Abstract
Isosteviol has been indicated as a cardiomyocyte protector. However, the underlying mechanism remains unclear. Thus, we sought to confirm the protective effect of isosteviol after myocardial infarction in a model of permanent coronary artery occlusion and investigate the potential proangiogenic activity in vitro and in vivo. A 4-week permanent coronary artery occlusion rat model was generated, and the protective effect of isosteviol was evaluated by echocardiographic imaging and hemodynamics assays. The coronary capillary density was tested by immunochemistry and micro-computed tomography (μCT) imaging. The effect of isosteviol on endothelial cells was determined in human umbilical vein endothelial cells (HUVECs) in vitro and Tg (kdrl: EGFP) zebrafish in vivo. We also examined the expression of related transcription factors by real-time polymerase chain reaction (RT-qPCR). Isosteviol increased ejection fraction (EF), fractional shortening (FS), cardiac systolic index (CI), maximum rate of increase of left ventricular pressure (Max dp/dt), and left ventricular systolic pressure (LVSP) by 32%, 40%, 25%, 26%, and 10%, respectively, in permanent coronary artery occlusion rats. Interestingly, it also promoted coronary capillary density by 2.5-fold. In addition, isosteviol promoted the proliferation and branching of HUVECs in vitro. It also rescued intersegmental vessel (ISV) development and improved endothelial cell proliferation by approximately fivefold (4–6) in zebrafish embryos in vivo. Isosteviol also upregulated the expression of hypoxia inducible factor-1α (HIF-1α) and vascular endothelial growth factor A (VEGFA) in zebrafish by fourfold and 3.5-fold, respectively. Our findings suggest that isosteviol is a proangiogenic agent and that this activity is related to its protective effects against myocardial ischemia.
Graphical abstract
After using the permanent coronary artery occlusion model, we demonstrated that isosteviol promotes angiogenesis directly and increases capillary density in myocardial ischemia rats. Isosteviol promotes angiogenesis in zebrafish in vivo and increases vascular endothelial cell proliferation in HUVECs and zebrafish. The angiogenesis activity of isosteviol may be correlated with VEGFA and HIF-1α signaling.

Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Myocardial ischemia (MI) is the leading cause of death and disability in the world (Liu et al. 2009; Hashimoto et al. 2018). Increasing angiogenesis could enrich coronary branching and restore the blood supply, which could benefit MI (Pagliaro et al. 2020), especially during cardiac remodeling after myocardial infarction (Goodwill et al. 2007). Hence, promoting angiogenesis may be a potential target for myocardial ischemia treatment. The research on proangiogenesis mainly focuses on either growth factors (Fu and Ou 2020) or gene therapy (Yuan et al. 2018). Vascular endothelial growth factor (VEGF) and fibroblast growth factor (FGF) as proangiogenic growth factors have been used in several clinical trials and achieved some good results in small-scale experiments (Lederman et al. 2002; Lazarous et al. 1996), while they also have obvious shortcomings, such as poor stability, short half-life, and considerable side effects (Timar et al. 2001). Gene therapy mainly involves recombination of various growth factor genes into vectors and delivery them into patients to activate downstream angiogenesis-promoting signaling pathways and the therapeutic effects are long-lasting (Albrecht-Schgoer et al. 2014). However, the introduced genes may integrate into the genome at unexpected sites, which may activate proto-oncogenes. The viral vector may induce inflammation or trigger an immune response (Eibel et al. 2017). Furthermore, no small molecule medicine targeting proangiogenesis has been approved. Thus, discovering novel small molecule medicines targeting angiogenesis is still needed.
Isosteviol, a worldwide food sweeter, is a potential drug candidate with many biological activities (Fan et al. 2016). In recent years, many studies have demonstrated that isosteviol has obvious therapeutic effects on heart and cerebrovascular diseases, including myocardial ischemia (Ke et al. 2021; Sun et al. 2018), diabetic heart disease (Tang et al. 2018), stroke (Yang et al. 2018), and myocardial hypertrophy (Liu et al. 2020). A recent study suggested that isosteviol protects heart against myocardial ischemia and reperfusion injury by remodeling cardiomyocyte metabolism (Mei et al. 2020). However, the underlying mechanism is still unclear. In this study, we discovered that coronary capillary density significantly increased after treatment with isosteviol in permanent coronary artery occlusion rats. We hypothesize that isosteviol may act as an angiogenesis activator in myocardial ischemia. Therefore, we further investigated the proangiogenic activity and the underlying mechanism in HUVECs and zebrafish.
Materials and methods
Echocardiography
Male Wistar rats (8–10 weeks of age) were purchased from Guangdong Medical Laboratory Animal Center and randomly grouped (n = 8–10). The permanent coronary artery occlusion model was generated by ligation operation with 10% chloral hydrate (0.3 ml/100 g) anesthetized. After ligation, 4 mg/kg isosteviol was administered twice a day by intraperitoneal injection, continuously for 4 weeks. In the sham and model group, the same volume of saline alone was injected as solvent control. Four weeks after treatment, transthoracic echocardiographic images were obtained by the VEVO 2100 high-resolution small animal ultrasound imaging system with a 30-MHz probe ms400 as previously described (Liu et al. 2014). Briefly, left ventricular end-diastolic posterior wall thickness (LVPW;d), left ventricular end-systolic posterior wall thickness (LVPW;s), left ventricular end-diastolic internal dimension (LVID;d) and left ventricular end-systolic internal dimension (LVID;s) were obtained by M-mode. Left ventricle volume diastole (LV Vol;d), left ventricle volume systole (LV Vol;s), EF, and FS were calculated automatically by the VEVO 2100 system.
Hemodynamics assays
Cardiac function was measured in rats anesthetized with 10% chloral hydrate (0.3 ml/100 g) as described before (Tang et al. 2018). Briefly, rats were sedated and mechanically ventilated; the hemodynamic function was recorded by inserting a microtip catheter equipped with Powerlab into left ventricle (LV) via the right carotid artery. LV systolic functions were evaluated by assessment CI, Max dp/dt, and LVSP in each study group.
Immunohistochemistry
Tissues were obtained from the hearts of permanent coronary artery occlusion rats and were paraformaldehyde-fixed, paraffin-embedded, and stained by immunohistochemistry for platelet endothelial cell adhesion molecule-1 (CD31, dilution 1:200) at 4 °C overnight and then incubated with biotin-conjugated goat anti-rabbit IgG (1:600) for 1 h at RT and then washed and stained with DAB kit for 5 min. At last, counterstaining was performed with hematoxylin and examined by light microscopy.
Micro-computed tomography imaging of cardiac vessels
The permanent coronary artery occlusion model was generated, and 4 mg/kg isosteviol was administered twice a day by intraperitoneal injection for 6 weeks. After treatment, μCT imaging was performed as previously described (Kivela et al. 2014). Briefly, the hearts were perfused with heparin (100 IU/kg) in 0.9% saline followed by adenosine (1 mg/ml) then perfusion-fixed with 4% paraformaldehyde, and the coronary arterial was filled with a contrast agent consisting of 20% bismuth oxychloride in 5% gelatin. The hearts were placed on ice to solidify the contrast medium for 30 min and imaged with a Small Animal CT Imaging System.
Cell viability
HUVECs were seeded at a density of 5 × 103 cells/well in a 96-well tissue culture dish and incubated at 37 °C in humidified conditions (5% CO2 and 95% air) with various concentrations of isosteviol; then, a CCK-8 assay was used to determine the cell viability following the manufacturer’s instructions and cells were visualized at 450 nm with an Enspire-2300 Multimode Reader.
Tube formation assay
The HUVECs were seeded at a density of 2 — × 104 cells/well on a 24-well culture dish. Each well was supplemented with 200 µl medium containing 10% serum and different concentrations of isosteviol. The plate was placed at 37 °C for 4 h and measured by microscope at 5 × magnification. Three random fields of vision were chosen in each well, and the images were captured using an AxioCam camera. These images were analyzed using Adobe Photoshop CS6 by creating a new transparent layer to cover the original layer in each image. In this new layer, the branches were traced to reveal outlines on the original cell images. The pixel value of the transparent layer was used to define the length of branches.
Zebrafish angiogenesis inhibition model
Zebrafish angiogenesis inhibition model was constructed as described before (Zhao et al. 2016). Briefly, 20 healthy Tg (kdrl: EGFP) zebrafish (http://zfin.org/ZDB-FISH-150901-14755) (Beis et al. 2005) embryos were selected and placed into a 24-well culture dish at 28.5 °C. Different concentrations of isosteviol were added together with 450 nmol/L VEGFR tyrosine kinase inhibitor II (VRI) at 24-h post fertilization (hpf) and then added again without VRI at 30 hpf. The embryos were observed under an Axioshop 2 Plus microscope at 72 hpf, and the images were captured using an AxioCam camera. These images were analyzed using Adobe Photoshop CS6. The pixel value of the transparent layer was used to define the length of ISV.
Fluorescence-activated cell sorting (FACS)
Tg (kdrl: EGFP) zebrafish embryos were used to establish this assay. At 72 hpf, all the embryos were homogenized and a mixture solution contains trypsin–EDTA and collagenase was added to make the embryos completely dissolved. Then, FBS was added to stop trypsin digestion, the cell suspensions were centrifuged and gently mixed, and this assay was determined by flow cytometry. In this study, wild-type zebrafish embryos were used as unstained control. P2 was gated based on the population in the unstained control (0.3%, close to 0%). The gating for model and isosteviol groups was determined by referring to the unstained control (supplemental Fig. 1). The flow cytometric analysis was performed using FlowJo 7.6.1.
Isosteviol improves systolic function in myocardial ischemia rats. The permanent coronary artery occlusion model was generated by ligation operation, and after 4 weeks of treatment, transthoracic echocardiographic images and hemodynamic function were recorded by the VEVO 2100 high-resolution small animal ultrasound imaging system and Powerlab respectively. (a–a″) Echocardiography of rat heart demonstrated that isosteviol rescued systolic function. (b) Ejection fraction (EF). (c) Fractional shortening (FS). (d) Cardiac systolic index (CI). (e) Maximum rate of increase of left ventricular pressure (Max dp/dt). (f) Left ventricular systolic pressure (LVSP). **P < 0.01, vs model group, #P < 0.05, ##P < 0.05 vs sham group, n = 8–10 per group
RNA isolation and real-time PCR analysis
HUVECs were seeded at a density of 2 × 104 cells/well in a 24-well tissue culture dish and incubated at 37 °C in humidified conditions (5% CO2 and 95% air) with various concentrations of isosteviol for 24 h. Trizol reagent was used to isolating total RNA from HUVECs cells followed by cDNA synthesis using the M-MLV 1st Strand Kit from Invitrogen. Quantitative real-time PCR was performed with the SYBR Green Mix. The mRNA levels of the relevant markers of angiogenesis, such as HIF-1α, VEGFA, and Notch homolog 1, translocation-associated (Drosophila) (Notch1) were compared against the level of β-actin with 2−△△Ct cycle threshold method. The sequences of the primer used are listed in Table 1.
Statistical Analysis
Statistical analysis was performed using GraphPad Prism 5 (GraphPad Software). Statistical analysis of multiply groups was performed with one-way ANOVA, followed by Tukey or Bonferroni post hoc tests. A P value of less than 0.05 was considered statistically significant. Measurements are expressed as means ± standard errors of the mean (SEM).
Results
Isosteviol improves systolic function in myocardial ischemia rats
To evaluate the potential cardioprotective activity of isosteviol in myocardial ischemia rats, we assessed systolic function by echocardiography and hemodynamics assays. Echocardiography of rat heart demonstrated that isosteviol rescued systolic function (Fig. 1(a–a″)). There was no significant change of LVPW;d, LVPW;s, and LVID;d after MI or isosteviol treatment (supplemental Fig. 2a–c). The LVID;s increased by 48% (#P < 0.05) in the model group compared with the sham group, and isosteviol decreased it by 15% compared with the model group, but had no significant difference (supplemental Fig. 2d). The ejection fraction and fractional shortening in the model group were 65% and 38%, respectively. Isosteviol rescued EF and FS to 86% (*P < 0.05) (Fig. 1(b)) and 53% (*P < 0.05) respectively (Fig. 1(c)).
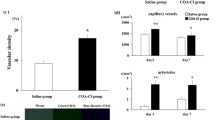
Isosteviol improves coronary angiogenesis in myocardial ischemia rats. a–c CD31 was stained to determine the tube number by immunohistochemistry; the arrows point to the coronary capillary; the bar is 20 × magnification. d Average tube number per section of different group. Isosteviol increased capillary significantly. ***P < 0.001, vs model group. n = 6 per group. e–h μCT was used to further determine the effect of isosteviol on arterial. The magnification image is 40 × . n = 4–5 per group
The hemodynamic function was recorded by inserting a micro tip catheter equipped with Powerlab into left ventricle (LV) via the right carotid artery. As shown in Fig. 1(d–f), the CI, Max dp/dt, and LVSP in the model group were 182 1/s, 9305 mmHg/s, and 93 mmHg,respectively, and isosteviol rescued them by 25% (**P < 0.01), 26% (**P < 0.01), and 10% (**P < 0.01) respectively. Therefore, a significant correlation was found between isosteviol and cardioprotective activity.
Isosteviol improves coronary angiogenesis in myocardial ischemia rats
Interestingly, we found that the coronary capillary density increased after treatment with isosteviol in myocardial ischemia rats. CD31 was stained to determine the tube number by immunohistochemistry. As shown in Fig. 2a–c, the arrow indicates the blood vessels stained by immunohistochemistry. Isosteviol increased the average tube number by 2.5-fold compared with the model group (***P < 0.001; Fig. 2d). To determine the effects of isosteviol on the entire coronary arterial, we filled coronary arteries with a contrast agent and analyzed them with µCT. The results indicated that isosteviol significantly increased coronary angiogenesis compared with the model group (Fig. 2e–h), which is consistent with the immunohistochemistry results. However, RQ was similar in all groups (supplemental Fig. 3a). Isosteviol moderately rescued EE compared with model group but had no significant difference (supplemental Fig. 3b).
Isosteviol enhances angiogenesis in HUVECs in vitro. Three random fields of vision were chosen in each well, and the images were captured using an AxioCam camera. These images were analyzed using Adobe Photoshop CS6 by creating a new transparent layer to cover the original layer in each image. In the new layer, the branches were traced to reveal outlines on the original cell images. The pixel value of the transparent layer was used to define the length of branches. a–d Differentiation of HUVECs in three-dimensional Matrigel culture with isosteviol (0 µmol/L, 1.25 µmol/L, 5 µmol/L, 20 µmol/L); the bar is 200 µm. The arrows point to the branching sprouts. e Effects of isosteviol on the proliferation of HUVECs. *P < 0.05, vs blank group, n = 4 per group. f Increased branch length in isosteviol-treated HUVECs. ***P < 0.001, vs blank group, n = 18 per group. Three individual experiments repeat. g Increased branch numbers in isosteviol-treated HUVECs. ***P < 0.001, vs blank group, n = 18 per group. Three individual experiments were conducted
Isosteviol promotes angiogenesis in zebrafish in vivo. Zebrafish angiogenesis inhibition model was constructed, and the embryos were observed under an Axioshop 2 Plus microscope at 72 hpf, and the images were captured using an AxioCam camera. These images were analyzed using Adobe Photoshop CS6. The pixel value of the transparent layer was used to define the length of ISV. (a) Experiment procedure of the zebrafish angiogenesis evaluation of isosteviol. (b–e) Different groups of zebrafish embryos at 72 hpf. Model (b–b″), 3.125 µmol/L isosteviol (c–c″), 12.5 µmol/L isosteviol (d–d″), and 50 µmol/L isosteviol (e–e″). The bar is 500 µm. The arrows point to sprouts of the intersegmental blood vessels (ISV). The magnification image is 10 × . n = 20 per group. Three individual experiments were conducted. (f) More than three ISV sprouts as recovered embryos, and the recovery rate was measured. The graph shows ISV-recovered rate. n = 20 per group. (g) Average branch length in different groups. (h) Average branch numbers in different groups. ***P < 0.001, **P < 0.01, *P < 0.05, vs model group, n = 20 per group
Isosteviol increases vascular endothelial cell proliferation in zebrafish. Tg (kdrl: EGFP) zebrafish embryos were used to conduct this assay, wild type zebrafish embryos were used as unstained control. P2 was gated based on the population in the unstained control. The flow cytometric analysis was performed using FlowJo 7.6.1. (a–a″) The red points represent the cells, and the points in box P2 are fluorescent cells in different group. The scatter plots show the percentage of fluorescent intensity in the model (450 nmol/L VRI) and isosteviol (12.5 µmol/L and 50 µmol/L) groups. Three individual experiments repeat. (b) The bar graph displays the average fluorescent intensity after treatment in different groups. (c) The fluorescent intensity of overlay histograms is shown. The model group (red line) on the leftmost had the lowest fluorescent intensity, while the 12.5 µmol/L isosteviol group (blue line) on the rightmost had the highest
Isosteviol increases the expression of HIF-1α, VEGFA, and Notch1. The mRNA levels of HIF-1α (a), VEGFA (b), and Notch1 (c) in HUVECs cells were conducted by RT-qPCR. The values shown in this graph represent means ± S.D. from three independent experiments. *P < 0.05, **P < 0.01 vs blank group. (d–d″) Mechanistic illustration of isosteviol improves cardiac function and promotes angiogenesis by improving vascular endothelial cell proliferation and tube formation
Isosteviol enhances angiogenesis in HUVECs in vitro
The process of angiogenesis is complicated, mainly including proliferation and tube formation. Thus, we first used HUVECs to examine the effect of isosteviol on proliferation by the CCK8 assay. Isosteviol at 0.3125 μmol/L, 1.25 μmol/L, and 5 μmol/L increased the cell viability by 25.6% (*P < 0.05), 26.1% (*P < 0.05), and 28.8% (*P < 0.05), respectively, compared with the blank group. No significant increase in the cell viability was observed in the group treated with 20 μmol/L isosteviol (Fig. 3e). Thus, isosteviol improves the proliferation of HUVECs.
We further tested the tube formation of HUVECs after treatment with different concentrations of isosteviol. As shown in Fig. 3a–d, the arrows point to the newly formed branches. The branch length in 1.25 μmol/L and 5 μmol/L isosteviol groups increased by 80% (***P < 0.001) and 93% (***P < 0.001), respectively, compared with the blank control, but no significant difference was observed in the 20 μmol/L isosteviol group (Fig. 3f). As shown in Fig. 3g, Isosteviol at 1.25 μmol/L, 5 μmol/L, and 20 μmol/L increased the branch number by 34% (***P < 0.001), 38% (***P < 0.001), and 46% (***P < 0.001), respectively, compared with the blank control. Thus, isosteviol enhances the branches of HUVECs. Overall, no matter the tube length or tube number, isosteviol was confirmed to improve angiogenesis in HUVECs in vitro.
Isosteviol promotes angiogenesis in zebrafish in vivo
After experiments with HUVECs in vitro, the Tg (kdrl: EGFP) zebrafish was used as an in vivo model to evaluate the angiogenesis effects of isosteviol. As shown in Fig. 4(a), we first treated embryos with VRI and isosteviol for 6 h (24 hpf to 30 hpf) to prepare zebrafish embryos that had sprouting of intersegmental blood vessels (ISV) pre-inhibited. Then VRI was washed away, and isosteviol (0 µmol/L, 3.125 µmol/L, 12.5 µmol/L, 50 µmol/L) was added. As shown in Fig. 4(b–b″), the model group induced a loss of almost all their ISV (20/20) and a majority of the head vessels (20/20) at 72 hpf. As shown in Fig. 4(c–e″), we observed that ISV sprouts extended from the dorsal aorta and head vessels were recovered after treatment with different concentrations of isosteviol. These results indicated that isosteviol promotes angiogenesis. We defined embryos with more than three ISV sprouts as recovered embryos, and the recovery rate (recovered embryos count/total embryos) was measured. Isosteviol at 3.125 μmol/L, 12.5 μmol/L, and 50 μmol/L increased the recovery rate by 2.4-fold, 2.4-fold, and 3.1-fold, respectively, compared with the model group (Fig. 4(f)). The tube length in the 3.125 μmol/L, 12.5 μmol/L, and 50 μmol/L isosteviol group increased by fourfold (*P < 0.05), sixfold (***P < 0.001), and fivefold (**P < 0.01), respectively, compared with the model group (Fig. 4(g)). Isosteviol also increased the tube number by twofold (*P < 0.05), fourfold (***P < 0.001), and threefold (**P < 0.01), respectively, compared with the model group (Fig. 4(h)). These results indicate that isosteviol improved angiogenesis in zebrafish embryos no matter in tube length or tube number, consistent with the data in vitro.
Isosteviol increases vascular endothelial cell proliferation in zebrafish
Angiogenesis is a complicated process, mainly including proliferation and tube formation. Since we demonstrated that isosteviol promotes angiogenesis in zebrafish at 72 hpf, we investigated the process involved in isosteviol mediated angiogenesis. Endothelial cell count is one of the major ways to quantify proliferation. Thus, we performed flow cytometry at 72 hpf to measure endothelial cell count. Tg (kdrl: EGFP) zebrafish embryos were used to conduct this assay, and the wild-type zebrafish embryos were used as unstained control. In Fig. 5(a–a″), the red points represent the cells and the points in box P2 are fluorescent cells. Isosteviol at 12.5 µmol/L and 50 µmol/L increased the fluorescent percentage to 10% and 9%, respectively, compared with the model group (3.9%). Isosteviol at 12.5 µmol/L and 50 µmol/L increased fluorescent intensity by 62% and 16%, respectively, compared with the model group (Fig. 5(b)). In Fig. 5(c), the fluorescent intensity of overlay histograms is shown. The model group (red line) on the left most had the lowest fluorescent intensity, while the 12.5 µmol/L isosteviol group (blue line) on the right most had the highest. Thus, these results demonstrated that isosteviol improves endothelial cell proliferation.
Isosteviol upregulates the expression of HIF-1α, VEGFA, and Notch1
To study the potential molecular pathway of the proangiogenic activity of isosteviol, we examined the expression of HIF-1α, VEGFA, and Notch1 factors for angiogenesis using RT-qPCR. HIF-1α and VEGFA in the group after treatment with 5 µmol/L isosteviol increased by fourfold (**P < 0.01) and threefold (*P < 0.05), respectively, compared with the blank control; however, there is no significant increase in HIF-1α and VEGFA observed in 1.25 µmol/L and 20 µmol/L isosteviol group (Fig. 6(a–b)). Notch1 had a 1.5-fold increase in the 5 µmol/L isosteviol group compared with the blank group but had no significant difference (Fig. 6(c)). Isosteviol improves cardiac function and promotes angiogenesis by improving the vascular endothelial cell proliferation and tube formation through upregulation of HIF-1α and VEGFA expression but not Notch1 (Fig. 6(d–d″)).
Discussion
Myocardial ischemia is a severe threat to human health and life [1]. Even though there are various drugs targets (De Vries et al. 2018), most of them have unavoidable limitations. For example, β-receptor blocker reduces myocardial oxygen consumption. Meanwhile, it may inhibit cardiac function (Garcia-Prieto et al. 2017). Nitrates may produce drug resistance in the long term (Muenzel and Daiber 2018). Thus, the development of novel drugs with new targets for myocardial ischemia is still in need.
Isosteviol is a cardiomyocyte protector (Ke et al. 2021; Sun et al. 2018; Liu et al. 2020; Mei et al. 2020; Chen et al. 2019), but the underlying mechanism is still unclear. No reports have demonstrated if isosteviol or its analogues may directly interact with the vascular endothelial cells. Our previous studies indicated that isosteviol and its analogues might increase mitochondrial function and prevent reactive oxygen species damage in cardiomyocytes (Liu et al. 2020; Zhang et al. 2019); in this study, we report that isosteviol improved cardiac function in permanent coronary artery occlusion by improving the vascular endothelial cell proliferation and tube formation.
Furthermore, a higher density of coronary capillary was observed after isosteviol treatment. Thus, we hypothesize that isosteviol may directly promote angiogenesis based on this result, and that the newly formed capillaries may support sufficient oxygen and nutrition and compensate for the energy consumed by increasing mitochondrial function. However, there are obvious differences between rat and human heart coronary arteries. Thus, it is necessary to investigate the effect of isosteviol on promoting coronary angiogenesis in pigs and other mammals.
Angiogenesis, a complicated process of forming blood vessels from existing vessels (Risau et al. 1997), plays a critical role in various biological processes such as embryonic development, tissue repair, and wound healing (Nowak-Sliwinska et al. 2018) Currently, proangiogenic therapy trials have focused on the use of growth factors or gene therapy. Toldo et al. (2016) demonstrated that the recombinant human AAT-Fc reduced the acute myocardial inflammatory injury after ischemia–reperfusion in the mouse. Yang et al. (2015) found that the modified VEGF decreased scar size, enhanced angiogenesis, and improved cardiac function. Therefore, angiogenesis could be a potential therapy for cardiovascular diseases including myocardial ischemia (Mitsos et al. 2012), and small molecules with different functions that enhance angiogenesis have become another direction for therapeutic strategy (Huang et al. 2012). Considering the new finding that isosteviol promotes coronary capillary density, we hypothesize that isosteviol may directly improve angiogenesis. Thus, we further evaluated this activity in vitro and in vivo.
Our experimental results indicated that isosteviol markedly improved the proliferation and branch formation of HUVECs in vitro. Isosteviol enhanced endothelial cell proliferation. Likewise, isosteviol treatment caused a noticeable increase in the number of branches. Furthermore, isosteviol treatment in zebrafish embryos also induced an extraordinary proangiogenic phenotype, in which longer and more vessels were formed. These data clearly demonstrated that the isosteviol promotes endothelial cell proliferation, sprouting, and branching in HUVECs in vitro and zebrafish in vivo. In addition, the endothelial cell counts of zebrafish were determined by flow cytometry. The results suggested that isosteviol increased endothelial cell proliferation, further examining the angiogenic activity of isosteviol.
However, the molecular mechanism is still unclear. Our experimental results suggested that isosteviol may promote the expression of HIF-1α and VEGFA. As VEGFA is a master regulator of angiogenesis (Milincovici et al. 2018), the upregulation of VEGFA may be the core factor responsible for this activity. Interestingly, HIF-1α, a well-known hypoxia-sensitive pathway regulator that plays a vital role in proangiogenesis (Serocki et al. 2018), was significantly increased. In addition, HIF-1α is an antioxidant on ischemia injure via targeting mitochondria (Li et al. 2019; Jiang et al. 2019). Thus, the up-regulation of HIF-1α may be a considerable mechanism for the anti-oxygen stress activity of isosteviol.
Conclusion
In conclusion, our study, for the first time, demonstrated that isosteviol promotes angiogenesis directly and increases capillary density in myocardial ischemia rats. Isosteviol also ameliorates cardiac function by improving vascular endothelial cell proliferation and tube formation. The angiogenesis activity of isosteviol may be correlated with VEGFA and HIF-1α signaling.
References
Albrecht-Schgoer K, Schgoer W, Theurl M et al (2014) Topical secretoneurin gene therapy accelerates diabetic wound healing by interaction between heparan-sulfate proteoglycans and basic FGF. Angiogenesis 17(1):27–36
Beis D, Bartman T, Jin SW et al (2005) Genetic and cellular analyses of zebrafish atrioventricular cushion and valve development. Development 132(18):4193–4204
Chen Y, Beng H, Su H et al (2019) Isosteviol prevents the development of isoprenaline induced myocardial hypertrophy. Int J Mol Med 44(5):1932–1942
De Vries M, Seppala LJ, Daams JG et al (2018) Fall-risk-increasing drugs: a systematic review and meta-analysis: I. Cardiovascular drugs. J Am Med Dir Assoc 19(4), 371:1–9
Eibel B, Markoski MM, Rodrigues CG et al (2017) VEGF Gene Therapy Cooperatively Recruits Molecules from the Immune System and Stimulates Cell Homing and Angiogenesis in Refractory Angina. Cytokine 91:44–50
Fan Z, Wen T, Chen Y et al (2016) Isosteviol sensitizes sarcK(ATP) channels towards Pinacidil and potentiates mitochondrial uncoupling of diazoxide in guinea pig ventricular myocytes. Oxid Med Cell Longev 2016:6362812
Fu X, Ou B (2020) miR-152/LIN28B axis modulates high-glucose-induced angiogenesis in human retinal endothelial cells via VEGF signaling. J Cell Biochem 121(2):954–962
Garcia-Prieto J, Villena-Gutierrez R, Gomez M et al (2017) Neutrophil stunning by metoprolol reduces infarct size. Nat Commun 4(8):14780
Goodwill AG, Dick GM, Kiel AM et al (2007) Regulation of coronary blood flow. Compr Physiol 7(2):321–382
Hashimoto H, Olson EN, Bassel-Duby R (2018) Therapeutic approaches for cardiac regeneration and repair. Nat Rev Cardiol 15(10):585–600
Huang H, Lindgren A, Wu X et al (2012) High-throughput screening for bioactive molecules using primary cell culture of transgenic zebrafish embryos. Cell Rep 2(3):695–704
Jiang L, Zeng H, Ni L et al (2019) HIF-1α preconditioning potentiates antioxidant activity in ischemic injury: the role of sequential administration of dihydrotanshinone I and protocatechuic aldehyde in cardioprotection. Antioxid Redox Signal 31(3):227–242
Ke Q, Liu F, Tang Y et al (2021) The protective effect of isosteviol sodiumon cardiac function and myocardial remodeling in transverse aortic constriction rat. J Cell Mol Med 25(2):1166–1177
Kivela R, Bry M, Robciuc MR et al (2014) VEGF-B-induced vascular growth leads to metabolic reprogramming and ischemia resistance in the heart. EMBO Mol Med 6(3):307–321
Lazarous DF, Shou M, Scheinowitz M et al (1996) Comparative effects of basic fibroblast growth factor and vascular endothelial growth factor on coronary collateral development and the arterial response to injury. Circulation 94(5):1074–1082
Lederman RJ, Mendelsohn FO, Anderson RD et al (2002) Therapeutic angiogenesis with recombinant fibroblast growth factor-2 for intermittent claudication (the TRAFFIC study): a randomized trial. Lancet 359(9323):2053–2058
Li H, Zhou Y, Li L et al (2019) HIF-1α protects against oxidative stress by directly targeting mitochondria. Redox Biol vol. 25 101109
Liu F, Su H, Liu B et al (2020) STVNa attenuates isoproterenol-induced cardiac hypertrophy response through the HDAC4 and Prdx2/ROS/Trx1 pathways. Int J Mol Sci 21:6822
Liu N, Wu M, Chen C et al (2009) Novel molecular targets participating in myocardial ischemia-reperfusion injury and cardioprotection. Cardiol Res Pract 2019:6935147
Liu Y, Asnani A, Zou L et al (2014) Visnagin protects against doxorubicin-induced cardiomyopathy through modulation of mitochondrial malate dehydrogenase. Sci Transl Med 6:70266
Mei Y, Liu B, Su H et al (2020) Isosteviol sodium protects the cardiomyocyte response associated with the SIRT1/PGC-1 alpha pathway. J Cell Mol Med 24(18):10866–10875
Melincovici CS, Bosca AB, Susman S et al (2018) Vascular endothelial growth factor (VEGF) — key factor in normal and pathological angiogenesis. Rom J Morphol Embryol 2:455–467
Mitsos S, Katsanos K, Koletsis E et al (2012) Therapeutic angiogenesis for myocardial ischemia revisited: basic biological concepts and focus on latest clinical trials. Angiogenesis 15(1):1–22
Muenzel T, Daiber A (2018) Inorganic nitrite and nitrate in cardiovascular therapy: a better alternative to organic nitrates as nitric oxide donors? Vascul Pharmacol 102:1–10
Nowak-Sliwinska P, Alitalo K, Allen E et al (2018) Consensus guidelines for the use and interpretation of angiogenesis assays. Angiogenesis 21(3):425–532
Pagliaro BR, Cannata F, Stefanini GG et al (2020) Myocardial ischemia and coronary disease in heart failure. Heart Fail Rev 25(1SI):53–65
Risau W (1997) Mechanisms of angiogenesis. Nature 386(6626):671–674
Serocki M, Bartoszewska S, Janaszak-Jasiecka A et al (2018) MiRNAs regulate the HIF switch during hypoxia: a novel therapeutic target. Angiogenesis 21(2):183–202
Sun X, Yang Y, Xie Y et al (2018) Protective role of STVNa in myocardial ischemia reperfusion injury by inhibiting mitochondrial fission. Oncotarget 9(2):1898–1905
Tang S, Liu X, Ye J et al (2018) Isosteviol ameliorates diabetic cardiomyopathy in rats by inhibiting ERK and NF-kappa B signaling pathways. J Endocrinol 238(1):47–60
Timar J, Dome B, Fazekas K et al (2001) Angiogenesis-dependent diseases and angiogenesis therapy. Pathol Oncol Res: POR 7(2):85–94
Toldo S, Mauro AG, Marchetti C et al (2016) Recombinant HumanAlpha-1 Antitrypsin-Fc fusion protein reduces mouse myocardial inflammatory injury after ischemia-reperfusion independent of elastase inhibition. J Cardiovasc Pharmacol 68(1):27–32
Yang Y, Shi C, Hou X et al (2015) Modified VEGF targets the ischemic myocardium and promotes functional recovery after myocardial infarction. J Control Release 213:27–35
Yang Y, Zhong Q, Zhang H et al (2018) Lipidomics study of the protective effects of isosteviol sodium on stroke rats using ultra high-performance supercritical fluid chromatography coupling with ion-trap and time-of-flight tandem mass spectrometry. J Pharm Biomed Anal 157:145–155
Yuan R, Xin Q, Shi W et al (2018) Vascular endothelial growth factor gene transfer therapy for coronary artery disease: a systematic review and meta-analysis. Cardiovasc Ther 36:e124615
Zhang H, Liu B, Xu G et al (2019) Synthesis and in vivo screening of isosteviol derivatives as new cardioprotective agents. Eur J Med Chem vol. 219 2021:113396
Zhao H, Osborne OJ, Lin S et al (2016) Lanthanide hydroxide nanoparticles induce angiogenesis via ROS-sensitive signaling. Small 12(32):4404–4411
Acknowledgements
We thank Dr. Khaja Shameem Mohammed Abdul and Dr. Shanping Wang for his assistance with the language editing in the manuscript.
Funding
This study was funded by the National Natural Science Foundation of China (Grant No. 81903600 and No.81802123), China Postdoctoral Science Foundation (Grant No. 2018M643026, the Science and Technology Innovation Project of Foshan (Grant No. 2017IT100162), and Technology Major Projects for “Major New Drugs Innovation and Development (2019ZX09301120).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical approval
All procedures performed in the studies involving animals were in accordance with the ethical standards of the Institutional Animal Care and Use Committee of Guangdong Pharmaceutical University (Approval No. 20180714–03).
Informed consent
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
441_2021_3559_MOESM1_ESM.tif
Supplementary file1 Fig. 1 The embryos of wild type and Tg (kdrl: EGFP) zebrafish were used for gating in flow cytometry. (a) The embryos of wild type zebrafish were used as unstained control. P2 was gated based on the population in the unstained control (0.3%, close to 0%). (b) The gating for Tg (kdrl: EGFP) zebrafish groups was determined by referring to the unstained control. The representative image of isosteviol shown that the fluorescent intensity increased (7%) (TIF 1721 KB)
441_2021_3559_MOESM2_ESM.tif
Supplementary file2 Fig. 2 The left ventricular wall thickness and dimension had no significant difference after treatment with isosteviol. (a) Left ventricular end-diastolic posterior wall thickness (LVPW;d) had no significant difference after treatment with isosteviol. n = 8–10 per group. (b) Left ventricular end-systolic posterior wall thickness (LVPW;s) had no significant change after treatment with isosteviol. n = 8–10 per group. (c) Left ventricular end-diastolic internal dimension (LVID;d) had no significant difference after treatment with isosteviol. n = 8–10 per group; (d) Left ventricular end-systolic internal dimension (LVID;s) decreased in the isosteviol group compared with the model group but had no significant difference. #P < 0.05, vs sham group n = 8–10 per group (TIF 3786 KB)
441_2021_3559_MOESM3_ESM.tif
Supplementary file3 Fig. 3 The energy statue had no significant difference after treatment with isosteviol. (a) Respiratory quotient (RQ) was similar in all groups; (b) Isosteviol moderately rescued EE compared with model group but had no significant difference. n = 8–10 per group (TIF 1964 KB)
Rights and permissions
About this article
Cite this article
Liu, F., Song, L., Lu, Z. et al. Isosteviol improves cardiac function and promotes angiogenesis after myocardial infarction in rats. Cell Tissue Res 387, 275–285 (2022). https://doi.org/10.1007/s00441-021-03559-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-021-03559-9