Eleusine coracana) husk is used as a substrate for enzyme production. Ragi husk was received after the dehusking process from the agriculture field of Chikmagalur District, State of Karnataka, which is a major ragi (or finger millet) producing state in India [10]. To optimize process parameters, different approaches were implemented such as one-factor-at-a-time (OFAT) and statistical design to maximize the individual responses of CMCase and β-glucosidase with process variables such as temperature, substrate concentration, pH, and process time duration [11,12,13,14,15].
To our knowledge, there are no reports of using ragi husk together with fungal cultures for cellulase production. Therefore, the objective of the present study was to investigate CMCase and β-glucosidase production by directly utilizing agro-industrial waste ragi husk as a substrate for the microbial fermentation process. Besides, a statistical design was used to optimize the process variables that have a significant role in enzyme production to reduce energy requirements and cost.
Materials and methods
Microbial strain
The fungal strain was isolated from a soil sample collected from Warangal District, Telangana, India. The strain was identified as Aspergillus fumigatus JCM 10253 (Accession no. LC228711) based on 18S rRNA sequencing performed by the National Fungal Culture Collection of India (NFCCI), Agharkar Research Institute, Pune, India. The isolated fungal strain was maintained on Emerson YSS agar and stored at 4 °C for further studies [9].
Scanning electron microscopy analysis
Czapek–Dox broth medium with addition of 1% (w/v) carboxymethyl cellulose (CMC) was prepared in 250 mL of Erlenmeyer flasks. The prepared medium was autoclaved and then inoculated a loopful of isolated fungal spores into a prepared medium was incubated for 20 days at 30 °C, 150 rpm. After 20 days of incubation, fungal biomass was filtered by Whatman filter paper and treated with 3% glutaraldehyde and 2% formaldehyde for 5 h. The samples were washed with distilled water three-to-four times and treated with ethanol for dehydration. After that, the samples were kept in the oven to remove the moisture content. The prepared samples were carried out for high magnification images under scanning electron microscopy (SEM) at different voltage and magnifications [16].
Processing of substrate
Ragi husk is an outer covering or protective layer of ragi grains. The ragi husk was obtained after the dehusking process from the agriculture field. The dried husk with the particle size of 1.33 mm was sieved and stored at room temperature.
Enzyme production in shake flasks
Cellulase production was performed in the Mandels and Weber medium [17]. The Mandels and Weber production medium was carried out in 250 mL of Erlenmeyer flasks which composed of: KH2PO4, 2 gm/L; CaCl2∙2H2O, 0.3 gm/L; Urea, 0.3 gm/L; MgSO4∙7H2O, 0.3 gm/L; (NH4)2SO4, 1.4 gm/L; Peptone, 0.25 gm/L; Yeast extract, 0.1 gm/L; FeSO4∙7H2O, 0.005 gm/L; MnSO4∙H2O, 0.0016 gm/L; ZnSO4∙7H2O, 0.0014 gm/L; CoCl2∙6H2O, 0.002 gm/L; Tween-80, 1 mL; pH 2.0, and supplemented with 1.5% (w/v) ragi husk as a substrate and 1% (w/v) CMC as a carbon source. The prepared medium was sterilized by autoclaving at 121 °C under a pressure of 15 psi for 15 min. A loopful of isolated fungal spores from the agar plate was inoculated into the above-sterilized medium and incubated at 50 °C, 160 rpm for 12 days. During the fermentation process, enzyme activity was measured by collecting the sample for every 24 h of interval and centrifugation at 10,000 rpm for 20 min at 4 °C to separate the pellet from a liquid medium. Therefore, the supernatant was collected for further analysis.
Protein quantification and enzyme assays
Bradford assay was used to determine the extracellular total protein content [18]. Carboxymethyl cellulase (CMCase) and filter paper activity (FPase) were measured at 540 nm wavelength using UV–visible spectrophotometer (Model: Jasco-V-630) as the method described by Ghose [19]. β-glucosidase activity was analyzed at a wavelength of 400 nm using the substrate as p-nitrophenyl-β-d-glucopyranoside (pNPG) [20]. Enzyme activity was evaluated in international units (IU) per mL. The quantity of enzyme required to liberate 1 µmol of glucose or p-nitrophenol from their respective substrates per min under the standard assay conditions was defined as one unit (IU) of enzyme activity [21].
Optimization of cellulase production
Optimization of the process variables influencing the enzyme production was performed using the one-factor-at-a-time (OFAT) method. The process variables selected for investigation were temperature (30–60 °C), substrate concentration [0.25–3% (w/v)], pH (1‒6), and incubation time (1–12 days). All experimental tests were performed in triplicate at an agitation rate of 160 rpm.
Experimental design
Based on the result obtained from the one-factor-at-a-time approach, response surface methodology (RSM) was employed. The experimental central composite design (CCD) was used to determine the effect of variables and optimize the variables that had a significant effect on enzyme production. Statistical software Design-Expert7® was used to analyze and fit a second-order quadratic model to four independent variables [i.e., temperature (A), substrate concentration (B), pH (C), and incubation time (D)], with the experimental ranges denoted in Table 1A and B. Selections of actual and coded factors were based on the premise that has a significant influence on enzyme production. Table 2A and B shows the 30 experimental setup of CCD with respect to the variables. The response surface plots were obtained to determine their optimum levels and also to understand the effect of variables individually and in combination [22].
Fourier transform infrared spectroscopy
The characterization of the crude enzyme was performed by Fourier transform infrared spectroscopy (FTIR) (Model: 3000 Hyperion Microscope with Vertex 80 FTIR system, Bruker, Germany). The pellet was obtained by adding the sample to KBr salt (5% sample, 95% KBr), and the spectrum scan range was from 4000 to 400 cm−1 [23]. Disks were prepared in triplicates to obtain a constant spectrum.
Results and discussion
SEM analysis
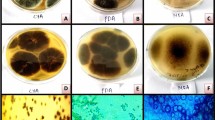
The morphological characterization of fungal biomass from Aspergillus fumigatus JCM 10253 was grown on the CMC-based medium for 20 days which found to be the most significant for micro- or nano-scale visualizing of surface morphology and structures such as conidial heads, conidia, and ascospores. The SEM analysis determined that it can reproduce sexually by developing cleistothecium with ascospores and asexually by developing conidia (Fig. 1). Such observations play a crucial role in the effective cultivation of fungi and also in optimization at different parameters for fermentation processes [16, 24]. The previous results described different stages of Aspergillus sp. biofilm that was analyzed through SEM [16, 25]. The reports revealed that the morphological and physiological studies of Aspergillus biofilms are beneficial for industrial aspects [26, 27].
Effect of physiological parameters on cellulase production
Temperature is the most significant physical variable affecting enzyme production, because enzymes are sensitive to temperature [28, 29]. The optimal production of CMCase (17.1 IU/mL), FPase (4.40 IU/mL), and β-glucosidase (0.14 IU/mL) were obtained at 50 °C which was in the temperature range favorable to thermophilic fungi [30]. Enzyme production gradually increased in the temperature range from 30 to 50 °C, and maximum enzyme production occurred at high temperature, which is suitable for the hydrolysis of cellulosic materials. However, a further increase in temperature beyond 50 °C results in a drastic decline in enzyme activities (Fig. 2a). The thermostable enzymes have been reported previously as a great potential towards lignocellulosic biomass conversion into simple sugars for the bioenergy sector due to persisting non-denatured under a longer duration of hydrolysis condition [31]. Gautam et al. reported optimum cellulase production using municipal solid waste residue by Trichoderma sp. at 45 °C [32]. Liu et al. exhibited optimum CMCase and FPase activities from Aspergillus fumigatus Z5 were obtained at 50 °C [33]. On the other hand, Soni et al. [34] described optimum cellulase level achieved at 45 °C and Mehboob et al. demonstrated the maximal cellulase activity from Aspergillus fumigatus at 55 °C [35]. Mallek-Fakhfakh et al. also reported that maximum β-glucosidase activity was achieved at 50 °C [36]. The obtained results suggest that cellulases from Aspergillus fumigatus JCM 10253 perform well under thermophilic conditions. Hence, these types of enzymes are favored to employ for commercial purposes or other biotechnological applications.
Profiles of cellulase production by Aspergillus fumigatus JCM 10253 using ragi husk as a substrate. a Effect of temperature, b effect of substrate concentration, c effect of pH, and d effect of time course on CMCase, FPase, and β-glucosidase activity. Cellulase production measured by its activity as described in the Materials and methods section. Results are the mean of triplicate experiments with standard deviation represented by error bars
The level of the solid substrate acts as a crucial role in enzyme production during fermentation. Substrate concentration ranging from 0.25 to 3% (w/v) was used to determine the substrate effect on enzyme production in the medium (Fig. 2b). The optimal substrate concentrations for cellulase production were determined at 1.5% (w/v) for CMCase (34.4 IU/mL), 1% (w/v) for FPase (21.7 IU/mL), and β-glucosidase (0.151 IU/mL). The different substrate concentrations were used to enhance the production of enzymes. The utilization of agro-waste materials in fermentation provides aeration and growth to the microbes which survive in between substrate particles. Moreover, the availability of the nutrients in medium absorbed by a solid substrate through which microbes maintain their growth in the medium for enzyme production [37, 38]. There are various factors which influence enzyme production like accessibility, biodegradability, chemical structure, and the physiochemical linkage between the components of the substrate [37, 39,40,41]. Kumar et al. described cellulase producing fungi Schizophyllum commune NAIMCC-F-03379 which showed optimum CMCase and FPase activities with 1% (w/v) wheat bran [42]. The previous reports also showed that the supplement of 1% (w/v) sawdust supported optimum CMCase, FPase, and β-glucosidase activities by Aspergillus niger [43, 44]. The CMCase and FPase activities enhanced with 1% (w/v) soybean hulls by Penicillium sp.[45].
The pH is a critical environmental parameter that influences fungal growth and plays a major role in enzyme stability and production. The cellulase production by Aspergillus fumigatus JCM 10253 was optimized by varying pH from 1 to 6. The maximum CMCase (84.2 IU/mL) and FPase activities (27.2 IU/mL) occurred when the initial pH was 2, and β-glucosidase (0.163 IU/mL) showed the maximum activity at pH 4 (Fig. 2c). The results demonstrated that the cellulase activities decreased as the pH increased towards alkalinity. The optimum cellulase production determined at low pH indicates that fungal strain is acidophilic in nature. This property suggests that the enzymes could be useful to degrade cellulosic materials in acidic conditions. In the previous studies, optimum pH values ranging from 1 to 6 were determined for cellulase production [46,47,48]. El-Nahrawy et al. reported optimum cellulase production at pH 4.0 by Aspergillus tubingensis KY615746 [49]. Liu et al. described maximum CMCase and FPase production by Aspergillus fumigatus Z5 at pH 4.0 [33]. Simultaneously, Xu et al. described β-glucosidase production by Inonotus obliquus exhibited maximum activity at pH 3.5 [50]. The report revealed by Delabona et al. that the maximum FPase and β-glucosidase activities were obtained at pH 4.0 and 3.0, respectively [51]. Similarly, the thermo-acidophilic fungus Aspergillus terreus M11 showed high-level cellulase activity at pH 3 [39].
The effect of the incubation period on cellulase production was investigated and the results are shown in Fig. 2d. CMCase activity (84.6 IU/mL) showed optimum on the 7th day of the incubation period; whereas the maximum FPase and β-glucosidase activities were 27.6 IU/mL and 0.169 IU/mL occurred on the 9th and 8th day of incubation period, respectively. Extending the incubation time beyond these values did not have any significant effect on enzyme production due to the various reasons behind it such as spontaneous depletion of nutrients or either toxic substances produced in the medium during the fermentation process which affects fungal growth as well as enzyme production [37, 40]. Silva et al. demonstrated optimum cellulase activity by thermophilic fungus Myceliophthora heterothallica F.2.1.4 exhibited after 192 h of incubation period [52]. The previous report showed that optimum cellulase production by Trichoderma reesei was achieved after the 8th day of incubation period [53]. However, in another study, CMCase, FPase, and β-glucosidase from Penicillium oxalicum HC6 showed maximal activities on the 8th day of incubation time [54]. Simultaneously, Penicillium sp. AKB-24 exhibited maximum β-glucosidase activity obtained on the 9th day of the incubation period [16].
Optimization of enzyme production using Design-Expert® statistical software
Table 3A and B shows the analysis of variance (ANOVA) for the response surface quadratic model to evaluate the significant effects of variables and to confirm the adequacy of the model. The regression models developed showed responses as functions of temperature (A), substrate concentration (B), pH (C), and incubation time (D). A polynomial equation for the model describes the relationship between the responses and the independent variables:
Low probability value of p [(pmodel > F) < 0.0005] shows that the model terms are significant. The fitness of the models was indicated by the coefficients (R2) of 95.46% for CMCase and 92.38% for β-glucosidase activity. Furthermore, only 4.5% and 7.6% of the variation in the enzyme activities are not described by the model, respectively. In the current study, the pred. R2 values for Eq. 1 (84.1%) and Eq. 2 (80.1%) show that the sample variations for CMCase and β-glucosidase of 95.46% and 92.38%, respectively, were attributed to independent variables. The adjusted coefficient (adj. R2 = 91.2% and R2 = 85.2% for Eq. 1 and Eq. 2, respectively) was also satisfactory and corroborated the significance of the models.
Interactive effects of independent process variables on cellulase activity
The interactive effect of process variables on cellulase activity is illustrated in Fig. 3a–l. The highest cellulase activity was achieved in the temperature range of 40–60 °C, substrate concentration from 0.5 to 2% (w/v), pH range of 1–5, and incubation time from 6 to 9 days. The three-dimensional surface plots were employed to investigate the mutual interactions among the independent variables to develop the correlation between the selected parameters. Each response surface for a particular enzyme represents different combinations of two test variables simultaneously while holding the other variables at zero.
Three-dimensional surface plots used to explore the interactions between the variables: CMCase activity vs. substrate and temperature (a), β-glucosidase activity vs. substrate and temperature (b), CMCase activity vs. pH and temperature (c), β-glucosidase activity vs. pH and temperature (d), CMCase activity vs. incubation time and temperature (e), β-glucosidase activity vs. incubation time and temperature (f), CMCase activity vs. pH and substrate (g), β-glucosidase activity vs. pH and substrate (h), CMCase activity vs. incubation time and substrate (i), β-glucosidase activity vs. incubation time and substrate (j), CMCase activity vs. incubation time and pH (k), β-glucosidase activity vs. incubation time and pH (l)
Model validation
The statistical response surface methodology proposed the desirable theoretical models for enzyme production. The most highly desirable theoretical model was selected to validate the models. The optimal values for CMCase production were 48.6 °C at 1.6% (w/v) substrate concentration, pH 2.0, and 8.5 days of process time. Correspondingly, this combination resulted in a predicted maximum CMCase activity of 97.06 IU/mL, which was quite close to the experimental observation of 95.2 IU/mL. Similarly, a temperature of 48.8 °C at 1.17% (w/v) substrate concentration, pH 3.8, and 7.6 days of process time resulted in a predicted maximum β-glucosidase activity of 0.19 IU/mL, which was relatively close to the experimental observation of 0.174 IU/mL, as described in Table 4. The predicted values from optimized parameters attain from the RSM models are closely agreed with the experimental observations, validating that the statistical software for experimental design can effectively optimize the process variables, and identify the significance of the individual, cumulative, and interaction effects of the independent variables in enzyme production [55].
The incorporation of the statistical method has proven to be a strong and beneficial tool for the enhancement of standard culture condition and to obtain minimal data with the less number of test trials for high titer production. However, the potential utilization of agricultural by-products ragi husk as a cheap commodity along with selecting an efficient fungal strain Aspergillus fumigatus JCM 10253 that effectively employed for microbial fermentation to scale up the production of cellulase that revealed as a potential feedstock for enzyme production. In the development of more robust processes, these thermostable enzymes can play a key role as a biocatalyst in sustainable bio-based processes for the production of fuels and chemicals. Therefore, the production values of CMCase and β-glucosidase were compared with those for previously reported enzymes produced by other strains of Aspergillus fumigatus using different substrates, which are described in Table 5.
FTIR characterization
The characterization of optimized crude CMCase and β-glucosidase enzymes was performed by FTIR spectroscopy. The FTIR spectrum of the crude enzymes showed the most intense absorption band for protein helical structures (Fig. 4a, b). For alpha structure, the frequency of 1639.89 and 1607.37 cm−1 for CMCase and β-glucosidase showed amide I vibration, respectively, which are considered for protein secondary structure components [23]. It is primarily governed by the stretching vibration of C = O which is related to backbone conformation. Amide II is found in the 1500 and 1400 cm−1 region which derives N–H-bending and C–N-stretching vibration [63, 64]. The degradation process of lignocellulosic materials to glucose required synergistic action between different enzymes. Therefore, the characterization of co-related enzymes is mandatory.
Conclusion
This study evaluated the potential use of ragi husk as a raw material for cellulase production. The present study highlighted the ability of an isolated thermo-acidophilic fungus strain Aspergillus fumigatus JCM 10253 which could produce cellulase under production medium with untreated ragi husk as a substrate which is an abundant and cost-effective commodity. Statistical design response surface methodology was employed to maximize CMCase and β-glucosidase production. The parameters (temperature, substrate, pH, and process time) showed significant effects on CMCase and β-glucosidase production. The experimental yield of CMCase and β-glucosidase was 95.2 IU/mL and 0.174 IU/mL, respectively, using the optimized process parameters which were closely predicted by models developed through RSM. Consequently, these results demonstrated that the agro-industrial waste ragi husk has potential use as a substrate to scale up the production of the highly efficient thermophilic enzymes for converting lignocellulosic wastes into fermentable sugars.
References
Zabed H, Faruq G, Sahu JN, Azirun MS, Hashim R, Nasrulhaq Boyce A (2014) Bioethanol production from fermentable sugar juice. Sci World J. 2014:11. https://doi.org/10.1155/2014/957102
Balan V (2014) Current challenges in commercially producing biofuels from lignocellulosic biomass. ISRN biotechnol. 2014:31. https://doi.org/10.1155/2014/463074
Latifian M, Hamidi-Esfahani Z, Barzegar M (2007) Evaluation of culture conditions for cellulase production by two Trichoderma reesei mutants under solid-state fermentation conditions. Biores Technol 98:3634–3637
Medina-Morales M, Martínez-Hernández JL, De La Garza H, Aguilar CN (2011) Cellulolytic enzymes production by solid state culture using pecan nut shell as substrate and support. AJABS 6:196–200
El-Shishtawy RM, Mohamed SA, Asiri AM, Abu-bakr MG, Ibrahim IH, Al-Talhi HA (2014) Solid fermentation of wheat bran for hydrolytic enzymes production and saccharification content by a local isolate Bacillus megatherium. BMC Biotechnol 14:29
Arora R, Behera S, Sharma NK, Singh R, Yadav YK, Kumar S (2014) Biochemical conversion of rice straw (Oryza sativa L.) to bioethanol using thermotolerant isolate K. marxianus NIRE-K3. In: Proceedings of exploring and basic sciences for Next Generation Frontiers. Elsevier, New Delhi, pp 143–146.
Mallerman J, Papinutti L, Levin L (2015) Characterization of β-glucosidase produced by the white rot fungus Flammulina velutipes. J Microbiol Biotechnol 25:57–65
Andrade CM, Pereira N Jr, Antranikian G (1999) Extremely thermophilic microorganisms and their polymer-hidrolytic enzymes. Rev Microbiol 30:287–298
Saroj P, Manasa P, Narasimhulu K (2018) Characterization of thermophilic fungi producing extracellular lignocellulolytic enzymes for lignocellulosic hydrolysis under solid-state fermentation. Bioresour Bioprocess 5:31
Bellundagi V, Umesh KB, Ravi SC (2016) Growth dynamics and forecasting of finger millet (ragi) production in Karnataka. Econ Aff 61(2):195–201
Jampala P, Tadikamalla S, Preethi M, Ramanujam S, Uppuluri KB (2017) Concurrent production of cellulase and xylanase from Trichoderma reesei NCIM 1186: enhancement of production by desirability-based multi-objective method. 3 Biotech 7:14
Srikanth R, Siddartha G, Reddy CHS, Harish B, Ramaiah MJ, Uppuluri KB (2015) Antioxidant and anti-inflammatory levan produced from Acetobacter xylinum NCIM2526 and its statistical optimization. Carbohyd Polym 123:8–16
Ahamed A, Vermette P (2008) Culture-based strategies to enhance cellulase enzyme production from Trichoderma reesei RUT-C30 in bioreactor culture conditions. Biochem Eng J 40:399–407
Ahamed A, Vermette P (2010) Effect of mechanical agitation on the production of cellulases by Trichoderma reesei RUT-C30 in a draft-tube airlift bioreactor. Biochem Eng J 49:379–387
Shajahan S, Moorthy IG, Sivakumar N, Selvakumar G (2017) Statistical modeling and optimization of cellulase production by Bacillus licheniformis NCIM 5556 isolated from the hot spring, Maharashtra, India. J King Saud Univ Sci 29:302–310
Kumar A, Gautam A, Dutt D (2016) Co-Cultivation of Penicillium sp. AKB-24 and Aspergillus nidulans AKB-25 as a cost-effective method to produce cellulases for the hydrolysis of pearl millet stover. Fermentation 2:12
Mandels M, Weber J (1969) Prod Cellul ACS Publ 95:391–414
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Ghose T (1987) Measurement of cellulase activities. Pure Appl Chem 59:257–268
Grover AK, MacMurchie DD, Cushley RJ (1977) Studies on almond emulsin β d-glucosidase I. Isolation and characterization of a bifunctional isozyme. Biochim et Biophys Acta (BBA)-Enzymol 482:98–108
Kovács K, Szakacs G, Zacchi G (2009) Comparative enzymatic hydrolysis of pretreated spruce by supernatants, whole fermentation broths and washed mycelia of Trichoderma reesei and Trichoderma atroviride. Biores Technol 100:1350–1357
Zakaria B, Nassar H, Amr SA, El-Gendy NS (2015) Applying factorial design and response surface methodology to enhance microbial denitrogenation by tween 80 and yeast extract. Pet Sci Technol 33:880–892
Bai H, Wang H, Junde Sun MI, Han M, Huang Y, Han X, Yang Q (2013) Production, purification and characterization of novel beta glucosidase from newly isolated Penicillium simplicissimum H-11 in submerged fermentation. EXCLI J 12:528
Manan M (2017) The morphology and structure of red pigment producing fungus: Monascus purpureus. J Microbiol Exp 5:00138
González-Ramírez AI, Ramírez-Granillo A, Medina-Canales MG, Rodríguez-Tovar AV, Martínez-Rivera MA (2016) Analysis and description of the stages of Aspergillus fumigatus biofilm formation using scanning electron microscopy. BMC Microbiol 16:243
Ramage G, Rajendran R, Gutierrez-Correa M, Jones B, Williams C (2011) Aspergillus biofilms: clinical and industrial significance. FEMS Microbiol Lett 324:89–97
Villena G, Gutiérrez-Correa M (2007) Morphological patterns of Aspergillus niger biofilms and pellets related to lignocellulolytic enzyme productivities. Lett Appl Microbiol 45:231–237
Krishna C (2005) Solid-state fermentation systems—an overview. Crit Rev Biotechnol 25:1–30
Herculano PN, Porto TS, Moreira KA, Pinto GA, Souza-Motta CM, Porto ALF (2011) Cellulase production by Aspergillus japonicus URM5620 using waste from castor bean (Ricinus communis L.) under solid-state fermentation. Appl Biochem Biotechnol 165:1057–1067
Maheshwari R, Bharadwaj G, Bhat MK (2000) Thermophilic fungi: their physiology and enzymes. Microbiol Mol Biol Rev 64:461–488
Olajuyigbe FM, Nlekerem CM, Ogunyewo OA (2016) Production and characterization of highly thermostable β-glucosidase during the biodegradation of methyl cellulose by Fusarium oxysporum. Biochem Res Int. https://doi.org/10.1155/2016/3978124
Gautam S, Bundela P, Pandey A, Khan J, Awasthi M, Sarsaiya S (2011) Optimization for the production of cellulase enzyme from municipal solid waste residue by two novel cellulolytic fungi. Biotechnol Res Int. https://doi.org/10.4061/2011/810425
Liu D, Zhang R, Yang X, Wu H, Xu D, Tang Z, Shen Q (2011) Thermostable cellulase production of Aspergillus fumigatus Z5 under solid-state fermentation and its application in degradation of agricultural wastes. Int Biodeterior Biodegrad 65:717–725
Soni R, Nazir A, Chadha B (2010) Optimization of cellulase production by a versatile Aspergillus fumigatus fresenius strain (AMA) capable of efficient deinking and enzymatic hydrolysis of solka floc and bagasse. Ind Crops Prod 31:277–283
Mehboob N, Asad MJ, Asgher M, Gulfraz M, Mukhtar T, Mahmood RT (2014) Exploring thermophilic cellulolytic enzyme production potential of Aspergillus fumigatus by the solid-state fermentation of wheat straw. Appl Biochem Biotechnol 172:3646–3655
Mallek-Fakhfakh H, Fakhfakh J, Masmoudi N, Rezgui F, Gargouri A, Belghith H (2017) Agricultural wastes as substrates for β-glucosidase production by Talaromyces thermophilus: role of these enzymes in enhancing waste paper saccharification. Prep Biochem Biotechnol 47:414–423
Bharti AK, Kumar A, Kumar A, Dutt D (2018) Exploitation of Parthenium hysterophorous biomass as low-cost substrate for cellulase and xylanase production under solid-state fermentation using Talaromyces stipitatus MTCC 12687. J Radiat Res Appl Sci 11:271–280
Richard TL, Veeken AH, De Wilde V, Hamelers H (2004) Air-filled porosity and permeability relationships during solid-state fermentation. Biotechnol Prog 20:1372–1381
Gao J, Weng H, Zhu D, Yuan M, Guan F, Xi Y (2008) Production and characterization of cellulolytic enzymes from the thermoacidophilic fungal Aspergillus terreus M11 under solid-state cultivation of corn stover. Biores Technol 99:7623–7629
Sandhya C, Sumantha A, Szakacs G, Pandey A (2005) Comparative evaluation of neutral protease production by Aspergillus oryzae in submerged and solid-state fermentation. Process Biochem 40:2689–2694
Gautam A, Kumar A, Dutt D (2015) Production of cellulase-free xylanase by Aspergillus flavus ARC-12 using pearl millet stover as the substrate under solid-state fermentation. J Adv Enzym Res 1:1–9
Kumar B, Bhardwaj N, Alam A, Agrawal K, Prasad H, Verma P (2018) Production, purification and characterization of an acid/alkali and thermo tolerant cellulase from Schizophyllum commune NAIMCC-F-03379 and its application in hydrolysis of lignocellulosic wastes. AMB EXPRESS 8:173
Narasimha G, Sridevi A, Buddolla V, Subhosh CM, Rajasekhar RB (2006) Nutrient effects on production of cellulolytic enzymes by Aspergillus niger. Afr J Biotech 5:472–476
Akula S, Golla N (2018) Optimization of cellulase production by Aspergillus niger isolated from forest soil. Open Biotechnol J 12:256–269
Fernandes TG, López JA, Silva LA, Polizeli MdLT, Silva DP, Ruzene DS, Carvalho ML, Carvalho ÍF (2018) Prospecting of soybean hulls as an inducer carbon source for the cellulase production. Prep Biochem Biotechnol 48:743–749
Romero M, Aguado J, González L, Ladero M (1999) Cellulase production by Neurospora crassa on wheat straw. Enzyme and Microb Technol 25:244–250
Xia L, Cen P (1999) Cellulase production by solid state fermentation on lignocellulosic waste from the xylose industry. Process Biochem 34:909–912
Grigorevski-Lima A, Da Vinha F, Souza D, Bispo A, Bon E, Coelho R, Nascimento R (2009) Aspergillus fumigatus thermophilic and acidophilic endoglucanases. Appl Biochem Biotechnol 155:18–26
El-Nahrawy S, Metwally M, El-Kodoos A, Rizk Y, Belal E-SB, Shabana SA, El-Refai IM (2017) Optimization of culture conditions for production of cellulase by Aspergillus tubingensis KY615746 using rice straw waste. Environ Biodivers Soil Security 1:177–189
Xu X, Lin M, Zang Q, Shi S (2018) Solid state bioconversion of lignocellulosic residues by Inonotus obliquus for production of cellulolytic enzymes and saccharification. Biores Technol 247:88–95
Delabona PS, Pirota RDPB, Codima CA, Tremacoldi CR, Rodrigues A, Farinas CS (2013) Effect of initial moisture content on two Amazon rainforest Aspergillus strains cultivated on agro-industrial residues: biomass-degrading enzymes production and characterization. Ind Crops Prod 42:236–242
Silva VCT, Coto ALS, Souza RC, Neves MBS, Gomes E, Bonilla-Rodriguez GO (2016) Effect of pH, temperature, and chemicals on the endoglucanases and β-glucosidases from the thermophilic fungus Myceliophthora heterothallica F.2.1.4. obtained by solid-state and submerged cultivation. Biochem Res Int. https://doi.org/10.1155/2016/9781216
Saini A, Aggarwal NK, Yadav A (2017) Cost-effective cellulase production using Parthenium hysterophorus biomass as an unconventional lignocellulosic substrate. 3 Biotech 7:12
Sun Y-X, Shen B-B, Han H-Y, Lu Y, Zhang B-X, Gao Y-F, Hu B-Z, Hu X-M (2018) Screening of potential IL-tolerant cellulases and their efficient saccharification of IL-pretreated lignocelluloses. RSC Adv 8:30957–30965
Preetha B, Viruthagiri T (2007) Application of response surface methodology for the biosorption of copper using Rhizopus arrhizus. J Hazard Mater 143:506–510
Sherief A, El-Tanash A, Atia N (2010) Cellulase production by Aspergillus fumigatus grown on mixed substrate of rice straw and wheat bran. Res J Microbiol 5:199–211
Sarkar N, Aikat K (2014) Aspergillus fumigatus NITDGPKA3 provides for increased cellulase production. Int J Chem Eng. 2014:1–9. https://doi.org/10.1155/2014/959845
Gilna V, Khaleel K (2011) Cellulase enzyme activity of Aspergillus fumigatus from mangrove soil on lignocellulosic substrate. Recent Res Sci Technol 3:132–134
Elsa C, Kumar M, Baskar G (2015) Optimized production of cellulase using fruit waste and its application in bioethanol production. Int J Pharma Bio Sci 6(2):1005–1013
Bagewadi ZK, Ninnekar HZ (2015) Production, purifiation and characterization of endoglucanase from Aspergillus fumigatus and enzymatic hydrolysis of lignocellulosic waste. Int J Biotechnol Biomed Sci 1:25–32
Ang S, Shaza E, Adibah Y, Suraini A, Madihah M (2013) Production of cellulases and xylanase by Aspergillus fumigatus SK1 using untreated oil palm trunk through solid state fermentation. Process Biochem 48:1293–1302
Mohapatra S, Padhy S, Mohapatra PKD, Thatoi H (2018) Enhanced reducing sugar production by saccharification of lignocellulosic biomass, Pennisetum species through cellulase from a newly isolated Aspergillus fumigatus. Biores Technol 253:262–272
Krimm S, Bandekar J (1986) Vibrational spectroscopy and conformation of peptides, polypeptides, and proteins. Adv Protein chem Elsevier.
Bai H, Wang H, Sun J, Irfan M, Han M, Huang Y, Han X, Yang Q (2013) Purification and characterization of beta 1,4-glucanases from Penicillium simplicissimum H-11. BioResources 8:3657–3671
Acknowledgments
The authors acknowledge the facility provided by SAIF, IIT Bombay for FTIR analysis, and also the support provided by the Department of Biotechnology, National Institute of Technology Warangal, India.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Saroj, P., P, M. & Narasimhulu, K. Assessment and evaluation of cellulase production using ragi (Eleusine coracana) husk as a substrate from thermo-acidophilic Aspergillus fumigatus JCM 10253. Bioprocess Biosyst Eng 44, 113–126 (2021). https://doi.org/10.1007/s00449-020-02428-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00449-020-02428-z