Abstract
Liver fibrogenesis is a dynamic process including quantitative and qualitative changes of the extracellular matrix, of which the most prominent is the deposition of type I collagen. These changes progressively disrupt normal liver architecture and result in cirrhosis formation. In the fibrotic liver, as in all other fibrotic tissues, the extracellular matrix is produced by cells usually characterized by the de novo expression of alpha-smooth muscle actin and known as myofibroblasts. Portal myofibroblasts (PMFs) appear to be critical in pathological angiogenesis, which constantly occurs in advanced liver fibrosis. Whereas the association between angiogenesis and fibrosis during the progression of liver diseases remains to be elucidated, we suggest that collagen-type-XV-alpha1-producing PMFs could provide an important link both by stabilizing newly formed vessels and by forming a scaffold for the deposition of interstitial collagen.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The incidence of chronic liver diseases keeps growing worldwide, largely because of the epidemic of metabolic syndrome and non-alcoholic fatty liver disease (NAFLD). To a great extent, the prognosis of chronic liver diseases is determined by the development of liver fibrosis. However, no anti-fibrotic drug that would prevent the progression of liver fibrosis towards cirrhosis is yet available and a better understanding of fibrogenesis in the liver is still needed. Liver fibrogenesis is a dynamic process including quantitative and qualitative changes of the extracellular matrix, of which the most prominent is the deposition of type I collagen. These changes progressively disrupt normal liver architecture and result in cirrhosis formation. In the fibrotic liver, as in all other fibrotic tissues, the extracellular matrix is produced by cells that are usually characterized by the de novo expression of alpha-smooth muscle actin (α-SMA) and that are known as myofibroblasts.
Liver myofibroblasts
Myofibroblasts form heterogeneous populations of cells, with different possible origins. Current evidence indicates that, in the liver, myofibroblasts are mainly derived from hepatic stellate cells (HSCs; Mederacke, et al. 2013). However, cells that are distinct from HSCs and located in the portal tract can also give rise to myofibroblasts that we now globally refer to as portal myofibroblasts (PMFs; Lemoinne et al. 2015; Lua et al. 2016). PMFs were first described in the setting of biliary-type liver fibrosis such as that induced by bile duct ligation (BDL) in rats or mice (Kinnman et al. 2003). PMFs outnumber hepatic-stellate-cell-derived myofibroblasts (HSC-MFs) at the onset of biliary-type liver injury (Beaussier et al. 2007) contributing by more than 70 % to liver myofibroblasts at 5 days after BDL (Iwaisako et al. 2014). Simultaneously, in sinusoids, HSCs undergo phenotypic changes including the overexpression of desmin and of platelet-derived growth factor receptor-beta (PDGFR-β) and increased DNA synthesis but yet without fully converting into myofibroblasts at this stage (Beaussier et al. 2007). Studies of PMFs in culture have demonstrated that they require transforming growth factor-beta (TGF-β) and that they are also dependent on mechanical tension for myofibroblastic differentiation (Li et al. 2007), a typical feature of myofibroblasts (Eyden 2008). In vivo, the emergence and expansion of PMFs can be triggered by interactions with ductular epithelial cells, which proliferate after bile duct injury in the so-called ductular reaction. Thus, a marked increase in the expression of αvβ6 integrin on the surface of ductular epithelial cells is induced, in mice, by acute bile duct obstruction, which directly triggers the periductal accumulation of myofibroblasts and fibrosis, through the activation of TGF-β (Wang et al. 2007). In human liver tissues, the expression of αvβ6 in ductular epithelial cells has also been found to be increased in acute but not chronic, biliary-type injury (Wang et al. 2007). Reactive ductules also express profibrogenic factors such as TGF-β itself or platelet-derived growth factor-BB (PDGF-BB), which also stimulates PMF expansion (Kinnman et al. 2003). Nevertheless, the biology of PMFs remains poorly known, mainly because of the lack of markers that would allow investigators to authenticate and distinguish them from HSC-MFs in the injured liver. In a majority of studies, HSC-MFs have been identified on the unique basis of their expression of α-SMA, one of the many markers that they have in common with PMFs. Therefore, the contribution of PMFs might have been underestimated and some of their properties improperly allocated to HSC-MFs. By comparing the transcriptome of PMFs and HSC-MFs, we have identified a marker of PMFs, almost not expressed in HSC-MFs or in any other liver cell type, i.e., collagen type XV alpha1 (COL15A1; Lemoinne et al. 2015) and this result has subsequently been confirmed by another group (Lua et al. 2016). We have shown that, both in animal models and in patients with chronic liver diseases, a marked increase in the hepatic expression of COL15A1 occurs at the time of progression to advanced liver fibrosis (Lemoinne et al. 2015). This is the case not only in biliary-type liver fibrosis (i.e., bile-duct-ligated rats and patients with primary biliary cholangitis) but also in post-necrotic liver fibrosis (i.e., carbon-tetrachloride-treated rats and patients with NAFLD), suggesting that PMFs or at least sub-populations of PMFs are involved in the progression of all types of liver fibrosis. In addition to α-SMA expression, myofibroblasts are characterized by ultrastructural features including a prominent rough endoplasmic reticulum, a Golgi apparatus producing collagen secretory granules and peripheral myofilaments (Eyden 2008). Yet another highly characteristic trait is a cell-to-matrix junction that consists in an aligned myofilament bundle and a fibronectin fibril contacting one another through a point at the cell surface termed a fibronexus (Eyden 2008). Of particular interest in this respect, we have found, in our previous comparative analyses, that PMFs compared with HSC-MFs express fibronectin at higher levels and virtually no desmin, both features belonging to the definition of myofibroblasts (Eyden 2008). Therefore, PMFs fulfil more criteria that define myofibroblasts than do HSC-MFs (Eyden 2008).
Liver angiogenesis
Angiogenesis is a dynamic process leading to the formation of news vessels from pre-existing vessels. In all tissues, angiogenesis is determined by two main pathways, i.e., hypoxia and inflammation and comprises the following steps: (1) sprouting and budding of endothelial cells; (2) degradation of the extracellular matrix by proteinases and migration of endothelial cells; (3) endothelial cell proliferation, tube formation and branching; and (4) vessel maintenance, maturation and stabilization (Elpek 2015). Vascular endothelial growth factor (VEGF) signaling has been implicated in virtually all steps of angiogenesis, whereas the recruitment of mural pericyte-like cells is required in the last step for nascent vessels to mature and stabilize. As is now well established, liver disease progression is accompanied by angiogenesis. The first demonstration was provided more than thirty years ago by Rappaport et al. (1983) who showed that human cirrhotic livers contained more vessels than healthy livers and that cirrhotic nodules were surrounded by a dense vascular plexus. Liver tissue hypoxia occurs at early stages of liver injury and increases with disease progression as a result of the structural and functional changes in the liver angioarchitecture (Corpechot et al. 2002). During liver fibrogenesis, fibrillar type I collagen progressively replaces type IV collagen in the perisinusoidal space of Disse; this replacement, together with the loss of endothelial fenestrations, causes sinusoidal capillarization. Therefore, fibrosis by itself can contribute to the development of hypoxia and, thereby, can promote angiogenesis. Vascular remodeling leading to the capillarization of the sinusoids and the generation of intrahepatic shunts characterizes hepatic angiogenesis. Such changes in angioarchitecture cause a decrease in hepatocyte perfusion that aggravates hypoxia. Liver angiogenesis is also stimulated by inflammation. The chemokine-dependent accumulation of monocyte-derived macrophages is an important mechanism of hepatic inflammation and fibrogenesis in human liver diseases and experimental mouse models. The chemokine receptor CCR2 and its ligand CCL2 (MCP-1) promote the accumulation of monocyte-derived macrophages releasing pro-inflammatory and pro-angiogenic cytokines in the liver. Infiltrating CCL2-dependent inflammatory monocytes also provide pro-angiogenic signals via the production of VEGF-A and matrix metalloproteinase-9 (MMP-9; Ehling et al. 2014). Three-dimensional micromorphological analyses in mouse models of carbon-tetrachloride- or BDL-induced liver injury have demonstrated that macrophage-dependent angiogenesis during chronic liver injury is largely confined to portal veins and that the pharmacological inhibition of CCL2-mediated inflammatory monocyte infiltration primarily reduces angiogenic vessel sprouting in the portal vein (Ehling et al. 2014). Therefore, infiltrating bone-marrow-derived inflammatory monocytes possibly mediate the induction of hepatic angiogenesis by effects that are primarily attributable to changes in the portal vein system.
Angiogenesis and fibrosis progression are closely correlated in experimental liver injury and human liver disease. Angiogenesis and fibrogenesis are also triggered by similar pathways in response to hypoxia and inflammation, so that a potential causal relationship between them has been difficult to establish. In addition, dichotomous effects of angiogenesis on fibrosis have now been reported in various tissues. The administration of VEGF, while stimulating angiogenesis, has been shown to reduce renal fibrosis and to stabilize renal function in the remnant kidney model of progressive renal failure (Kang et al. 2001). Proangiogenic activity induced in an adipocyte-specific VEGF-A overexpression model has been shown to suppress fibrosis, inflammation and insulin resistance during the early phase of high-fat-diet-induced adipose tissue expansion. Conversely, VEGF-A-VEGFR2 blockade has an aggravating effect in this context, whereas in ob/ob mice with pre-existing adipose tissue dysfunction, the anti-angiogenic action of VEGF-A-VEGFR2 blockade causes an improvement in metabolism and a decrease in inflammatory factors (Sun et al. 2012). Many studies in animal models of liver injury have shown that the inhibition of angiogenesis results in a decrease in liver fibrosis (Table 1). However, the pathways targeted in these studies might also have promoted fibrosis directly. In addition, a few studies have led to different conclusions. Thus, pharmacological inhibition of the vitronectin receptor integrin alphavbeta3, which stimulates endothelial cell proliferation and HSC activation, while suppressing hepatic angiogenesis, aggravates liver fibrosis induced by BDL or thioacetamide (TAA) (Patsenker et al. 2009). Pharmacological inhibition of the chemokine CCL2 reduces monocyte infiltration and angiogenesis but not fibrosis progression in mouse models of carbon tetrachloride or BDL-induced liver injury (Ehling et al. 2014). Dichotomous effects of angiogenesis have also been reported in a model of fibrosis resolution generated by cholecystojejunostomy, which restores bile flow after BDL (L. Yang et al. 2014). In this model, VEGF-induced angiogenesis promotes fibrogenesis after BDL but is also required for fibrosis resolution after cholecystojejunostomy. Accordingly, VEGF-neutralizing antibodies not only prevent the development of fibrosis but also disrupt hepatic tissue repair and fibrosis resolution (L. Yang et al. 2014).
Contribution of PMFs to liver angiogenesis
HSC-MFs and PMFs probably both contribute to liver angiogenesis, although at different stages. HSCs in their quiescent state act as pericytes that regulate the functions of sinusoidal endothelial cells. Following myofibroblastic differentiation, they acquire a proangiogenic phenotype and secrete proangiogenic factors such as angiopoietin-1 (Semela et al. 2008; Thabut et al. 2011). At an early stage of liver injury, HSC-MFs promote an enhanced coverage of sinusoids and angiogenesis in areas of active fibrogenesis at the leading edge of developing fibrotic septa (Novo et al. 2007). At later stages, endothelial cell proliferation is correlated with the expansion of PMFs suggesting a role for PMF in liver angiogenesis (Lemoinne et al. 2015). Further evidence that this is the case has been provided by the immunostaining of human cirrhotic livers showing that COL15A1-positive PMFs display a perivascular distribution and outline vascular capillaries within large fibrotic septa. Using a cell model of PMFs that we have previously described in detail, we have demonstrated that PMFs are able to enhance angiogenesis in vitro and in vivo by various mechanisms including the formation of direct intercellular junctions with endothelial cells and the production of VEGF-A-containing microparticles (Lemoinne et al. 2015). Compared with HSC-MFs, PMFs largely overexpress COL15A1 and also COL18A1, which are the α1 chains of collagen XV and collagen XVIII, respectively. Both collagens belong to the superfamily of multiplexins and their C-terminal parts, endostatin and restin, respectively, are anti-angiogenic, which could provide a negative retrocontrol in PMF-induced angiogenesis. Collagen XV provides stability and resilience to mechanical forces in the skeletal muscle and microvessels. It is contained in the basement membrane of continuous capillaries and serves as a scaffold that anchors the basement membrane to interstitial collagen; loss of its expression results in collapsed capillaries in mice. It is absent from specialized capillaries such as fenestrated liver sinusoids. The normal liver contains almost no collagen XV, with the exception of trace amounts in the portal and periportal area. Collagen XV forms a proangiogenic matrix for endothelial cells and thereby might contribute to the angiogenic properties of PMFs. Increased expression of collagen XV has been reported in hepatocellular carcinogenesis in mice and humans. In human hepatocellular carcinoma, collagen XV has been identified as a prominent histopathological component of intratumoral capillaries (Kimura et al. 2016).
Concluding remarks
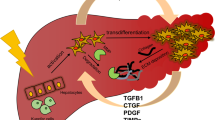
In summary, PMFs appear to be critical in pathological angiogenesis, which constantly occurs in advanced liver fibrosis. Although the way that angiogenesis and fibrosis are linked to each other during the progression of liver diseases remains to be elucidated, we suggest that COL15A1-producing PMFs provide an important link both by stabilizing newly formed vessels and by forming a scaffold for the deposition of interstitial collagen (Fig. 1).
Role of portal myofibroblasts in liver angiogenesis and fibrosis. a In normal liver, quiescent hepatic stellate cells (HSCs) form a continuum with portal mesenchymal cells, which include vascular smooth muscle cells (SMCs), portal fibroblasts and a few mesenchymal progenitor cells. The extracellular matrix is poorly abundant and contains trace amounts of collagen type XV alpha1 (COL15A1) along mesenchymal progenitors. b In advanced fibrosis, portal myofibroblasts (PMFs) proliferate and promote angiogenesis. COL15A1 expressed by PMFs provides a scaffold for interstitial collagen produced in excess mostly by hepatic stellate cell-derived myofibroblats (HSC-MFs)
Abbreviations
- α-SMA:
-
Alpha-smooth muscle actin
- BDL:
-
Bile duct ligation
- COL15A1:
-
Collagen type XV alpha1
- COX-2:
-
Cyclooxygenase-2
- HSC:
-
Hepatic stellate cell
- HSC-MF:
-
Hepatic-stellate-cell-derived myofibroblast
- MCD:
-
Methionine-choline-deficient
- MMP:
-
Matrix metalloproteinase
- NAFLD:
-
Non-alcoholic fatty liver disease
- PDGF-BB:
-
Platelet-derived growth factor-BB
- PDGFR-β:
-
Platelet-derived growth factor receptor-beta
- PlGF:
-
Placental growth factor
- PMF:
-
Portal myofibroblast
- TAA:
-
Thioacetamide
- TGF-β:
-
Transforming growth factor-beta
- VEGF:
-
Vascular endothelial growth factor
- VEGFR:
-
Vascular endothelial growth factor receptor
References
Beaussier M, Wendum D, Schiffer E, Dumont S, Rey C, Lienhart A, Housset C (2007) Prominent contribution of portal mesenchymal cells to liver fibrosis in ischemic and obstructive cholestatic injuries. Lab Invest 87:292–303
Corpechot C, Barbu V, Wendum D, Kinnman N, Rey C, Poupon R, Housset C, Rosmorduc O (2002) Hypoxia-induced VEGF and collagen I expressions are associated with angiogenesis and fibrogenesis in experimental cirrhosis. Hepatology 35:1010–1021
Ehling J, Bartneck M, Wei X, Gremse F, Fech V, Mockel D, Baeck C, Hittatiya K, Eulberg D, Luedde T, Kiessling F, Trautwein C, Lammers T, Tacke F (2014) CCL2-dependent infiltrating macrophages promote angiogenesis in progressive liver fibrosis. Gut 63:1960–1971
Elpek GO (2015) Angiogenesis and liver fibrosis. World J Hepatol 7:377–391
Eyden B (2008) The myofibroblast: phenotypic characterization as a prerequisite to understanding its functions in translational medicine. J Cell Mol Med 12:22–37
Gao JH, Wen SL, Yang WJ, Lu YY, Tong H, Huang ZY, Liu ZX, Tang CW (2013) Celecoxib ameliorates portal hypertension of the cirrhotic rats through the dual inhibitory effects on the intrahepatic fibrosis and angiogenesis. PloS one 8:e69309
Hennenberg M, Trebicka J, Kohistani Z, Stark C, Nischalke HD, Kramer B, Korner C, Klein S, Granzow M, Fischer HP, Heller J, Sauerbruch T (2011) Hepatic and HSC-specific sorafenib effects in rats with established secondary biliary cirrhosis. Lab Invest 91:241–251
Huang Y, Feng H, Kan T, Huang B, Zhang M, Li Y, Shi C, Wu M, Luo Y, Yang J, Xu F (2013) Bevacizumab attenuates hepatic fibrosis in rats by inhibiting activation of hepatic stellate cells. PloS one 8:e73492
Iwaisako K, Jiang C, Zhang M, Cong M, Moore-Morris TJ, Park TJ, Liu X, Xu J, Wang P, Paik YH, Meng F, Asagiri M, Murray LA, Hofmann AF, Iida T, Glass CK, Brenner DA, Kisseleva T (2014) Origin of myofibroblasts in the fibrotic liver in mice. Proc Natl Acad Sci U S A 111:E3297–E3305
Kang DH, Hughes J, Mazzali M, Schreiner GF, Johnson RJ (2001) Impaired angiogenesis in the remnant kidney model. II. Vascular endothelial growth factor administration reduces renal fibrosis and stabilizes renal function. J Am Soc Nephrol 12:1448–1457
Kimura K, Nakayama M, Naito I, Komiyama T, Ichimura K, Asano H, Tsukuda K, Ohtsuka A, Oohashi T, Miyoshi S, Ninomiya Y (2016) Human collagen XV is a prominent histopathological component of sinusoidal capillarization in hepatocellular carcinogenesis. Int J Clin Oncol 21:302–309
Kinnman N, Francoz C, Barbu V, Wendum D, Rey C, Hultcrantz R, Poupon R, Housset C (2003) The myofibroblastic conversion of peribiliary fibrogenic cells distinct from hepatic stellate cells is stimulated by platelet-derived growth factor during liver fibrogenesis. Lab Invest 83:163–173
Lemoinne S, Cadoret A, Rautou PE, El Mourabit H, Ratziu V, Corpechot C, Rey C, Bosselut N, Barbu V, Wendum D, Feldmann G, Boulanger C, Henegar C, Housset C, Thabut D (2015) Portal myofibroblasts promote vascular remodeling underlying cirrhosis formation through the release of microparticles. Hepatology 61:1041–1055
Li Z, Dranoff JA, Chan EP, Uemura M, Sevigny J, Wells RG (2007) Transforming growth factor-beta and substrate stiffness regulate portal fibroblast activation in culture. Hepatology 46:1246–1256
Liu C, Yang Z, Wang L, Lu Y, Tang B, Miao H, Xu Q, Chen X (2015) Combination of sorafenib and gadolinium chloride (GdCl3) attenuates dimethylnitrosamine(DMN)-induced liver fibrosis in rats. BMC Gastroenterol 15:159
Lua I, Li Y, Zagory JA, Wang KS, French SW, Sevigny J, Asahina K (2016) Characterization of hepatic stellate cells, portal fibroblasts, and mesothelial cells in normal and fibrotic livers. J Hepatol 64:1137–1146
May D, Djonov V, Zamir G, Bala M, Safadi R, Sklair-Levy M, Keshet E (2011) A transgenic model for conditional induction and rescue of portal hypertension reveals a role of VEGF-mediated regulation of sinusoidal fenestrations. PloS one 6:e21478
Mederacke I, Hsu CC, Troeger JS, Huebener P, Mu X, Dapito DH, Pradere JP, Schwabe RF (2013) Fate tracing reveals hepatic stellate cells as dominant contributors to liver fibrosis independent of its aetiology. Nat Commun 4:2823
Mejias M, Garcia-Pras E, Tiani C, Miquel R, Bosch J, Fernandez M (2009) Beneficial effects of sorafenib on splanchnic, intrahepatic, and portocollateral circulations in portal hypertensive and cirrhotic rats. Hepatology 49:1245–1256
Novo E, Cannito S, Zamara E, Valfre di Bonzo L, Caligiuri A, Cravanzola C, Compagnone A, Colombatto S, Marra F, Pinzani M, Parola M (2007) Proangiogenic cytokines as hypoxia-dependent factors stimulating migration of human hepatic stellate cells. Am J Pathol 170:1942–1953
Patsenker E, Popov Y, Stickel F, Schneider V, Ledermann M, Sagesser H, Niedobitek G, Goodman SL, Schuppan D (2009) Pharmacological inhibition of integrin alphavbeta3 aggravates experimental liver fibrosis and suppresses hepatic angiogenesis. Hepatology 50:1501–1511
Rappaport AM, MacPhee PJ, Fisher MM, Phillips MJ (1983) The scarring of the liver acini (cirrhosis). Tridimensional and microcirculatory considerations. Virchows Arch A Pathol Anat Histopathol 402:107–137
Sahin H, Borkham-Kamphorst E, Kuppe C, Zaldivar MM, Grouls C, Al-samman M, Nellen A, Schmitz P, Heinrichs D, Berres ML, Doleschel D, Scholten D, Weiskirchen R, Moeller MJ, Kiessling F, Trautwein C, Wasmuth HE (2012) Chemokine Cxcl9 attenuates liver fibrosis-associated angiogenesis in mice. Hepatology 55:1610–1619
Semela D, Das A, Langer D, Kang N, Leof E, Shah V (2008) Platelet-derived growth factor signaling through ephrin-b2 regulates hepatic vascular structure and function. Gastroenterology 135:671–679
Sun K, Wernstedt Asterholm I, Kusminski CM, Bueno AC, Wang ZV, Pollard JW, Brekken RA, Scherer PE (2012) Dichotomous effects of VEGF-A on adipose tissue dysfunction. Proc Natl Acad Sci U S A 109:5874–5879
Taura K, De Minicis S, Seki E, Hatano E, Iwaisako K, Osterreicher CH, Kodama Y, Miura K, Ikai I, Uemoto S, Brenner DA (2008) Hepatic stellate cells secrete angiopoietin 1 that induces angiogenesis in liver fibrosis. Gastroenterology 135:1729–1738
Thabut D, Routray C, Lomberk G, Shergill U, Glaser K, Huebert R, Patel L, Masyuk T, Blechacz B, Vercnocke A, Ritman E, Ehman R, Urrutia R, Shah V (2011) Complementary vascular and matrix regulatory pathways underlie the beneficial mechanism of action of sorafenib in liver fibrosis. Hepatology 54:573–585
Tugues S, Fernandez-Varo G, Munoz-Luque J, Ros J, Arroyo V, Rodes J, Friedman SL, Carmeliet P, Jimenez W, Morales-Ruiz M (2007) Antiangiogenic treatment with sunitinib ameliorates inflammatory infiltrate, fibrosis, and portal pressure in cirrhotic rats. Hepatology 46:1919–1926
Van Steenkiste C, Ribera J, Geerts A, Pauta M, Tugues S, Casteleyn C, Libbrecht L, Olievier K, Schroyen B, Reynaert H, Grunsven LA van, Blomme B, Coulon S, Heindryckx F, De Vos M, Stassen JM, Vinckier S, Altamirano J, Bataller R, Carmeliet P, Van Vlierberghe H, Colle I, Morales-Ruiz M (2011) Inhibition of placental growth factor activity reduces the severity of fibrosis, inflammation, and portal hypertension in cirrhotic mice. Hepatology 53:1629–1640
Wang B, Dolinski BM, Kikuchi N, Leone DR, Peters MG, Weinreb PH, Violette SM, Bissell DM (2007) Role of alphavbeta6 integrin in acute biliary fibrosis. Hepatology 46:1404–1412
Yan Z, Qu K, Zhang J, Huang Q, Qu P, Xu X, Yuan P, Huang X, Shao Y, Liu C, Zhang H, Xing J (2015) CD147 promotes liver fibrosis progression via VEGF-A/VEGFR2 signalling-mediated cross-talk between hepatocytes and sinusoidal endothelial cells. Clin Sci (Lond) 129:699–710
Yang L, Kwon J, Popov Y, Gajdos GB, Ordog T, Brekken RA, Mukhopadhyay D, Schuppan D, Bi Y, Simonetto D, Shah VH (2014) Vascular endothelial growth factor promotes fibrosis resolution and repair in mice. Gastroenterology 146:1339-1350
Yang YY, Liu RS, Lee PC, Yeh YC, Huang YT, Lee WP, Lee KC, Hsieh YC, Lee FY, Tan TW, Lin HC (2014) Anti-VEGFR agents ameliorate hepatic venous dysregulation/microcirculatory dysfunction, splanchnic venous pooling and ascites of NASH-cirrhotic rat. Liver Int 34:521–534
Yoshiji H, Kuriyama S, Yoshii J, Ikenaka Y, Noguchi R, Hicklin DJ, Wu Y, Yanase K, Namisaki T, Yamazaki M, Tsujinoue H, Imazu H, Masaki T, Fukui H (2003) Vascular endothelial growth factor and receptor interaction is a prerequisite for murine hepatic fibrogenesis. Gut 52:1347–1354
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lemoinne, S., Thabut, D. & Housset, C. Portal myofibroblasts connect angiogenesis and fibrosis in liver. Cell Tissue Res 365, 583–589 (2016). https://doi.org/10.1007/s00441-016-2443-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-016-2443-5