Abstract
Belowground bud banks play a crucial role in plant population regeneration, community dynamics, and functional responses of ecosystems to environmental change and disturbance. In mesic grasslands, belowground bud banks are largely resistant to short-term drought. However, the sensitivity of belowground bud banks to long-term extreme drought in semi-arid grasslands is less understood. We investigated the legacy effects of a four-year experimental drought (i.e., 66% reduction in growing season precipitation) on belowground bud density, aboveground shoot density, and the meristem limitation index (MLI; the ratio of bud to shoot density) in two semi-arid grasslands that differ in dominant grass species growth forms (i.e., rhizomatous vs. bunchgrasses). Measurements were made during the first recovery year following drought; thus, we report the legacy effects of drought on belowground bud banks. At the community level, drought reduced belowground bud density and aboveground shoot density with no change in MLI. However, drought had no significant influences on belowground buds, aboveground shoots and MLI of the dominant plant growth form in each community. The legacy effects of drought were largely dependent on plant community type and growth form. Specifically, bunchgrasses and bunchgrass-dominated communities were characterized by greater meristem limitation than rhizomatous grasses, likely due to their cluster/phalanx clonal growth. Overall, our study suggests bud banks may indeed be sensitive to long-term drought, although this depends on plant growth forms and community characteristics.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Plants can regenerate sexually through the production of seeds, or asexually via growth from belowground meristems (Harper 1977; Ott et al. 2019). In perennial grasslands, however, population dynamics and primary productivity are almost entirely driven by asexual reproduction via belowground bud banks (Benson et al. 2004; Benson and Hartnett 2006). Belowground bud banks regulate patterns of population regeneration, community dynamics and ecosystem functioning following disturbances (e.g., fire and grazing) and changes in soil moisture and nutrient availability (Dalgleish and Hartnett 2006, 2009; Qian et al. 2017a, b; Ma et al. 2019; Kühn et al. 2021). This suggests that belowground buds, as the primary source of tiller/ramet production in many grassland communities, could contribute to ecosystem recovery following extreme events, such as drought (VanderWeide et al. 2015; Ott et al. 2019).
Global climate change is expected to increase rainfall variability, potentially increasing the frequency of extreme drought events during this century (Huang et al. 2016; Williams et al. 2020). In most water-limited ecosystems such as grasslands, drought can alter structural attributes (e.g., tiller and bud density), reduce ecosystem functions (e.g., individual plant growth and aboveground net primary productivity (ANPP)), and shift biomass allocation patterns, which can have prolonged post-drought effects on ecosystem function (Evans et al. 2011; Hoover et al. 2014; Song et al. 2016; Luo et al. 2021; Zhang et al. 2021). Consequently, legacy effects of drought (i.e., drought-induced alterations in resources or ecosystem properties that continue to affect ecosystem function post-drought) are expected to become more common in these ecosystems (Yahdjian and Sala 2006; Griffin-Nolan et al. 2018; Zhang et al. 2021). However, grasslands are highly resilient ecosystems, meaning they have a high capacity for recovery of ecosystem functions following drought (Hoover et al. 2014; Stuart-Haëntjens et al. 2018). For example, ANPP of a mesic grassland recovered to pre-drought levels within one year after drought (Hoover et al. 2014; Wilcox et al. 2020). While legacy effects of drought are often negative (Sala et al. 2012), some grasslands are more productive than expected one year after drought (Griffin-Nolan et al. 2018). Understanding how bud banks respond to extreme drought may help explain the often observed high drought resilience of grassland communities (Dukes et al. 2014).
During drought, plants senesce aboveground tissue to prevent desiccation and mortality, which can influence growth and density of belowground buds (Knapp and Smith 2001; VanderWeide et al. 2014). Plants may also alter the translocation of carbon and nutrients from aboveground to belowground organs during drought, influencing the regrowth from belowground buds after drought (VanderWeide et al. 2014). Drought can have variable impacts on belowground bud bank dynamics, ranging from negative to positive effects with prolonged recovery (Dalgleish and Hartnett 2006; Qian et al. 2017a). This variability may be related to drought intensity, timing, and duration. For example, belowground bud density decreased with decreasing water availability along a natural precipitation gradient in North American grasslands (Dalgleish and Hartnett 2006). In grasslands of northern China, however, belowground bud density decreased slightly with the increased aridity in relatively mesic regions but increased with reduced water availability in more arid regions (Qian et al. 2017a). Additionally, a short-term (2-year) experimental drought in a North American tallgrass prairie had little effect on the belowground bud bank dynamics even though it altered community structure and ecosystem functioning aboveground (VanderWeide et al. 2014). The impact of extreme droughts of longer duration on bud bank dynamics remains understudied.
The sensitivity of belowground bud banks to extreme drought likely depends on plant functional groups (Qian et al. 2017a; Klimešová and Klimeš 2007, 2008)). For example, functional groups with greater bud production and/or resource conservative traits (e.g., high tissue dry matter content) may yield greater vegetation recovery following drought compared to other functional groups (Xu et al. 2017). Specifically, rhizomatous grasses are characterized by shallow buds, while bunchgrasses have deeper tiller buds (Luo et al. 2018; Ott et al. 2019) and these characteristics may lead to differential drought sensitivity between these two groups. Additionally, bulbous species, which have large buds that can conserve water and use thickened scale leaves to protect apical meristems, may be particularly well-adapted to periodic drought (Pate and Dixon 1982). Previous work suggests drought can differentially reduce the belowground bud bank density of rhizomatous grasses, bunchgrasses and forbs in a restored grassland, mediating shifts in population structure and community composition both during and after drought (Carter et al. 2012). Yet, the patterns of drought effects on belowground bud and shoot densities in different grassland ecosystems dominated by plants with different growth traits remain unclear.
Here, we studied the legacy effects of a four-year extreme drought (66% reduction in growing season precipitation) on belowground bud density and aboveground shoot density in two semi-arid grassland communities. The dominant perennial grasses in these communities differed in growth traits, one being dominated by a rhizomatous grass (Leymus chinensis, “rhizomatous grass community”), the other by a bunchgrass (Stipa grandis, “bunchgrass community”), but both species co-occurred in each community (Bai et al. 2004; Luo et al. 2018). Previous studies in this same region have shown negative effects of extreme drought on ecosystem functioning (Luo et al. 2021; Xu et al. 2021); the present work assesses the recovery of belowground bud density following a long-term drought and its relationships with aboveground shoot density.
We tested the hypothesis that drought has lagged negative effects on belowground bud banks. Specifically, we predicted that both belowground bud density and aboveground shoot density would be lower in plots previously drought compared to control plots (Dalgleish and Hartnett 2006; Qian et al. 2017a). We also hypothesized that the legacy effects on bud and shoot densities would differ between the two communities given the difference in the traits of the dominant grass species (rhizomatous vs bunchgrass). Specifically, we predicted that rhizomatous grass communities would recover faster than bunchgrass communities due to the shallower root and bud structures in the rhizomatous vs. bunchgrass-dominated community (Klimešová and Klimeš 2008) and tendency for near-surface soil moisture to rehydrate faster than deeper soil moisture layers post drought.
Materials and methods
Study sites
This study was conducted at two sites within the Inner Mongolia Grassland Ecosystem Research Station (IMGERS), located in the Xilin River Basin, Inner Mongolia Autonomous Region, China (116°33′E, 43°32′N) (Figure S1). These sites are part of the Extreme Drought in Grassland Experiment (EDGE) in China. The two sites are characterized as semi-arid grassland and share similar climatic conditions. The major soil types of this region are calcic chestnuts and calcic chernozems (Luo et al. 2018).
These two grasslands differ in species composition with one being dominated by a rhizomatous grass (L. chinensis) and the other by a bunchgrass (S. grandis) (Table 1). Importantly, each of these species also occur in the community where they are not dominant, but at lower abundance (Table 1). The rhizomatous grasses regenerate via rhizome buds distributed in the topsoil, while bunchgrasses mainly regenerate via deeper tiller buds, although some also have rhizome buds (Klimešová and Klimeš 2007, 2008). These rhizomatous- and bunchgrass-dominated grasslands are the most widely distributed grassland ecosystems across the steppe region of Eurasia, which represents the largest contiguous grassland area in the world (Kang et al. 2017). The rhizomatous grass and bunchgrass communities have been fenced to exclude large ungulate herbivores since 1999 and 1979, respectively (Bai et al. 2004). Both communities reach peak productivity (ANPP of about 193 g m− 2 for the L. chinensis dominated community, and 217 g m− 2 for the S. grandis dominated community) in mid-August (Kang et al. 2007).
Drought experiment
The experiment was established in April 2015 as a randomized block design with two precipitation levels. We established 12 plots (6 × 6 m in size; n = 6 control; n = 6 drought) in a topographically uniform area at each site and arranged plots in a randomized complete block design. We oriented plots such that each plot was separated from neighboring plots by at least 2 m. We inserted aluminum flashing to a depth of 1 m around each plot to prevent any lateral movement of soil water. We simulated extreme drought with passive rainfall manipulation in each grassland site (Figure S1). Shelters intercepted approximately 66% of growing season precipitation and permitted more than 90% transmittance of photosynthetically active radiation. We installed rainfall exclusion shelters on May 1st and removed them at the end of August for four years (2015–2018). We established a 1 m external buffer zone under each shelter to minimize edge effects associated with the rainout shelter. We designed the shelters with a slight slope toward a subtle topographic gradient to allow for rapid runoff of the intercepted precipitation. We obtained the long-term meteorological data from the weather stations of IMGERS.
Sampling and investigation
In early August of 2019 (the first recovery year following 4-year experimental drought), we sampled aboveground biomass by harvesting all plants within two 0.5 m × 0.5 m quadrats that were randomly located in each plot. We sorted all living vascular plants into species and dried these samples at 65 °C for 48 h before weighing. We used the dry mass of all plant species averaged over two quadrats to estimate aboveground community biomass in each plot.
We investigated the belowground bud bank and aboveground shoots within a 0.3 m × 0.3 m quadrat adjacent to the two 0.5 m × 0.5 m quadrats in each experimental plot. As most buds in this grassland are located in shallow soil profiles (0–30 cm), we excavated all belowground parts to a depth of 30 cm (Qian et al. 2017a, b). We kept the connection between below and aboveground plant parts intact to accurately identify the buds of different species. We stored samples in plastic bags, took them to lab and processed them within one week. Following the procedures of Qian et al. (2017a, 2021), we categorized belowground buds into four types: (1) tiller buds (axillary buds at the shoot base of caespitose and rhizomatous grasses), (2) rhizome buds (axillary buds and apical buds on hypogenous rhizomes sensu Klimešová and Klimeš (2008), (3) bulb buds (meristems wrapped in the swollen leaf base or scale leaf of bulb species) and (4) dicot buds (buds on belowground parts of dicotyledonous herbs). Given the different morphological characteristics among these four types of buds, we dissected the shoot and bulb bases to count tiller buds and bulb buds but counted rhizome and dicot buds without dissection. We also recorded the number of aboveground shoots (tillers or ramet) of each species within each quadrat. We sorted plant species into rhizomatous (with rhizome buds), bunchgrasses (with tiller buds and rhizome buds) and forbs (with bulb buds and dicot buds).
Metrics and statistical analysis
Given that > 90% of the total bud and shoot densities come from grasses in our studied grasslands (Figure S2), we only focused on grass responses (i.e., rhizomatous and bunchgrasses). We regarded the number of buds and shoots recorded in each quadrat as the measures of bud and shoot densities (per square meter), respectively. We calculated the meristem limitation index (MLI) as the ratio of belowground bud density to aboveground shoot density. This index is a measure of the degree of limitation that belowground buds imposed on aboveground population recruitment/regeneration in perennial grasslands (Benson et al. 2004).
We assessed the legacy effect of extreme drought on total belowground bud density, total aboveground shoot density, and total MLI as well as on those of rhizomatous and bunchgrasses across the two grasslands by conducting a mixed-model analysis of variance (ANOVA) with drought treatment and community as fixed factors and block as a random factor. When interactive effects of drought treatment and community were marginally significant, we separately applied the mixed models for each community (rhizomatous or bunchgrass community) with drought treatment as a fixed factor and block as a random factor.
We conducted Shapiro–Wilk and Levene’s tests to confirm all data were normally distributed and had equal variance, respectively, before statistical analysis. Hence, we used original data without transformation in our statistical analyses. We conducted all statistical analyses using the nlme package in R (R i386 3.1.1).
Results
Treatment effectiveness
The long-term (1982–2014) mean annual precipitation (MAP) for this region is 346 mm, 71% of which (i.e., 246 mm) falls during the growing season (May through August; Figure S1). The average annual precipitation and growing season precipitation during the four-year experimental period (2015–2018) were 312 mm and 199 mm, respectively (Figure S1). We effectively imposed an extreme drought during this 4-year period (i.e., precipitation close to the 10th percentile of the historical record) based on the estimated long-term normal distribution of precipitation in each grassland (Figure S1). For control treatments, growing season precipitation was near average in all years (approx. 50th percentile of historic amounts; Figure S1).
Community composition
Under the ambient precipitation conditions, grasses covered 78 and 81% of total community productivity in the rhizomatous and bunchgrass communities, respectively (Table 1). The rhizomatous and bunchgrasses covered 49 and 30% of total community productivity in the rhizomatous grass community, respectively, and covered 28 and 53% in the bunchgrass community (Table 1). Species richness of rhizomatous and bunchgrass communities was similar (i.e., 16 and 17 plant species, respectively) under the ambient precipitation conditions (Table 1). Grasses (i.e., rhizomatous and bunchgrasses) accounted for 91% of total bud density and 96% of total shoot density in the two grassland communities (Figure S2).
Bud bank dynamics
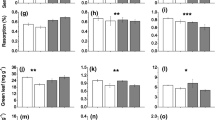
There was strong evidence that total bud density and MLI were higher in the rhizomatous grass community compared to the bunchgrass community under ambient conditions (F = 27.5, P < 0.001 and F = 30.79, P < 0.001, respectively), while total shoot density was similar between the two plant communities (Table 2 and Fig. 1). There was weak evidence that the drought had a negative legacy effect on total belowground bud densities in each grassland community (F = 3.61, P < 0.1); MLI was unaffected (Table 2 and Fig. 1). The interactive effects of drought treatment and community type were marginally significant for the aboveground shoot density (F = 3.82, P < 0.1), indicating a differential drought resilience of total shoot density between the two communities (Table 2). Specifically, there was moderate evidence for drought reducing the total shoot density of the bunchgrass community, but this was not the case in the rhizomatous grass community (Fig. 1).
Effects of drought legacy and community type on (a) total belowground bud density, (b) total aboveground shoot density and (c) the meristem limitation index (MLI) in two plant communities with different dominant species, rhizomatous grass community (RGC) and bunchgrass community (BGC). Each point represents the means with error bars indicating standard errors calculated from replicate plots for each treatment. Drought legacy effect, defined as drought-induced alterations in resources or ecosystem properties that continue to affect ecosystem function after drought, was estimated as the differences in bud or shoot densities between control and treatment plots. Different letters indicate significant differences between rhizomatous and bunchgrass communities at P < 0.05. Statistical significance of drought legacy effect is depicted *P < 0.1 and **P < 0.05. Con, control (i.e., the ambient precipitation); DRec, recovery following the drought treatment (i.e., 4-year reduction of 66% growing season precipitation)
We did not observe a legacy effect of drought on bud densities or MLI of rhizomatous or bunchgrasses separately across the two grassland communities (Table 2; Fig. 2). However, there was moderate evidence of a negative legacy effect on shoot density of bunchgrasses in the bunchgrass community (Fig. 2). The bud densities of both rhizomatous and bunchgrasses were significantly lower in the rhizomatous vs. bunchgrass community (F = 11.78, P < 0.01 and F = 18.00, P < 0.001, respectively), while no difference was observed for the shoot densities of bunchgrasses between the two communities (Fig. 2). Accordingly, the MLI in the rhizomatous grass community was significantly higher than that in the bunchgrass community (F = 22.54, P < 0.001; Fig. 2). Furthermore, we found no evidence that bud density differed between bunchgrass and rhizomatous grass, yet strong evidence that the shoot density of bunchgrasses was significantly higher (Figure S3) than that of rhizomatous grasses, which led to a higher MLI of rhizomatous grasses compared with bunchgrasses (Figure S3).
Effects of drought legacy and community type on (a–b) belowground bud densities, (c–d) aboveground shoot densities and (e–f) the meristem limitation index (MLI) of two dominant functional groups (rhizomatous vs bunchgrass) in two grassland communities with different dominant species, rhizomatous grass community (RGC) and bunchgrass community (BGC). Each point represents the means with error bars indicating standard errors calculated from replicate plots for each treatment. See Fig. 1 legend for the definition of drought legacy effect. Different letters indicate significant differences between rhizomatous and bunchgrass communities at P < 0.05. Statistical significance of drought legacy effect is depicted *P < 0.1 and **P < 0.05. Con, control (i.e., the ambient precipitation); DRec, recovery following the drought treatment (i.e., 4-year reduction of 66% growing season precipitation)
Discussion
Grassland recovery following extreme drought is critical for the long-term sustainability of ecosystem services under climate change and is likely limited by the drought resilience of belowground bud banks (Stampfli and Zeiter 2004; VanderWeide et al. 2014). We measured the density of belowground buds and aboveground shoots in two semi-arid grassland communities one year after a long-term (four-year) extreme drought (66% reduction in ambient precipitation). At both communities, we observed reduced belowground bud density in plots that had previously experienced drought. The observed negative legacy effect contradicts previous work in more mesic grasslands where bud density was unchanged following short-term drought of a similar intensity (Carder et al. 2012; VanderWeide et al. 2014). A negative drought legacy effect was also observed for shoot density, but only in the community dominated by bunchgrasses. We did not detect a legacy effect of extreme drought on MLI (i.e., bud to shoot ratio) in either community, likely due to the coinciding response of above- and belowground processes. This suggests that while extreme drought reduced bud and shoot densities, the resilience potential of these communities to further changes in precipitation was unaltered (Knapp and Smith 2001; Dalgleish and Hartnett 2006).
Overall, these results support our hypothesis that drought has negative lagged impacts on bud banks. Interestingly, these findings differ from those of studies conducted in more mesic grasslands where belowground bud densities were unaffected by one- (Carlter et al. 2012) or 2-year droughts of similar intensity (VanderWeide et al. 2014). This may simply reflect the longer duration of our drought treatments, which would suggest bud banks are resilient to short-term water limitation. Short-term droughts likely influence bud bank dynamics through intraspecific variation in bud density, while longer-term droughts impact bud density via species turnover (Smith 2011; Beier et al. 2012). Alternatively, such differential drought sensitivity between the current drought experiment and those of Carlter et al. (2012) and VanderWeide et al. (2014) may also be due to the differences in precipitation regimes among these studies (Huxman et al. 2004; Knapp et al. 2015). Previous studies have shown that belowground bud density decreases with increased aridity along the precipitation gradient (Dalgleish and Hartnett 2006) and that ecosystem functions of more arid grasslands are more sensitive to extreme drought than that of mesic grasslands (Knapp et al. 2015; Luo et al. 2021). Indeed, we observed that the sensitivity of belowground bud density to drought in our semi-arid grasslands with MAP of 330 mm was higher than that reported for more mesic grasslands with MAP of 880 mm (VanderWeide et al. 2014). This finding, however, contrasts with the prediction that vegetation chronically exposed to water limitations should be stress tolerant and more resistant to drought (Grime et al. 2000). Thus, more studies on short- and long-term responses of belowground buds to extreme drought across multiple grasslands spanning a broad aridity gradient are needed.
We noted significant differences between the two plant communities in belowground bud dynamics. Specifically, the belowground bud density and MLI in the rhizomatous grass community were significantly higher than those in the bunchgrass community (Fig. 1). Similarly, the negative legacy effect of extreme drought on the total shoot density was larger in the bunchgrass than the rhizomatous grass community (Fig. 1). In line with these results, the shoot density of the rhizomatous grasses growing in the bunchgrass-dominated community showed higher drought resilience than the co-occurring bunchgrasses (Fig. 2). This might be attributed to the differences in bud traits as rhizomatous grasses have relatively shallower buds compared to bunchgrasses (Klimešová and Klimeš 2008).
Our results indicate that bunchgrasses have fewer buds per stem, suggesting a greater meristem limitation for bunchgrasses than rhizomatous grasses (Figure S3). This highlights the importance of functional groups for understanding the differential capacity of different grasslands to respond to extreme drought (Cart et al. 2012). Tilman et al. (1996) showed that differential responses to environmental fluctuations among species and/or functional groups within plant communities could contribute to the temporal stability of ecosystem functions. Additionally, functional group differences impact the functional diversity of communities during and after drought events, exerting longer-term impacts on plant community structure (Xu et al. 2017). Our results suggest that rhizomatous grasses may persist and dominate in these grassland community for a longer period during and following drought compared to bunchgrasses. Wang et al. (2019) found that short-term summer drought decreased the bud bank density and shoot production of the rhizomatous grass, L. chinensis, and consequently constrained both its current and future productivity, but increased the proportion of buds which developed into rhizomes. Taken together, our data and that of Wang et al. suggest we must consider belowground bud and aboveground shoot demography in predictive models of how drought may alter community dynamics and ecosystem functions.
In conclusion, both belowground bud and aboveground shoot densities were reduced one year following drought, but meristem limitation was unaltered in the studied semi-arid grasslands. The legacy effects of extreme drought on belowground bud banks and the relationship with aboveground vegetation depended on plant community type and plant growth forms. Due to the growth habit of bunchgrasses (the cluster form/phalanx clonal growth), these communities face a greater meristem limitation than rhizomatous grass communities. Belowground bud bank dynamics may constrain community responses to climate change and human disturbances, and consequently has important implications on ecosystem function and services.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author.
References
Bai YF, Han XG, Wu JG, Chen ZZ, Li LH (2004) Ecosystem stability and compensatory effects in the inner mongolia grassland. Nature 431:181–184. https://doi.org/10.1038/nature02850
Beier C, Beierkuhnlein C, Wohlgemuth T et al (2012) Precipitation manipulation experiments-challenges and recommendations for the future. Ecol Lett 15:899–911. https://doi.org/10.1111/j.1461-0248.2012.01793.x
Benson EJ, Hartnett DC (2006) The role of seed and vegetative reproduction in plant recruitment and demography in tallgrass prairie. Plant Ecol 187:163–178. https://doi.org/10.1007/s11258-005-0975-y
Benson EJ, Hartnett DC, Mann KH (2004) Belowground bud banks and meristem limitation in tallgrass prairie plant populations. Am J Bot 91:416–421. https://doi.org/10.3732/ajb.91.3.416
Carter DL, Vanderweide B, Blair JM (2012) Drought-mediated stem and below-ground bud dynamics in restored grasslands. Appl Veg Sci 15:470–478. https://doi.org/10.1111/j.1654-109X.2012.01200.x
Dalgleish HJ, Hartnett DC (2006) Below-ground bud banks increase along a precipitation gradient of the North American Great Plains: a test of the meristem limitation hypothesis. New Phytol 171:81–89. https://doi.org/10.1111/j.1469-8137.2006.01739.x
Dalgleish HJ, Hartnett DC (2009) The effects of fire frequency and grazing on tallgrass prairie productivity and plant composition are mediated through bud bank demography. Plant Ecol 201:411–420. https://doi.org/10.1007/s11258-008-9562-3
Dukes JS, Classen AT, Wan S, Langley JA (2014) Using results from global change experiments to inform land model development and calibration. New Phytol 204:744–746. https://doi.org/10.1111/nph.13083
Evans SE, Byrne KM, Lauenroth WK, Burke IC (2011) Defining the limit to resistance in a drought-tolerant grassland: long-term severe drought significantly reduces the dominant species and increases ruderals. J Ecol 99:1500–1507. https://doi.org/10.1111/j.1365-2745.2011.01864.x
Griffin-Nolan RJ, Carroll CJ, Denton EM et al (2018) Legacy effects of a regional drought on aboveground net primary production in six central US grasslands. Plant Ecol 219(5):505–515. https://doi.org/10.1007/s11258-018-0813-7
Grime JP, Brown VK, Thompson K et al (2000) The response of two contrasting limestone grasslands to simulated climate change. Science 289:762–765. https://doi.org/10.1126/science.289.5480.762
Harper JL (1977) Population biology of plants. Academic Press, New York
Hoover DL, Knapp AK, Smith MD (2014) Resistance and resilience of a grassland ecosystem to climate extremes. Ecology 95:2646–2656. https://doi.org/10.1890/13-2186.1
Huang JH, Guan YX, Wang G, Guo R (2016) Accelerated dryland expansion under climate change. Nat Clim Change 6:166–171. https://doi.org/10.1038/nclimate2837
Huxman TE, Smith MD, Fay PA, Knapp AK et al (2004) Convergence across biomes to a common rain-use efficiency. Nature 429:651–654. https://doi.org/10.1038/nature02561
Kang L, Han XG, Zhang ZB, Sun OJ (2007) Grassland ecosystems in China: review of current knowledge and research advancement. Philos t r Soc B 362(1482):997–1008. https://doi.org/10.1098/rstb.2007.2029
Klimešová J, Klimeš L (2007) Bud banks and their role in vegetative regeneration-a literature review and proposal for simple classification and assessment. Perspect Plant Ecol 8(3):115–129. https://doi.org/10.1016/j.ppees.2006.10.002
Klimešová J, Klimeš L (2008) Clonal growth diversity and bud banks of plants in the Czech flora: an evaluation using the CLO-PLA3 database. Preslia 80(3):255–275. https://doi.org/10.1055/s-2008-1081305
Knapp AK, Smith MD (2001) Variation among biomes in temporal dynamics of aboveground primary production. Science 291(5503):481–484. https://doi.org/10.1126/science.291.5503.481
Knapp AK, Carroll CJW, Denton EM, La Pierre KJ, Collins SL, Smith MD (2015) Differential sensitivity to regional-scale drought in six central US grasslands. Oecologia 177(4):949–957. https://doi.org/10.1007/s00442-015-3233-6
Kühn N, Tovar Ingar C, Carretero J, Vandvik V, Enquist B, Willis K (2021) Globally important plant functional traits for coping with climate change. Front Biogeogr 13(04):e53774. https://doi.org/10.21425/F5FBG53774
Luo WT, Xu C, Ma W et al (2018) Effects of extreme drought on plant nutrient uptake and resorption in rhizomatous vs bunchgrass-dominated grasslands. Oecologia 188(2):633–643. https://doi.org/10.1007/s00442-018-4232-1
Luo WT, Griffin-Nolan RJ, Ma W et al (2021) Plant traits and soil fertility mediate productivity losses under extreme drought in C3 grasslands. Ecology 102(10):e03465. https://doi.org/10.1002/ecy.3465
Ma Q, Qian JQ, Tian L, Liu ZM (2019) Responses of belowground bud bank to disturbance and stress in the sand dune ecosystem. Ecol Indic 106:105521. https://doi.org/10.1016/j.ecolind.2019.105521
Ott JP, Klimešová J, Hartnett DC (2019) The ecology and significance of below-ground bud banks in plants. Ann Bot 123(7):1099–1118. https://doi.org/10.1093/aob/mcz051
Pate JS, Dixon KW (1982) Tuberous, cormous and bulbous plants biology of an adaptive strategy in Western Australia. University of Western Australia Press, Nedlands
Qian JQ, Wang ZW, Klimesova J et al (2017a) Differences in below-ground bud bank density and composition along a climatic gradient in the temperate steppe of northern China. Ann Bot 120(5):755–764. https://doi.org/10.1093/aob/mcx072
Qian JQ, Wang ZW, Liu ZM, Busso CA (2017b) Belowground bud bank responses to grazing intensity in the Inner-Mongolia steppe. China Land Degrad Dev 28(3):822–832. https://doi.org/10.1002/ldr.2300
Qian JQ, Wang ZW, Klimesova J, Lü XT, Zhang CY (2021) Belowground bud bank and its relationship with aboveground vegetation under watering and nitrogen addition in temperate semiarid steppe. Ecol Indic 125:107520. https://doi.org/10.1016/j.ecolind.2021.107520
Sala OE, Gherardi LA, Reichmann L, Jobbagy E, Peters D (2012) Legacies of precipitation fluctuations on primary production: theory and data synthesis. Philos t r Soc B 367(1606):3135–3144. https://doi.org/10.1098/rstb.2011.0347
Smith MD (2011) An ecological perspective on extreme climatic events: a synthetic definition and framework to guide future research. J Ecol 99(3):656–663. https://doi.org/10.1111/j.1365-2745.2011.01798.x
Song B, Niu S, Wan S (2016) Precipitation regulates plant gas exchange and its long-term response to climate change in a temperate grassland. J Plant Ecol 9(5):531–541. https://doi.org/10.1093/jpe/rtw010
Stampfli A, Zeiter M (2004) Plant regeneration directs changes in grassland composition after extreme drought: a 13-year study in southern Switzerland. J Ecol 92(4):568–576. https://doi.org/10.1111/j.0022-0477.2004.00900.x
Stuart-Haëntjens E, De Boeck HJ, Lemoine NP et al (2018) Mean annual precipitation predicts primary production resistance and resilience to extreme drought. Sci Total Environ 636:360–366. https://doi.org/10.1016/j.scitotenv.2018.04.290
Tilman D, Wedin D, Knops J (1996) Productivity and sustainability influenced by biodiversity in grassland ecosystems. Nature 379(6567):718–720. https://doi.org/10.1016/j.scitotenv.2018.04.290
VanderWeide BL, Hartnett DC (2015) Belowground bud bank response to grazing under severe short-term drought. Oecologia 178(3):795–806. https://doi.org/10.1007/s00442-015-3249-y
VanderWeide BL, Hartnett DC, Carter DL (2014) Belowground bud banks of tallgrass prairie are insensitive to multi-year, growing-season drought. Ecosphere 5(8):103. https://doi.org/10.1890/ES14-00058.1
Wang JF, Shi YJ, Ao YN et al (2019) Summer drought decreases Leymus chinensis productivity through constraining the bud, tiller and shoot production. J Agron Crop Sci 205(6):554–561. https://doi.org/10.1111/jac.12354
Wilcox KR, Koerner SE, Hoover DL et al (2020) Rapid recovery of ecosystem function following extreme drought in a South African savanna grassland. Ecology 101(4):e02983. https://doi.org/10.1002/ecy.2983
Williams AP, Cook ER, Smerdon JE et al (2020) Large contribution from anthropogenic warming to an emerging North American megadrought. Science 370(6516):314–318. https://doi.org/10.1126/science.abf3676
Xu X, Polley HW, Hofmockel K, Wilsey BJ (2017) Species composition but not diversity explains recovery from the 2011 drought in Texas grasslands. Ecosphere 8(3):e01704. https://doi.org/10.1002/ecs2.1704
Xu C, Ke Y, Zhou W et al (2021) Resistance and resilience of a semi-arid grassland to multi-year extreme drought. Ecol Indic 131:108139. https://doi.org/10.1016/j.ecolind.2021.108139
Yahdjian L, Sala OE (2006) Vegetation structure constrains primary production response to water availability in the Patagonian steppe. Ecology 87(4):952–962. https://doi.org/10.1890/0012-9658(2006)87[952:VSCPPR]2.0.CO;2
Zhang Y, Keenan TF, Zhou S (2021) Exacerbated drought impacts on global ecosystems due to structural overshoot. Nat Ecol Evol 5(11):1490–1498. https://doi.org/10.1038/s41559-021-01551-8
Acknowledgements
This study was financially supported by the National Natural Science Foundation of China (41877542, 32171549 and 31971465), Liaoning Provincial Science and Technology Plan Projects (2020JH1/10300006), Strategic Priority Research Program of Chinese Academy of Sciences (XDA23080401), and Youth Innovation Promotion Association CAS (2020199).
Author information
Authors and Affiliations
Contributions
JQ and WL conceived and designed the experiments. ZG, NT, LS, CX, ZZ and QY performed the experiments. WL, JQ and ZZ analyzed the data. JQ, TOM RJG and WL wrote the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Consent for publication
All patients provided written informed consent to publish the data contained within this article.
Additional information
Communicated by Brian J. Wilsey.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Qian, J., Guo, Z., Muraina, T.O. et al. Legacy effects of a multi-year extreme drought on belowground bud banks in rhizomatous vs bunchgrass-dominated grasslands. Oecologia 198, 763–771 (2022). https://doi.org/10.1007/s00442-022-05133-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-022-05133-8