Abstract
One of the main objectives of community ecology is to understand the conditions allowing species to coexist. However, few studies have investigated the role of fine-scale habitat use segregation in the functioning of guild communities in relatively homogeneous landscapes where opportunities for coexistence are likely to be the most restrictive. We investigate how the process of habitat use differentiation at the home range level according to the degree of specialism/generalism of species can lead to coexistence between guild species. We examine differences in fine-scale habitat use and niche separation as potential mechanisms explaining the coexistence of five sympatric carnivore species that differ in life history traits (Iberian lynx, Eurasian badger, Egyptian mongoose, common genet and red fox) by collecting data from systematic track censuses in a relatively homogeneous Mediterranean landscape. We found that a higher degree of specialism determines the segregation of species among the fine-scale ecological niche dimensions defined using quantitative elements associated with vegetation, landscape, prey availability and human disturbance. The species with the lowest total performance over the set of variables did not exhibit segregation in the use of habitat at this level. Our study indicates that in relatively homogeneous landscapes, there exist subtle patterns of habitat partitioning over small-scale gradients of habitat determinants as a function of the degree of specialism of carnivore species within a guild. Our results also suggest that coexistence between generalist species may be permitted by fine-scale spatial–temporal segregation of activity patterns or trophic resource consumption, but not fine-scale habitat use differentiation.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The competitive exclusion principle states that two ecologically similar species cannot coexist unless they exhibit niche separation (Gause 1934; Hardin 1960). Thus, some degree of partitioning must occur in the realised niche of coexisting species, which can occur at the temporal, trophic and/or habitat selection level. This is particularly obvious in heterogeneous landscapes where spatial heterogeneity foments coexistence between similar species (i.e. within the same trophic level), with the selection of different habitats being one of the main processes promoting sympatry (Levin 1974; Rosenzweig et al. 1984). Nevertheless, in relatively homogeneous landscapes, where a general habitat type dominates, similar species may use different patches that support a set of resources sufficient to fulfil their requirements.
We define a relatively homogeneous landscape as an area largely covered by one single land cover class with interspersed patches of different vegetation types. This definition implies low spatial variation, both in terms of composition (i.e. diversity) and configuration (i.e. patch density) and takes into account body size and the spatial scale of movements of small- to medium-sized mammal carnivores. The body size of larger species may influence their scale of perception. Hence, larger mammals such as carnivores with large home ranges perceive a more homogeneous landscape (Lidicker and Koening 1996) composed of fewer isolated patches than smaller species (Addicott el al. 1987; Gehring and Swihart 2003).
Although fine-scale habitat use differentiation can also occur in heterogeneous landscapes, it may appear as a key mechanism, apart from trophic and/or activity pattern segregation, allowing the coexistence of species, particularly in these relatively homogeneous landscapes. The analyses of fine-scale habitat use segregation between species, until now poorly explored, requires more detailed landscape information for a more accurate discrimination and quality assessment of suitable habitats and the detection of crucial habitat features not obvious at broader scales (Fernández et al. 2003; Martin et al. 2010).
Additionally, life history trait differences between coexisting species in relatively homogeneous landscapes can help us understand the way species exploit resources. Hence, besides fine-scale habitat use segregation, coexistence is therefore possible if species exhibit differences in their life history traits that allow niche differences in space (Brown and Wilson 1956; Hutchinson 1959; Chesson 2000). In other words, cohabitation may be privileged when species with different degrees of habitat and/or trophic specialisation are present.
To test the hypothesis of fine-scale spatial partitioning as a function of the species’ degree of specialisation, we compared the habitat use of five sympatric carnivore species (Iberian lynx Lynx pardinus, Eurasian badger Meles meles, Egyptian mongoose Herpestes ichneumon, common genet Genetta genetta and red fox Vulpes vulpes) within a Mediterranean protected area in south-western Spain. The study is focused on the main habitat type used by the target species within the protected area, the Mediterranean woodland. We hypothesised that in an area with a main habitat type or land cover class, fine-scale habitat use differences may allow sympatry of species within the same trophic level but that differ in their degree of specialisation. If we graph the ecological space filled by each species within an n-dimensional hypervolume representing environmental variability, spatial segregation in their realised niches as a function of their degrees of specialisation may be revealed. We first developed habitat models at fine spatial scales to assess the environmental features that best describe the relationship between each species and its environment. Secondly, we used an ordination method to illustrate the ecological space filled by each species in a multidimensional space. We fitted a set of fine-scale habitat models by taking into consideration prior knowledge of species ecology to test five hypotheses:
-
1.
Vegetation and landscape structure are the best explanatory factors of species’ habitat use.
-
2.
Since habitat used by carnivores in our study area has a relatively homogeneous vegetation, we hypothesised that prey availability is the single explanatory factor of habitat use.
-
3.
Human disturbance is the most important variable describing habitat selection.
-
4.
Vegetation and landscape structure and prey availability interact to explain habitat use.
-
5.
All landscape descriptors, prey availability and human disturbance are relevant.
The study of the carnivore guild considered here is particularly interesting because it allows a comparison of different species within a continuum of specialisation ranging from complete specialisation (i.e. the Iberian lynx) to full generalisation (i.e. the red fox).
The Eurasian badger and the red fox are generalists in their habitat use and diet (Amores 1975; Kruuk and Parish 1985; Cavallini and Lovari 1991; Roper and Lups 1995; Carvalho and Gomes 2001; Balestrieri et al. 2005; Rosalino et al. 2005; Delibes-Mateos et al. 2008), although a certain degree of local feeding specialisation has been reported for several badger populations and for different resources (Kruuk and De Kock 1981; Kruuk and Parish 1981; Martín et al. 1995) as well as negative responses to certain types of landscape fragmentation patterns (Virgós 2002). The Egyptian mongoose is a trophic generalist (Delibes and Palomares 1991a; Zapata et al. 2007), but they are habitat specialists in Mediterranean areas (Palomares and Delibes 1993a). Mongooses actively avoid open areas and select those with dense vegetation (thickets) for foraging and resting (Palomares and Delibes 1993b). The Iberian lynx is the most specialised in terms of habitat use and trophic niche in this carnivore guild (Palomares et al. 1991; Ferreras et al. 1997; Delibes et al. 2000, 2007a, 2007b; Palomares et al. 2001). Lynxes feed almost exclusively upon European wild rabbits (Oryctolagus cuniculus) and need shrub vegetation patches to rest and breed. Habitats sustaining stable lynx populations should ideally include 40 % cover of understorey vegetation, half of which should be bushes (Delibes et al. 2000, 2007a, 2007b; Palomares 2001). Moreover, the density of ecotones between shrubland and pastureland is also a robust predictor of territory occurrence (Fernández et al. 2003, 2007). The common genet is considered to be intermediate between a typical generalist and a typical specialist species (Virgós et al. 1999). Genets need bushes and hollow trees as sites for nocturnal and diurnal resting and feed mainly on small mammals such as the long-tailed field mouse (Apodemus sylvaticus) (Palomares 1986; Delibes and Palomares 1988, 1991b).
We predicted that habitat use in lynxes and genets would be explained by multiple variables due to their higher degree of habitat and trophic specialisation, whereas badgers and foxes should have fewer requirements and respond to single explanatory factors, such as prey availability or vegetation and landscape structure, because of their omnivorous diets and habitat generalism. For mongooses, as habitat specialists but trophic generalists, vegetation and landscape structure should explain their pattern of habitat use. Therefore, genets and lynxes should show the most marginal (i.e. located in extreme values of variable gradients) and narrowest realised niches, whereas foxes and badgers should have the most widespread and widest niches along the small-scale gradients of habitat determinants. Meanwhile, mongooses may show a non-marginal niche, wider than those of lynxes and genets but narrower than those of foxes and badgers.
Materials and methods
Study area
This study was located in Doñana National Park (DNP), a fully protected 550-km2 area in south-western Spain (37°9′N, 6°26′W). DNP is a flat sandy area at sea level bordered to the south and west by the Atlantic Ocean and to the east by the Guadalquivir River mouth. The climate is Mediterranean subhumid (i.e. characterised by mild wet winters and hot dry summers), with an average annual rainfall of approximately 550 mm. Approximately half of the surface of DNP is covered by Mediterranean scrubland, and the other half by marshland whose clay soils make it unsuitable for track censuses. There is a dune system in the southern part of the scrubland area. Track censuses were restricted to the scrubland biotope, as it comprises the most frequently used environmental unit by our study species.
Mediterranean scrubland is dominated by hygrophytic species consisting of very dense clumps of heather Erica sp. up to 3 m high [Erica scoparidae—Ullicetum australis and Erica ciliaris—Ullicetum (minaris) lusitanici associations] and xerophytic species of up to 1.5 m high, mainly consisting of Halimium sp. and others such as Cistus sp., gorses Ulex sp. and rosemary Rosmarinus officinalis (Halimio halimifolii—Stauracanthetum genistoidis association). More mature shrubland areas with Pistacia lentiscus and Myrtus communis can be found mainly in the north and in the valleys of the dune system. Interspersed between purely scrubland areas are a few, small patches of Eucalyptus camaldulensis and pine Pinis pinea plantations. Most of the dune valleys are also colonised by pines P. pinea.
Human access to the park is regulated, but some low-impact traditional uses are maintained under control including cattle raising, apiculture, and fishing with traditional methods. The northern and western edges of the protected area are in close contact with human settlements, crop fields and an intensively used paved road.
Study design
The study area was divided into sixty-nine 2 × 2-km quadrants following Universal Transverse Mercator (UTM) grid coordinates. Each quadrant was sampled twice (2007–2008 and 2008–2009) throughout the wet season (October–April). A 3-km-long survey route was slowly walked to search for carnivore and prey tracks within each quadrant. We counted the total number of tracks for each carnivore species within each sampling unit. The survey routes were located along sandy paths (firebreaks and car roads) between 2 and 12 m wide. Once a continuous track that crossed from one side to the other across the pathway was detected, we georeferenced it using a global positioning system. We always carried out surveys at least 3 days after any rainfall. We also sampled by track censuses potential target prey species of the carnivores studied.
We sampled tracks of small mammals (most of which probably belonged to the long-tailed field mouse Apodemus sylvaticus according to Kufner and Moreno (1989), but likely included the Western Mediterranean mouse Mus spretus, white-toothed shrew Crocidura russula, and garden dormouse Eliomys quercinus, European rabbit Oryctolagus cuniculus, red partridge Alectoris rufa, domestic cow Bos taurus and horse Equus caballus; and wild ungulates such as the fallow deer Dama dama, the red deer Cervus elaphus and the wild boar Sus scrofa. Wild and domestic ungulates are rarely preyed upon by any of the carnivore species studied here, but we sampled them since they may occasionally provide an important food source as carrion for some of them. We concentrated prey sampling in a 1-month period to avoid particularly apparent inter-monthly variations in abundance for some species (i.e. small mammals and European rabbits) (Kufner 1986; Palomares et al. 2001). Thus, we carried out the sampling of prey tracks in April (corresponding to the intra-annual abundance peak in both species) along transects in every quadrant sampled for carnivore tracks. These transects for prey species were sandy paths, 25 m in length and approximately 1.7 m wide (i.e. the area of a four-wheel-drive car), separated by at least 300 m and located on the same routes walked for carnivore tracks. Thus, between seven and ten prey censuses were carried out per quadrant. Due to the ephemeral character of tracks, these surveys may be linked to a certain underestimation of particularly small species such as small mammals. Nevertheless, the sandy substrate of our study area is highly appropriate for this type of survey due to the visibility of tracks. Additionally, we always conducted prey surveys under favourable tracking conditions. Hence, transects were located on sandy paths close to the nearest vegetation border, as this likely exerts a protective effect against wind and maintains a higher level of moisture in the sand, thereby increasing substrate quality for the detection of tracks (Soto et al. 2012). We considered unsuitable for prey counts transects in which grassy groundcover was more than 30 % in some part of the transect, and we improved reliability by suspending searches of tracks for ≥78 h after precipitation or periods of high winds (≥24 km/h) and by making only early morning observations.
We also recorded habitat structure at each quadrant in circles of 25-m diameter around sampling points located every 300 m along the survey transects where we censused prey species. Here, we visually estimated the percentage of three categories of vegetation: short shrubs (xerophytic species such as Halimium sp. and Cistus sp.), tall shrubs (species such as Erica sp., Juniperus phoenica and Pistacia lentiscus), and trees. In addition, we measured other variables related to habitat structure, i.e. average tree height as well as average height of tall shrub and short shrub species.
Fine-scale habitat modelling design
We selected 14 variables to study habitat selection patterns for each species (see Table 1 for model definitions; see next section for arguments on variable selection).
Each quadrant was associated with values of abundance for different prey and percentages of coverage of different vegetation types. We averaged the value obtained at the sampling points both for prey (abundance index of different preys per kilometre) and vegetation indexes (percentage of coverage).
In addition to the distance from the centre of each grid to water, we calculated distance to the ecotone between marshland and scrubland and distance to the anthropogenic edge (see Table 1) from digitised roads, urban settlements, and water source cover layers in DNP using a Euclidean distance-based approach (Perkins and Conner 2004; Benson and Chamberlain 2007). Urban settlements included towns and villages surrounding the national park. Water sources included natural and artificial ponds (i.e. dug for the cattle in zones where the water table is higher) that were permanently flooded. Traffic index (T) per quadrant was derived from data in a previous study on the effect of traffic on biodiversity in DNP (Román et al. 2010).
Ecotones between pastureland and scrubland were defined using a 1:10,000 fine-scale vegetation map for 1996–2006 obtained from the Sistema de Información Ambiental de Andalucía for the Doñana area. We reclassified vegetation units or polygons based on four vegetation attributes of physiognomy, species composition and density of each vegetation layer within the polygon: (a) trees (P. pinea, Quercus suber and Eucaliptus spp.); (b) tall shrubs (subsequently referred to as ‘bushes’) of mature Mediterranean shrubland (e.g. P. lentiscus, M. communis) and tall thicket Erica spp.; (c) short shrubs (H. halimifolium, Ulex spp., Stauracanthus genistoides); and (d) pastures. The result was a reclassified vegetation digital map with 316 polygons and four vegetation attributes per polygon. The projection for all geographic information system (GIS) layers and data were UTM 30S, datum European 1950. Hawth’s tools and geoprocessing extension in ArcInfo 9.3 (ESRI, Redlands, CA) were used to calculate distance-based variables, to identify ecotones, and to calculate their density (Table 1).
Statistical analysis
Fine-scale habitat modelling
We based our analyses on information-theoretic methods guided by the view that ecological inference can best be approached by weighing evidence for multiple working hypotheses simultaneously (Hilborn and Mangel 1997; Burnham and Anderson 1998; Johnson and Omland 2004). These methods consist of identifying a priori the alternative hypotheses for habitat selection and their mathematical formulation, and then testing their support by fitting the relevant equations to species distribution data and examining penalised maximum-likelihood estimates (e.g. Fernández et al. 2003; Johnson et al. 2004).
We first specified a set of 16 candidate models that could potentially predict species habitat use and distribution in DNP, therefore restricting the model selection process to a few meaningful combinations of predictors of the species. For selecting predictor variables and formulating the candidate models, we considered five working hypotheses addressing the critical points of the different species’ life history traits and requirements: (a) vegetation and landscape structure, (b) prey availability, (c) human disturbance, (d) vegetation and landscape structure plus prey availability, and (e) global.
Considering previous knowledge of the ecology of the species under study in the Doñana area, we designed fine-scale habitat models aimed to offer insight into the mechanisms that structure the carnivore guild at the DNP. Fine-scale habitat models developed based on fine-scale habitat data are particularly useful for an understanding of relevant ecological factors for habitat selection of the species under study along small-scale gradients.
We also hypothesised that distance to permanent water resources may be an important factor for all species, particularly during the hottest months when many surface water sources dry out in the area. Additionally, proximity to humans and infrastructure derived from their activity as well as the T inside the protected area may be detrimental to all the species because they produce higher mortality, degrade the original Mediterranean ecosystems and involve a higher risk of predation or competition with non-native carnivores (i.e. domestic dogs).
Pearson’s correlation coefficients among the variables and predictors were computed. Predictors with r > 0.6 were never included in the same model. In addition, we fitted an intercept-only equation to test improvement over the null model of no effect. Fitted models were compared using the second-order Akaike information criterion (AICc) and model weights (Burnham and Anderson 2002). We ranked models by their AICc values and determined the model-averaged parameter estimates (Burnham and Anderson 2002). The relative variable importance of predictor variable j(w j ) was determined as the sum of the w i across all models where j occurs. Larger w j values indicate a higher relative importance of variable j compared to other variables (Burnham and Anderson 2002).
Candidate model equations were fitted using generalised linear mixed models (GLMM) in SAS 9.2 with logit-link and binomial (for lynxes and genets) or negative binomial (for badgers, mongooses and foxes) error structure (McCullagh and Nelder 1989). Observer and quadrants were modelled as random effects, humidity as a covariate (Soto et al. 2012), and the distance covered per quadrant during track censuses as an offset in all models.
Additionally, although we selected 4-km2 grids for sampling in order to diminish the possibility of the same individuals being sampled in neighbouring grids [several of the carnivore species sampled may have home ranges of this size (Palomares 1994; Delibes and Palomares 1994)], track counts at neighbouring grids can be expected to show spatial autocorrelation. Therefore, we checked for autocorrelation in our data through inspection of semi-variograms and Moran’s I correlograms of non-spatial negative binomial and logistic generalised model residuals. We performed analyses using an autocovariate (AC) method in the GLIMMIX procedure (SAS 9.2, Littell et al. 1996) when spatial autocorrelation in non-spatial generalised model residuals was detected (Hosmer and Lemeshow 2000). The autocovariate method accounts for fine-scale spatial variation in the data by estimating how much the response at each location reflects response values at surrounding locations (Dormann et al. 2007). This extra parameter is intended to capture spatial autocorrelation originating from endogenous processes such as movement of censused individuals between sampling sites (Smith 1994; Keitt et al. 2002; Yamaguchi et al. 2003). For every quadrant in our study area, we calculated the AC i in ArcGIS 9.3 (ESRI) as:
where y j is the number of tracks at quadrant j and w j is the inverse Euclidean distance between locations i and j. Hence, an autocovariate at location i is defined as a weighted sum of observation records y at locations j in a neighbourhood determined by N i (Miller 2005).
The neighbourhood size (N i ) is often chosen arbitrarily and optimised (by trial and error) to best capture spatial autocorrelation (Augustin et al. 1996). Nevertheless, it may be informed as well by biological parameters, such as species’ movements or dispersal capacity (Knapp et al. 2003) if the cause of spatial autocorrelation is known (or at least suspected). Hence, we hypothesised that autocorrelation in our data may partially originate from movement of censused individuals between sampling sites. We therefore set the neighbourhood size to two quadrants from each quadrant border to capture the average home range for all species. We incorporated each autocovariate as an additional explanatory variable in the GLMM models to account for the variation explained by space while maintaining the same variable selection procedures as for spatially invariant models. Finally, we tested autocovariate models for autocorrelation in the Pearson residuals, using Moran’s I correlograms and semivariograms of the most parsimonious model. We used the variogram procedure in SAS 9.2 to conduct these tests.
Outlying mean index analysis
To separate the ecological space filled by each species within an n-dimensional hypervolume representing environmental variability at DNP, we used the outlying mean index analysis (OMI) ordination method (Dolédec et al. 2000). This method measures the marginality of a species’ habitat distribution (niche position), i.e. the distance between the mean habitat conditions used by a species (species centroid) and the mean habitat conditions across the study area (origin of the niche hyperspace) as well as the species tolerance (niche breadth), i.e. the amplitude in the distribution of each species along the sampled environmental gradients (Hurlbert 1978). The OMI index measures the niche position of each species. Species with high values of OMI have marginal niches and are assumed to be influenced by a subset of the measured environmental variables (they occur in atypical habitats in a region), and those with low values have non-marginal niches and indicate no specific response to the environmental variables (they occur in typical habitats in a region); such species tend to be more common throughout the study area. The niche breadth or species tolerance is measured by an additional variance term provided by this method. Low values of species tolerance mean that a species is distributed across habitats with a limited range of conditions (specialist species), while high values imply that a species is distributed across habitats with widely varying environmental conditions (generalist species). Residual tolerance is the variation in species occurrence not accounted for by the main gradient. Outlying mean index is robust to unimodal, linear, or a mixture of species response curves and is not biased against species-poor or low-abundance sites on the synthetic gradient. Its interpretations are also robust to multicollinearity among the explanatory variables (Dolédec et al. 2000). We determined significance of the outlying mean index analysis at α = 0.05 based upon a Monte Carlo simulation (Metropolis and Ulam 1949), in which observed marginalities were statistically compared to 10,000 random permutation values of species marginalities or the null hypothesis that species are distributed equivalently in relation to the environmental variables. The OMI analysis was performed with the ADE4 library (Thioulouse et al. 1997) in the R software (Ihaka and Gentleman 1996).
Results
We surveyed 471 km and 8,373 carnivore tracks were found, with foxes, badgers and mongooses being the most frequent species (Online Resource 1). For prey, 5000 tracks were detected over the 11.6 km sampled. The most common prey species were wild ungulates and rabbits. The variables kilometric abundance index of rabbits per quadrant (Ra) and kilometric abundance index of rabbits, partridges, small mammals and ungulates per quadrant (Tot) were correlated (r = 0.669, P < 0.001).
Based on the P-value of Moran’s I (P > 0.05) and the semivariograms of residuals, habitat use models for badgers, mongooses and foxes showed spatial autocorrelation, while this was not detected for lynxes and genets. We therefore fitted spatial negative binomial generalised models (i.e. using an autocovariate) for badgers, mongooses and foxes and non-spatial logistic generalised models for lynxes and genets.
The best approximating models (∆AICc < 2) for mongooses, badgers and foxes belonged to the set of candidates designed with the hypothesis of vegetation and landscape structure plus prey availability, as well as with the hypothesis of prey availability (Table 2). For mongooses, top models included distance to ecotone (DE), distance to water (DW), Tot, Ra, mean cover of short shrub and bushes per quadrant (%SB), mean cover of bushes per quadrant (%B) and edges (linear) between tall shrub and pastureland (eBP) as predictors of their relative abundance. Variables DW and DE had the highest weights and were positively associated with the number of mongoose tracks (Table 4). Variables Tot and %B were also positive and had relatively high weights (wj > 0.4) (Table 4). For badgers, models included as predictors Ra, SM, %SB, %B, mean cover of trees per quadrant (%T), DW, DE and eBP. All of these predictors were positive except for the distance to the ecotone between marshland and scrubland. Ra was the variable with the highest weight (wj = 0.577) (Table 4) and was positively associated with the number of badger tracks. For the red fox, only one model was supported by the data (Table 2). This model included as predictors %B, %T, DW and SM, and were all positively associated with the number of fox tracks. Only two variables showed high weights: %B (wj = 0.809) and DW (wj = 0.871) (Table 4).
The best approximating models for lynxes and genets belonged to the set of candidates designed with the hypothesis of vegetation and landscape structure, vegetation and landscape structure plus prey availability as well as with the global hypothesis (Table 3). For genets, top models included as predictors %B, %T, DW, SM, DH and T. Four variables included in top models (%B, %T, DW and SM) had high weights (>0.812; Table 4). %B, %T and SM were positively associated with the presence of genet tracks while DW was negatively associated. Finally, for lynxes, top models included as predictors %SB, %B, DW, eBP, T and Ra. Variables included in the best models showed high weights (wj > 0.8) (Table 4). %SB, %B, DW, eBP and Ra were positively associated with the presence of lynx tracks while DE was negatively associated.
To illustrate niche separation between species, we used the first two axes of the outlying mean index analysis, which accounted for 99.95 % of the total explained environmental variability (Online Resource 2). The overall outlying mean index analysis (i.e. sensitivity of carnivores to environmental variables) was significant (P < 0.0001).
The position of the species along the first two OMI axes is presented in Fig. 1. Genets and lynxes were separated from mongooses, badgers and foxes. Genets and lynxes had the highest marginality values and the lowest tolerance to average habitat conditions of the synthetic gradients (Online Resource 3). Badgers and foxes had the lowest marginality and the highest tolerance values, whereas mongooses exhibited relatively low marginality and high tolerance. Although the overall response of mongooses was non-significant, their marginality and tolerance levels were between those of genets/lynxes and badgers/foxes, as predicted.
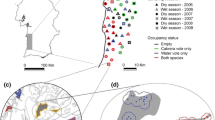
Outlying mean index (OMI) analysis of carnivore track censuses. Weighted positions of the species along the first two axes of the OMI analysis. The figure presents the ecological position of the five carnivore species in the n-dimensional hypervolume representing fine-scale environmental variability in Doñana National Park. a Iberian lynx, b common genet, c Egyptian mongoose, d Eurasian badger, and e red fox. The set of N resource units available to the species of the study defines a cloud of N points in the P-dimensional space defined by the P variables (‘ecological space’). For each species, a set of utilisation weights (indicated by circles with diameter proportional to the weights) allows us to define its niche, which are the available points that are actually used. Species that had low marginality were located near the origin and those that had high marginality were located far from the origin
Ordination diagrams on the first two axes of the OMI describe the environmental gradients that best discriminated the occurrences of the five carnivore species in DNP (Fig. 2). The first OMI axis was most influenced by prey availability and shrub cover far from human nuclei and the proportion of tall shrubs and trees at the opposing end of the gradient. The second OMI axis was most influenced by distance to the main ecotone between marshland and scrubland and prey availability and percentage of ecotones between pastureland and scrubland at the opposite end of the gradient (Online Resource 2). The presence of genet tracks was negatively associated with axis 1 (Fig. 1), and thus was positively related to patches distant from human nuclei and with high tree and tall shrub coverage (Fig. 2). Lynxes were positively associated with axis 1 and negatively with axis 2 and thus positively related to patches with high prey availability (mainly rabbits) close to the ecotone between marshland and scrubland. Conversely, mongooses, badgers and foxes were more likely to be found across a wide range of habitat types with varying environmental conditions.
Canonical weights of environmental variables. Contribution of environmental, human and prey variables to the definition of the niche parameters of species in the OMI analysis. The length of the arrow describes the relative importance of each variable in the analysis, and the direction of the arrow indicates among-variable correlations. For abbreviations, see Table 1)
Discussion
Heterogeneous landscapes offer many choices in habitat selection (i.e. different axes over which species can differ), thus broadening opportunities for coexistence. However, in more homogeneous landscapes where a general habitat type dominates, the distribution of the resources available for species is limited and opportunities for coexistence are likely to be most restrictive. Hence, subtle patterns of habitat partitioning as well as coexistence between species with different degrees of specialisation may promote ecological separation and thus community functioning under this scenario. A parsimony-based strategy for confronting different model hypotheses allowed us to assess fine-scale landscape attributes linked to five carnivore species in a protected area where species coexist in a largely similar habitat type, the Mediterranean scrubland. These species exhibited spatial storage or separate niches along a gradient of environmental variability according to their degrees of specialisation.
On the basis of an OMI analysis, lynxes and genets are marked specialists and showed the narrowest and most marginal niches throughout DNP’s environmental conditions (Fig. 1). Additionally, lynxes and genets showed niche segregation as a result of their differences in ecological preferences (Online Resource 3), which is supported by the fine-scale habitat use results. Results of fine-scale habitat use models for lynxes and genets were probably related to the availability of prime resources for the two species such as refuges and prey. Lynxes and genets showed the most restricted habitat use pattern depending on vegetation, landscape, prey and human-related variables. Lynxes were associated with zones with a high density of bushes likely representing areas of late-successional Mediterranean communities, an uncommon microhabitat in the area due to the human transformation of the autochthonous vegetation (García-Novo and Martín-Cabrera 2005). The density of ecotones between bushes and pastureland, which in turn favour abundance of the main prey of lynx, the European rabbit (Fernández et al. 2006, 2007), was also a robust predictor of the presence of lynx tracks. This was also true of the distance to the ecotone band between the Mediterranean scrubland, where refuge and grass availability favour a high rabbit density in Doñana (Moreno and Villafuerte 1997; Fernández and Palomares 2000). As expected, genets showed associations with areas with high small mammal abundance (a community mostly composed of the long-tailed field mouse), important prey items for the species in Mediterranean areas (Delibes and Palomares 1991b), as well as preferences for pine forest areas with undergrowth bushes. These results agree with previous findings for the radio-tracked population of the Iberian lynx (Palomares et al. 2000; Palomares 2001; Fernández et al. 2003) and the common genet (Delibes and Palomares 1988, 1991b, 1994; Palomares et al. 1996) in the protected area. Genets and lynxes showed low abundance and a clearly spatially structured distribution in DNP (Online Resource 4) that could therefore be explained by the fact that genets and lynxes may be limited in their home ranges to areas of simultaneous convergence of high small mammal or rabbit availability, as well as a high percentage of tall shrub cover and overall understorey or a high percentage of tree cover, respectively.
Foxes and badgers showed a non-specific habitat use pattern and a wide distribution across the environmental conditions sampled in our study area as shown by the low weights of predictors in the fine-scale habitat models (Table 4) and the OMI analysis (Fig. 1). Rabbits, as well as bush density and distance to water sources, were the most important predictors associated with numbers of badger and red fox tracks, respectively. Badgers are trophic generalists but in Mediterranean areas rabbits may constitute an important prey for the species (Palomares and Revilla 2002).
Foxes exhibited higher relative abundances in areas with dense bush cover, a microhabitat that could potentially offer higher protection.
Mongooses were positively associated with dense bush cover and high availability of different prey items as a whole (i.e. rabbits, small mammals and partridges) as well as with high availability of rabbits. This could be explained by the fact that in spite of the trophic generalism of the species (Santos et al. 2007), wild rabbits constitute an important prey item for the species in Mediterranean areas (Palomares and Delibes 1991b).
Foxes, badgers and mongooses overlapped in niche position, but foxes and badgers showed broader niches than mongooses (Fig. 1; Online Resource 3). In fact, mongooses exhibited an intermediate niche position and niche breadth between that of genets/lynxes and badgers/foxes (Online Resource 3). These results suggest that mongooses exhibit a certain degree of habitat specialisation.
The wider spatial niches exhibited by mongooses, foxes and badgers may be a response to fluctuations in resource availability, characteristic of Mediterranean environments. Mediterraneity (Virgós and Casanovas 1999) may result in an opportunistic behaviour in food gathering and habitat use broadening realised niches of species and allowing coexistence between species with similar requirements by the switch to other temporarily available resources.
In summary, sympatry of carnivore species in DNP appears to be mediated by fine-scale habitat selection only for the most specialist species (lynxes and genets). Badgers and foxes meanwhile did not show any clear pattern of specialisation at a fine scale as expected due to their high levels of habitat and trophic generalism. Although mongooses were more specialised in habitat selection than badgers and foxes, for areas such as that studied here, the three species can coexist with no apparent differences in habitat use and/or realised niche segregation. Differences in activity patterns of these three species may help to explain the coexistence among them. Mongooses are almost exclusively diurnal in the study area (Palomares and Delibes 1993b), while badgers are exclusively nocturnal (Palomares and Revilla 2002) and foxes nocturnal and crepuscular. Therefore, the carnivore community structure in DNP does not seem to be defined only by habitat use partitioning.
Interspecific interactions among predators are another aspect that should be explored and can also greatly shape the community structure. Mongooses, for example, were strongly negatively associated with distance to the ecotone between marshland and scrubland, which may be related to the high use that lynxes made of this area (Palomares et al. 1996; Viota et al. 2012).
Under a landscape analysis scheme that we have defined as coarse scaled, where Mediterranean scrubland can be considered as a homogeneous landscape, we have been able to detect differences in suitability for carnivores in DNP after examining fine-scale habitat variables. Differences in fine-scale habitat use and ecological separation through segregation along the ecological niche dimensions have been shown as key mechanisms for the most specialised species allowing coexistence, as for lynxes and genets. Hence, the study of spatial heterogeneity at small scales, particularly in largely homogeneous areas, is essential to understanding the community structure and functioning of similar, coexisting species. To manage conservation areas that protect coexisting carnivore species, we must understand guild community functioning and species-specific habitat use patterns and evaluate the role that competitors play in determining these patterns.
Author contribution statement
F. P. originally formulated the idea, F. P. and C. S. developed the methodology, C. S. conducted the fieldwork, developed the mathematical models and performed the statistical analyses, and F. P. and C. S. wrote the manuscript.
References
Amores F (1975) Diet of the red fox (Vulpes vulpes) in the western Sierra Morena (south Spain). Doñana Acta Vertebrata 2:221–239
Addicott JF, Aho JM, Antolin MF, Padilla DK, Richardson JS, Soluk DA (1987) Ecological neighborhoods: scaling environmental patterns. Oikos 49:340–346
Balestrieri A, Remonti L, Prigioni C (2005) Diet of the Eurasian badger (Meles meles) in an agricultural riverine habitat (NW Italy). Hystrix 15
Benson JF, Chamberlain MJ (2007) Space use and habitat selection by female Louisiana black bears in the Tensas River Basin of Louisiana. J Wildl Manage 71:117–126. doi:10.2193/2005-580
Brown WL, Wilson EO (1956) Character displacement. Syst Zool 5:49–64
Burnham KP, Anderson DR (1998) Model Selection and Inference: A Practical Information- Theoretical Approach. Springer-Verlag, New York
Burnham KP, Anderson DR (2002) Model Selection and Multimodel Inference: A Practical Information-TheoreticalApproach, 2d edn. Springer-Verlag, New York
Carvalho JC, Gomes P (2001) Food habits and trophic niche overlap of the red fox, European wild cat and common genet in the Peneda-Geres National Park. Galemys 13:39–48
Cavallini P, Lovari S (1991) Environmental factors influencing the use of habitat in the red fox, Vulpes vulpes. J Zool 223:323–339. doi:10.1111/j.1469-7998.1991.tb04768.x
Chesson P (2000) General theory of competitive coexistence in spatially-varying environments. Theor Popul Biol 58:11–237
Delibes M, Palomares F (1988) Time and space use by two common genets (Genetta genetta) in the Doñana National Park, Spain. J Mammal 69:635–637
Delibes M, Palomares F (1991a) Dieta del meloncillo (Herpestes ichneumon), en el Coto del Rey (Norte del Parque Nacional de Doñana, S.O. España). Doñana Acta Vertebrata 18:187–194
Delibes M, Palomares F (1991b) Ecología comparada de la gineta Genetta genetta (L.) y el meloncillo Herpestes ichneumon (L.) (Mammalia, Viverridae) en Doñana (SO de la Península Ibérica). Boletín de la Real Sociedad Española de Historia Natural (Sec. Biológica) 87:257-266
Delibes M, Palomares F (1993) Key habitats for Egyptian mongooses in Doñana National Park, south-western Spain. J Appl Ecol 30:752–758
Delibes M, Palomares F (1994) Spatio-temporal ecology and behavior of european genets in southwestern Spain. J Mammal 75:714–724
Delibes M, Laffitte R, Palomares F, Rodríguez A (1991) The status and distribution of the iberian lynx felis pardina (Temminck) in Coto Doñana Area, SW Spain. Biol Conserv 57:159–169
Delibes M, Fedriani JM, Ferreras P, Palomares F (1996) Spatial relationships between iberian lynx and other carnivores in an area of south-western Spain. J Appl Ecol 33:5–13
Delibes M, Rodríguez A, Ferreras P (2000) Action plan for the conservation of the Iberian lynx in Europe (Lynx pardinus). Council of Europe Publishing, Strasbourg
Delibes M, Fernández N, Mladenoff D, Palomares F (2003) Identifying breeding habitat for the iberian lynx: inferences from a fine-scale spatial analysis. Ecol Appl 13:1310–1324
Delibes M, Fernández N, Palomares F (2007a) Habitat-related heterogeneity in breeding in a metapopulation of the Iberian lynx. Ecography 30:431–439
Delibes M, Fernández N, Palomares F (2007b) Landscape evaluation in conservation: molecular sampling and habitat modeling for the Iberian lynx. Ecol Appl 16:1037–1049
Delibes-Mateos M, Fernandez de Simon J, Villafuerte R, Ferreras P (2008) Feeding responses of the red fox (Vulpes vulpes) to different wild rabbit (Oryctolagus cuniculus) densities: a regional approach. Eur J Wildl Res 54:71–78. doi:10.1007/s10344-007-0111-5
Dolédec S, Chessel D, Gimaret-Carpentier C (2000) Niche separation in community analysis: a new method. Ecology 81:2914–2927. doi:10.1890/0012-9658(2000)081[2914:NSICAA]2.0.CO;2
Dormann CF et al (2007) Methods to account for spatial autocorrelation in the analysis of species distributional data: a review. Ecography 30:609–628. doi:10.1111/j.2007.0906-7590.05171.x
Fernández N, Palomares F (2000) The selection of breeding dens by the endangered Iberian lynx (Lynx pardinus): implications for its conservation. Biol Conserv 94:51–61. doi:10.1016/S0006-3207(99)00164-0
Fernández N, Delibes M, Palomares F (2006) Landscape evaluation in conservation: molecular sampling and habitat modeling for the Iberian lynx. Ecol Appl 16:1037–1049. doi:10.1890/1051-0761(2006)016[1037:LEICMS]2.0.CO;2
Ferreras P, Beltrán JF, Aldama JJ, Delibes M (1997) Spatial organization and land tenure system of the endangered Iberian lynx (Lynx pardinus). J Zool 243:163–189
García-Novo F, Martín-Cabrera C (2005) Doñana: Agua y Biosfera. Doñana 2005, Confederación hidrográfica del Guadalquivir, Ministerio de Medio Ambiente. Madrid
Gause GF (1934) The struggle for existence. Williams & Wilkins, Baltimore
Gehring TM, Swihart RK (2003) Body size, niche breadth, and ecologically scaled responses to habitat fragmentation: Mammalian predators in an agricultural landscape. Biol Conserv 109:283–295
Hardin G (1960) The competitive exclusion principle. Science 131:1292–1297. doi:10.1126/science.131.3409.1292
Hilborn R, Mangel M (1997) The ecological detective. Confronting models with data. Monogr Popul Biol 28:1–315
Hosmer DW, Lemeshow S (2000) Applied Logistic Regression. Wiley, NewYork
Hurlbert SH (1978) The measurement of niche overlap and some relatives. Ecology 59:67–77. doi:10.2307/1936632
Hutchinson GE (1959) Homage to Santa Rosalia or why are there so many kinds of animals? Am Nat 93:145–159. doi:10.2307/2458768
Ihaka R, Gentleman R (1996) R: a language for data analysis and graphics. J Comput Graph Stat 5:299–314. doi:10.1080/10618600.1996.10474713
Johnson JB, Omland KS (2004) Model selection in ecology and evolution. Trends in Ecol Evol 19:101–108. doi:10.1016/j.tree.2003.10.013
Johnson CJ, Seip DR, Boyce MS (2004) A quantitative approach to conservation planning: using resource selection functions to map the distribution of mountain caribou at multiple spatial scales. J Appl Ecol 41:238–251. doi:10.1111/j.0021-8901.2004.00899.x
Keitt TH, Bjørnstad ON, Dixon PM, Citron-Pousty S (2002) Accounting for spatial pattern when modeling organism-environment interactions. Ecography 25:616–625. doi:10.1034/j.1600-0587.2002.250509.x
Knapp RA, Matthews KR, Preisler HK, Jellison R (2003) Developing probabilistic models to predict amphibian site occupancy in a patchy landscape. Ecol Appl 13:1069–1082. doi:10.1890/1051-0761(2003)13[1069:DPMTPA]2.0.CO;2
Kruuk H, De Kock L (1981) Food and habitat of badgers (Meles meles L.) on Monte Baldo, northern Italy. Z für Säugetierkd 46:295–301
Kruuk H, Parish T (1981) Feeding specialization of the European badger Meles meles in Scotland. J Anim Ecol 50:773–788. doi:10.2307/4136
Kruuk H, Parish T (1985) Food, food availability and weight of badgers (Meles meles) in relation to agricultural changes. J Appl Ecol 22:705–715. doi:10.2307/2403223
Kufner MB (1986) Tamaño actividad, densidad relativa y preferencias de hábitat de los pequeños y medianos mamíferos de Doñana como factores condicionantes de su tasa de predación. Universidad Autónoma de Madrid, Tesis doctoral
Kufner MB, Moreno S (1989) Abundancia y amplitud de los desplazamientos de Apodemus sylvaticus en cuatro biotopos de Doñana que difieren en cobertura vegetal. Doñana Acta Vertebr 16:179–181
Lidicker WZ, Koenig WD (1996) Responses of terrestrial vertebrates to habitat edges and corridors. In: McCullough DR (ed) Metapopulation and wildlife conservation. Island Press, Washington, DC, pp 85–109
Littell RC, Milliken GA, Stroup WW, Wolfinger RD (1996) SAS System for Mixed Models. SAS Institute Inc, Cary
Martin J, Basille M, Van Moorter B, Kindberg J, Allainé D, Swenson JE (2010) Coping with human disturbance: spatial and temporal tactics of the brown bear (Ursus arctos). Can J Zool 88:875–883. doi:10.1139/Z10-053
Martín R, Rodríguez A, Delibes M (1995) Local feeding specialization by badgers (Meles meles) in a mediterranean environment. Oecologia 101:45–50. doi:10.1007/BF00328898
McCullagh P, Nelder JA (1989) Generalized Linear Models, 2nd edn. Chapman and Hall, London
Metropolis N, Ulam S (1949) The monte carlo method. J Am Stat Assoc 44:335–341. doi:10.1080/01621459.1949.10483310
Miller J (2005) Incorporating spatial dependence in predictive vegetation models: residual interpolation methods. Prof Geogr 57:169–184. doi:10.1111/j.0033-0124.2005.00470.x
Moreno S, Villafuerte R (1997) Predation risk, cover type, and group size in European rabbits in Doñana (SW Spain). Acta Theriol 42:225–230
Palomares F (1986) Ecología de la gineta y del meloncillo en el Parque Nacional de Doñana. Universidad de Granada, Tesis de licenciatura
Palomares F (1994) Site fidelity and effects of body mass on home-range size of Egyptian mongooses. Can J Zool 72:465–469. doi:10.1139/z94-065
Palomares F (2001) Vegetation structure and prey abundance requirements of the Iberian lynx: implications for the design of reserves and corridors. J Appl Ecol 38:9–18
Palomares F, Delibes M (1993) Resting ecology and behaviour of Egyptian mongooses (Herpestes ichneumon) in southwestern Spain. J Zool 230:557–566. doi:10.1111/j.1469-7998.1993.tb02706.x
Palomares F, Revilla E (2002) Does local feeding specialization exist in Eurasian badgers? Can J Zool 80:83–93
Palomares F, Delibes M, Ferreras P, Fedriani JM, Calzada J, Revilla E (2000) Iberian lynx in a fragmented landscape: predispersal dispersal, and postdispersal habitats. Conserv Biol 14:809–818. doi:10.1046/j.1523-1739.2000.98539.x
Palomares F, Delibes M, Revilla E, Calzada J, Fedriani JM (2001) Spatial ecology of iberian lynx and abundance of European rabbits in southwestern Spain. Wildl Monogr :1–36. doi:10.2307/3830752
Perkins MW, Conner LM (2004) Habitat use of fox squirrels in southwestern Georgia. J Wildl Manage 68:509–513. doi:10.2193/0022-541X(2004)068[0509:HUOFSI]2.0.CO;2
Román J, Barón A, Revilla E (2010) Evaluación de los efectos del tránsito a motor sobre especies y comunidades de interés en el Espacio Natural de Doñana. Consejería de Medio Ambiente, Junta de Andalucía. Estación Biológica de Doñana, CSIC
Roper TJ, Lups P (1995) Diet of badgers (Meles meles) in central Switzerland—an analysis of stomach contents. Z Saugetierkd Int J Mamm Biol 60:9–19
Rosalino LM, Loureiro F, Macdonald DW, Santon-Reis M (2005) Dietary shifts of the badger (Meles meles) in Mediterranean woodlands: an opportunistic forager with seasonal specialisms. Mamm Biol Z Säugetierkd 70:12–23. doi:10.1078/1616-5047-00172
Rosenzweig ML, Abramsky Z, Brand S (1984) Estimating species interactions in heterogeneous environments. Oikos 43:329–340. doi:10.2307/3544150
Santos MJ, Pinto BM, Santos-Reis M (2007) Trophic niche partitioning between two native and two exotic carnivores in SW Portugal. Web Ecol 7:53–62. doi:10.5194/we-7-53-2007
Simon AL (1974) Dispersion and population interactions. Am Nat 108:207–228. doi:10.2307/2459851
Smith PA (1994) Autocorrelation in logistic regression modelling of species’ distributions. Glob Ecol Biogeogr Lett 4:47–61. doi:10.2307/2997753
Soto Navarro C, Desniça S, Palomares Fernández F (2012) Nonbiological factors affecting track censuses: implications for sampling design and reliability. Eur J Wildl Res 58:117–126. doi:10.1007/s10344-011-0551-9
Thioulouse J, Chessel D, Dole´dec S, Olivier JM (1997) ADE-4: a multivariate analysis and graphical display software. Stat Comput 7:75–83. doi:10.1023/A:1018513530268
Viota M, Rodríguez A, López-Bao JV, Palomares F (2012) Shift in microhabitat use as a mechanism allowing the coexistence of victim and killer carnivore predators. Open J Ecol 2:115–120. doi:10.4236/oje.2012.23014
Virgós E (2002) Are habitat generalists affected by forest fragmentation? A test with Eurasian badgers (Meles meles) in coarse-grained fragmented landscapes of central Spain. J Zool 258:313–318. doi:10.1017/S0952836902001449
Virgos E, Casanovas JG (1999) Environmental constraints at the edge of a species distribution, the Eurasian badger (Meles meles L.): a biogeographic approach. J Biogeogr 26:559–564. doi:10.2307/2656143
Virgós E, Llorente M, Cortésá Y (1999) Geographical variation in genet (Genetta genetta L.) diet: a literature review. Mamm Rev 29:117–126. doi:10.1046/j.1365-2907.1999.00041.x
Yamaguchi N, Rushton S, MacDonald DW (2003) Habitat preferences of feral American mink in the upper thames. J Mammal 84:1356–1373. doi:10.2307/1384021
Zapata S, Travaini A, Ferreras P, Delibes M (2007) Analysis of trophic structure of two carnivore assemblages by means of guild identification. Eur J Wildl Res 53:276–286. doi:10.1007/s10344-007-0095-1
Acknowledgments
This research was funded by the projects CGL2004-00346/BOS (Spanish Ministry of Education and Science) and 17/2005 (Spanish Ministry of the Environment; National Parks Research Program). Land-Rover Spain lent us two vehicles for this work. We are especially grateful to J. C. Rivilla and S. Desniça for assistance during fieldwork and to M. González and N. Fernández for their valuable suggestions on earlier versions of the manuscript. C. Soto was also supported by a JAE-Predoc grant from the Consejo Superior de Investigaciones Científicas.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Andreas Zedrosser.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Soto, C., Palomares, F. Coexistence of sympatric carnivores in relatively homogeneous Mediterranean landscapes: functional importance of habitat segregation at the fine-scale level. Oecologia 179, 223–235 (2015). https://doi.org/10.1007/s00442-015-3311-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00442-015-3311-9