Abstract
Myostatin, a member of the TGF-β superfamily of secreted growth factors, is a negative regulator of skeletal muscle growth. In the heart, it is expressed at lower levels compared to skeletal muscle but up-regulated under disease conditions. Cre recombinase-mediated inactivation of myostatin in adult cardiomyocytes leads to heart failure and increased mortality but cardiac function of surviving mice is restored after several weeks probably due to compensatory expression in non-cardiomyocytes. To study long-term effects of increased myostatin expression in the heart and to analyze the putative crosstalk between cardiomyocytes and fibroblasts, we overexpressed myostatin in cardiomyocytes. Increased expression of myostatin in heart muscle cells caused interstitial fibrosis via activation of the TAK-1-MKK3/6-p38 signaling pathway, compromising cardiac function in older mice. Our results uncover a novel role of myostatin in the heart and highlight the necessity for tight regulation of myostatin to maintain normal heart function.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Intercellular communication between cardiomyocytes and cardiac fibroblasts plays an important role in pathologic remodeling of the heart. Cross-talk between different cell populations in the heart is regulated via release of proteins regulating tissue homeostasis and response to cardiac injury (Shimano et al. 2012). A major mediator of interactions between cardiomyocytes and fibroblasts is TGF-β1 (transforming growth factor β1). Myostatin (also called GDF-8) belongs to the TGF-β superfamily of secreted growth factors and was first identified as a negative regulator of skeletal muscle growth (McPherron et al. 1997). Inactivation of myostatin by targeted deletion or by naturally occurring mutations leads to a dramatic skeletal muscle hypertrophy in several species including mice, cattle, sheep, dogs and human beings (McPherron et al. 1997; Rodgers and Garikipati 2008). In addition to its function in restricting skeletal muscle growth, myostatin also enables profibrotic responses in skeletal muscle. Genetic inactivation of myostatin or treatment with inhibitory peptides decreases fibrosis after musculoskeletal injury or in dystrophic mice (Bogdanovich et al. 2002; McCroskery et al. 2005; Nakatani et al. 2008; Wagner et al. 2002). Furthermore, myostatin promotes proliferation, differentiation and expression of extracellular matrix proteins of fibroblasts in skeletal muscles (Li et al. 2008; Zhu et al. 2007).
Recently, we demonstrated that inactivation of myostatin in adult cardiomyocytes leads to cardiac hypertrophy and heart failure. Interestingly, cardiac dysfunction is rescued after inactivation of myostatin in cardiomyocytes within a matter of several weeks, probably by compensatory expression of myostatin by non-cardiomyocytes indicating a close interaction between cardiomyocytes and fibroblasts (Biesemann et al. 2014). Analysis of the effects of myostatin on cardiac fibroblasts using germ-line myostatin knockout mice has not so far yielded conclusive results, revealing reduced fibrosis in aging mice (Morissette et al. 2009) but unchanged cardiac fibrosis in dystrophin-deficient mdx mice (Cohn et al. 2007). Furthermore, it has been recently postulated that increased expression of GDF11, which is highly similar to myostatin, reverses age-related cardiac hypertrophy (Loffredo et al. 2013), implying potential benefits of increased expression of GDF11 and myostatin that are both signaling via the same receptors (Andersson et al. 2006; Oh et al. 2002). To better understand the crosstalk between cardiomyocytes and fibroblasts, we asked whether cardiomyocyte-derived myostatin has an impact on non-cardiomyocytes in the myocardium. Therefore, we specifically overexpressed myostatin in cardiomyocytes in mice, which was associated with activation of the TAK1-MKK3/6-p38 signaling pathway and caused an increase in interstitial fibrosis compromising cardiac function in older mice.
Materials and methods
Generation and treatment of transgenic mouse lines
All procedures were performed in accordance with the guidelines for animal experimentation of the local authorities. All animals were kept on the identical C57/BL6/J background. For all studies, male mice were used. Transgenic mice were generated as described before (Biesemann et al. 2014). Cre recombination in αMyHC-MCM mice was achieved by intraperitoneal administration of tamoxifen for five consecutive days.
Physiological measurements
Cardiac MRI measurements were performed on a 7.0 T Bruker Pharmascan, equipped with a 300mT/m gradient system, using a custom-built circularly polarized birdcage resonator and the IntraGate™ self-gating tool (Bruker, Ettlingen, Germany). Mice were analyzed under volatile isoflurane (1.5–2.0 %) anaesthesia. Measurements were based on the gradient echo method (repetition time = 6.02 ms; echo time = 1.6 ms; field of view = 2.20 × 2.20 cm; slice thickness = 1.0 mm; matrix = 128 × 128; repetitions = 100). The imaging plane was localized using scout images showing 2- and 4-chamber views of the heart, followed by acquisition in the short axis view, orthogonal on the septum in both scouts. Multiple contiguous short-axis slices were acquired for coverage of the left ventricle. MRI data were analysed using Qmass digital imaging software (Medis, Leiden, Netherlands).
RT-PCR analysis
For treatment with pharmacological inhibitors, cardiomyocytes were stimulated with different reagents as indicated. RNA was isolated from either ventricular tissue or isolated cell population according to standard protocols using the Trizol reagent (Invitrogen, Karlsruhe, Germany) and subjected to RT-PCR analysis using a Bio-Rad iCYCLER iQ5 Real-time PCR machine with Arp (acidic ribosomal protein) as internal standard, as described before (Neuhaus et al. 2003).
Western blot analysis
Ventricular tissue was minced in liquid nitrogen and lysed by standard procedures. Next, 10–20 μg of lysed ventricular tissue or isolated cardiomyocytes were separated by SDS-PAGE and incubated with different antibodies. Antibodies recognizing p-p38 (Thr180/Tyr182), p38, p-TAK1 (Thr187), p-MKK3/6 (Ser189/207) and p-HSP27 (Ser82) were from Cell Signaling. The antibody against RALA was from BD and anti-α-tubulin from Sigma. Immunoreactive proteins were visualized on a VersaDoc (BioRad) and quantified with the QuantityOne software (BioRad).
Morphological analysis
Midventricular parts of the heart were cut into 10-μm-thick sections using a Leica CM 1950 cryostat (Leica). Masson’s Trichrome staining was performed according to the ACCUSTAIN® protocol (Sigma). Images were taken with an Axio Imager.Z1 microscope (Carl Zeiss). The extent of fibrosis was determined by analyzing ×10 images with Masson’s Trichrome using ImageJ software. To measure cardiomyocyte area, sections were fixed and incubated with TRITC-labeled Lectin from Triticum vulgaris (Sigma, Steinheim, Germany) followed by analysis of four random areas (×20 images) per tissue block (n = 3 per mouse model) using ImageJ software. For Collagen I staining, cryosections were probed with a Collagen I antibody (Rockland) and secondary antibodies. Sections without primary antibodies served as negative controls. For each mouse, at least five random microscopic fields per each tissue block (n = 3 per mouse model and time point) were analyzed with a Leica SP2 confocal scanning microscope using a ×40 Planapo objective (Leica). Immunolabeled cryosections were analyzed using “Quantification” and “VoxelShop” options of Imaris 5 (Bitplane) and Image J software. The area of specific labeling for Collagen I was calculated as percent of positive labeling per tissue area as previously described (Vakhrusheva et al. 2008).
Statistical analysis
Data are shown as mean ± SEM. Statistical analysis was performed using GraphPad Prism (v.5.0; GraphPad Software). Two groups were compared using the Student’s t test, three or more groups using one-way ANOVA followed by Bonferroni’s multiple comparison test. Values of P < 0.05 were considered as statistically significant.
Results
Local increase of myostatin signaling causes interstitial fibrosis in the heart
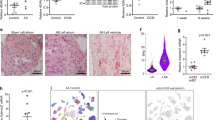
Previous studies indicated that myostatin promotes fibrosis in skeletal muscle (Wagner et al. 2002) and that constitutive loss of myostatin protects from cardiac fibrosis during aging (Morissette et al. 2009). To analyze whether increased expression of myostatin in cardiomyocytes induces cardiac fibrosis, we generated αMyHC-Cre/CAGG-Mstn mice, in which the myostatin cDNA is expressed under control of the CAG promoter following removal of a flox-stop cassette by cre recombinase specifically in cardiomyocytes. Interestingly, we detected increased interstitial fibrosis in αMyHC-Cre/CAGG-Mstn mice starting at 4.5 months (Fig. 1a”–c”, d). In contrast, age-matched αMyHC-Cre controls (Fig. 1a–d) and αMyHC-Cre/Mstn fl/fl mice (Fig. 1a’–c’, d), in which the myostatin gene was constitutively deleted in the cardiomyocyte lineage, did not develop fibrosis.
Overexpression of myostatin in cardiomyocytes induces cardiac interstitial fibrosis. Immunofluorescence staining for F-Actin/DAPI (a–a”), Collagen-I/DAPI (b–b”) and F-Actin/Collagen-I/DAPI (c–c”) in hearts of 4.5-month-old αMyHC-Cre (a–c), αMyHC-Cre/Mstn fl/fl (a’–c’) and αMyHC-Cre/CAGG-Mstn (a”–c”) mice. Scale bar 20 μm. (d) Analysis of collagen content in hearts from αMyHC-Cre, αMyHC-Cre/Mstnfl/fl and αMyHC-Cre/CAGG-Mstn mice. Collagen content was analyzed out of ×10 pictures from trichrome staining. Values represent means ± SEM; **P < 0.01
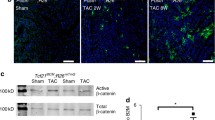
Next, we wanted to know whether myostatin needs to be delivered by cardiomyocytes to induce cardiac fibrosis or whether increased expression of myostatin in non-cardiomyocytes of the heart exerts similar effects. For this purpose, we used αMyHC-MCM/Mstn fl/fl mice (Biesemann et al. 2014), which are characterized by a massive increase of myostatin expression in non-cardiomyocytes, 6 weeks after induced deletion of myostatin in cardiomyocytes (Fig. 2a). We observed an increased collagen I deposition and interstitial fibrosis 6 weeks after tamoxifen administration (Fig. 2b, d–d”, e–e”, f–f”). The increase in collagen I deposition paralleled the increase of myostatin expression in non-cardiomyocytes (Fig. 2a). No fibrosis was detected in tamoxifen-treated and untreated αMyHC-MCM control mice (Fig. 2b, c–c”).
Compensatory increase of myostatin expression in non-cardiomyocytes after deletion of myostatin in adult cardiomyocytes induces cardiac interstitial fibrosis. a Quantitative RT-PCR analysis of myostatin expression in ventricles of αMyHC-MCM and αMyHC-MCM/Mstn fl/fl mice (three independent experiments, n = 3). b Analysis of collagen I content in the mid-ventricular area of αMyHC-MCM and αMyHC-MCM/Mstn fl/fl mice. Mean collagen I content was measured in five different microscopic fields per heart section (n = 3). c–f” Immunofluorescence staining for collagen I in hearts of αMyHC-MCM mice 84 days after tamoxifen injection (c–c”) and of αMyHC-MCM/Mstn fl/fl mice 10 (d–d”), 42 (e–e”) and 120 (f–f”) days after tamoxifen injection. Scale bar 30 μm. Values represent means ± SEM; *P < 0.05, **P < 0.01, ****P < 0.0001
Long-term overexpression of myostatin slightly impairs cardiac function
Despite the development of interstitial fibrosis, we did not detect impaired cardiac function in myostatin overexpressing male αMyHC-Cre/CAGG-Mstn mice at 4.5 months of age. Therefore, we wondered whether older αMyHC-Cre/CAGG-Mstn mice (9 months), which maintain high levels of myostatin in the heart (Fig. 3d) and show large patches of fibrotic areas in the myocardium together with interstitial and perivascular fibrosis in the heart (Fig. 3b’), develop cardiac dysfunction. Assessment of cardiac function by MRI revealed significantly decreased ejection fraction and stroke volume as well as increased endsystolic and enddiastolic volumes at 9 months of age (Fig. 3e f, h–i). Since the long-term overexpression of myostatin did not affect the cardiomyocyte area (Fig. 3a’, c), we concluded that the impaired cardiac function in 9-month old αMyHC-Cre/CAGG-Mstn mice is due to increased cardiac fibrosis.
Long-term overexpression of myostatin induces cardiac fibrosis and favors impaired contractility. a–a’ Representative WGA staining of CAGG-Mstn (a) and αMyHC-Cre/CAGG-Mstn (a’) mid-ventricular heart sections at 9 months of age. Scale bar 50 μm. b–b’ Trichrome staining of CAGG-Mstn (b) and αMyHC-Cre/CAGG-Mstn (b’) mid-ventricular heart sections at 9 months of age. Scale bar 20 μm. c Analysis of cardiomyocyte cross-sectional area (CSA). CSA was determined by analysis of four different microscopic fields (×20) per heart section (n = 3). d Quantitative RT-PCR analysis of myostatin expression in CAGG-Mstn and αMyHC-Cre/CAGG-Mstn ventricles. Arp (acidic ribosomal protein) was used for normalization. (n = 3). e, f, h–j MRI analysis of CAGG-Mstn and αMyHC-Cre/CAGG-Mstn mice. Male mice at 9 months of age were used (n = 3). e Ejection fraction. f Stroke volume. h Endsystolic volume (ESV). i Enddiastolic volume (EDV). j Left ventricular (LV) mass. g Body weight of CAGG-Mstn and αMyHC-Cre/CAGG-Mstn mice. Male mice at 9 months of age were used (n = 6 (CAGG-Mstn), n = 4 (αMyHC-Cre/CAGG-Mstn)). All measurements represent means ± SEM. *P < 0.05, **P < 0.01
Myostatin activates TAK1-MKK3/6-p38 MAPK pathway
To characterize the molecular mechanism causing cardiac fibrosis in αMyHC-MCM/Mstn fl/fl and αMyHC-Cre/CAGG-Mstn mice, we screened for activation of putative pro-fibrotic pathways focusing on p38, a well-known activator of cardiac fibrosis (Turner 2011; Zhang et al. 2003). So far, the effects of myostatin on p38 activation have been controversial: in vitro data obtained in skeletal muscle C2C12 cells suggested that myostatin activates p38 (Philip et al. 2005) but opposite effects were observed in neonatal rat cardiomyocytes (Morissette et al. 2006). In vivo, we detected a strong up-regulation of p38 phosphorylation in αMyHC-Cre/CAGG-Mstn as well as in αMyHC-MCM/Mstn fl/fl mice, which show a massive compensatory increase of myostatin expression in non-cardiomyocytes (Fig. 4a–b’). Since p38 is strongly activated by TGF-beta activated kinase 1 (TAK1) and mitogen-activated protein kinase kinase 3/6 (MKK3/6) (Sorrentino et al. 2008; Yamaguchi et al. 1995), we examined the phosphorylation status of TAK1 and MKK3/6 in hearts of αMyHC-Cre/CAGG-Mstn and αMyHC-MCM/Mstn fl/fl mice. We observed an increase of TAK1 and MKK3/6 phosphorylation in both strains (Fig. 4a–b’), which corresponds well to our previous findings that effects of myostatin on AMPK are mediated by TAK1 (Biesemann et al. 2014). However, in contrast to the significant increase of MKK3/6 phosphorylation the increase in TAK1 phosphorylation was not seen in all αMyHC-Cre/CAGG-Mstn and αMyHC-MCM/Mstn fl/fl animals impeding statistical significance.
Myostatin activates TAK1-MKK3/6-p38 pathway. a–a’ Western blot analysis (a) and quantitation (a’) of p-TAK1 (Thr187), p-MKK3/6 (Ser189/297), p-p38 (Thr180/Tyr182) and p38 in αMyHC-Cre and αMyHC-Cre/CAGG-Mstn hearts (three independent experiments, n ≥ 3). b–b’p-TAK1 (Thr187), p-MKK3/6 (Ser189/297), p-p38 (Thr180/Tyr182) and p38 in αMyHC-MCM and αMyHC-MCM/Mstn fl/fl hearts 6 weeks after tamoxifen administration (three independent experiments, n ≥ 3). Values represent means ± SEM; *P < 0.05, **P < 0.01, ****P < 0.0001
Myostatin induces interstitial fibrosis via p38 activation
To demonstrate that myostatin induces fibrosis and activates collagen I expression via stimulation of p38 phosphorylation, we treated isolated primary adult ventricular cardiomyocytes in culture for 30 min with recombinant myostatin. We detected a strong activation of p38 phosphorylation in isolated cardiomyocytes (Fig. 5a, a’) by myostatin, concomitant with a clear up-regulation of collagen I expression (Fig. 5b). Interestingly, myostatin-induced expression of collagen I was blocked by treatment of cardiomyocytes with SB203580 (Fig. 5b). SB203580 efficiently inhibits actions of activated p38 as demonstrated by suppression of HSP27 phosphorylation, a direct target of p38 (Fig. 5a). Taken together, our results indicate that continuous stimulation of the myocardium by myostatin induces cardiac fibrosis most likely via TAK1 and p38 (Fig. 5c).
Myostatin induces fibrosis via the p38 signaling pathway. a, a’, b Murine adult ventricular wildtype cardiomyocytes (1 day in culture) were stimulated with DMSO (1 h), 30 ng/ml myostatin (Mstn, 30 min), 10 μM SB203580 (SB, 1 h) or preincubated with 10 μM SB203580 (SB, 30 min) and then stimulated with 30 ng/ml myostatin (30 min). a p-p38 (Thr180/Tyr182) and p-Hsp27 (Ser82) in treated cardiomyocytes (three independent experiments, n = 3). a’ Quantitation of p-p38 in non-stimulated and myostatin-stimulated cardiomyocytes (three independent experiments, n = 3). b Quantitative RT-PCR of Col1a1 expression (three independent experiments, n = 3) in treated cardiomyocytes. c Simplified model of myostatin signaling inducing fibrosis mechanism: myostatin stimulates p38 via activation of TAK1 and MKK3/6, which then promotes collagen I expression. Values represent means ± SEM; *P < 0.05, ***P < 0.001
Discussion
We demonstrated here that increased expression of myostatin in the heart promotes fibrosis after both directed expression in cardiomyocytes and compensatory increase in non-cardiomyocytes after genetic inactivation in cardiomyocytes. These observations correspond well to the established profibrotic role of myostatin in skeletal muscle (Bogdanovich et al. 2002; McCroskery et al. 2005). However, in the heart, the situation has been less clear and a direct proof of a profibrotic effect of myostatin was missing (Cohn et al. 2007; Morissette et al. 2009). Interestingly, activation of myostatin in the heart is also increased during heart failure in human patients suffering from ischemic and dilated cardiomyopathy, which has been attributed to an anti-hypertrophic role of myostatin counteracting increased IGF-1 signaling (George et al. 2010). Our own findings demonstrating a transient increase in cardiomyocyte hypertrophy after induced inactivation of myostatin in adult cardiomyocytes support this view (Biesemann et al. 2014). However, cardiac fibrosis is an unwanted side effect of increased myostatin signaling, which is otherwise beneficial and prevents heart failure. In this context, it is important to note that increased expression of myostatin does not compromise cardiac function in 4.5-month-old mice despite a moderate increase in fibrosis. The situation is different in 9-month-old mice, which show increased fibrosis with large patches of fibrotic tissue in the myocardium and impaired cardiac function. Tight regulation of myostatin up-regulation seems mandatory to restrict cardiac hypertrophy and prevent organ fibrosis in a pathological situation in order to maintain cardiac function. We hypothesize that a limited up-regulation of myostatin in pathological conditions in the heart might help to restrict hypertrophy and improve cardiac contractility with only minor adverse effects, while negative side effects such as cardiac fibrosis seem to dominate after extended activation of myostatin (Breitbart et al. 2011; George et al. 2010; Sharma et al. 1999). GDF11 has been claimed to reverse age-related cardiac hypertrophy arguing for continuous administration of GDF11 to prevent age-related organ dysfunction (Loffredo et al. 2013; Sinha et al. 2014). Since GDF11 utilizes the same receptors as myostatin (Andersson et al. 2006; Oh et al. 2002), it seems likely that GDF11 will also induce fibrosis when administered at high doses for an extended time. Careful adjustment of GDF11 levels might prevent adverse consequences and limit counter-regulatory effects, although much needs to be learned about the intricate regulatory network that regulates GDF11 and myostatin levels in different organs.
Activation of p38 and its upstream kinases MKK3/6 are well known to induce organ fibrosis (Liao et al. 2001; Turner 2011; Zhang et al. 2003). However, activation of p38 signaling has not been linked to the profibrotic activities of myostatin in skeletal muscle (Wagner et al. 2002). Our data clearly indicate that increased expression of myostatin results in enhanced phosphorylation of TAK1, MKK3/6 and p38, which was further validated in isolated adult cardiomyocytes by demonstrating that inhibition of p38 blocked myostatin-depended stimulation of Collagen I expression. In contrast to our results, Morissette et al. (2006) observed that adenoviral expression of myostatin did not increase p38 phosphorylation but reversed phenylephrine-induced activation of p38 in neonatal rat cardiomyocytes in vitro. Since we analyzed adult murine heart samples and primary mouse cardiomyocytes in our experiments, this discrepancy might reflect stage-dependent differences of myostatin on p38 signaling. Further support for this idea comes from the analysis of the adult murine myoblast cell line C2C12, in which myostatin also activates p38 (Philip et al. 2005).
Taken together, our study establishes a profibrotic role of myostatin in the heart, which is exerted via the p38 signaling pathway, compromising cardiac functions upon extended exposure to myostatin. We postulate that myostatin levels in the heart need to be tightly regulated in pathological conditions to avoid adverse effects. Long-term therapeutic interventions by continuous manipulation of myostatin or GDF11 levels might initiate unwanted side effects such as fibrosis or exacerbate an already existing organ fibrosis.
References
Andersson O, Reissmann E, Ibanez CF (2006) Growth differentiation factor 11 signals through the transforming growth factor-beta receptor ALK5 to regionalize the anterior-posterior axis. EMBO Rep 7:831–837
Biesemann N, Mendler L, Wietelmann A, Hermann S, Schafers M, Kruger M, Boettger T, Borchardt T, Braun T (2014) Myostatin regulates energy homeostasis in the heart and prevents heart failure. Circ Res 115:296–310
Bogdanovich S, Krag TO, Barton ER, Morris LD, Whittemore LA, Ahima RS, Khurana TS (2002) Functional improvement of dystrophic muscle by myostatin blockade. Nature 420:418–421
Breitbart A, Auger-Messier M, Molkentin JD, Heineke J (2011) Myostatin from the heart: local and systemic actions in cardiac failure and muscle wasting. Am J Physiol Heart Circ Physiol 300:H1973–H1982
Cohn RD, Liang HY, Shetty R, Abraham T, Wagner KR (2007) Myostatin does not regulate cardiac hypertrophy or fibrosis. Neuromuscul Disord 17:290–296
George I, Bish LT, Kamalakkannan G, Petrilli CM, Oz MC, Naka Y, Sweeney HL, Maybaum S (2010) Myostatin activation in patients with advanced heart failure and after mechanical unloading. Eur J Heart Fail 12:444–453
Li ZB, Kollias HD, Wagner KR (2008) Myostatin directly regulates skeletal muscle fibrosis. J Biol Chem 283:19371–19378
Liao P, Georgakopoulos D, Kovacs A, Zheng M, Lerner D, Pu H, Saffitz J, Chien K, Xiao RP, Kass DA, Wang Y (2001) The in vivo role of p38 MAP kinases in cardiac remodeling and restrictive cardiomyopathy. Proc Natl Acad Sci U S A 98:12283–12288
Loffredo FS, Steinhauser ML, Jay SM, Gannon J, Pancoast JR, Yalamanchi P, Sinha M, Dall'Osso C, Khong D, Shadrach JL, Miller CM, Singer BS, Stewart A, Psychogios N, Gerszten RE, Hartigan AJ, Kim MJ, Serwold T, Wagers AJ, Lee RT (2013) Growth differentiation factor 11 is a circulating factor that reverses age-related cardiac hypertrophy. Cell 153:828–839
McCroskery S, Thomas M, Platt L, Hennebry A, Nishimura T, McLeay L, Sharma M, Kambadur R (2005) Improved muscle healing through enhanced regeneration and reduced fibrosis in myostatin-null mice. J Cell Sci 118:3531–3541
McPherron AC, Lawler AM, Lee SJ (1997) Regulation of skeletal muscle mass in mice by a new TGF-beta superfamily member. Nature 387:83–90
Morissette MR, Cook SA, Foo S, McKoy G, Ashida N, Novikov M, Scherrer-Crosbie M, Li L, Matsui T, Brooks G, Rosenzweig A (2006) Myostatin regulates cardiomyocyte growth through modulation of Akt signaling. Circ Res 99:15–24
Morissette MR, Stricker JC, Rosenberg MA, Buranasombati C, Levitan EB, Mittleman MA, Rosenzweig A (2009) Effects of myostatin deletion in aging mice. Aging Cell 8:573–583
Nakatani M, Takehara Y, Sugino H, Matsumoto M, Hashimoto O, Hasegawa Y, Murakami T, Uezumi A, Takeda S, Noji S, Sunada Y, Tsuchida K (2008) Transgenic expression of a myostatin inhibitor derived from follistatin increases skeletal muscle mass and ameliorates dystrophic pathology in mdx mice. FASEB J 22:477–487
Neuhaus P, Oustanina S, Loch T, Kruger M, Bober E, Dono R, Zeller R, Braun T (2003) Reduced mobility of fibroblast growth factor (FGF)-deficient myoblasts might contribute to dystrophic changes in the musculature of FGF2/FGF6/mdx triple-mutant mice. Mol Cell Biol 23:6037–6048
Oh SP, Yeo CY, Lee Y, Schrewe H, Whitman M, Li E (2002) Activin type IIA and IIB receptors mediate Gdf11 signaling in axial vertebral patterning. Genes Dev 16:2749–2754
Philip B, Lu Z, Gao Y (2005) Regulation of GDF-8 signaling by the p38 MAPK. Cell Signal 17:365–375
Rodgers BD, Garikipati DK (2008) Clinical, agricultural, and evolutionary biology of myostatin: a comparative review. Endocr Rev 29:513–534
Sharma M, Kambadur R, Matthews KG, Somers WG, Devlin GP, Conaglen JV, Fowke PJ, Bass JJ (1999) Myostatin, a transforming growth factor-beta superfamily member, is expressed in heart muscle and is upregulated in cardiomyocytes after infarct. J Cell Physiol 180:1–9
Shimano M, Ouchi N, Walsh K (2012) Cardiokines: recent progress in elucidating the cardiac secretome. Circulation 126:e327–e332
Sinha M, Jang YC, Oh J, Khong D, Wu EY, Manohar R, Miller C, Regalado SG, Loffredo FS, Pancoast JR, Hirshman MF, Lebowitz J, Shadrach JL, Cerletti M, Kim MJ, Serwold T, Goodyear LJ, Rosner B, Lee RT, Wagers AJ (2014) Restoring systemic GDF11 levels reverses age-related dysfunction in mouse skeletal muscle. Science 344:649–652
Sorrentino A, Thakur N, Grimsby S, Marcusson A, von Bulow V, Schuster N, Zhang S, Heldin CH, Landstrom M (2008) The type I TGF-beta receptor engages TRAF6 to activate TAK1 in a receptor kinase-independent manner. Nat Cell Biol 10:1199–1207
Turner NA (2011) Therapeutic regulation of cardiac fibroblast function: targeting stress-activated protein kinase pathways. Future Cardiol 7:673–691
Vakhrusheva O, Smolka C, Gajawada P, Kostin S, Boettger T, Kubin T, Braun T, Bober E (2008) Sirt7 increases stress resistance of cardiomyocytes and prevents apoptosis and inflammatory cardiomyopathy in mice. Circ Res 102:703–710
Wagner KR, McPherron AC, Winik N, Lee SJ (2002) Loss of myostatin attenuates severity of muscular dystrophy in mdx mice. Ann Neurol 52:832–836
Yamaguchi K, Shirakabe K, Shibuya H, Irie K, Oishi I, Ueno N, Taniguchi T, Nishida E, Matsumoto K (1995) Identification of a member of the MAPKKK family as a potential mediator of TGF-beta signal transduction. Science 270:2008–2011
Zhang S, Weinheimer C, Courtois M, Kovacs A, Zhang CE, Cheng AM, Wang Y, Muslin AJ (2003) The role of the Grb2-p38 MAPK signaling pathway in cardiac hypertrophy and fibrosis. J Clin Invest 111:833–841
Zhu J, Li Y, Shen W, Qiao C, Ambrosio F, Lavasani M, Nozaki M, Branca MF, Huard J (2007) Relationships between transforming growth factor-beta1, myostatin, and decorin: implications for skeletal muscle fibrosis. J Biol Chem 282:25852–25863
Acknowledgments
This work was supported by the Max Planck Society, the Excellence Initiative “Cardiopulmonary System”, the German Center for Cardiovascular Research (DZHK) and the Leducq Foundation.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Biesemann, N., Mendler, L., Kostin, S. et al. Myostatin induces interstitial fibrosis in the heart via TAK1 and p38. Cell Tissue Res 361, 779–787 (2015). https://doi.org/10.1007/s00441-015-2139-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-015-2139-2