Abstract
In the developing mammalian nervous system, common progenitors integrate both cell extrinsic and intrinsic regulatory programs to produce distinct neuronal and glial cell types as development proceeds. This spatiotemporal restriction of neural progenitor differentiation is enforced, in part, by the dynamic reorganization of chromatin into repressive domains by Polycomb repressive complexes, effectively limiting the expression of fate-determining genes. Here, we review the distinct roles that Polycomb repressive complexes play during neurogenesis and gliogenesis, while also highlighting recent work describing the molecular mechanisms that govern their dynamic activity in neural development. Further investigation of the way in which Polycomb complexes are regulated in neural development will enable more precise manipulation of neural progenitor differentiation facilitating the efficient generation of specific neuronal and glial cell types for many biological applications.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
During development of the nervous system, developmental potential is progressively restricted as pluripotent cells of the early embryo give rise to multi-potent progenitor cells, and as these progenitors differentiate into neurons and glia. By definition, this is an epigenetic phenomenon, whereby cells with the same genome acquire and maintain distinct gene expression patterns that differentiate them with regard to form and function. Mechanisms that reorganize chromatin structure play an essential role in this process. The basic unit of chromatin is the nucleosome consisting of DNA wrapped around core histones, which can be assembled together with non-histone proteins into the complex topology of higher order chromatin structures characteristic of eukaryotic genomes. In its simplest form, the topological arrangement of chromatin partitions the genome into sterically open (euchromatic) and compact (heterochromatic) compartments promoting or inhibiting transcriptional initiation and elongation leading to the patterning of gene expression in the cell (Armstrong 2012; Olynik and Rastegar 2012; Wutz 2013).
Multipotent stem and progenitor cells have a distinct chromatin structure that facilitates their maintenance of developmental plasticity. In the pluripotent “ground” state of embryonic stem cells (ESCs, derived from the inner cell mass of the early embryo), chromatin is decondensed and histone proteins are loosely bound, exhibiting hyperdynamic exchange rates (Meshorer et al. 2006; Meshorer 2007). During differentiation, histone exchange becomes less dynamic, and the chromatin becomes more condensed as foci of heterochromatin form and spread (Meshorer et al. 2006; Meshorer 2007). The exact placement and organization of heterochromatin constrains the competence of a cell by limiting the gene programs available for transcription (Francastel et al. 2000; Arney and Fisher 2004; Bernstein et al. 2007; Campos and Reinberg 2009; Zhou et al. 2011). Understanding the way that heterochromatin is successively patterned in different progenitors is therefore essential for comprehension of (1) how cell fate is controlled during development and (2) how it may be modified ex vivo for experimental and therapeutic purposes.
A number of different regulatory mechanisms have been described that contribute to the formation and dynamic rearrangement of heterochromatin during neural development. These include enzymatic machineries that methylate DNA or covalently modify the amino-terminal tails of histone proteins after translation, namely by acetylating, ubiquitylating, phosphorylating, or methylating specific residues (Campos and Reinberg 2009; Zhou et al. 2011). Many of these modifications are well correlated with specific biological functions, including transcriptional activation, repression, and enhancer activity. Whereas the exact consequences of the various post-translation modifications (PTMs) of histone tails is an area of active research, they generally influence transcription by altering nucleosome compaction or mobility, and by modulating the recruitment of non-histone effector proteins (Taverna et al. 2007; Yun et al. 2011; Zhou et al. 2011).
Efforts to unravel the way in which the chromatin state is regulated during development have been complicated by the fact that many chromatin-modifying proteins are expressed in multiple cell and tissue types. Even within a single cell lineage, these chromatin modifiers can act with temporal specificity, targeting distinct suites of genes during each developmental transition. Therefore, a major current challenge lies in understanding the means by which such spatially and temporally controlled targeting of chromatin modifiers is achieved during development. Here, we will address some of the key histone modification state changes that accompany mammalian neurogenesis and gliogenesis, focusing in particular on temporally distinct roles that the Polycomb repressor complexes play in these processes and on recent advances in research aimed at unraveling the long-standing enigma of how these complexes recognize different genomic targets in different neural cell lineages.
Regulation of developmental gene expression by Polycomb and trithorax proteins
Among the most well characterized chromatin modifiers are the Trithorax group (TrxG) and Polycomb group (PcG) proteins. TrxG and PcG proteins were originally discovered in Drosophila melanogaster as multimeric protein complexes that work in opposition respectively to activate or repress Hox gene expression (Schuettengruber et al. 2007). TrxG protein complexes are likewise critical activators of gene expression in mammals in which they catalyze trimethylation of histone 3 lysine 4 (H3K4me3) at promoters to stimulate active transcription. The enzymatic ortholog of Drosophila TrxG in mammals is variable and can include mixed lineage leukemia (MLL) proteins 1–4, Set1A, and Set1B, all of which bind additional activating proteins to form the multi-subunit MLL1–4 and Set1A/B complexes (Schuettengruber et al. 2011).
In contrast, repression of many developmental loci is mediated by the activity of PcG protein complexes. PcG-mediated repression of developmental genes directly antagonizes TrxG-mediated gene activation, preventing ectopic expression of genes associated with alternative lineages (Margueron and Reinberg 2011; Simon and Kingston 2013). The diverse proteins that contribute to PcG-mediated transcriptional regulation are traditionally subdivided into two complexes, Polycomb repressive complex 1 (PRC1) and Polycomb repressive complex 2 (PRC2), on the basis of their associated enzymatic activity. Mammalian PRC2 consists of three core subunits that are essential for proper catalytic activity and gene repression in vivo: enhancer of zeste 2 (Ezh2) or its homolog Ezh1, embryonic ectoderm development (Eed), and suppressor of zeste 12 (Suz12). Ezh2 and Ezh1 contain a conserved SET domain capable of catalyzing the mono-, di-, and tri-methylation of H3K27 (Margueron et al. 2008; Shen et al. 2008). Trimethylation of histone H3 lysine 27 (H3K27me3) is the most well characterized histone PTM catalyzed by PRC2, and this mark plays a crucial role in the establishment of facultative heterochromatin throughout development (Cao et al. 2002; Czermin et al. 2002; Kuzmichev et al. 2002; Kirmizis et al. 2004).
The deposition of H3K27me3 by PRC2 promotes the recruitment of a second Polycomb complex, PRC1, at a subset of targeted loci characterized by longer tracts of GC-rich DNA (Ku et al. 2008). PRC1 complexes are considerably more heterogeneous in composition than PRC2 (for a review, see Simon and Kingston 2013) and are discussed in some detail below. However, all mammalian PRC1 complexes include a homolog of the Drosophila Ring protein, which catalyzes the mono-ubiquitylation of lysine 119 of histone H2A (H2AK119ub). Whereas the exact consequences of H2AK119ub are not clear, PRC1 is thought to inhibit gene expression through a number of mechanisms, including by impairing transcriptional elongation, increasing chromatin compaction, decreasing nucleosome turnover, and modifying higher order chromatin structure (Simon and Kingston 2013).
Some vertebrates, including mammals and zebrafish, have been shown to utilize an additional mechanism of gene regulation involving combinatorial TrxG and PcG activity (Voigt et al. 2013). In stem and progenitor cells, genes that promote cell-type-specific fate acquisition and differentiation are repressed in a readily reversible manner through the co-modification of their promoters with both the “active” H3K4me3 and “repressive” H3K27me3 modifications (Azuara et al. 2006; Bernstein et al. 2006; Pan et al. 2007). The promoters of many key developmental regulatory genes are regulated by this “bivalent” histone modification signature, which maintains their expression in a restrained intermediary state characterized by extremely low levels of transcription (Bernstein et al. 2006; Pan et al. 2007; Zhao et al. 2007). This bivalent state resolves into sets of loci that are either activated or more stably repressed, depending on the lineage specified for a given progenitor (Mikkelsen et al. 2007; Alder et al. 2010). Thus, bivalent promoters appear to be poised to enable rapid expression once a specific lineage is selected, a process that might involve the loading of a stalled form of RNA polymerase II that could facilitate swift activation as differentiation is initiated (Brookes et al. 2012).
Dynamic Polycomb activity regulates the differentiation of neural stem cells in vitro
Bivalency is not restricted to ESCs but is found in other multipotent cell populations, including multipotent neural and glial progenitor cells (Mohn et al. 2008; Cui et al. 2009; Xie et al. 2013; Zhu et al. 2013). Formation of lineage-restricted progenitors during development involves both the loss and de novo acquisition of bivalently modified loci: some existing PcG targets are resolved to completely active or inactive states, whereas PcG complexes also relocate to new progenitor-specific targets that might be expressed in the subsequent stage of differentiation (Mohn et al. 2008). For instance, as pluripotent ESCs undergo neural fate specification, bivalent developmental genes that must be activated in neural progenitors lose the repressive H3K27me3 modification to become actively transcribed (Burgold et al. 2008; Mohn et al. 2008). This process is mediated, at least in part, by the Jmjd3 H3K27me3 demethylase, whose ability to facilitate the activation of neural-progenitor-associated genes such as Nestin is indispensable for neural fate acquisition (Burgold et al. 2008). Other activities also contribute to alleviating Polycomb-mediated gene repression during neural specification. For example, Zuotin-related factor 1 (Zrf1) was recently found to be required for the chromatin displacement of PRC1 to activate neural genes such as Pax6 during neural cell specification (Aloia et al. 2014).
Once specified, neural progenitors are restrained from further differentiation by Polycomb-mediated repression of genes associated with neuronal or glial differentiation. This involves both the maintenance of bivalent domains originally established in ESCs and the acquisition of new bivalent domains at previously unmarked genes in neural progenitors (Mohn et al. 2008; see examples in Fig. 1). Successive recruitment of PcG complexes to form new bivalent domains is therefore a recurring process, one that is used by multiple progenitor cell types to establish developmental potential by priming new groups of genes for rapid expression or repression (Fig. 1).
Changes in histone modification state accompany cell state transitions during neural development. a Pluripotent cells of the early embryo (ICM inner cell mass) and embryonic stem cells (ESC) can differentiate into multiple neuronal and glial cell types. This process involves cellular transitions through distinct progenitor cell states. Micrographs show human ESCs and their directed differentiation into Nestin-expressing neural stem cells (NSC; green signal in cytoplasm, with blue counterstaining of nuclei), beta-III-tubulin-expressing neurons, and glial fibrillary acidic protein (GFAP)-expressing astrocytes (NPC neural progenitor cell, OPC oligodendrocyte precursor cell). b Chromatin modification state changes occur at lineage-specific genes during the transition of pluripotent ESCs into differentiated derivatives including neurons. Hallmarks of this process include the acquisition of a bivalent modification state by new sets of genes in each progenitor type. Bivalent genes are silent but are primed for rapid expression as differentiation occurs. As a progenitor cell differentiates, bivalent genes associated with the selected lineage resolve into an active chromatin state, whereas genes associated with alternative lineages adopt more stable heterochromatic configurations (blue open arrows: transitions to a poised bivalent state, gray arrows: transitions from a bivalent to an active [H3K4me3 only] state). Examples for each type of gene are from Mohn et al. (2008)
During neurogenesis, a subset of progenitors activates neuronal genes in response to external and internal cues, whereas other progenitors maintain the repression of neuronal genes and have the capacity to contribute to gliogenesis. As neurogenesis is completed, and as gliogenesis subsequently begins, these gliogenic progenitors resolve bivalent genes with roles in astrocyte or, subsequently, oligodendrocyte differentiation to an actively transcribed state. This process appears to be particularly important for the specification of oligodendrocyte precursor cells (OPCs), which maintain substantially elevated levels of Ezh2 in comparison with differentiating neurons and astrocytes (Sher et al. 2008). Ectopic expression of Ezh2 in differentiating neural progenitor cell (NPCs) drives the formation of oligodendrocytes, while loss of Ezh2 has the opposite effect. Ezh2 expression remains high until late stages of oligodendrocyte differentiation, implying a role for PRC2, not just in OPCs, but throughout the multistep process of oligodendrocyte maturation (Sher et al. 2012). Indeed, analysis of the genome-wide distribution of Ezh2 in cultured murine neural stem cells (NSCs) and premature oligodendrocytes (pOLs) indicates that pOLs retain Ezh2 at a subset of targets involved in neuronal or astrocyte fate acquisition, supporting the hypothesis that the varying competence of the different progenitor cell populations is determined by the Polycomb-mediated repression of distinct targets.
Polycomb complexes regulate multiple distinct cell state transitions in the developing nervous system
Although the directed differentiation of cultured ESCs and neural progenitors has provided a tractable experimental model in which to dissect the molecular dynamics of chromatin regulation by PcG protein complexes, in vitro models are necessarily limited in their capacity to recapitulate the in vivo dynamics of mammalian neural development. The mammalian nervous system is among the most complex biological systems in existence. The human brain is composed of billions of neuronal and glial cells of numerous subtypes, all arranged in an intricate three-dimensional topology essential for proper function. The formation of this system requires multiple cell state transitions as progenitor pools proliferate, migrate, differentiate, and integrate to form circuits. Below, we review evidence demonstrating diverse roles for Polycomb complexes throughout this process.
Polycomb complexes regulate cortical progenitor renewal and differentiation
The mammalian nervous system begins its development as a simple neuroepithelial sheet that will subsequently be organized along the dorsoventral and rostrocaudal axes through complex interplay between extracellular morphogens with asymmetric spatial distributions. Cells located at the rostral extent of the neural plate will give rise to the brain, including the cortex, whereas more caudally positioned cells will eventually form the spinal cord.
After neurulation, cells of the rostral neuroepithelium (i.e., the ventricular zone) undergo symmetric self-renewing divisions to expand the neuroepithelial cell pool. At the beginning of cortical neurogenesis, neuroepithelial cells become multipotent neural progenitors called radial glial (RG) stem cells. RG stem cells undergo asymmetric divisions to generate additional RG stem cells and neurons, either directly or through the production of fate-restricted basal progenitors that leave the apical surface of the ventricular zone and move into the subventricular zone (SVZ). Basal progenitors usually undertake only a single symmetric division to generate two neurons, although they can also undergo asymmetric divisions to generate a basal progenitor and a neuron. This differentiation program follows a stereotyped inside-to-out pattern to generate the six layers of the cortex, with neurons located in the deep layers of the cortex being produced prior to neurons found in more superficial layers (Kriegstein and Alvarez-Buylla 2009; Martynoga et al. 2012; Greig et al. 2013; MuhChyi et al. 2013).
After its formation, the RG stem cell population must give rise to a large number of different neuronal and glial cell types. In addition to spatial information, which confers a dorsoventral and rostrocaudal identity upon neuronal populations, temporal information contributes to this diversity. Throughout the central nervous system (CNS), NPCs produce subtypes of neurons in a defined order before astrogliogenesis is initiated, and astrocytes are formed before most oligodendrogenesis is initiated (Walsh and Cepko 1992; Qian et al. 2000; Hirabayashi and Gotoh 2005; Shen et al. 2006; Noctor et al. 2008; Costa et al. 2009).
Although secreted extracellular signals guide the patterning of the developing cortex in vivo, clonal analyses of individual murine NPCs indicate that cell intrinsic mechanisms also play an instrumental role in controlling the differentiation potential of these cells, progressively restricting their competence as development proceeds. Neo-cortical progenitors generate lower-layer neurons after fewer cell divisions than upper-layer neurons, and progenitors from older mice exhibit a reduced capacity to generate earlier-born neuronal subtypes (Shen et al. 2006). Furthermore, experiments on mouse and human NSCs have demonstrated that the temporal order in which neural progenitors generate subtypes of neocortical neurons in vivo is retained in vitro, implying that the progressive cell-intrinsic restriction of neural progenitor competence is a general feature of mammalian neural development (Eiraku et al. 2008; Gaspard et al. 2008).
In addition to cortical neuron subtype specification, the switch from neurogenesis to gliogenesis also appears to involve cell-intrinsic mechanisms. At the onset of the neurogenic phase, extracellular Wnt signaling initiates the expression of the transcription factors Neurogenin 1 and 2 (Ngn1/2), which activate the expression of other neurogenesis-promoting genes (Hirabayashi et al. 2004; Israsena et al. 2004). The transition from neurogenesis to gliogenesis involves the activation of astrocytic genes such as glial fibrillary acidic protein (GFAP) by the Jak-STAT signaling pathway, signaling that is stimulated by extracellular signals including ciliary neurotrophic factor (CNTF), leukemia inhibitory factor (LIF), and bone morphogenetic protein 2 (BMP2; Johe et al. 1996; Bonni et al. 1997; Rajan and McKay 1998; Nakashima et al. 1999; He et al. 2005; Hsieh and Gage 2005; Shen et al. 2006; Yoshimatsu et al. 2006). However, despite the presence of gliogenic cytokines during early corticogenesis, early cortical progenitors do not generate glia (Uemura et al. 2002; Derouet et al. 2004), and young cortical progenitors cultured in vitro are less responsive to gliogenic cytokines than older progenitors (He et al. 2005). Conversely, neurogenic Wnt ligands continue to be expressed during astrogliogenesis, suggesting the existence of a cell-intrinsic mechanism for regulating the switch from neurogenic to gliogenic competence in neural progenitors (Shimogori et al. 2004).
The processes that limit cellular competence in cortical progenitors involve negative crosstalk between regulatory pathways. For example, Ngn1/2 blocks gliogenesis by sequestering the coactivators CBP/p300 and Smad1, which are required to promote transcriptional activation of glial genes such as GFAP in response to pro-astrocytic STAT signaling (Sun et al. 2001). However, a variety of epigenetic mechanisms have also been implicated. In particular, mounting evidence suggests that the Polycomb complexes form one of the key regulatory axes responsible for the sequential limitation of the gliogenic and neurogenic competence of neural progenitors in the developing cortex. Ezh2 is highly expressed in neural progenitors but is down-regulated during the differentiation of cortical neurons (Pereira et al. 2010). Premature loss of Ezh2 from the start of the neurogenic period (through conditional deletion from embryonic day 9.5 [E9.5] in the mouse) accelerates neurogenesis and neuronal differentiation, exhausting the apical and basal progenitor pools and resulting in a thinned cortex (Pereira et al. 2010; Testa 2011). The accelerating effect of Ezh2 deletion is not limited to neurogenic precursors but includes glial lineages, as evinced by the drastically expedited appearance of mature astrocytes in the mouse E16 ventricular zone (over 4 days ahead of schedule; Pereira et al. 2010; Testa 2011).
In addition to limiting the onset of neural progenitor differentiation, evidence exists that Polycomb activity regulates the developmental transition from neurogenesis to astrogliogenesis. Loss of Ezh2 or the PRC1 component Ring1b at later stages of cortical development prolongs neurogenesis, rather than accelerating it. This protracted neurogenic phase occurs at the expense of astrogliogenesis, the onset of which is delayed (Hirabayashi et al. 2009). Control of the transition between the neurogenic versus the gliogenic phase of development is exerted by the direct recruitment of PcG complexes to the promoters of the Ngn1/2 genes, progressively inhibiting their expression as corticogenesis proceeds (Hirabayashi et al. 2009). Polycomb-mediated repression of neurogenic genes in turn facilitates activation of astrocytic genes such as GFAP, e.g., by alleviating the Neurogenin sequestration of coactivators of the pro-astrocytic Stat3 transcription factor (Sun et al. 2001).
Some data also suggest that Polycomb activity continues to be important in later stages of cortical gliogenesis, as the production of astrocytes gives way to oligodendrogenesis. In the ventral telencephalon, the Dlx1 and Dlx2 transcription factors are required both to produce inhibitory GABA (gamma-aminobutyric acid)-ergic neurons and to repress oligodendrocyte formation promoted by Olig2 (Petryniak et al. 2007). Repression of neurogenic Dlx1/2 activity is therefore one requirement for the developmental transition to oligodendrogenesis. In adult NSCs, MLL activity maintains Dlx2 expression by antagonizing PcG-mediated repression (Lim et al. 2009), whereas in the embryonic cortex, loss of Polycomb activity (Ring1b) de-represses Dlx2 (Hirabayashi et al. 2009). These findings suggest that cell intrinsic regulatory mechanisms involving PcG also contribute in vivo to the developmental transition to oligodendrogenesis.
Roles for Polycomb function beyond cortical neurogenesis and gliogenesis
Beyond their functions as regulators of progenitor specification in the embryonic cortex, PcG complexes continue to play roles in adult neural progenitors and differentiated neurons. In the adult, a subset of astrocytes in the SVZ acts as NSCs, with a neurogenic competence that is lacking in other adult astrocytes. Recent work indicates that continued expression of Ezh2 distinguishes these neurogenic astrocytes from their glial-restricted counterparts. Specifically, Ezh2 controls the neurogenic competence of adult SVZ-NSCs by repressing the expression of Olig2 to permit neuronal differentiation, while simultaneously preventing the activation of genes associated with non-SVZ neuronal subtypes (Hwang et al. 2014). PRC1 also participates in the regulation of adult NSCs by modulating proliferation and self-renewal. The PRC1 component Bmi1 can maintain the proliferation and self-renewal of adult NSCs and NPCs through the repression of the cell cycle inhibitors p16Ink4a, p19Arf, and p21 (Molofsky et al. 2003, 2005; Bruggeman et al. 2005; Fasano et al. 2007, 2009; Román-Trufero et al. 2009). Accordingly, depletion of the PRC1 protein Ring1b from cultured adult olfactory bulb NSCs impairs NSC proliferation and self-renewal (Román-Trufero et al. 2009).
Finally, recent research has expanded the contexts in which PcG complexes function to include terminally differentiated neurons. During tangential migration of precerebellar neurons and formation of connections with the cortex, Ezh2-dependent regulation of transcriptional programs is required to maintain positional information to control topographic neuronal guidance and connectivity (Di Meglio et al. 2013). Mouse knockout studies of Jmjd3, the histone demethylase primarily responsible for the removal of H3K27me3, further demonstrate the importance of the precise regulation of H3K27me3-dependent gene repression for neuronal maintenance. Inactivation of Jmjd3 in the mouse leads to perinatal lethality as a result of disrupted maintenance of the pre-Botzinger complex (PBC), the pacemaker of the respiratory rhythm generator (Burgold et al. 2012). Specifically, whereas the early formation of the respiratory rhythm generator is not affected by the loss of Jmjd3, the maintenance of the PBC is perturbed, due to aberrant silencing of PBC-related genes. Among the genes dysregulated by the loss of Jmjd3 are reelin, a glycoprotein involved in neuronal migration, and Neph2, a transmembrane protein with roles in synaptogenesis (Burgold et al. 2012). In differentiated neurons, PRC2 also modulates neuronal activity-dependent processes including dendritic arborization (Qi et al. 2014). Together, these results support a continuing role for PcG complexes as regulators of neural circuit formation and maintenance, above and beyond their developmental functions as regulators of neuronal and glial fate specification.
Polycomb complexes regulate motor neuron subtype specification in spinal cord
During the period of embryogenesis when patterning of the rostral neural tube into the brain occurs, the caudal neuroepithelium is also patterned as the neural plate closes to form the spinal neural tube. The ventral region of the spinal neural tube is colonized by a progenitor population that will give rise to diverse subtypes of neurons, including motor neurons (MN). Spinal MN progenitors are grouped into columns on the basis of their rostocaudal location along the neuraxis, which is established by the expression of region-specific Hox transcription factors (Philippidou and Dasen 2013; Davis-Dusenbery et al. 2014). MN columns are comprised of sets of MNs arranged longitudinally along the rostrocaudal axis of the spinal cord, and neurons of each column project to distinct regions in the periphery. For example, the preganglionic motor column is located at the thoracic level of the neuraxis and contains visceral MNs that innervate sympathetic ganglia, whereas MNs of the lateral motor column span both the brachial and lumbar levels to innervate the limbs.
Hox gene expression in the spinal neural tube is patterned in two phases. First, extracellular signals secreted from the adjacent rostral somitic mesoderm and caudal presomitic mesoderm create opposing morphogen gradients that establish broad territories of Hox gene expression in proliferating spinal progenitors (Philippidou and Dasen 2013; Davis-Dusenbery et al. 2014). After spinal progenitors undergo their final mitosis, the boundaries of posterior Hox domains are further refined by cross-repressive interactions between the transcriptional programs originally induced by the various extracellular signals, a process that is required to sharpen and maintain expression borders.
Recent work has demonstrated the involvement of Polycomb repressive complexes at both steps of spinal progenitor differentiation. In one study, PRC2 was found to be essential for the initial repression of Hox gene expression prior to regionalization of spinal MNs by gradients of retinoic acid, Wnt, and fibroblast growth factor, and for maintaining the repression of alternative Hox codes after regionalization (Mazzoni et al. 2013). Treatment of differentiating mouse neural progenitors with retinoic acid results in the binding of retinoic acid receptors to the Hox1-Hox5 genes, triggering the rapid domain-wide clearance of PcG-dependent H3K27me3 repression to enable Hox gene activation and acquisition of cervical spinal identity. Wnt and fibroblast growth factor signals instead activate the expression of the Cdx2 transcription factor, whose subsequent binding to the Hox1-Hox9 genes clears H3K27me3 from these domains to specify brachial or thoracic spinal identity.
Polycomb also appears to be critical for the cross-repressive refinement of Hox expression boundaries in post-mitotic spinal neurons, with PRC1 exhibiting a dose-dependent regulation of MN subtype differentiation (Golden and Dasen 2012). Depletion of Bmi1 from the developing spinal cord results in the de-repression of more posterior Hox genes and alters MN fate, converting forelimb lateral motor column MNs to a thoracic preganglionic motor column identity. Intriguingly, ectopic expression of Bmi1 at thoracic levels has the opposite effect, converting preganglionic motor column MNs to a lateral motor column identity. The dose-dependent roles of PRC1 in the developing spinal cord imply that absolute levels of Polycomb activity are an important determinant of its regulatory targets in this context.
Molecular mechanisms governing PcG protein activity in neural development
The previous examples emphasize the multiple, temporally distinct roles played by the PcG complexes in neural development. Polycomb functions differ, not just between stem cells of different types, but also within the same cell type at different developmental stages. These observations imply the existence of molecular machinery capable of modifying the cohort of genes targeted by Polycomb in response to intrinsic and environmental cues. In Drosophila, specific DNA sequences called Polycomb response elements (PREs) recruit PcG protein complexes to their targets. However, in vertebrates, the isolation of response elements with similar functionality to fly PREs has proven to be exceedingly difficult, and no consensus motif for a DNA element capable of recruiting PRC2 or PRC1 has been identified. In the absence of a clear association between transcription factors and Polycomb binding, several alternative mechanisms contributing to Polycomb complex recruitment have been proposed. The complex biochemistry governing the general recruitment of mammalian Polycomb complexes is outside the scope of this review and has been described elsewhere (for an excellent review, see Simon and Kingston 2013). Instead, we will focus on the progress that has been made toward uncovering context-specific mechanisms that govern Polycomb recruitment specifically in the nervous system.
Several types of mechanisms for Polycomb recruitment have been proposed. In general, these involve: (1) the role of the chromatin environment and histone modification state in Polycomb recruitment, (2) the ability of several proteins to interact with either PRC2 or PRC1 complexes to mediate their recruitment to specific target sites, or (3) the role of interactions between Polycomb complexes and long non-coding RNAs in Polycomb recruitment. In the sections below, we discuss each of these potential recruitment mechanisms in detail and describe their known or probable roles in PcG complex recruitment during nervous system development.
Local chromatin environment influences activity of PRC2
Recently, the chromatin environment and histone modification state have been shown to influence recruitment of PcG complexes to and activity on chromatin. In addition to the enzymatic subunit Ezh2, PRC2 complexes include the proteins Eed and Suz12 in stoichiometric ratios, and both proteins are required for effective enzymatic activity (Fig. 2a, b). The latest research suggests that both Suz12 and Eed function as adaptors that bind to and modify Ezh2 catalysis in response to local chromatin cues.
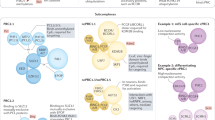
Polycomb complex diversity. a H3K27me3 modification of chromatin by the EZH1/2 subunit of Polycomb repressive complex 2 (PRC2) promotes recruitment of the “canonical” PRC1 complex, which catalyzes ubiquitylation of H2AK119 to repress gene expression. Additional “non-canonical” forms of the PRC1 complex can be recruited to chromatin in a PRC2-independent manner. b PRC2 associates with several non-stoichiometric proteins (yellow). PRC2 subunits EZH2 and SUZ12 also have functional domains (gray dashed areas), including RNA-binding domains (RBD) that can associate with long non-coding RNAs (lncRNA). Both protein and lncRNA associations have the potential to facilitate context-dependent recruitment to chromatin. c “Canonical” PRC1 complexes have multiple forms, through inclusion of specific subunit variants. The chromodomain (CD) of the Cbx subunit can also associate with lncRNAs. d “Non-canonical” PRC1 complexes have an even more diverse potential subunit composition with some complex variants as shown
Suz12 is a zinc-finger protein whose cumulative interactions with Ezh2 and neighboring histones (via an amino-terminal VEFS-BOX domain) can positively and negatively regulate PRC2 activity (Schmitges et al. 2011; Chan et al. 2012; Ciferri et al. 2012; Yuan et al. 2012). The ability of Suz12 to bind neighboring histone H3 confers upon PRC2 a substantial preference for densely packed polynucleosomes relative to dispersed substrates. Indeed, increased polynucleosome density augments PRC2 catalysis up to 30-fold over controls in vitro (Chan et al. 2012; Yuan et al. 2012). Eed instead seems to be primarily involved in the feed-forward of PRC2 activity via the ability of its WD40 β-propeller to recognize H3K27me3 and positively to stimulate Ezh2 (Margueron et al. 2009; Xu et al. 2010). The addition of K27me3-containing peptide to purified PRC2 complexes in vitro results in an up to seven-fold increase in PRC2 catalytic activity, consistent with experiments demonstrating the essential requirement of Eed for proper PRC2 function (Montgomery et al. 2005; Margueron et al. 2009). The potent allosteric activation of Ezh2 catalysis induced by EED binding to H3K27me3 might explain why acetylation of H3K27 inhibits PRC2 activity, and accordingly, why the removal of H3K27ac by the nucleosome remodeling and deacetylation (NuRD) complex is essential for PRC2 recruitment during ES differentiation (Tie et al. 2009; Pasini et al. 2010b; Reynolds et al. 2012).
Given the considerable influence of the non-catalytic subunits of PRC2 on methyltransferase activity, PRC2 appears to function as a complex holoenzyme, integrating the regulatory inputs from the core subunits and other cofactors to sense the local chromatin environment and adjust activity accordingly (Margueron and Reinberg 2011; Ciferri et al. 2012). The ability of PRC2 to sense the local chromatin environment might also link PRC2 target selection in neural development to the activities of other chromatin regulators with well-described functions in neural differentiation, such as the ATP-dependent BAF chromatin remodeling complexes (Ronan et al. 2013; Narayanan and Tuoc 2014).
PRC1 subunit composition confers cell-state-specific activity
Compared with PRC2, PRC1 complexes are highly heterogeneous in composition. “Canonical” PRC1 complexes have four core subunit types, each of which can be represented by several different PcG proteins: Cbx2/4/6/7/8, Ring1a/b, Pcgf1/2/3/4/5/6, and Ph1/2/3 (Fig. 2c; Levine et al. 2002). Canonical PRC1 complexes can be recruited to chromatin through Cbx protein binding to PRC2-deposited H3K27me3 (Wang et al. 2004), although H3K27me3-independent targeting of PRC1 complexes has also been documented. Once chromatin-bound, the Ring1a/b subunit ubiquitylates H2A lysine 119 (H2AK119Ub) (de Napoles et al. 2004; Cao et al. 2005; Fig. 2a). This ubiquitylation event promotes gene repression (Endoh et al. 2012) and can impair transcriptional elongation (Stock et al. 2007). PRC1 can also repress gene expression through Ring1b/Rnf2-mediated chromatin compaction (Francis et al. 2004; Eskeland et al. 2010; Endoh et al. 2012).
Evidence suggests that subunit variants are not redundant, but rather function in distinct complexes. For example, mutation of the PRC1 protein Ring1b results in embryonic lethality during gastrulation (Voncken et al. 2003), whereas Ring1a mice are viable (de Napoles et al. 2004). Although the roles of many of the PRC1 subunit variants in development remain uncharacterized, they presumably contribute to context-specific functions of PRC1 (Turner and Bracken 2013). For instance, the replacement of Cbx7-containing PRC1 complexes by Cbx2/4-containing PRC1 complexes has recently been shown to mediate a transition from pluripotency to fate acquisition (Morey et al. 2012; O'Loghlen et al. 2012). This example illustrates the potential for variant subunits to endow PRC1 with cell-state-specific functions.
PRC1 complexes also exist in non-canonical forms in which the Cbx subunit is substituted for alternative proteins (Fig. 2d; García et al. 1999; Sánchez et al. 2007; Gao et al. 2012; Hisada et al. 2012; Junco et al. 2013). Notably, Pcgf-Ring1a/b complexes lacking a Cbx subunit can instead associate with Rybp or its homolog Yaf2, in which case Rybp stimulates Ring1b-mediated ubiquitylation of H2AK119 in a PRC2/H3K27me3-independent manner (Gao et al. 2012; Tavares et al. 2012).
Many PRC2 and PRC1 core subunits, variant subunits, and associated recruitment factors or accessory proteins are expressed in the developing nervous system. The Gene Expression Database at the Mouse Genome Informatics Resource catalogs published temporal and spatial expression patterns of Polycomb protein expression reported to date for the mouse nervous system (Fig. 3). Some Polycomb subunits are expressed in the CNS from the onset of neural plate formation (embryonic day 8–8.5 (E8–8.5)), with other subunits being detected from the onset of neurogenesis (around E10.5) and through post-natal stages. Within the nervous system, the expression of many subunits is reported in the fore-, mid-, and hindbrain regions, with core PcG subunits frequently showing enrichment in the ventricular zone of the cortex. Expression of some subunits has also been reported in the spinal cord. These data support potential roles for both canonical and non-canonical Polycomb complexes in neural development.
Expression of Polycomb complex proteins and recruitment factors in the developing nervous system. a–a’’’ In situ hybridization data for four core Polycomb proteins at embryonic day 14.5 (E14.5; Genepaint database) showing robust expression in the ventricular zone of the cortex and other locations in the central nervous system (CNS). b, c The Gene Expression Database at the Mouse Genome Informatics Resource was used to catalog (b) temporal and (c) regional expression of Polycomb subunits reported for the mouse CNS between embryonic day 8 (E8) and post-natal day 7 (P7). Heat maps indicate developmental time windows (b) or CNS regions (c) in which expression is documented as present (red) or absent (green), or in which no data are reported (black). CNS expression of some Polycomb core subunits can be seen from the onset of neural plate formation, with expression of other subunits being detected during neurogenesis and through post-natal stages. Within the CNS, expression of many subunits is reported in the fore-, mid-, and hindbrain regions, with core PcG subunits, in particular, showing enrichment in the ventricular zone of the cortex. Expression of some subunits has also been reported in the spinal cord
Evidence from mouse knockout models suggests that non-canonical PRC1 complexes play specific roles in neural development. Whereas constitutive knockout of Rybp results in embryonic lethality at early post-implantation stages, chimeric embryos show myriad phenotypes indicative of aberrant neural development, including forebrain overgrowth and localized regions of disrupted neural tube closure (Pirity et al. 2005).
Protein recruitment factors regulate Polycomb complex targeting in neural development
Both PRC2 and PRC1 have been shown to interact directly with proteins that can facilitate their recruitment to chromatin through distinct mechanisms. Below, we discuss the way that some of these proteins might regulate the recruitment of Polycomb complexes to specific target subsets in neural development.
Jarid2 might regulate context-dependent PRC2 activity during neural development
Among the most prominent of the non-core PRC2-associated proteins is Jarid2, a catalytically inactive member of the jumonji family of histone demethylases that directly interacts with PRC2 in nearly stoichiometric ratios in ESCs (Peng et al. 2009; Shen et al. 2009; Li et al. 2010; Pasini et al. 2010a). Jarid2 has well-established roles in neurulation in mouse models and in directed differentiation of ESCs, including differentiation toward neuronal lineages: Jarid2 (also called Jumonji) was initially identified in the mouse based upon a gene trap mutation that resulted in defects in neural tube closure in the midbrain region, demonstrating strain-dependent requirements for neurulation (Takeuchi et al. 1995, 1999). Jarid2 was further shown to be required to repress cyclin D1 expression to coordinate cell cycle exit and neuronal migration during neurogenesis in the mouse hindbrain (Takahashi et al. 2007). In addition, several studies have examined the role of Jarid2 during directed differentiation of mouse ESCs. In this context, Jarid2 maps to many PcG target genes (Pasini et al. 2010a), and its activity is required for the induction of differentiation-related genes (Peng et al. 2009; Shen et al. 2009; Pasini et al. 2010a), including the expression of the neuronal marker Sox11 (Shen et al. 2009). Although both Jarid2 and PRC2 are required for neural development, whether the Jarid2-PRC2 interaction is required for PcG recruitment or for activity in the developmental contexts described above remains unknown.
Several studies do suggest that Jarid2 is important for the recruitment of PRC2 to its targets in ESCs, although whether this involves the direct binding of Jarid2 to DNA via its zinc finger or ARID domains, or whether some other mechanism is involved remains unclear. However, Jarid2 is unlikely to be the sole factor responsible for PRC2 recruitment in ESCs: the loss of Jarid2 does not result in the extensive re-expression of PRC2 target genes, as is observed when ablating a core PRC2 subunit, and the overall impact of Jarid2 loss upon H3K27me3 levels in cells is decidedly mild (Landeira et al. 2010; Margueron and Reinberg 2011).
A recent study suggests that conflicting reports regarding the importance of Jarid2 to PRC2 function might be reconciled by the newly discovered ability of Ezh1 to compensate for Jarid2: both Ezh1 and Jarid2 have innate nucleosome-binding capacity, and PRC2 recruitment and enzymatic activity in Jarid2-deficient myoblasts has been shown to depend on the expression of Ezh1 but not Ezh2 (Son et al. 2013). Jarid2 and Ezh1 also display reciprocal expression patterns: Jarid2 is expressed most highly in pluripotent and early-lineage-committed cells, whereas Ezh1 is most highly expressed in later, more differentiated cell types. Thus, Ezh1 might have functions that are non-redundant with Ezh2, including the promotion of the access of Ezh2-containing PRC2 complexes to chromatin in committed cell types that lack Jarid2 (Son et al. 2013). The testing of this hypothesis in models of lineage commitment other than myoblast differentiation will be important. In the particular context of neural development, the determination of whether a developmental time occurs at which the dependence of PRC on Jarid2 is supplanted by Ezh1, and whether this switch is associated with progressive restriction of neural progenitor competence, will be of interest.
Chd4 and Chd5 regulate PRC2 recruitment to promote neurogenesis in cortical progenitors
Effector proteins endowed with H3K27me3-“reader” domains can impinge on PRC2 activity in a cell-type-specific manner. For example, chromodomain helicase DNA-binding protein 5 (Chd5) is a protein that has the ability to remodel nucleosomes and that is frequently deleted in aggressive neuroblastoma (Koyama et al. 2012). Chd5 is characterized by two chromodomains that bind H3K27me3 and are essential for its function as a regulator of cortical neurogenesis (Egan et al. 2013). Depletion of Chd5 in differentiating neural progenitors leads to de-repression of a subset of PRC2 targets and the failure to activate the expression of key neuronal genes (Egan et al. 2013).
Another chromodomain helicase DNA-binding protein, Chd4, has also recently been shown to be a critical interaction partner of Ezh2 in cortical progenitors, in which it is required specifically for the PRC2-mediated suppression of the astrogliogenic marker gene GFAP. Accordingly, experimental depletion of Chd4 RNA from cortical progenitors in the developing neocortex promotes astrogliogenesis in vivo. Whereas Chd4 is a frequent component of the NuRD complex, depletion of other NuRD components do not result in increased astrogliogenesis, suggesting that Chd4 functions independently of the NuRD complex to regulate neural progenitor competence. Together, these data suggest that target gene-specific mechanisms involving crosstalk with other chromatin “readers” can influence Polycomb activity to control neural cell fate transitions.
Aebp2 is a co-activator of PRC2 and might regulate recruitment in neural development
Aebp2 is a Gli-type zinc finger frequently found in association with PRC2 and appears to enhance PRC2 enzymatic activity (Cao et al. 2002; Cao and Zhang 2004; Ciferri et al. 2012). Whereas homozygous loss of Aebp2 in mouse models is embryonic lethal, heterozygous animals have phenotypes suggesting a role for Aebp2 in the regulation of neural crest development (Kim et al. 2011). At present, whether Aebp2 primarily influences PRC2 enzymatic activity or recruits PRC2 to some of its targets through its ability to bind DNA remains unclear. Analysis of Aebp2 target sites in brains of 1-month-old mice has revealed a considerable overlap between Aebp2 and PRC2 target genes, supportive of a role for Aebp2 in the recruitment of PRC2 complexes during neural development (Kim et al. 2009). However, because this study relied on the cloning of DNA fragments isolated by chromatin immunoprecipitation (ChIP), combined with Sanger sequencing, this analysis should be performed on a genome-wide scale to validate the extent of Aebp2 and PRC2 overlap in bound chromatin locations. Moreover, if Aebp2 is required for PRC2 recruitment, the loss of Aebp2 should abrogate PRC2 recruitment specifically at common target sites, without affecting sites targeted only by PRC2; this remains to be tested. Therefore, while not definitive, the current evidence is supportive of a role for Aebp2 in the targeting of PRC2 during the development of the brain and neural crest.
Kdm2b regulates recruitment of non-canonical PRC1 complexes
Kdm2b (also known as Fbxl10 or Jhdm1b) is an H3K36-specific histone demethylase that was initially identified as a factor controlling cell proliferation and senescence by regulating the Ink4a-ARF-Ink4b locus (He et al. 2008). Recent work has revealed that Kdm2b also facilitates the recruitment of PRC1 in some contexts (X. Wu et al. 2013). Kdm2b-containing PRC1 complexes constitute a distinct type of non-canonical complex containing Ring1b, Pcgf1 (Nspc1), and Rybp, but not Cbx proteins (X. Wu et al. 2013; Fig. 2a, d). Kdm2b binds non-methylated CpG island sequences (CGIs) via its CxxC-type zinc-finger domain, promoting PRC1-mediated H2AK119 ubiquitylation at a subset of its target sites (Koyama-Nasu et al. 2007; Farcas et al. 2012; He et al. 2013; X. Wu et al. 2013). Although Kdm2b does indeed bind the CGIs of Polycomb-repressed genes, it also binds CGIs throughout the genome, a large fraction of which corresponds to actively transcribed genes that are not PRC1 targets. These observations indicate that the presence of Kdm2b at unmethylated CGIs is not sufficient for stable PRC1 recruitment.
Interestingly, although the process of PRC1 recruitment to target sites has been generally considered a hierarchical event that depends on prior PRC2-mediated H3K27me3, Kdm2b has recently been shown to recruit a Pcgf1-containing variant PRC1 complex to CGIs, with PRC1 chromatin binding leading to subsequent PRC2 recruitment and H3K27 methylation (Blackledge et al. 2014). Another recent study has also found that PRC1 recruitment to and H2A ubiquitylation at unmethylated CpG-rich chromatin regions in ESCs is sufficient to recruit PRC2 to chromatin (Cooper et al. 2014). This work supports a role for PRC1 recruitment in directing PRC2-mediated H3K27me3 and indicates that non-canonical complexes that associate with Kdm2b act in this manner.
Kdm2b appears to play essential roles in neural development. Whereas the loss of Kdm2b in cultured ESCs can cause de-repression of lineage-specific genes and precocious differentiation (He et al. 2013), Kdm2b mutant embryos die perinatally with defects specifically in neural development, including incomplete neural tube closure, exencephaly, and increased proliferation and apoptosis of NPCs (Fukuda et al. 2011). However, at a mechanistic level, the role of Kdm2b in non-canonical PRC1 targeting in the context of neural and glial development has yet to be elucidated.
Methylation of CGIs flanking promoters inhibits PRC2 in neural progenitors
With regard to the chromatin state, another aspect that can influence Polycomb recruitment is methylation of CGIs. Highly methylated CGIs are strongly anti-correlated with H3K27me3 and PRC2 binding, a finding supported by multiple studies; this might partially explain the restricted repertoire of PRC2 targets in various lineages (Mohn et al. 2008; Brinkman et al. 2012; Lynch et al. 2012; Xie et al. 2013). Whereas sequential ChIP and bisulfite sequencing experiments confirm that H3K27me3 and DNA methylation can indeed co-localize in the genome, this co-localization is never seen in regions with high CpG density, implying that high levels of DNA methylation somehow inhibit the local activity of PRC2 (Brinkman et al. 2012; Statham et al. 2012).
Loss-of-function studies of Dnmt3a and Dnmt3b also support the link between DNA methylation and PRC2 recruitment, particularly in neural development. In murine ESCs, the simultaneous knockout of Dnmt3a/b results in the depletion of DNA methylation from intragenic and intergenic CGIs, concomitant with an increase of H3K27me3 (Hagarman et al. 2013). Consistent with these results, a study examining the role of Dnmt3a in murine NSCs has found that, whereas Dnmt3a is dispensable for NSC renewal and proliferation, the knockout of Dnmt3a causes stunted neurogenic potential both in vitro and in vivo (H. Wu et al. 2010). Loss of Dnmt3a significantly decreases the expression of neurogenic genes, whereas genes involved in gliogenesis are significantly upregulated, even though both classes of targets experience a decrease in DNA methylation.
The results above are inconsistent with the straightforward interpretation of DNA methylation as a repressive mark. However, they might be explained by the striking observation that most genes that are down-regulated after Dnmt3a ablation have H3K4me-rich CGIs in their proximal promoters, whereas Dnmt3a binding (and associated DNA methylation) is present only in the inter- and intragenic regions flanking their promoter CGIs (H. Wu et al. 2010). In contrast, genes that are up-regulated after the loss of Dnmt3a feature CpG-poor proximal promoters, with low expression and little modification by H3K4me3. This led the authors to hypothesize that DNA methylation in regions flanking CpG-rich proximal promoters acts as an activating signal, preventing repression by some alternative mechanism (H. Wu et al. 2010). Genome-wide profiling of H3K27me3 in Dnmt3a-knockout NSCs has confirmed that genes that are down-regulated in response to DNA hypomethylation exhibit increased PRC2 activity. These results suggest that DNA methylation in gene bodies and in intragenic regions flanking the proximal promoter activates gene expression by inhibiting the enzymatic activity of PRC2 (Fig. 4). Future experiments exploring the dynamics of genome-wide DNA methylation in models of neural development will be required to determine the extent to which changing patterns of DNA methylation can explain the cell-state-specific selection of targets by otherwise ubiquitous chromatin modifiers such as PRC2.
Relationship between DNA methylation and Polycomb recruitment. Promoters of active genes are frequently (a) unmethylated (open circles), although (b) DNA methylation (closed circles) flanking the promoter can also facilitate the active state by preventing PRC2-mediated repression. Repressed genes might have methylated promoter DNA (c), although demethylation (d) allows PRC2-mediated silencing (arrows: transcription start site, rectangles: gene bodies)
Long non-coding RNAs in neural development
A host of studies have also implicated long non-coding RNAs (lncRNAs) in the targeting of PRC2. Whereas the investigation of lncRNAs in the CNS is still in its infancy, studies have emerged implicating lncRNAs in neurodegenerative, neuro-oncological, and psychiatric disorders (Johnson 2012; Qureshi and Mehler 2013; Tushir and Akbarian 2013; Ziats and Rennert 2013; Zhang and Leung 2014). Therefore, many additional connections between lncRNAs and Polycomb recruitment will probably be defined in the context of neural development. Below, we first highlight roles of lncRNAs in neural development and nascent connections to Polycomb recruitment in this context. We then discuss lncRNA-dependent Polycomb PRC2 recruitment mechanisms. For more comprehensive information regarding the roles of lncRNAs in CNS development, we refer the reader to several recent reviews (Qureshi and Mehler 2012; Ng et al. 2013a, 2013b; Fatica and Bozzoni 2014).
Given the high complexity of the vertebrate nervous system, developing neural cells unsurprisingly exhibit intense transcriptional activity, including the expression of a large number of lncRNAs. Initial attempts to characterize the breadth and specificity of lncRNA expression in the brain include high-throughput in situ hybridization data from the Allen Brain Atlas; this has found regional, cell-type, and subcellular specificity in the expression patterns of over 849 lncRNAs from diverse locations in the genome (Mercer et al. 2008). Similarly, microarray analysis of lncRNA expression in human brain samples detected thousands of lncRNA transcripts with regionally restricted expression, many of which are primate-specific (Derrien et al. 2012). The enormous lncRNA transcriptome of the mammalian CNS and the preponderance of primate-specific lncRNAs expressed in the human brain suggest that functionalization of non-coding RNAs is essential for the evolution of higher brain function. This hypothesis is consistent with an apparent correlation between the proportion of non-coding DNAs in the genome and organismal complexity (Taft et al. 2011).
In one of the earliest studies to assess the neural lncRNA transcriptome, researchers used custom microarrays to evaluate the expression of a large set of lncRNAs in embryonic forebrain-derived murine NSCs and in NSCs induced in vitro to become Nkx2.1-expressing bipotent neuronal-glial progenitors, which can produce both cortical GABAergic neurons and oligodendrocytes (Mercer et al. 2010). Interestingly, unique subsets of lncRNAs were differentially expressed throughout the process of neuro-gliogenesis, including during bipotent progenitor specification, GABAergic neurogenesis, the switch to oligodendrogenesis, and the maturation of myelin-producing oligodendrocytes. While none of the transcripts identified in this study were functionally characterized, extensive differential expression of lncRNAs during neural fate specification was later confirmed in human cells (Ng et al. 2012). In this work, custom microarrays were used to examine the expression of a set of previously identified lncRNAs in human ESC-derived NPCs, both in the process of in vitro NPC specification and during the differentiation of NPCs to produce dopaminergic neurons (Ng et al. 2012). Furthermore, a subset of four intergenic lncRNAs that were preferentially enriched in differentiated neurons was also selected for transient depletion in neural progenitors using short interfering RNAs (siRNAs). Depletion of each of these four intergenic lncRNAs inhibited neurogenesis, creating an average of a five-fold reduction in the numbers of Tuj1-positive neurons after differentiation, compared with non-targeting siRNA controls. Subsequent expression analysis of lncRNA-depleted NPCs by quantitative polymerase chain reaction revealed an apparent switch from a neurogenic to a gliogenic program, as indicated by the reduced expression of neurogenic markers such as Neurog2 and the concomitant increase in expression of gliogenic marker genes such as PDGFR-alpha and myelin basic protein.
In addition to establishing that lncRNAs can indeed act as critical regulators of neurogenic and gliogenic homeostasis, at least in vitro, Ng et al. (2012) have also demonstrated that neurogenic lncRNAs identified in this study bind selectively to the Suz12 subunit of PRC2 and to a subunit of the REST transcriptional repressor complex, implying a role in the epigenetic control of transcription (Ng et al. 2012). A follow-up study by the same group has revealed that one of the previously identified neurogenic lncRNAs that does not bind to either PRC2 or REST (previously dubbed RMST for rhabdomyosarcoma 2-associated transcript) nevertheless regulates neurogenesis at the transcriptional level by binding to the Sox2 transcription factor (Ng et al. 2013a, 2013b). Like previous examples of lncRNAs that contribute to transcriptional regulation, RMST has been found to act as a physical bridge between Sox2 and chromatin, effectively functioning as a molecular scaffold that facilitates the activation of key neurogenic genes. Taken together, these data reinforce the notion that lncRNAs can act as key transcriptional regulators in neural development, and that this regulation involves the recruitment of nuclear proteins to their genomic targets.
Finally, recent experiments in mouse models have provided insight into the neural lncRNA transcriptome and roles for lncRNAs in the nervous system in vivo (Aprea et al. 2013; Ramos et al. 2013; Sauvageau et al. 2013). In one study, researchers combined laser capture microdissection with RNA-sequencing to profile the expression of lncRNAs in the adult mouse SVZ, uncovering over 3000 completely novel lncRNAs and identifying the complex isoforms of many others (Ramos et al. 2013). By combining expression data with chromatin state maps, this study also identified a subset of approximately 100 lncRNAs with putative roles as regulators of neural development. These lncRNAs exhibit the bivalent (H3K27me3 and H3K4me3, repressed but poised) chromatin state in ESCs that resolves to H3K4me3-only (active expression) specifically in SVZ-NSCs (Ramos et al. 2013). The set of genes that are bivalent in ESCs is highly enriched for lineage-determining factors, leading to the hypothesis that bivalency pre-patterns lineage specification. Indeed, chromatin state maps have been used successfully to identify novel proteins that act as regulators of cell fate in a variety of tissue contexts (Paige et al. 2012; Wamstad et al. 2012; Xu and Zaret 2012; Xie et al. 2013). Application of this chromatin state model to predict functional lncRNAs in the adult SVZ also seems fruitful. For example, two lncRNAs identified as putative regulators of neural cell fate on the basis of chromatin state transitions during neural development, namely Six3os and Dlx1as, have been found to increase the production of astrocytes at the expense of neurons when depleted from cultured SVZ-NSCs (Ramos et al. 2013).
Long non-coding RNAs might influence recruitment of PRC2 in neural development
Although investigations are ongoing, the section above highlights roles that have previously been defined for lncRNAs in neural and glial development. Evidence in multiple contexts also indicates that some lncRNAs can control gene expression by recruiting transcription factors and complexes that modify chromatin, including Polycomb complexes, to their genomic targets. The original inspiration for an lncRNA-based model of Polycomb complex recruitment is the lncRNA Xist, which directly recruits PRC2 in cis to initiate the process of X chromosome inactivation in mammals (Engreitz et al. 2013; Froberg et al. 2013). The idea that lncRNAs might contribute to epigenetic regulation by PcG complexes has subsequently led to the identification of the lncRNA Hotair, which is transcribed from the HoxD locus and directly binds PRC2 to regulate the HoxC locus in trans (Rinn et al. 2007; Chu et al. 2011; Li et al. 2013). These findings have stimulated additional work investigating the entire population of RNAs bound by PRC2. RNA immunoprecipitation followed by microarray analysis (RIP-chip) or sequencing (RIP-seq) of PRC2 components including Ezh2 suggest that as many as 20 % of the known lncRNAs bind to PRC2 (Khalil et al. 2009; Zhao et al. 2010). These observations, combined with the findings that lncRNAs exhibit highly tissue- and cell-type-specific expression patterns, have led researchers to propose an attractive model, whereby the changing expression of cell-type-specific lncRNAs as development progresses alters the recruitment of PRC2 and other chromatin modifiers to their targets (Lee 2012; Nakagawa and Kageyama 2014).
The role of lncRNAs as general recruiters of PRC2 remains controversial, primarily because of the limited availability of experimental evidence directly supporting the model. The overwhelming number of putative RNA-PRC2 interactions predicted by RIP has led some to question the specificity of the assay. Indeed, native RIP (RIP performed without crosslinking, as performed in Khalil et al. 2009; Zhao et al. 2010) suffers from a notoriously high background signal (Mili and Steitz 2004; Brockdorff 2013). Although these caveats and limitations must be kept in mind, the results of high-stringency RIP of PRC2 from cells fixed by a variety of strategies (including formaldehyde fixation and UV crosslinking) recapitulate the large number of PRC2-interacting RNAs found by prior studies (Guttman et al. 2011; Davidovich et al. 2013). Many of the identified RNAs are therefore likely to be bona fide interactors with Polycomb, regardless of the functional relevance of this interaction.
Nevertheless, the overwhelming number of RNAs that bind PRC2 poses a significant challenge to experimentalists. No single RNA motif has been identified that mediates PRC2 binding. Although, for example, Xist and Hotair have specific sequence tracts that mediate binding (Cui et al. 2009; Engreitz et al. 2013; L. Wu et al. 2013), these tracts are not particularly similar at the level of the primary sequence. In the absence of a defined RNA motif, how does PRC2 recognize such a vast repertoire of RNAs, and how is the necessary specificity for a subset of functionally relevant RNAs achieved? Recent work from two different groups illustrates that PRC2 might have a generic RNA affinity that is important for its function (Davidovich et al. 2013; Kaneko et al. 2013). In one study, mammalian PRC2 was shown to bind RNA promiscuously and with submicromolar affinity, including exogenous RNA from ciliates and bacteria (Davidovich et al. 2013). Whereas sequence appears to be largely irrelevant, size matters: longer RNAs are bound with higher affinity than shorter RNAs in a salt-independent-manner, illustrating that the interaction with PRC2 is not merely electrostatic and might involve a more complex mechanism, such as base stacking with aromatic amino acid side chains (Davidovich et al. 2013).
These data demonstrate the relatively high affinity of PRC2 for RNA and suggest its biological significance, but they also challenge the simplistic model of lncRNAs as a specialized class of molecular recruiter for PRC2. ChIP-seq has verified that most promoters with Ezh2 are enriched for H3K27me3 and correspond to repressed genes, whereas RIP-seq indicates that Ezh2 tends to bind nascent RNAs from actively transcribed genes at which H3K27me3 is absent (Davidovich et al. 2013; Kaneko et al. 2013). These observations suggest that PRC2 globally surveys transcription by interacting with nascent RNAs, including protein coding and noncoding transcripts, whereas actual repression only occurs if other factors (e.g., the local chromatin context) are conducive to PRC2 transference from RNA to chromatin.
On top of this basic level of regulation by transcriptional surveillance, some lncRNAs (e.g., Xist and Hotair) have clearly evolved to achieve higher affinity and specificity for PRC2. Both Xist and Hotair have specific domains that are required for their interaction with PRC2, suggesting that their structures are optimized preferentially to recruit PRC2 with higher affinity and thereby to modify the distribution of PRC2 across the genome in biologically significant ways (Cui et al. 2009; Engreitz et al. 2013; L. Wu et al. 2013). Given the diversity of processes regulated by these two transcripts alone and the sheer abundance of lncRNAs that bind PRC2, other RNAs have probably evolved a similar, uniquely potent, PRC2 affinity that might modulate its activity in other contexts. Such a mechanism might be particularly important in the nervous system in which lncRNA transcription is exceptionally abundant and complex, and in which chromatin-interacting lncRNAs that are essential for neurogenesis have previously been identified in vitro and in vivo (Dinger et al. 2008; Derrien et al. 2012; Ng et al. 2013a, b; Ramos et al. 2013; Sauvageau et al. 2013).
Identification of the subset of “high affinity” PRC2-recruiting lncRNAs, should they exist, is an ongoing challenge. To truly demonstrate a direct role for a given RNA in the recruitment of a chromatin complex such as PRC2, the simple illustration that a given RNA binds to PRC2, or that its depletion affects the deposition of H3K27me3, is insufficient. For example, some lncRNAs have been shown to antagonize DNA methylation by inhibiting the activity of DNMT1, and lncRNA binding has also been suggested to modulate the function of other chromatin complexes, underscoring the myriad ways that the expression of a given lncRNA might be linked to PRC2 recruitment through any of several alternative models (Wang et al. 2011; Di Ruscio et al. 2013). Distinguishing between the direct recruitment of PRC2 and the other complex ways in which lncRNAs can regulate chromatin structure will necessitate experimental evidence that a putative RNA recruitment factor is located in physical proximity to the supposed sites of recruitment, as has been demonstrated for the lncRNAs Xist and Hotair (Chu et al. 2011; Engreitz et al. 2013). Many groups have reported technical advancements that will facilitate future research, including a variety of techniques involving the use of tiled biotinylated oligonucleotide probes to immunoprecipitate cross-linked lncRNA-chromatin complexes, a method that can be combined with high-throughput DNA sequencing to characterize the genome-wide localization of a lncRNA (Simon et al. 2011; Chu et al. 2012; Engreitz et al. 2013). Annotation of the cistrome of PRC2-interacting lncRNAs in neural and glial progenitors and their derivatives will provide invaluable information for modeling the way and the extent to which lncRNAs contribute to PcG complex function in neural development.
Concluding remarks
The multiplicity of Polycomb activities in the developing nervous system is readily apparent, even from the relatively limited data available. To date, these activities include the maintenance of the balance between NPC self-renewal and the onset of neurogenesis in the cortex, the promotion of the transition from neurogenesis to gliogenesis, the direction of the astrocyte versus the oligodendrocyte precursor fate switch, and even the regulation of transcriptional events in terminally differentiated neurons. Although more work is required to tease apart the exact functions of individual PcG proteins at the various stages of neural development, the dynamic activity of Polycomb complexes clearly contributes to the regulation of neural progenitor fate specification at nearly every step. The precise manner in which Polycomb complexes are recruited to regulate unique targets across the spectrum of neural fates is among the most important, unanswered questions in the field.
In recent years, a number of potential mechanisms have emerged that can influence the dynamic recruitment of Polycomb to chromatin. These mechanisms include facilitators of Polycomb recruitment, such as unmethylated CGI sequences and the proteins that maintain their unmethylated status, protein recruitment factors that can sense the local chromatin environment to influence the recruitment of PcG complexes in a context-dependent manner, and lncRNAs. Additionally, the targeting of Polycomb occurs in a milieu of multiple chromatin modifications that can directly or indirectly modulate the recruitment process, consistent with the increasingly appreciated multidimensional complexity of chromatin structure. However, much of what is known about Polycomb recruitment is derived from in vitro studies of ESCs, an extremely useful model that nevertheless cannot fully recapitulate the complexity of Polycomb activity during the generation of complex tissues such as the nervous system.
Recent work, such as the finding that Chd4 promotes neurogenesis by specifically recruiting PRC2 to silence gliogenic target genes, underscores the fact that unique PcG recruitment mechanisms are used to specify distinct neural cell fates. Continued research to identify these unique methods of PcG recruitment is a necessary precursor to the design of experimental and therapeutic approaches that can target specific aspects of PcG recruitment, rather than globally affecting PcG activity. For instance, the outcome of cortical progenitor differentiation is not dictated solely by the absolute and relative concentrations of instructive extracellular signals, but also by an intrinsic mechanism that selectively recruits Polycomb to specific targets in order to modify the response of the cells to such signals. Targeted manipulation of PcG recruitment should therefore enable more precise control of the timing and efficiency of neural progenitor differentiation into therapeutically relevant cell types than would be achievable by the manipulation of extracellular signaling pathways alone.
As an example, current protocols for the production of OPCs from human neural progenitors can take months, with relatively low efficiency yields. Manipulating PcG recruitment to accelerate the developmental “clock” could potentially permit the more rapid production of homogeneous populations of OPCs for use in applications such as in vitro disease modeling or the development of cell transplantation therapies. Therefore, the expansion of our knowledge of the way that PcG proteins are regulated during neural fate acquisition will, in all probability, simultaneously expand the molecular toolkit available for this research and its application to regenerative medicine.
References
Alder O, Lavial F, Helness A, Brookes E, Pinho S, Chandrashekran A, Arnaud P, Pombo A, O'Neill L, Azuara V (2010) Ring1B and Suv39h1 delineate distinct chromatin states at bivalent genes during early mouse lineage commitment. Development 137:2483–2492
Aloia L, Di Stefano B, Sessa A, Morey L, Santanach A, Gutierrez A, Cozzuto L, Benitah SA, Graf T, Broccoli V, Di Croce L (2014) Zrf1 is required to establish and maintain neural progenitor identity. Genes Dev 28:182–197
Aprea J, Prenninger S, Dori M, Ghosh T, Monasor LS, Wessendorf E, Zocher S, Massalini S, Alexopoulou D, Lesche M, Dahl A, Groszer M, Hiller M, Calegari F (2013) Transcriptome sequencing during mouse brain development identifies long non-coding RNAs functionally involved in neurogenic commitment. EMBO J 32:3145–3160
Armstrong L (2012) Epigenetic control of embryonic stem cell differentiation. Stem Cell Rev 8:67–77
Arney KL, Fisher AG (2004) Epigenetic aspects of differentiation. J Cell Sci 117:4355–4363
Azuara V, Perry P, Sauer S, Spivakov M, Jorgensen HF, John RM, Gouti M, Casanova M, Warnes G, Merkenschlager M, Fisher AG (2006) Chromatin signatures of pluripotent cell lines. Nat Cell Biol 8:532–538
Bernstein BE, Mikkelsen TS, Xie X, Kamal M, Huebert DJ, Cuff J, Fry B, Meissner A, Wernig M, Plath K, Jaenisch R, Wagschal A, Feil R, Schreiber SL, Lander ES (2006) A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125:315–326
Bernstein BE, Meissner A, Lander ES (2007) The mammalian epigenome. Cell 128:669–681
Blackledge NP, Farcas AM, Kondo T, King HW, McGouran JF, Hanssen LL, Ito S, Cooper S, Kondo K, Koseki Y, Ishikura T, Long HK, Sheahan TW, Brockdorff N, Kessler BM, Koseki H, Klose RJ (2014) Variant PRC1 complex-dependent H2A ubiquitylation drives PRC2 recruitment and polycomb domain formation. Cell 157:1445–1459
Bonni A, Sun Y, Nadal-Vicens M, Bhatt A, Frank DA, Rozovsky I, Stahl N, Yancopoulos GD, Greenberg ME (1997) Regulation of gliogenesis in the central nervous system by the JAK-STAT signaling pathway. Science 278:477–483
Brinkman AB, Gu H, Bartels SJ, Zhang Y, Matarese F, Simmer F, Marks H, Bock C, Gnirke A, Meissner A, Stunnenberg HG (2012) Sequential ChIP-bisulfite sequencing enables direct genome-scale investigation of chromatin and DNA methylation cross-talk. Genome Res 22:1128–1138
Brockdorff N (2013) Noncoding RNA and Polycomb recruitment. RNA 19:429–442
Brookes E, de Santiago I, Hebenstreit D, Morris KJ, Carroll T, Xie SQ, Stock JK, Heidemann M, Eick D, Nozaki N, Kimura H, Ragoussis J, Teichmann SA, Pombo A (2012) Polycomb associates genome-wide with a specific RNA polymerase II variant, and regulates metabolic genes in ESCs. Cell Stem Cell 10:157–170
Bruggeman SWM, Valk-Lingbeek ME, van der Stoop PPM, Jacobs JJL, Kieboom K, Tanger E, Hulsman D, Leung C, Arsenijevic Y, Marino S, van Lohuizen M, (2005) Ink4a and Arf differentially affect cell proliferation and neural stem cell self-renewal in Bmi1-deficient mice. Genes Dev 19:1438–1443
Burgold T, Spreafico F, De Santa F, Totaro MG, Prosperini E, Natoli G, Testa G (2008) The histone H3 lysine 27-specific demethylase Jmjd3 is required for neural commitment. PLoS One 3:e3034
Burgold T, Voituron N, Caganova M, Tripathi PP, Menuet C, Tusi BK, Spreafico F, Bevengut M, Gestreau C, Buontempo S, Simeone A, Kruidenier L, Natoli G, Casola S, Hilaire G, Testa G (2012) The H3K27 demethylase JMJD3 is required for maintenance of the embryonic respiratory neuronal network, neonatal breathing, and survival. Cell Rep 2:1244–1258
Campos EI, Reinberg D (2009) Histones: annotating chromatin. Annu Rev Genet 43:559–599
Cao R, Zhang Y (2004) SUZ12 is required for both the histone methyltransferase activity and the silencing function of the EED-EZH2 complex. Mol Cell 15:57–67
Cao R, Wang L, Wang H, Xia L, Erdjument-Bromage H, Tempst P, Jones RS, Zhang Y (2002) Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science 298:1039–1043
Cao R, Tsukada Y-I, Zhang Y (2005) Role of Bmi-1 and Ring1A in H2A ubiquitylation and Hox gene silencing. Mol Cell 20:845–854
Chan THM, Chen L, Liu M, Hu L, Zheng B-J, Poon VK-M, Huang P, Yuan Y-F, Huang J-D, Yang J, Tsao GS-W, Guan X-Y (2012) Translationally controlled tumor protein induces mitotic defects and chromosome missegregation in hepatocellular carcinoma development. Hepatology 55:491–505
Chu C, Qu K, Zhong FL, Artandi SE, Chang HY (2011) Genomic maps of long noncoding RNA occupancy reveal principles of RNA-chromatin interactions. Mol Cell 44:667–678
Chu C, Quinn J, Chang HY (2012) Chromatin isolation by RNA purification (ChIRP). J Vis Exp 61:e3912
Ciferri C, Lander GC, Maiolica A, Herzog F, Aebersold R, Nogales E (2012) Molecular architecture of human polycomb repressive complex 2. Elife 1:e00005
Cooper S, Dienstbier M, Hassan R, Schermelleh L, Sharif J, Blackledge NP, De Marco V, Elderkin S, Koseki H, Klose R, Heger A, Brockdorff N (2014) Targeting polycomb to pericentric heterochromatin in embryonic stem cells reveals a role for H2AK119u1 in PRC2 recruitment. Cell Rep 7:1456–1470
Costa MR, Bucholz O, Schroeder T, Gotz M (2009) Late origin of glia-restricted progenitors in the developing mouse cerebral cortex. Cereb Cortex 19 (Suppl 1):i135–i143
Cui K, Zang C, Roh TY, Schones DE, Childs RW, Peng W, Zhao K (2009) Chromatin signatures in multipotent human hematopoietic stem cells indicate the fate of bivalent genes during differentiation. Cell Stem Cell 4:80–93
Czermin B, Melfi R, McCabe D, Seitz V, Imhof A, Pirrotta V (2002) Drosophila enhancer of Zeste/ESC complexes have a histone H3 methyltransferase activity that marks chromosomal Polycomb sites. Cell 111:185–196
Davidovich C, Zheng L, Goodrich KJ, Cech TR (2013) Promiscuous RNA binding by Polycomb repressive complex 2. Nat Struct Mol Biol 20:1250–1257
Davis-Dusenbery BN, Williams LA, Klim JR, Eggan K (2014) How to make spinal motor neurons. Development 141:491–501
de Napoles M, Mermoud JE, Wakao R, Tang YA, Endoh M, Appanah R, Nesterova TB, Silva J, Otte AP, Vidal M, Koseki H, Brockdorff N (2004) Polycomb group proteins Ring1A/B link ubiquitylation of histone H2A to heritable gene silencing and X inactivation. Dev Cell 7:663–676
Derouet D, Rousseau F, Alfonsi F, Froger J, Hermann J, Barbier F, Perret D, Diveu C, Guillet C, Preisser L, Dumont A, Barbado M, Morel A, deLapeyriere O, Gascan H, Chevalier S (2004) Neuropoietin, a new IL-6-related cytokine signaling through the ciliary neurotrophic factor receptor. Proc Natl Acad Sci U S A 101:4827–4832
Derrien T, Johnson R, Bussotti G, Tanzer A, Djebali S, Tilgner H, Guernec G, Martin D, Merkel A, Knowles DG, Lagarde J, Veeravalli L, Ruan X, Ruan Y, Lassmann T, Carninci P, Brown JB, Lipovich L, Gonzalez JM, Thomas M, Davis CA, Shiekhattar R, Gingeras TR, Hubbard TJ, Notredame C, Harrow J, Guigó R (2012) The GENCODE v7 catalog of human long noncoding RNAs: analysis of their gene structure, evolution, and expression. Genome Res 22:1775–1789
Di Meglio T, Kratochwil CF, Vilain N, Loche A, Vitobello A, Yonehara K, Hrycaj SM, Roska B, Peters AH, Eichmann A, Wellik D, Ducret S, Rijli FM (2013) Ezh2 orchestrates topographic migration and connectivity of mouse precerebellar neurons. Science 339:204–207
Di Ruscio A, Ebralidze AK, Benoukraf T, Amabile G, Goff LA, Terragni J, Figueroa ME, De Figueiredo Pontes LL, Alberich-Jorda M, Zhang P, Wu M, D'Alo F, Melnick A, Leone G, Ebralidze KK, Pradhan S, Rinn JL, Tenen DG (2013) DNMT1-interacting RNAs block gene-specific DNA methylation. Nature 503:371–376
Dinger ME, Amaral PP, Mercer TR, Pang KC, Bruce SJ, Gardiner BB, Askarian-Amiri ME, Ru K, Soldà G, Simons C, Sunkin SM, Crowe ML, Grimmond SM, Perkins AC, Mattick JS (2008) Long noncoding RNAs in mouse embryonic stem cell pluripotency and differentiation. Genome Res 18:1433–1445
Egan CM, Nyman U, Skotte J, Streubel G, Turner S, O'Connell DJ, Rraklli V, Dolan MJ, Chadderton N, Hansen K, Farrar GJ, Helin K, Holmberg J, Bracken AP (2013) CHD5 is required for neurogenesis and has a dual role in facilitating gene expression and polycomb gene repression. Dev Cell 26:223–236
Eiraku M, Watanabe K, Matsuo-Takasaki M, Kawada M, Yonemura S, Matsumura M, Wataya T, Nishiyama A, Muguruma K, Sasai Y (2008) Self-organized formation of polarized cortical tissues from ESCs and its active manipulation by extrinsic signals. Cell Stem Cell 3:519–532
Endoh M, Endo TA, Endoh T, Isono K, Sharif J, Ohara O, Toyoda T, Ito T, Eskeland R, Bickmore WA, Vidal M, Bernstein BE, Koseki H (2012) Histone H2A mono-ubiquitination is a crucial step to mediate PRC1-dependent repression of developmental genes to maintain ES cell identity. PLoS Genet 8:e1002774
Engreitz JM, Pandya-Jones A, McDonel P, Shishkin A, Sirokman K, Surka C, Kadri S, Xing J, Goren A, Lander ES, Plath K, Guttman M (2013) The Xist lncRNA exploits three-dimensional genome architecture to spread across the X chromosome. Science 341:1237973
Eskeland R, Leeb M, Grimes GR, Kress C, Boyle S, Sproul D, Gilbert N, Fan Y, Skoultchi AI, Wutz A, Bickmore WA (2010) Ring1B compacts chromatin structure and represses gene expression independent of histone ubiquitination. Mol Cell 38:452–464
Farcas AM, Blackledge NP, Sudbery I, Long HK, McGouran JF, Rose NR, Lee S, Sims D, Cerase A, Sheahan TW, Koseki H, Brockdorff N, Ponting CP, Kessler BM, Klose RJ (2012) KDM2B links the Polycomb repressive complex 1 (PRC1) to recognition of CpG islands. Elife 1:e00205
Fasano CA, Dimos JT, Ivanova NB, Lowry N, Lemischka IR, Temple S (2007) shRNA knockdown of Bmi-1 reveals a critical role for p21-Rb pathway in NSC self-renewal during development. Cell Stem Cell 1:87–99
Fasano CA, Phoenix TN, Kokovay E, Lowry N, Elkabetz Y, Dimos JT, Lemischka IR, Studer L, Temple S (2009) Bmi-1 cooperates with Foxg1 to maintain neural stem cell self-renewal in the forebrain. Genes Dev 23:561–574
Fatica A, Bozzoni I (2014) Long non-coding RNAs: new players in cell differentiation and development. Nat Rev Genet 15:7–21
Francastel C, Schubeler D, Martin DI, Groudine M (2000) Nuclear compartmentalization and gene activity. Nat Rev Mol Cell Biol 1:137–143
Francis NJ, Kingston RE, Woodcock CL (2004) Chromatin compaction by a polycomb group protein complex. Science 306:1574–1577
Froberg JE, Yang L, Lee JT (2013) Guided by RNAs: X-inactivation as a model for lncRNA function. J Mol Biol 425:3698–3706
Fukuda T, Tokunaga A, Sakamoto R, Yoshida N (2011) Fbxl10/Kdm2b deficiency accelerates neural progenitor cell death and leads to exencephaly. Mol Cell Neurosci 46:614–624
Gao Z, Zhang J, Bonasio R, Strino F, Sawai A, Parisi F, Kluger Y, Reinberg D (2012) PCGF homologs, CBX proteins, and RYBP define functionally distinct PRC1 family complexes. Mol Cell 45:344–356
García E, Marcos-Gutiérrez C, del Mar Lorente M, Moreno JC, Vidal M (1999) RYBP, a new repressor protein that interacts with components of the mammalian Polycomb complex, and with the transcription factor YY1. EMBO J 18:3404–3418
Gaspard N, Bouschet T, Hourez R, Dimidschstein J, Naeije G, van den Ameele J, Espuny-Camacho I, Herpoel A, Passante L, Schiffmann SN, Gaillard A, Vanderhaeghen P (2008) An intrinsic mechanism of corticogenesis from embryonic stem cells. Nature 455:351–357
Golden MG, Dasen JS (2012) Polycomb repressive complex 1 activities determine the columnar organization of motor neurons. Genes Dev 26:2236–2250
Greig LC, Woodworth MB, Galazo MJ, Padmanabhan H, Macklis JD (2013) Molecular logic of neocortical projection neuron specification, development and diversity. Nat Rev Neurosci 14:755–769
Guttman M, Donaghey J, Carey BW, Garber M, Grenier JK, Munson G, Young G, Lucas AB, Ach R, Bruhn L, Yang X, Amit I, Meissner A, Regev A, Rinn JL, Root DE, Lander ES (2011) lincRNAs act in the circuitry controlling pluripotency and differentiation. Nature 477:295–300
Hagarman JA, Motley MP, Kristjansdottir K, Soloway PD (2013) Coordinate regulation of DNA methylation and H3K27me3 in mouse embryonic stem cells. PLoS One 8:e53880
He F, Ge W, Martinowich K, Becker-Catania S, Coskun V, Zhu W, Wu H, Castro D, Guillemot F, Fan G, de Vellis J, Sun YE (2005) A positive autoregulatory loop of Jak-STAT signaling controls the onset of astrogliogenesis. Nat Neurosci 8:616–625
He J, Kallin EM, Tsukada YI, Zhang Y (2008) The H3K36 demethylase Jhdm1b/Kdm2b regulates cell proliferation and senescence through p15(Ink4b). Nat Struct Mol Biol 15:1169–1175
He J, Shen L, Wan M, Taranova O, Wu H, Zhang Y (2013) Kdm2b maintains murine embryonic stem cell status by recruiting PRC1 complex to CpG islands of developmental genes. Nat Cell Biol 15:373–384
Hirabayashi Y, Gotoh Y (2005) Stage-dependent fate determination of neural precursor cells in mouse forebrain. Neurosci Res 51:331–336
Hirabayashi Y, Itoh Y, Tabata H, Nakajima K, Akiyama T, Masuyama N, Gotoh Y (2004) The Wnt/beta-catenin pathway directs neuronal differentiation of cortical neural precursor cells. Development 131:2791–2801
Hirabayashi Y, Suzki N, Tsuboi M, Endo TA, Toyoda T, Shinga J, Koseki H, Vidal M, Gotoh Y (2009) Polycomb limits the neurogenic competence of neural precursor cells to promote astrogenic fate transition. Neuron 63:600–613
Hisada K, Sanchez C, Endo TA, Endoh M, Roman-Trufero M, Sharif J, Koseki H, Vidal M (2012) RYBP represses endogenous retroviruses and preimplantation- and germ line-specific genes in mouse embryonic stem cells. Mol Cell Biol 32:1139–1149
Hsieh J, Gage FH (2005) Chromatin remodeling in neural development and plasticity. Curr Opin Cell Biol 17:664–671
Hwang WW, Salinas RD, Siu JJ, Kelley KW, Delgado RN, Paredes MF, Alvarez-Buylla A, Oldham MC, Lim DA (2014) Distinct and separable roles for EZH2 in neurogenic astroglia. Elife 3:e02439
Israsena N, Hu M, Fu W, Kan L, Kessler JA (2004) The presence of FGF2 signaling determines whether beta-catenin exerts effects on proliferation or neuronal differentiation of neural stem cells. Dev Biol 268:220–231
Johe KK, Hazel TG, Muller T, Dugich-Djordjevic MM, McKay RD (1996) Single factors direct the differentiation of stem cells from the fetal and adult central nervous system. Genes Dev 10:3129–3140
Johnson R (2012) Long non-coding RNAs in Huntington's disease neurodegeneration. Neurobiol Dis 46:245–254
Junco SE, Wang R, Gaipa JC, Taylor AB, Schirf V, Gearhart MD, Bardwell VJ, Demeler B, Hart PJ, Kim CA (2013) Structure of the polycomb group protein PCGF1 in complex with BCOR reveals basis for binding selectivity of PCGF homologs. Structure 21:665–671
Kaneko S, Bonasio R, Saldaña-Meyer R, Yoshida T, Son J, Nishino K, Umezawa A, Reinberg D (2013) Interactions between JARID2 and noncoding RNAs regulate PRC2 recruitment to chromatin. Mol Cell 53:290–300
Khalil AM, Guttman M, Huarte M, Garber M, Raj A, Rivea Morales D, Thomas K, Presser A, Bernstein BE, van Oudenaarden A, Regev A, Lander ES, Rinn JL (2009) Many human large intergenic noncoding RNAs associate with chromatin-modifying complexes and affect gene expression. Proc Natl Acad Sci U S A 106:11667–11672
Kim H, Kang K, Kim J (2009) AEBP2 as a potential targeting protein for Polycomb repression complex PRC2. Nucleic Acids Res 37:2940–2950
Kim H, Kang K, Ekram MB, Roh T-Y, Kim J (2011) Aebp2 as an epigenetic regulator for neural crest cells. PLoS One 6:e25174
Kirmizis A, Bartley SM, Kuzmichev A, Margueron R, Reinberg D, Green R, Farnham PJ (2004) Silencing of human polycomb target genes is associated with methylation of histone H3 Lys 27. Genes Dev 18:1592–1605
Koyama H, Zhuang T, Light JE, Kolla V, Higashi M, McGrady PW, London WB, Brodeur GM (2012) Mechanisms of CHD5 inactivation in neuroblastomas. Clin Cancer Res 18:1588–1597
Koyama-Nasu R, David G, Tanese N (2007) The F-box protein Fbl10 is a novel transcriptional repressor of c-Jun. Nat Cell Biol 9:1074–1080
Kriegstein A, Alvarez-Buylla A (2009) The glial nature of embryonic and adult neural stem cells. Annu Rev Neurosci 32:149–184
Ku M, Koche RP, Rheinbay E, Mendenhall EM, Endoh M, Mikkelsen TS, Presser A, Nusbaum C, Xie X, Chi AS, Adli M, Kasif S, Ptaszek LM, Cowan CA, Lander ES, Koseki H, Bernstein BE (2008) Genomewide analysis of PRC1 and PRC2 occupancy identifies two classes of bivalent domains. PLoS Genet 4:e1000242
Kuzmichev A, Nishioka K, Erdjument-Bromage H, Tempst P, Reinberg D (2002) Histone methyltransferase activity associated with a human multiprotein complex containing the Enhancer of Zeste protein. Genes Dev 16:2893–2905
Landeira D, Sauer S, Poot R, Dvorkina M, Mazzarella L, Jørgensen HF, Pereira CF, Leleu M, Piccolo FM, Spivakov M, Brookes E, Pombo A, Fisher C, Skarnes WC, Snoek T, Bezstarosti K, Demmers J, Klose RJ, Casanova M, Tavares L, Brockdorff N, Merkenschlager M, Fisher AG (2010) Jarid2 is a PRC2 component in embryonic stem cells required for multi-lineage differentiation and recruitment of PRC1 and RNA Polymerase II to developmental regulators. Nat Cell Biol 12:618–624
Lee JT (2012) Epigenetic regulation by long noncoding RNAs. Science 338:1435–1439
Levine SS, Weiss A, Erdjument-Bromage H, Shao Z, Tempst P, Kingston RE (2002) The core of the polycomb repressive complex is compositionally and functionally conserved in flies and humans. Mol Cell Biol 22:6070–6078
Li G, Margueron R, Ku M, Chambon P, Bernstein BE, Reinberg D (2010) Jarid2 and PRC2, partners in regulating gene expression. Genes Dev 24:368–380
Li L, Liu B, Wapinski OL, Tsai M-C, Qu K, Zhang J, Carlson JC, Lin M, Fang F, Gupta RA, Helms JA, Chang HY (2013) Targeted disruption of Hotair leads to homeotic transformation and gene derepression. Cell Rep 5:3–12
Lim DA, Huang YC, Swigut T, Mirick AL, Garcia-Verdugo JM, Wysocka J, Ernst P, Alvarez-Buylla A (2009) Chromatin remodelling factor Mll1 is essential for neurogenesis from postnatal neural stem cells. Nature 458:529–533
Lynch MD, Smith AJ, De Gobbi M, Flenley M, Hughes JR, Vernimmen D, Ayyub H, Sharpe JA, Sloane-Stanley JA, Sutherland L, Meek S, Burdon T, Gibbons RJ, Garrick D, Higgs DR (2012) An interspecies analysis reveals a key role for unmethylated CpG dinucleotides in vertebrate Polycomb complex recruitment. EMBO J 31:317–329
Margueron R, Reinberg D (2011) The Polycomb complex PRC2 and its mark in life. Nature 469:343–349
Margueron R, Li G, Sarma K, Blais A, Zavadil J, Woodcock CL, Dynlacht BD, Reinberg D (2008) Ezh1 and Ezh2 maintain repressive chromatin through different mechanisms. Mol Cell 32:503–518
Margueron R, Justin N, Ohno K, Sharpe ML, Son J, Drury WJ 3rd, Voigt P, Martin SR, Taylor WR, De Marco V, Pirrotta V, Reinberg D, Gamblin SJ (2009) Role of the polycomb protein EED in the propagation of repressive histone marks. Nature 461:762–767
Martynoga B, Drechsel D, Guillemot F (2012) Molecular control of neurogenesis: a view from the mammalian cerebral cortex. Cold Spring Harb Perspect Biol 4:a008359
Mazzoni EO, Mahony S, Peljto M, Patel T, Thornton SR, McCuine S, Reeder C, Boyer LA, Young RA, Gifford DK, Wichterle H (2013) Saltatory remodeling of Hox chromatin in response to rostrocaudal patterning signals. Nat Neurosci 16:1191–1198
Mercer TR, Dinger ME, Sunkin SM, Mehler MF, Mattick JS (2008) Specific expression of long noncoding RNAs in the mouse brain. Proc Natl Acad Sci U S A 105:716–721
Mercer TR, Qureshi IA, Gokhan S, Dinger ME, Li G, Mattick JS, Mehler MF (2010) Long noncoding RNAs in neuronal-glial fate specification and oligodendrocyte lineage maturation. BMC Neurosci 11:14
Meshorer E (2007) Chromatin in embryonic stem cell neuronal differentiation. Histol Histopathol 22:311–319
Meshorer E, Yellajoshula D, George E, Scambler PJ, Brown DT, Misteli T (2006) Hyperdynamic plasticity of chromatin proteins in pluripotent embryonic stem cells. Dev Cell 10:105–116
Mikkelsen TS, Ku M, Jaffe DB, Issac B, Lieberman E, Giannoukos G, Alvarez P, Brockman W, Kim T-K, Koche RP, Lee W, Mendenhall E, O'Donovan A, Presser A, Russ C, Xie X, Meissner A, Wernig M, Jaenisch R, Nusbaum C, Lander ES, Bernstein BE (2007) Genome-wide maps of chromatin state in pluripotent and lineage-committed cells. Nature 448:553–560
Mili S, Steitz JA (2004) Evidence for reassociation of RNA-binding proteins after cell lysis: implications for the interpretation of immunoprecipitation analyses. RNA 10:1692–1694
Mohn F, Weber M, Rebhan M, Roloff TC, Richter J, Stadler MB, Bibel M, Schübeler D (2008) Lineage-specific polycomb targets and de novo DNA methylation define restriction and potential of neuronal progenitors. Mol Cell 30:755–766
Molofsky AV, Pardal R, Iwashita T, Park I-K, Clarke MF, Morrison SJ (2003) Bmi-1 dependence distinguishes neural stem cell self-renewal from progenitor proliferation. Nature 425:962–967
Molofsky AV, He S, Bydon M, Morrison SJ, Pardal R (2005) Bmi-1 promotes neural stem cell self-renewal and neural development but not mouse growth and survival by repressing the p16Ink4a and p19Arf senescence pathways. Genes Dev 19:1432–1437
Montgomery ND, Yee D, Chen A, Kalantry S, Chamberlain SJ, Otte AP, Magnuson T (2005) The murine polycomb group protein Eed is required for global histone H3 lysine-27 methylation. Curr Biol 15:942–947
Morey L, Pascual G, Cozzuto L, Roma G, Wutz A, Benitah SA, Di Croce L (2012) Nonoverlapping functions of the Polycomb group Cbx family of proteins in embryonic stem cells. Cell Stem Cell 10:47–62
MuhChyi C, Juliandi B, Matsuda T, Nakashima K (2013) Epigenetic regulation of neural stem cell fate during corticogenesis. Int J Dev Neurosci 31:424–433
Nakagawa S, Kageyama Y (2014) Nuclear lncRNAs as epigenetic regulators-Beyond skepticism. Biochim Biophys Acta 1839:215–222
Nakashima K, Yanagisawa M, Arakawa H, Kimura N, Hisatsune T, Kawabata M, Miyazono K, Taga T (1999) Synergistic signaling in fetal brain by STAT3-Smad1 complex bridged by p300. Science 284:479–482
Narayanan R, Tuoc TC (2014) Roles of chromatin remodeling BAF complex in neural differentiation and reprogramming. Cell Tissue Res 356:575–584
Ng S-Y, Johnson R, Stanton LW (2012) Human long non-coding RNAs promote pluripotency and neuronal differentiation by association with chromatin modifiers and transcription factors. EMBO J 31:522–533
Ng S-Y, Bogu GK, Soh B-S, Stanton LW (2013a) The long noncoding RNA RMST interacts with SOX2 to regulate neurogenesis. Mol Cell 51:349–359
Ng S-Y, Lin L, Soh B-S, Stanton LW (2013b) Long noncoding RNAs in development and disease of the central nervous system. Trends Genet 29:461–468
Noctor SC, Martinez-Cerdeno V, Kriegstein AR (2008) Distinct behaviors of neural stem and progenitor cells underlie cortical neurogenesis. J Comp Neurol 508:28–44
O'Loghlen A, Munoz-Cabello AM, Gaspar-Maia A, Wu HA, Banito A, Kunowska N, Racek T, Pemberton HN, Beolchi P, Lavial F, Masui O, Vermeulen M, Carroll T, Graumann J, Heard E, Dillon N, Azuara V, Snijders AP, Peters G, Bernstein E, Gil J (2012) MicroRNA regulation of Cbx7 mediates a switch of Polycomb orthologs during ESC differentiation. Cell Stem Cell 10:33–46
Olynik BM, Rastegar M (2012) The genetic and epigenetic journey of embryonic stem cells into mature neural cells. Front Genet 3:81
Paige SL, Thomas S, Stoick-Cooper CL, Wang H, Maves L, Sandstrom R, Pabon L, Reinecke H, Pratt G, Keller G, Moon RT, Stamatoyannopoulos J, Murry CE (2012) A temporal chromatin signature in human embryonic stem cells identifies regulators of cardiac development. Cell 151:221–232
Pan G, Tian S, Nie J, Yang C, Ruotti V, Wei H, Jonsdottir GA, Stewart R, Thomson JA (2007) Whole-genome analysis of histone H3 lysine 4 and lysine 27 methylation in human embryonic stem cells. Cell Stem Cell 1:299–312
Pasini D, Cloos PAC, Walfridsson J, Olsson L, Bukowski J-P, Johansen JV, Bak M, Tommerup N, Rappsilber J, Helin K (2010a) JARID2 regulates binding of the Polycomb repressive complex 2 to target genes in ES cells. Nature 464:306–310
Pasini D, Malatesta M, Jung HR, Walfridsson J, Willer A, Olsson L, Skotte J, Wutz A, Porse B, Jensen ON, Helin K (2010b) Characterization of an antagonistic switch between histone H3 lysine 27 methylation and acetylation in the transcriptional regulation of Polycomb group target genes. Nucleic Acids Res 38:4958–4969
Peng JC, Valouev A, Swigut T, Zhang J, Zhao Y, Sidow A, Wysocka J (2009) Jarid2/Jumonji coordinates control of PRC2 enzymatic activity and target gene occupancy in pluripotent cells. Cell 139:1290–1302
Pereira JD, Sansom SN, Smith J, Dobenecker M-W, Tarakhovsky A, Livesey FJ (2010) Ezh2, the histone methyltransferase of PRC2, regulates the balance between self-renewal and differentiation in the cerebral cortex. Proc Natl Acad Sci U S A 107:15957-15962
Petryniak MA, Potter GB, Rowitch DH, Rubenstein JL (2007) Dlx1 and Dlx2 control neuronal versus oligodendroglial cell fate acquisition in the developing forebrain. Neuron 55:417–433
Philippidou P, Dasen JS (2013) Hox genes: choreographers in neural development, architects of circuit organization. Neuron 80:12–34
Pirity MK, Locker J, Schreiber-Agus N (2005) Rybp/DEDAF is required for early postimplantation and for central nervous system development. Mol Cell Biol 25:7193–7202
Qi C, Liu S, Qin R, Zhang Y, Wang G, Shang Y, Wang Y, Liang J (2014) Coordinated regulation of dendrite arborization by epigenetic factors CDYL and EZH2. J Neurosci 34:4494–4508
Qian X, Shen Q, Goderie SK, He W, Capela A, Davis AA, Temple S (2000) Timing of CNS cell generation: a programmed sequence of neuron and glial cell production from isolated murine cortical stem cells. Neuron 28:69–80
Qureshi IA, Mehler MF (2012) Emerging roles of non-coding RNAs in brain evolution, development, plasticity and disease. Nat Rev Neurosci 13:528–541
Qureshi IA, Mehler MF (2013) Long non-coding RNAs: novel targets for nervous system disease diagnosis and therapy. Neurotherapeutics 10:632–646
Rajan P, McKay RD (1998) Multiple routes to astrocytic differentiation in the CNS. J Neurosci 18:3620–3629
Ramos AD, Diaz A, Nellore A, Delgado RN, Park K-Y, Gonzales-Roybal G, Oldham MC, Song JS, Lim DA (2013) Integration of genome-wide approaches identifies lncRNAs of adult neural stem cells and their progeny in vivo. Cell Stem Cell 12:616–628
Reynolds N, Salmon-Divon M, Dvinge H, Hynes-Allen A, Balasooriya G, Leaford D, Behrens A, Bertone P, Hendrich B (2012) NuRD-mediated deacetylation of H3K27 facilitates recruitment of Polycomb repressive complex 2 to direct gene repression. EMBO J 31:593–605
Rinn JL, Kertesz M, Wang JK, Squazzo SL, Xu X, Brugmann SA, Goodnough LH, Helms JA, Farnham PJ, Segal E, Chang HY (2007) Functional demarcation of active and silent chromatin domains in human HOX loci by noncoding RNAs. Cell 129:1311–1323
Román-Trufero M, Méndez-Gómez H, Pérez C, Hijikata A, Fujimura Y, Endo T, Koseki H, Vicario-Abejón C, Vidal M (2009) Maintenance of undifferentiated state and self-renewal of embryonic neural stem cells by polycomb protein Ring1B. Stem Cells 27:1559–1570
Ronan JL, Wu W, Crabtree GR (2013) From neural development to cognition: unexpected roles for chromatin. Nat Rev Genet 14:347–359
Sánchez C, Sánchez I, Demmers JAA, Rodriguez P, Strouboulis J, Vidal M (2007) Proteomics analysis of Ring1B/Rnf2 interactors identifies a novel complex with the Fbxl10/Jhdm1B histone demethylase and the Bcl6 interacting corepressor. Mol Cell Proteomics 6:820–834
Sauvageau M, Goff LA, Lodato S, Bonev B, Groff AF, Gerhardinger C, Sanchez-Gomez DB, Hacisuleyman E, Li E, Spence M, Liapis SC, Mallard W, Morse M, Swerdel MR, Dapos MF, Ecclessis, Moore JC, Lai V, Gong G, Yancopoulos GD, Frendewey D, Kellis M, Hart RP, Valenzuela DM, Arlotta P, Rinn JL (2013) Multiple knockout mouse models reveal lincRNAs are required for life and brain development. Elife 2:e01749
Schmitges FW, Prusty AB, Faty M, Stützer A, Lingaraju GM, Aiwazian J, Sack R, Hess D, Li L, Zhou S, Bunker RD, Wirth U, Bouwmeester T, Bauer A, Ly-Hartig N, Zhao K, Chan H, Gu J, Gut H, Fischle W, Müller J, Thomä NH (2011) Histone methylation by PRC2 is inhibited by active chromatin marks. Mol Cell 42:330–341
Schuettengruber B, Chourrout D, Vervoort M, Leblanc B, Cavalli G (2007) Genome regulation by polycomb and trithorax proteins. Cell 128:735–745
Schuettengruber B, Martinez AM, Iovino N, Cavalli G (2011) Trithorax group proteins: switching genes on and keeping them active. Nat Rev Mol Cell Biol 12:799–814
Shen Q, Wang Y, Dimos JT, Fasano CA, Phoenix TN, Lemischka IR, Ivanova NB, Stifani S, Morrisey EE, Temple S (2006) The timing of cortical neurogenesis is encoded within lineages of individual progenitor cells. Nat Neurosci 9:743–751
Shen X, Liu Y, Hsu YJ, Fujiwara Y, Kim J, Mao X, Yuan GC, Orkin SH (2008) EZH1 mediates methylation on histone H3 lysine 27 and complements EZH2 in maintaining stem cell identity and executing pluripotency. Mol Cell 32:491–502
Shen X, Kim W, Fujiwara Y, Simon MD, Liu Y, Mysliwiec MR, Yuan G-C, Lee Y, Orkin SH (2009) Jumonji modulates polycomb activity and self-renewal versus differentiation of stem cells. Cell 139:1303–1314
Sher F, Rössler R, Brouwer N, Balasubramaniyan V, Boddeke E, Copray S (2008) Differentiation of neural stem cells into oligodendrocytes: involvement of the polycomb group protein Ezh2. Stem Cells 26:2875–2883
Sher F, Boddeke E, Olah M, Copray S (2012) Dynamic changes in Ezh2 gene occupancy underlie its involvement in neural stem cell self-renewal and differentiation towards oligodendrocytes. PLoS One 7:e40399
Shimogori T, VanSant J, Paik E, Grove EA (2004) Members of the Wnt, Fz, and Frp gene families expressed in postnatal mouse cerebral cortex. J Comp Neurol 473:496–510
Simon JA, Kingston RE (2013) Occupying chromatin: Polycomb mechanisms for getting to genomic targets, stopping transcriptional traffic, and staying put. Mol Cell 49:808–824
Simon MD, Wang CI, Kharchenko PV, West JA, Chapman BA, Alekseyenko AA, Borowsky ML, Kuroda MI, Kingston RE (2011) The genomic binding sites of a noncoding RNA. Proc Natl Acad Sci U S A 108:20497–20502
Son J, Shen SS, Margueron R, Reinberg D (2013) Nucleosome-binding activities within JARID2 and EZH1 regulate the function of PRC2 on chromatin. Genes Dev 27:2663–2677
Statham AL, Robinson MD, Song JZ, Coolen MW, Stirzaker C, Clark SJ (2012) Bisulfite sequencing of chromatin immunoprecipitated DNA (BisChIP-seq) directly informs methylation status of histone-modified DNA. Genome Res 22:1120–1127
Stock JK, Giadrossi S, Casanova M, Brookes E, Vidal M, Koseki H, Brockdorff N, Fisher AG, Pombo A (2007) Ring1-mediated ubiquitination of H2A restrains poised RNA polymerase II at bivalent genes in mouse ES cells. Nat Cell Biol 9:1428–1435
Sun Y, Nadal-Vicens M, Misono S, Lin MZ, Zubiaga A, Hua X, Fan G, Greenberg ME (2001) Neurogenin promotes neurogenesis and inhibits glial differentiation by independent mechanisms. Cell 104:365–376
Taft RJ, Hawkins PG, Mattick JS, Morris KV (2011) The relationship between transcription initiation RNAs and CCCTC-binding factor (CTCF) localization. Epigenetics Chromatin 4:13
Takahashi M, Kojima M, Nakajima K, Suzuki-Migishima R, Takeuchi T (2007) Functions of a jumonji-cyclin D1 pathway in the coordination of cell cycle exit and migration during neurogenesis in the mouse hindbrain. Dev Biol 303:549–560
Takeuchi T, Yamazaki Y, Katoh-Fukui Y, Tsuchiya R, Kondo S, Motoyama J, Higashinakagawa T (1995) Gene trap capture of a novel mouse gene, jumonji, required for neural tube formation. Genes Dev 9:1211–1222
Takeuchi T, Kojima M, Nakajima K, Kondo S (1999) Jumonji gene is essential for the neurulation and cardiac development of mouse embryos with a C3H/He background. Mech Dev 86:29–38
Tavares L, Dimitrova E, Oxley D, Webster J, Poot R, Demmers J, Bezstarosti K, Taylor S, Ura H, Koide H, Wutz A, Vidal M, Elderkin S, Brockdorff N (2012) RYBP-PRC1 complexes mediate H2A ubiquitylation at polycomb target sites independently of PRC2 and H3K27me3. Cell 148:664–678
Taverna SD, Li H, Ruthenburg AJ, Allis CD, Patel DJ (2007) How chromatin-binding modules interpret histone modifications: lessons from professional pocket pickers. Nat Struct Mol Biol 14:1025–1040
Testa G (2011) The time of timing: how Polycomb proteins regulate neurogenesis. Bioessays 33:519–528
Tie F, Banerjee R, Stratton CA, Prasad-Sinha J, Stepanik V, Zlobin A, Diaz MO, Scacheri PC, Harte PJ (2009) CBP-mediated acetylation of histone H3 lysine 27 antagonizes Drosophila Polycomb silencing. Development 136:3131–3141
Turner SA, Bracken AP (2013) A "complex" issue: deciphering the role of variant PRC1 in ESCs. Cell Stem Cell 12:145–146
Tushir JS, Akbarian S (2013) Chromatin-bound RNA and the neurobiology of psychiatric disease. Neuroscience 264:131–141
Uemura A, Takizawa T, Ochiai W, Yanagisawa M, Nakashima K, Taga T (2002) Cardiotrophin-like cytokine induces astrocyte differentiation of fetal neuroepithelial cells via activation of STAT3. Cytokine 18:1–7
Voigt P, Tee WW, Reinberg D (2013) A double take on bivalent promoters. Genes Dev 27:1318–1338
Voncken JW, Roelen BA, Roefs M, de Vries S, Verhoeven E, Marino S, Deschamps J, van Lohuizen M (2003) Rnf2 (Ring1b) deficiency causes gastrulation arrest and cell cycle inhibition. Proc Natl Acad Sci U S A 100:2468–2473
Walsh C, Cepko CL (1992) Widespread dispersion of neuronal clones across functional regions of the cerebral cortex. Science 255:434–440
Wamstad JA, Alexander JM, Truty RM, Shrikumar A, Li F, Eilertson KE, Ding H, Wylie JN, Pico AR, Capra JA, Erwin G, Kattman SJ, Keller GM, Srivastava D, Levine SS, Pollard KS, Holloway AK, Boyer LA, Bruneau BG (2012) Dynamic and coordinated epigenetic regulation of developmental transitions in the cardiac lineage. Cell 151:206–220
Wang KC, Yang YW, Liu B, Sanyal A, Corces-Zimmerman R, Chen Y, Lajoie BR, Protacio A, Flynn RA, Gupta RA, Wysocka J, Lei M, Dekker J, Helms JA, Chang HY (2011) A long noncoding RNA maintains active chromatin to coordinate homeotic gene expression. Nature 472:120–124
Wang L, Brown JL, Cao R, Zhang Y, Kassis JA, Jones RS (2004) Hierarchical recruitment of polycomb group silencing complexes. Mol Cell 14:637–646
Wu H, Coskun V, Tao J, Xie W, Ge W, Yoshikawa K, Li E, Zhang Y, Sun YE (2010) Dnmt3a-dependent nonpromoter DNA methylation facilitates transcription of neurogenic genes. Science 329:444–448
Wu L, Murat P, Matak-Vinkovic D, Murrell A, Balasubramanian S (2013) Binding interactions between long noncoding RNA HOTAIR and PRC2 proteins. Biochemistry 52:9519–9527
Wu X, Johansen JV, Helin K (2013) Fbxl10/Kdm2b recruits polycomb repressive complex 1 to CpG islands and regulates H2A ubiquitylation. Mol Cell 49:1134–1146
Wutz A (2013) Epigenetic regulation of stem cells: the role of chromatin in cell differentiation. Adv Exp Med Biol 786:307–328
Xie W, Schultz MD, Lister R, Hou Z, Rajagopal N, Ray P, Whitaker JW, Tian S, Hawkins RD, Leung D, Yang H, Wang T, Lee AY, Swanson SA, Zhang J, Zhu Y, Kim A, Nery JR, Urich MA, Kuan S, Yen C-A, Klugman S, Yu P, Suknuntha K, Propson NE, Chen H, Edsall LE, Wagner U, Li Y, Ye Z, Kulkarni A, Xuan Z, Chung W-Y, Chi NC, Antosiewicz-Bourget JE, Slukvin I, Stewart R, Zhang MQ, Wang W, Thomson JA, Ecker JR, Ren B (2013) Epigenomic analysis of multilineage differentiation of human embryonic stem cells. Cell 153:1134–1148
Xu CR, Zaret KS (2012) Chromatin "pre-pattern" and epigenetic modulation in the cell fate choice of liver over pancreas in the endoderm. Nucleus 3:150–154
Xu C, Bian C, Yang W, Galka M, Ouyang H, Chen C, Qiu W, Liu H, Jones AE, MacKenzie F, Pan P, Li SS-C, Wang H, Min J (2010) Binding of different histone marks differentially regulates the activity and specificity of polycomb repressive complex 2 (PRC2). Proc Natl Acad Sci U S A 107:19266–19271
Yoshimatsu T, Kawaguchi D, Oishi K, Takeda K, Akira S, Masuyama N, Gotoh Y (2006) Non-cell-autonomous action of STAT3 in maintenance of neural precursor cells in the mouse neocortex. Development 133:2553–2563
Yuan W, Wu T, Fu H, Dai C, Wu H, Liu N, Li X, Xu M, Zhang Z, Niu T, Han Z, Chai J, Zhou XJ, Gao S, Zhu B (2012) Dense chromatin activates Polycomb repressive complex 2 to regulate H3 lysine 27 methylation. Science 337:971–975
Yun M, Wu J, Workman JL, Li B (2011) Readers of histone modifications. Cell Res 21:564–578
Zhang XQ, Leung GK (2014) Long non-coding RNAs in glioma: Functional roles and clinical perspectives. Neurochem Int (in press)
Zhao J, Ohsumi TK, Kung JT, Ogawa Y, Grau DJ, Sarma K, Song JJ, Kingston RE, Borowsky M, Lee JT (2010) Genome-wide identification of polycomb-associated RNAs by RIP-seq. Mol Cell 40:939–953
Zhao XD, Han X, Chew JL, Liu J, Chiu KP, Choo A, Orlov YL, Sung WK, Shahab A, Kuznetsov VA, Bourque G, Oh S, Ruan Y, Ng HH, Wei CL (2007) Whole-genome mapping of histone H3 Lys4 and 27 trimethylations reveals distinct genomic compartments in human embryonic stem cells. Cell Stem Cell 1:286–298
Zhou VW, Goren A, Bernstein BE (2011) Charting histone modifications and the functional organization of mammalian genomes. Nat Rev Genet 12:7–18
Zhu J, Adli M, Zou JY, Verstappen G, Coyne M, Zhang X, Durham T, Miri M, Deshpande V, De Jager PL, Bennett DA, Houmard JA, Muoio DM, Onder TT, Camahort R, Cowan CA, Meissner A, Epstein CB, Shoresh N, Bernstein BE (2013) Genome-wide chromatin state transitions associated with developmental and environmental cues. Cell 152:642–654
Ziats MN, Rennert OM (2013) Aberrant expression of long noncoding RNAs in autistic brain. J Mol Neurosci 49:589–593
Acknowledgments
We thank Kesavan Meganathan for providing micrographs of hESCs undergoing directed differentiation (Fig. 1) and Ethan Patterson, Bryan Teets, Laura Waller, and Kesavan Meganathan for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work was funded by grants from the NIH (GM66815), the March of Dimes (FY13-413), and the Association for Research on Childhood Cancer to K.K. and a Cell and Molecular Biology Training Grant (GM007067) to M.C.
Rights and permissions
About this article
Cite this article
Corley, M., Kroll, K.L. The roles and regulation of Polycomb complexes in neural development. Cell Tissue Res 359, 65–85 (2015). https://doi.org/10.1007/s00441-014-2011-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-014-2011-9