Abstract
Recovery of ecosystem properties following removal of invasive plants likely varies with characteristics of the plant and the relative soil quality at a given site. These factors may influence the occurrence of soil legacies and secondary invasions, hindering the effectiveness of restoration strategies. We assessed the potential for ecosystem recovery following removal of N-fixing Scotch broom for 4 years at two sites that contrasted strongly in soil quality in western Washington and Oregon, USA. Comparisons were made among plots, where Scotch broom was never present (uninvaded), retained, or removed. Scotch broom removal increased PAR and soil temperature but had limited effects on soil moisture. Concentrations of soil Ca, Mg, K, and P were significantly lower with Scotch broom removal, with the effect being most pronounced at the low-quality site. NMS ordinations indicated that the treatments differed in vegetation composition, with limited recovery following broom removal. Non-native and native species varied inversely in their abundance responses, where non-native species abundance was greatest in the removal treatment, intermediate in the retained treatment, and lowest in the uninvaded treatment, indicating occurrence of a secondary invasion following removal. As with the soil response, effects were more pronounced at the low-quality site. Our findings indicate that Scotch broom removal exacerbates negative effects on soil chemistry and plant communities, with little evidence of recovery over our study period. These findings highlight the importance of controlling Scotch broom invasions immediately after the species establishes, especially on low-quality sites that are more susceptible to Scotch broom invasion.
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Efforts to understand and mitigate the impacts of invasive species are ubiquitous globally in response to unprecedented changes in ecosystem conditions associated with climate change, globalization, and land management. Against this backdrop is a scarcity of resources for mitigative actions, which limits the capacity for managers to address impacts to ecosystem functions across a given land base (Simberloff et al. 2005). There is a continued need for information that can guide managers on determining, where their efforts are most likely to be effective and achieve ecosystem recovery (McGeoch et al. 2016). In this context, we define ecosystem recovery as a return to pre-invasion conditions inferred through either direct observation or via comparison to areas never invaded (cf. Prior et al. 2018).
Invasive species can alter soil C and nutrient pools, which may alter resource availability to plants via direct (e.g., nutrient availability) and indirect means (e.g., C-induced changes in microbial activity). Soil N enrichment associated with invasions by N-fixing species is particularly problematic because it can facilitate the establishment and success of other non-native invaders (Walker and Vitousek 1991, Weidenhamer and Callaway 2010, Maron and Jefferies 1999, Carter et al. 2019c). Given this, it is likely that increased N pools will hamper recovery of site productivity and native plant community composition even after removal of the invader (i.e., soil legacies, Corbin and Antonio 2012). Legacy effects of invasive N-fixing species on other soil nutrients are also possible. For example, some studies have shown lower soil available P in the presence of invasive N-fixing species (Caldwell 2006; Shaben and Meyers 2010; Bateman and Vitousek 2018) which can vary depending on site or soil quality (Slesak et al. 2016). Such changes in soil nutrient availability and soil C may alter plant community composition and its recovery, but the direction and magnitude of change is likely variable depending on characteristics of the invasive plant and site factors (e.g., soils). For example, the effect of N-fixing invasive species on N availability and cycling can last for a decade or longer following removal (Bateman and Vitousek 2018; Maron and Jefferies 1999).
Removal of invasive plants may result in recovery of native biodiversity, but positive outcomes do not always occur (Prior et al. 2018; Kettenring and Adams 2011) and control efforts can often lead to secondary invasions following removal of the dominant invasive (Kuebbing and Nunez 2015; Reid et al. 2009; Yelenik et al. 2004). These secondary invasions are most commonly associated with changes in resource availability combined with changes in microclimate that allow initially minor invasive species to become more dominant. For example, Maron and Jefferies (1999) showed how mortality and loss of the N-fixing yellow bush lupine (Lupinus arboreus Sims) increased N and light availability, leading to invasion by non-native grasses that dominated the plant community. Other studies have shown that increased shade and litter accumulation associated with invasive species can modify germination and establishment of native species (Egunjobi 1971, Waterhouse 1988, Wearne and Morgan 2004). The likelihood of secondary invasions likely increases when the removed invader strongly influenced resource availability and microclimate, such as situations, where its cover was extensive or particularly dense.
Although ecosystem recovery can be variable following invasive species control, it is likely to depend on site (soil) quality (e.g., texture and its influence on water and nutrient availability). For example, Dassonwille et al. (2008) found that invasive plant feedbacks to soil depended on initial soil quality, being generally positive (increased nutrient pools) at sites with low soil nutrient status but negative at sites with initially high nutrient status. Other work has shown that soil quality can also influence the occurrence and spread of invasive species (Williamson and Harrison 2002; Czortek et al. 2020; Lázaro-Lobo et al. 2021). Carter et al. (2021) showed that site susceptibility to invasion was dependent on soil texture and its effect on competitive ability of the invasive and native plants. Such differences in the magnitude and direction of invasion response are likely to lead to different recovery trajectories, but it is not clear how these differences will vary with initial soil quality. Understanding the effects of soil quality on ecosystem recovery is important for the development of restoration strategies across a range of site conditions.
Scotch broom (Cytisus scoparius (L.) Link) is an N-fixing invasive species of major ecological concern capable of dominating sites (Bossard and Rejmanek 1994; Richardson et al. 2002) and altering ecosystem function (Parker et al. 1997). The species is currently increasing its range globally and is a major problem for production forestry in New Zealand, Australia, Canada, and the United States (Potter et al. 2009). Scotch broom is an aggressive invader in early successional coast Douglas-fir (Pseudotsuga menziesii (Mirb.) Franco var. menziesii) forests of the Pacific Northwest (PNW), USA, where it can impact soil properties (Slesak et al. 2016; Grove et al. 2015; Caldwell 2006), alter vegetation communities (Carter et al. 2019c; Shaben and Meyers 2010), and reduce site productivity (Harrington et al. 2018). Because of this, it is common for managers to aggressively control Scotch broom when present, but the potential for and patterns of ecosystem recovery following removal of this invasive species are unknown. Given its ability to fix moderate quantities of N (20–100 kg ha−1 yr−1, Carter et al. 2019a, Watt et al. 2003a) and its propensity to form dense and extensive thickets (Waterhouse 1988), the potential for recovery of soils and vegetation following broom removal may be lower compared to other invasive species.
Here, we report on changes in soil chemical properties, microclimate, and plant community structure over a 4-year period following Scotch broom removal at two Douglas-fir sites that contrasted in soil quality. In a prior assessment at these sites comparing areas with broom present to areas, where it was absent, Slesak et al. (2016) found that Scotch broom presence was associated with higher total C and N, lower nutrient cations (particularly K), and lower intermediately available P, but the magnitude of these effects varied and was generally more pronounced at the higher quality site. In this study, our primary objective was to assess the potential for soil and plant community recovery over a period of 4 years following removal of Scotch broom, and how these responses vary with initial soil quality. Based on previous work evaluating recovery following removal of N-fixing invasive species, we expected limited recovery of soil properties and plant communities to uninvaded conditions. We also expected that levels of recovery would vary depending on initial site (soil) quality, being greatest at the higher quality site, where effects on soil properties were also greatest (Slesak et al. 2016).
Methods
Site descriptions
The study was conducted at two Douglas-fir sites affiliated with the North American Long-Term Soil Productivity (LTSP) study located near Matlock, Washington (WA) and Molalla, Oregon (OR) USA (Harrington and Schoenholtz 2010) (Table 1). Soil characteristics and stand productivity varied strongly between sites (Harrington et al. 2020), with the WA site having lower site quality and the OR site generally having high site quality as evidenced by large differences in limiting resources including available water and total soil N (Table 1). These differences in soil properties are associated with large differences in productivity; at age 15, the high-quality OR site had more than twice the Douglas-fir stand volume found at the lower quality WA site (Harrington et al. 2020). Soils at the WA site are classified as sandy-skeletal, mixed, mesic, Dystric Xerorthents formed in glacial outwash with slopes ranging from 0 to 3% (Soil Survey Staff, USDA-NRCS 2021). Soils at the OR site are classified as fine-loamy, isotic, mesic Andic Dystrudepts formed in basic agglomerate residuum with slopes ranging from 2 to 40% (Soil Survey Staff, USDA-NRCS 2021). The regional climate is Mediterranean, characterized by cool, wet winters and warm, dry summers with periods of prolonged drought. Potential natural vegetation includes the western hemlock (Tsuga heterophylla (Raf.) Sarg.)/salal (Gaultheria shallon Pursh) plant association at the WA site (Henderson et al. 1989) and the western hemlock/Oregon-grape (Mahonia nervosa (Pursh) Nutt.)/swordfern (Polystichum munitum (Kaulf.) Presl) and western hemlock/Oregon grape-salal plant associations at the OR site (Halverson et al. 1986).
Experimental design
Each of the sites was clearcut harvested in spring 2003 and planted with Douglas-fir seedlings in spring 2004. Following forest harvesting, Scotch broom began to proliferate across each of the sites. In the fall of 2013, 3-m radius circular plots were replicated 10 times at each site for each of the following Scotch broom treatments: (i) Removed: Scotch broom present since harvest and then removed in 2013 (see details below), (ii) Retained: Scotch broom present since harvest or (iii) Uninvaded: Scotch broom absent since harvest. We were able to locate areas, where Scotch broom was present or uninvaded with high confidence due to our past work at these sites (Harrington et al. 2010). Because we could not randomly assign the presence or absence of Scotch broom to a given replication, we identified double the number of candidate replications for each treatment and then randomly selected half for inclusion in this study. This approach allowed for unbiased estimation of soil conditions within each of the Scotch broom conditions. For removal treatments, Scotch broom was cut near ground surface, aboveground biomass was removed from the plots, and stumps were sprayed with a 20% solution of Garlon®4 (triclopyr ester) herbicide in an oil carrier to ensure complete mortality. Thereafter, any Scotch broom germinants in the plots were removed by hand throughout the duration of the study. Scotch broom basal area (at 15-cm above ground), stem density, average height, and crown cover were measured in both the removed and retained treatment plots prior to treatment application (Table 1).
Microclimate measurements
Soil moisture was monitored in each of the Scotch broom removal and retained treatment plots with soil moisture sensors (model EC-5, Meter Corporation, Pullman WA, USA) attached to dataloggers (model EM-5, Meter Corporation, Pullman WA, USA). Sensors were installed on a randomly selected azimuth 50 cm from the center of the plot at a depth of 30 cm. Soil moisture readings were logged every 4 h throughout the duration of the study. Soil temperature was also measured in each of the Scotch broom removal and retained treatment plots with iButton dataloggers (model DS1921G, Maxim Integrated Technologies, Inc., Sunnyvale CA, USA) that were installed 50 cm from plot center (opposite from the soil moisture sensor) and at a depth of 5 cm. The iButtons were programmed to log temperature at 2-h intervals throughout the duration of the study. Light availability was assessed with a linear PAR sensor (Accupar LP-80 ceptometer, Meter Corporation, Pullman WA, USA) at each of the plots once during each year from 2015 to 2017. Readings were taken around the summer solstice between 11:00 and 14:00 Pacific Daylight Time at six locations in each plot on a 1-m grid centered on plot center. Readings were taken at 0.5 m height and expressed relative to an above canopy reading taken immediately prior to below canopy readings.
Soil collection, processing, and analytical procedures
Soil samples were collected in the fall of 2013, 2015, and 2017 at both sites using bucket augers. At each treatment replication, three samples were collected at 0–15 and 15–30-cm depth increments, composited in a bucket by depth increment, and thoroughly mixed; a subsample was placed in a sealed plastic bag for transport. Soil samples were returned to the laboratory, air-dried, and sieved to pass a 2-mm mesh. Total soil C and N were measured on a 1-g subsample that was ground with a mortar and pestle to pass a 0.25-mm mesh, followed by dry combustion using a LECO Dumas combustion technique on a Fisons NA1500 NCS Elemental Analyzer (ThermoQuest Italia, Milan, Italy). The Mehlich method was used to extract Ca, Mg, and K, and extract concentrations were measured with inductively coupled plasma spectroscopy (Varian Vista MPX, Varian, Palo Alto, CA, USA). Available soil P was estimated using the Bray extraction followed by calorimetric estimation on a spectrophotometer (Spectronic 20 Genesys, Model 4001, Thermo Electron Corporation, Waltham, MA, USA).
Vegetation measurements
Vegetation measurements were conducted once per year in each 3-m radius plot in June–July of 2014–2017. For each measurement, visual estimates of percent cover were made for each species in the entire plot (nearest 1% for covers < 5% and nearest 5% for covers between 5 and 100%). In this method, cover for a single species is constrained to 100%, but the sum of covers for multiple species on a given plot can exceed 100% due to crown overlap. The same observer performed all vegetation measurements to maintain consistency in visual estimates over time.
Analysis
Data from each site were analyzed independently to assess treatment effects on response variables. Treatment effects on soil chemistry (total C and N, extractable Ca, Mg, K, and P), microclimate (soil moisture, temperature, and PAR), and the richness and abundance (cover) of native and non-native species in each plot were assessed with mixed effects repeated measures analysis of variance (ANOVA). Plot within treatment was modeled as a random effect and treatment, time, and their interaction were modeled as fixed effects. For all models, we used a first order autoregressive covariance matrix to account for serial correlation among successive measurements. For soil moisture and temperature, data were first averaged by day and then by month; models were run independently for each year to conform with the chosen covariance matrix (i.e., avoid high correlation for a given month across years). Soil chemistry responses were analyzed separately for each depth increment to simplify interpretation of model output. We used the USDA Plants Database (The Plants Database 2018) for our taxonomic treatment and to code each plant species according to their life form and whether they were native or non-native. For each plot, we calculated total cover for native and non-native species groups and used ANOVA to compare treatment effects between the two groups. We also analyzed differences in cover by life form among treatments. All mixed effects analysis was conducted in SAS 9.4 using an alpha of 0.05 (SAS Institute, Inc., 2013). When F statistics indicated significant treatment or treatment-by-time effects, we used Tukey’s adjustment (for soil chemistry and plant responses) or slicing (for microclimate variables) to determine differences among treatments.
Nonmetric multidimensional scaling (NMS) ordination was used to graphically assess gradients in plant community composition across treatments and explore relationships with soil chemical properties. Rare species found on < 5% of plots were deleted prior to analysis given their disproportionate effect on NMS solutions. The community matrix consisted of measurements from 2014 to 2017 for each site. Ordinations were run in PC-ORD (Version 7.08, McCune and Mefford 2018) on autopilot mode using Sørenson (Bray–Curtis) distance measures with 500 iterations. Community composition was displayed graphically by taking the mean of plot values in each treatment and year and plotting them against the ordination axes. Treatment effects on plant communities were assessed with permutational multivariate analysis of variance (PERMANOVA) conducted in PC-ORD. PERMANOVA is a non-parametric test used to compare groups of objects and their dispersal based on a given dissimilarity measure around a central point. When significant treatment effects were observed, post hoc comparisons among treatments and years were conducted using a Bonferroni adjustment.
Relationships between the ordination axes and soil chemical properties were made using biplot overlays using an r2 cutoff of 0.2 (McCune et al. 2002). For these analyses, we explored relationships between pretreatment soil properties and vegetation composition in each of the measurement years and determined relationships across years using soil data collected from the associated year (i.e., 2014 (using pretreatment), 2015, and 2017). We also evaluated the effect of SWC and PAR on community composition with Pearson correlation analysis between the ordination axis values for each plot and year and the associated plot values of PAR and mean growing season SWC (May–Sept.).
Results
Microclimate responses
There was significant interaction between treatment and year for PAR at the low-quality site (Table S1). PAR in the Scotch broom removed and uninvaded treatments was 1.5 to 3.3 times greater than in the retained treatment in each year, except for 2017 when the uninvaded treatment was not different from the retained treatment (Fig. 1). At the high-quality site, there was a main effect of treatment and no interaction between treatment and year on PAR (Table S1). At that site, PAR in the removed treatment was 3.2 and 4.6 times greater than that in the retained and uninvaded treatments, respectively.
Soil temperature results largely mirrored those for PAR. At both sites and in each year of the study, there was a significant interaction between treatment and measurement month on soil temperature (p < 0.025, Table S2). At the low-quality site, the Scotch broom retained treatment had significantly lower soil temperature than the other treatments during growing season months (May–Sept, Fig. S2). Mean monthly differences averaged between 2 and 3 ℃ depending on month and year. At the high-quality site, the Scotch broom removed treatment had significantly greater soil temperature than the other treatments during growing season months. Mean monthly differences at that site ranged from 0.5 to 1.5 ℃.
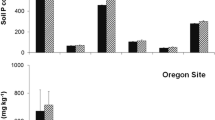
Treatment effects on soil water were small and limited to the first year after treatment application and only at the low-quality site (Fig. 2, Table S3). At that site, there was a main effect of treatment (p = 0.021) on soil water content, but multiple comparisons failed to identify differences among treatments. At the high-quality site, there was no effect of treatment on soil water content in the first year (p = 0.087) or any year thereafter (Table S3). At both sites, mean soil water content was higher in the removed treatment than in the retained treatment in the first year, but differences were only significant at the low-quality site.
Average monthly soil moisture content at 30 cm depth in Scotch broom removal and retention treatments during 2014–2017 at the a low-quality and b high-quality sites. Differences were not significantly different between treatments at any time period. Note that soil moisture content was not measured in the uninvaded treatment
Soil responses
Main effects of treatment on soil chemical properties were found at both sites but were more common at the low-quality site (Table S4). At the high-quality site at 0–15 cm depth, total C concentration was higher, where broom was retained compared to where it was uninvaded, but there was no difference between broom removal and uninvaded treatments. There were no other treatment effects on total C or total N at either depth increment or site. At the low-quality site, treatment effects on extractable P, Mg, K, and Ca were consistent and manifested as lower concentrations in the broom removal treatment relative to the broom retained and uninvaded treatments (Table 2). This pattern was consistent across depth increments (except for P at 15–30 cm), and lower concentrations in the removal treatment ranged from ~ 25% (relative to the uninvaded treatment) for P to ~ 40% for K. For K at 0–15 cm depth, there was a significant interaction between treatment and year which was caused by no difference at time 0 among treatments, followed by the same pattern that occurred for main effects in later years (i.e., broom removed < broom retained and uninvaded treatments). Similar trends were observed at the high-quality site, but the main effect of treatment was only significant for Ca at 15–30 cm depth increment (Table 3).
Vegetation response
There was a total of 70 and 76 plant species observed at the high- and low-quality sites, respectively, over the study period. A complete list of species, the number of times they were observed, and their associated cover by site and treatment can be found in the Supplemental material (Table S7). There were main effects of treatment on the richness and cover of non-native and native plant species at both sites (P < 0.001, Table S5) and only one interaction between treatment and year (see below). In general, non-native and native species varied inversely in their abundance responses to treatment, and the responses were consistent at both sites. At both sites, non-native species richness and cover were lowest in the uninvaded treatment, intermediate in the broom retained treatment, and highest in the broom removal treatment (Table 4). Note that non-native richness and cover estimates did not include broom. In the case of non-native species cover at the low-quality site, there was a significant interaction between treatment and year, where the broom removal treatment started out similar to the uninvaded treatment, but then non-native cover steadily increased until it exceeded the broom retained treatment by the end of the study (Fig. 3). There was no effect of treatment on native species richness at the high-quality site (p = 0.456), but native species richness at the low-quality site and native cover at both sites all responded in the same manner. In those cases, native species richness and cover was significantly greater in the uninvaded treatments, intermediate in broom retained treatments, and lowest in broom removal treatments (Table 4).
The gradients in vegetation community composition at each site were best explained by a three-dimensional NMS ordination solution. There was clear separation among treatments in community composition at both sites, but differences among years within a treatment were limited to the low-quality site (Fig. 4). At the low-quality site, the ordination had a final stress of 13.43 and instability < 0.001. Axis 1 (accounting for 23.1% of variance) was associated with plots dominated by native forbs, ferns, and shrubs (Linnaea borealis L., Gaultheria shallon Pursh, Pteridium aquilinum (L.) Kuhn, Hieracium albiflorum Hook.) in the negative portion to those dominated by the non-native species Scotch broom and velvet grass (Holcus lanatus L.) in the positive portion. Axis 2 (accounting for 17.1% of variance) was associated with plots containing native forbs (Crepis capillaris (L.) Wallr.) and graminoids (Luzula comosa E. Mey.) in the negative portion to those dominated by native shrubs and trees in the positive portion (Amelanchier alnifolia (Nutt.) Nutt. ex M. Roem., Frangula purshiana (DC.) Cooper, Pseudotsuga menziesii (Mirb.) Franco var. menziesii, Symphoricarpos albus (L.) S.F. Blake). At the high-quality site, the ordination had a final stress of 14.89 and instability < 0.001. At that site, Axis 1 (accounting for 33.3% of variance) trends from plots dominated by native trees in negative portions (Frangula purshiana (DC.) Cooper, Pseudotsuga menziesii (Mirb.) Franco var. menziesii) to non-native grasses and forbs in positive portions (Holcus lanatus L., Leucanthemum vulgare Lam., Hypericum perforatum L.). Axis 2 (accounting for 17.2% of variance) trends from plots dominated by Scotch broom in negative portions to those dominated by native vegetation in positive portions (Pteridium aquilinum (L.) Kuhn, Viola sempervirens Greene, Prunus emarginata (Douglas ex Hook.) D. Dietr.). At both sites, composition in the removal treatment tended to move along Axis 1 toward the uninvaded treatment over time (towards higher abundance of native forbs, herbs and shrubs on the positive portion of the axis), possibly indicating limited recovery in that treatment.
PERMANOVA results indicated that compositional differences among Scotch broom treatments were significant at each site (p < 0.001, Table S6). There was a main effect of year at the low-quality site, but no interaction between treatment and year at either site. Pairwise comparisons indicated all treatments were significantly different (p < 0.001) in their community composition at each of the sites (Fig. 4). Correlations between the ordination axis values and soil chemical properties did not exceed an r2 of 0.2 in any year (data not shown). There was no significant correlation between mean growing season SWC and plot ordination scores at either site (p > 0.10). PAR was negatively correlated with Axis 2 at the low-quality site (p < 0.001; r2 = 0.46) and positively correlated with Axis 1 at the high-quality site (p < 0.001; r2 = 0.62).
Discussion
There is a growing body of evidence that removal of invasive species does not always lead to recovery, with delays in the progress of plant communities and soil properties to pre-invasion conditions (D’Antonio et al. 2017; Nsikani et al. 2018). Here, we monitored the response of soil and vegetation for 4 years after the removal of Scotch broom to assess the potential for ecosystem recovery at two sites that contrasted in soil quality. Our results indicate that broom removal can decrease soil C compared to where it is retained and decrease nutrient cations relative to where it was retained or uninvaded. However, Scotch broom removal resulted in a distinctly different community composition compared to where it was retained or uninvaded, probably because removal increased the richness and cover of non-native species and decreased the richness and cover of native species. Taken together, our findings indicate that Scotch broom removal exacerbates negative effects on soil and plant communities, with little evidence of ecosystem recovery over our study period. Effects were consistent between sites but generally more pronounced at the low-quality site, highlighting the role of site quality on responses following Scotch broom removal. We note that physical removal of the broom biomass in our treatment likely influenced the response (see below), and different responses may occur with control efforts, where broom biomass is retained.
Microclimate responses
The significant effect of Scotch broom removal on PAR and soil temperature was a direct result of reduced canopy cover that occurred in that treatment. Scotch broom tends to form dense thickets which shade the soil surface and lower soil temperatures (Waterhouse 1988). Here, canopy removal resulted in relatively large increases in PAR and soil temperature, especially at the low-quality site, where PAR was increased by a factor of three or more. At the high-quality site, effects of Scotch broom removal on PAR and soil temperature were more muted because of uniform cover from the 10- to 14-year-old planted Douglas-fir. Effects of the Douglas-fir canopy are apparent in that there was no difference in PAR or soil temperature between the retained and uninvaded treatments at that site (Figs. 1 and S1). Still, the observed increases in PAR following Scotch broom removal at both sites would be likely to facilitate the release or establishment of vegetation in the absence of Scotch broom. The significant correlation between PAR and NMS ordinations at both sites supports this, demonstrating the importance of changes in light availability to plant community responses.
The lack of any treatment effect on soil water content was surprising, as broom has been shown to be effective at acquiring soil water (Carter et al. 2019b) and we expected that its removal would result in a concurrent increase in soil water availability associated with a reduction in transpiration losses. The lack of treatment effect indicates that any increase in soil water availability was quickly utilized by established and colonizing vegetation or offset by increased surface evaporation following canopy removal. It is difficult to quantify the relative contribution of these processes, but Carter et al. (2019c) showed that background vegetation in the presence of Scotch broom can have a pronounced effect on soil water content in this region. The lack of any treatment effect could also be because of the depth at which soil water content was measured (30 cm), as Scotch broom may acquire water from deeper portions of the soil profile. Regardless, the lack of any correlation between SWC and the ordination axis values indicates that SWC is not a large factor influencing the response of community composition to broom removal.
Soil responses
Given that Scotch broom is an N-fixing plant capable of fixing moderate quantities of N (20–100 kg ha−1 year, Carter et al. 2019a, Watt et al. 2003a), there is potential for Scotch broom to increase soil N pools over time (Fogarty and Facelli 1999) with concurrent increases in soil C, as these elements are inextricably linked in soil. The significantly higher soil C concentration at the high-quality site, where Scotch broom was retained supports this general reasoning and agrees with previous work conducted at these sites evaluating effects of Scotch broom absence or presence on soil chemical properties (Slesak et al. 2016). In contrast, there was no significant effect of Scotch broom on soil C and N at the low-quality site, which is consistent with past work in these areas (Slesak et al. 2016), likely because Scotch broom effectively retains fixed N in its biomass at sites with low soil quality (i.e., soils with low initial N, Carter et al. 2019a). The lack of larger or more significant effects of broom on soil N was unexpected given findings from previous studies (Maron and Jeffries 1999; D’Antonio et al. 2017; Nisikani et al. 2018) and may be a result of our focus on total N instead of more dynamic plant available pools. Regardless, to the extent that Scotch broom may influence soil C and N at these sites, trends in response from this study suggest that C and N pools are recovering following Scotch broom removal (Tables 2 and 3).
The lower amounts of extractable nutrient cations and P associated with Scotch broom removal indicate that negative effects on soil chemistry are being amplified in that treatment. Increased N availability associated with symbiotic N fixation is known to increase nutrient cation mobility and loss (Van Miegrot and Cole 1984; Montagnini et al. 1991), and Carter et al. (2019c) observed higher concentrations of soil water Ca and Mg, where Scotch broom was experimentally planted, which would be susceptible to leaching. It seems plausible that leaching contributed to a loss of nutrient cations when broom was present, but it is not clear why continued or accelerated loss would occur after broom removal. Most of the removal effects occurred at the low-quality site which is inherently more susceptible to leaching losses because of its coarse texture and low cation exchange capacity (Slesak et al. 2009). Furthermore, removal of the dense broom canopy would have decreased rainfall interception, favoring increased leaching. Alternatively, it is possible that colonizing vegetation in the removal treatment increased net nutrient uptake, resulting in a reduction in soil pools. Uptake may have been particularly high because we physically removed Scotch broom from the plot, and any nutrients bound within its biomass would have been effectively lost, leading to increased demand from the soil nutrient pool. On the other hand, the lack of any significant correlations in the NMS ordinations with soil variables does not provide evidence in support of increased uptake. The underlying mechanism leading to greater nutrient loss following Scotch broom removal has important implications in evaluating recovery and deserves further study.
Plant community responses
Treatment clearly influenced plant community structure, resulting in distinctly different communities among the three treatments (Fig. 4). Given this, it is apparent that vegetation communities have not recovered in the 4 years, since Scotch broom was removed. A lack of recovery following removal is not uncommon, especially when the invasive plant has altered soil properties (Corbin and Antonio 2012) which can lead to modified plant competitive relationships (Weidenhamer and Callaway 2010). Scotch broom is also capable of allelopathy (Haubensak and Parker 2004; Slesak et al. 2021) which could hamper plant community recovery. We note that our study period was only 4 years in length, and recovery could possibly occur at longer durations (Bateman and Vitousek 2018). The trend of the removal treatment communities at each site moving along Axis 1 towards the uninvaded treatment community provides support for this possibility (Fig. 4).
The lack of plant community recovery we observed occurred, because Scotch broom removal facilitated the establishment of non-native species and a reduction in native species abundance. Secondary invasions, where removal of an invasive species leads to an increase in other invasive species, have been reported in a number of studies (Kuebbing and Nunez 2015, Prior et al. 2018), and it appears that Scotch broom removal can also facilitate secondary invasion. Carter et al. (2019c) previously showed that Scotch broom can promote the establishment of non-native species via its influence on the microenvironment. The increase in richness and cover of non-native species we observed following Scotch broom removal is likely a result of higher initial abundances of non-native species, where Scotch broom was present (i.e., in both the retained and removed treatments; Table 4) combined with an increase in light availability following its removal. The increase in light could also have been detrimental to shade-tolerant native species present on these sites, providing more opportunity for the invasive species to establish. The correlations between PAR and the NMS ordination axis values support this given the negative relationship with axis 2 at the low-quality site (negative influence on native species) and the positive relationship with axis 1 at the high-quality site (positive influence on non-native species). Changes in nutrient availability (e.g., N) following broom removal may have also contributed to the response (Grove et al. 2015), but our measures of nutrient status were not related to the NMS ordinations. Other measures of nutrient availability (e.g., available N forms; ammonium and nitrate) would have been useful to determine if changes in nutrient availability influenced plant community dynamics.
There were clear differences between sites in the response of native and non-native species following Scotch broom removal. Shaben and Myers (2010) attributed differential response of non-native species response to Scotch broom at two sites to differences in the abundance of non-native species initially present at each site. Changes in resource availability following broom invasion or removal cannot favor other invasive species if those species are not present on the site (Maron and Jefferies 1999). The relatively larger response at the low-quality site is likely associated with a larger abundance of non-native species present (Table 4), the larger response in PAR following Scotch broom removal at that site (Fig. 1), and constrained soil resource supply (water and nutrients) associated with the coarse soil texture (Table 1). The likelihood of Scotch broom invasion and re-invasion has been greatest at the low-quality-site (Harrington and Schoenholtz 2010; Harrington et al. 2020), likely because of broom’s generalist ecology and physiological adaptations which make it particularly competitive on poor-quality sites with coarse textured soils (Carter et al. 2019b; Watt et al. 2003b). Thus, low-quality sites are both more susceptible to broom invasion and long-term, negative effects from the species.
Conclusion
Scotch broom is an invasive species of major concern and managers often vigorously try to control broom to restore and maintain ecosystem functions. Our results indicate that Scotch broom can negatively affect soil properties and plant communities, and these effects can be exacerbated following the species removal. Effects on soil nutrient cations and the abundance of native vs. non-native species are likely to hamper ecosystem recovery, and it is unclear when these negative effects will begin to diminish. Given our results, management actions are likely to be more effective if they focus on preempting establishment of Scotch broom or aggressively controlling any populations immediately after establishment. Low-quality sites are both more susceptible to invasion and subsequent negative ecosystem effects following the species removal, suggesting that similar site types be targeted for control efforts.
Availability of data and materials
All data associated with this study are available from the corresponding author on reasonable request.
References
Bateman JB, Vitousek PM (2018) Soil fertility response to Ulex europaeus invasion and restoration efforts. Biol Invasions 20:2777–2791. https://doi.org/10.1007/s10530-018-1729-9
Bossard CC, Rejmánek M (1994) Herbivory, growth, seed production, and resprouting of an exotic invasive shrub Cytisus scoparius. Biol Conserv 67:193–200
Caldwell BA (2006) Effects of invasive scotch broom on soil properties in a Pacific coastal prairie soil. Appl Soil Ecol 32:149–152
Carter DR, Slesak RA, Harrington TB, D’Amato AW (2019a) Effects of irrigation and phosphorus fertilization on physiology, growth, and nitrogen-fixation of Scotch broom (Cytisus scoparius). Plant Physiol Rep. https://doi.org/10.1007/s40502-019-00459-7
Carter DR, Slesak RA, Harrington TB, D’Amato AW (2019b) Comparative effects of soil resource availability on physiology and growth of Scotch broom (Cytisus scoparius) and Douglas-fir (Pseudotsuga menziesii) seedlings. For Ecol Manage. https://doi.org/10.1016/j.foreco.2019.117580
Carter DR, Slesak RA, Harrington TB, Peter DH, D’Amato AW (2019c) Scotch broom (Cytisus scoparius) modifies microenvironment to promote a non-native community structure. Biol Invasions. https://doi.org/10.1007/s10530-018-1885-y
Carter DR, Slesak RA, Harrington TB, D’Amato AW (2021) Soil texture and other site-level factors differentially affect growth of Scotch broom and Douglas-fir seedlings in the western Pacific Northwest. Can J for Res. https://doi.org/10.1139/cjfr-2021-0011
Corbin JD, D’Antonio CM (2012) Gone but not forgotten? Invasive plants legacies on community and ecosystem properties. Invasive Plant Sci Manage 5:117–124
Czortek P, Królak E, Borkowska L, Bielecka A (2020) Impacts of soil properties and functional diversity on the performance of invasive plant species Solidago canadensis L. on post-agricultural wastelands. Sci Total Environ. https://doi.org/10.1016/j.scitotenv.2020.139077
D’Antonio CM, Ostertag R, Cordell S, Yelenik S (2017) Interactions among invasive plants: lessons from Hawaii. Annu Rev Ecol Evol Syst 48:521–541. https://doi.org/10.1146/annurev-ecolsys-110316-022620
Dassonwille N, Vanderhoven S, Vanparys V, Hayez M, Gruber W, Meerts P (2008) Impacts of alien invasive plants on soil nutrients are correlated with initial site conditions in NW Europe. Oecologia 133:206–214
Egunjobi J (1971) Ecosystem Processes in a Stand of Ulex Europaeus L.: I. Dry matter production, litter fall and efficiency of solar energy utilization. J Ecol 59(1):31–38. https://doi.org/10.2307/2258449
Fogarty G, Facelli JM (1999) Growth and competition of Cytisus scoparius, an invasive shrub, and Australian native shrubs. Plant Ecol 144:27–35
Grove S, Parker IM, Haubensak KA (2015) Persistence of a soil legacy following removal of a nitrogen-fixing invader. Biol Invasions 17:2621–2631
Halverson NM, Topik C, Van Vickle R (1986) Plant association and management guide for the western hemlock zone. Mount Hood National Forest. USDA Forest Service. Pacific Northwest Region, Portland, pp 62–65
Harrington TB, Schoenholtz SH (2010) Effects of logging debris treatments on five-year development of competing vegetation and planted Douglas-fir. Can J for Res 40:500–510
Harrington TB, Peter DH, Slesak RA (2018) Logging debris and herbicide treatments improve growing conditions for planted Douglas-fir on a droughty forest site invaded by Scotch broom. For Ecol Manage 417:31–39
Harrington TB, Slesak RA, Dollins J, Schoenholtz SH, Peter D (2020) Logging- debris configuration and vegetation control influence 15-year changes in soil C and N and stand characteristics of planted coast Douglas-fir in western Washington and Oregon. For Ecol Manage. https://doi.org/10.1016/j.foreco.2020.118288
Haubensak KA, Parker IM (2004) Soil changes accompanying invasion of the non-native shrub Cytisus scoparius in glacial outwash prairies of western Washington [USA]. Plant Ecol 175:71–79
Henderson JA, Peter DH, Lesher RD, Shaw DC (1989) Forested plant associations of the Olympic national forest. USDA Forest Service Report No. R6-ECOL-TP 001–88, pp 350–353
Kettenring KM, Adams CR (2011) Lessons learned from invasive plant control experiments: a systematic review and meta-analysis. J Appl Ecol 48:970–979
Kuebbing SE, Nunez MA (2015) Negative, neutral, and positive interactions among non-native plants: patterns, processes, and management implications. Glob Change Biol 21:926–934
Lázaro-Lobo A, Ramirez-Reyes C, Lucardi RD, Ervin GN (2021) Multivariate analysis of invasive plant species distributions in southern US forests. Landscape Ecol 36:3539–3555. https://doi.org/10.1007/s10980-021-01326-3
Maron JL, Jefferies RL (1999) Bush lupine mortality, altered resource availability, and alternative vegetation states. Ecology 80(2):443–454
McCune B, Mefford MJ (2018) PC-ORD. Multivariate Analysis of Ecological Data. Version 7.08
McCune B, Grace JB, Urban DL (2002) Analysis of Ecological Communities. MjM Software Design, Gleneden Beach
McGeoch MA, Genovesi P, Bellingham PJ, Costello MJ, McGrannachan C, Sheppard A (2016) Prioritizing species, pathways, and sites to achieve conservation targets for biological invasion. Biol Invasions 18:299–314. https://doi.org/10.1007/s10530-015-1013-1
Montagnini F, Haines B, Swank WT (1991) Soil solution chemistry in black locust, pine/mixed-hardwoods and oak/hickory forest stands in the southern Appalachians, USA. For Ecol Manage 40:199–208
Nisikani MM, van Wilgen BW, Gaertner M (2018) Barriers to ecosystem restoration presented by soil legacy effects of invasive alien N2-fixing woody species: implications for ecological restoration. Restor Ecol 26:235–244. https://doi.org/10.1111/rec.12669
Parker IM, Harpole W, Dionne D (1997) Plant community diversity and invasion of the exotic shrub Cytisus scoparius: testing hypotheses on invasibility and impact. In: Dunn PV, Ewing K (eds) Ecology and conservation of the Southern Puget Sound Prairie Landscape The Land Conservancy. Seattle, Washington, pp 149–161
Potter KJB, Kritcos DJ, Watt MS, Leriche A (2009) The current and future potential distribution of Cytisus scoparius: a weed of pastoral systems, natural ecosystems and plantation forestry. Weed Res 49:271–282
Prior KM, Adams DC, Klepzig KD, Huler J (2018) When does invasive species removal lead to ecological recovery? Implications for management success. Biol Invasions 20:267–283
PRISM Climate Group (2012) PRISM (Parameter-elevation regressions on independent slopes model) climate mapping system. Oregon State University. http://prism.oregonstate.edu
Reid AM, Morin L, Downey PO, French K, Virtue JG (2009) Does invasive plant management aid the restoration of natural ecosystems? Biol Conserv 142(10):2342–2349
Richardson B, Whitehead D, McCracken IJ (2002) Root-zone water storage and growth of Pinus radiata in the presence of a broom understorey. N Z J for Sci 32(2):2008–2220
SAS Institute Inc. (2013) The SAS System for Windows, version 9.4. Cary, North Carolina
Shaben J, Myers JH (2010) Relationships between Scotch broom (Cytisus scoparius), soil nutrients, and plant diversity in the Garry oak savannah ecosystem. Plant Ecol 207:81–91. https://doi.org/10.1007/s11258-009-9655-7
Simberloff D, Parker IM, Windle PN (2005) Introduced species policy, management, and future research needs. Front Ecol Environ 3(1):12–20. https://doi.org/10.1890/1540-9295(2005)003[0012:ISPMAF]2.0.CO;2
Slesak RA, Scheonholtz SH, Harrington TB, Strahm BD (2009) Dissolved carbon and nitrogen leaching following variable logging-debris retention and competing vegetation control in Douglas-fir plantations of western Oregon and Washington. Can J for Res 39:1484–1497
Slesak RA, Harrington TB, D’Amato AW (2016) Invasive Scotch broom alters soil chemical properties in Douglas-fir forests of the Pacific Northwest, USA. Plant Soil 398:281–289. https://doi.org/10.1007/s11104-015-2662-7
Slesak RA, Harrington TB, D’Amato AW, Carter DR (2021) Legacy effects of non-native Cytisus scoparius in glacial outwash soils: Potential impacts to forest soil productivity in western Washington. For Ecol Manage 481(2):1–8. https://doi.org/10.1016/j.foreco.2020.118733
Soil Survey Staff, USDA-NRCS. 2021. Official soil series descriptions. Available from https://www.nrcs.usda.gov/wps/portal/nrcs/detail/soils/survey/geo/?cid=nrcs142p2_053587 [accessed March 3, 2021].
The PLANTS database, 2018. USDA Nat. Resour. Conserv. Serv. <http://plants.usda.gov>
Van Miegrot H, Cole DW (1984) The impact of nitrification on soil acidification and cation leaching in a red alder ecosystem. J Environ Qual 13:586–590
Walker LR, Vitousek PM (1991) An invader alters germination and growth of a native dominant tree in Hawai’i. Ecology 72:1449–1455
Waterhouse BM (1988) Broom (Cytisus scoparius) at Barrington Tops, New South Wales. Aust Geogr Stud 26:239–248
Watt MS, Whitehead D, Mason EG, Richardson B, Kimberly MO (2003a) The influence of weed competition for light and water on growth and dry matter partitioning of young Pinus radiata, at a dryland site. For Ecol Manage 183:363–376
Watt MS, Clinton PW, Whitehead D, Richardson B, Mason EG, Leckie AC (2003b) Above-ground biomass accumulation and nitrogen fixation of broom (Cytisus scoparius L.) growing with juvenile Pinus radiata on a dryland site. For Ecol Manage 184:93–104
Wearne LJ, Morgan JW (2004) Community-level changes in Australian subalpine vegetation following invasion by the non-native shrub Cytisus scoparius. J Veg Sci 15:595–604
Weidenhamer JD, Callaway RM (2010) Direct and indirect effects of invasive plants on soil chemistry and ecosystem function. J Chem Ecol 36:59–69
Williamson J, Harrison S (2002) Biotic and abiotic limits to the spread of exotic revegetation species. Ecol Appl 12:40–51. https://doi.org/10.1890/1051-0761(2002)012[0040:BAALTT]2.0.CO;2
Yelenik SG, Stock WD, Richardson M (2004) Ecosystem level impacts of invasive Acacia saligna in the South African Fynbos. Restor Ecol 12(1):44–51. https://doi.org/10.1111/j.1061-2971.2004.00289.x
Acknowledgements
Financial support for this research was provided by the USDA National Institute for Food and Agriculture (Grants.gov number: GRANT11325729) and by the USDA Forest Service, Pacific Northwest Research Station. We thank Port Blakely and Green Diamond Resource Company for use of their land and logistical support. Thanks also to James Dollins for assistance with the field work.
Author information
Authors and Affiliations
Contributions
RAS, TBH, and AWD conceived and designed the experiment. TBH and DHP collected the data. RAS and TBH analyzed the data. RAS wrote the manuscript, and all other authors reviewed the paper and provided editorial comment. All authors approved of the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Communicated by Jennifer Funk.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Slesak, R.A., Harrington, T.B., D’Amato, A.W. et al. Removal of invasive Scotch broom increases its negative effects on soil chemistry and plant communities. Oecologia 198, 243–254 (2022). https://doi.org/10.1007/s00442-021-05099-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-021-05099-z