Abstract
Trichinella spiralis is a foodborne zoonotic nematode, which causes trichinellosis. During the infection, parasite evades the host immune responses by direct and indirect (through excretory-secretory products) contact with host immune cells. One of the main targets for immunomodulation induced by helminths are macrophages. In this study, we examined whether direct contact of different stages of T. spiralis can affect the polarization of human THP-1 macrophages. Co-culture of adult parasite stage and cells in direct contact without LPS addition had a significant impact on TNFα levels. Interestingly, in settings with the addition of LPS, the levels of IL-1β and TNFα significantly increased in adult parasite and newborn larvae (NBL) but not for muscle larvae (ML). While we tested muscle larvae ESP products to compare its effect with whole ML parasite, we detect an increase of pro-inflammatory cytokines like IL-1β and TNFα in no LPS conditions. Whereas, muscle larvae ESP significantly suppressed the inflammatory response measured by IL-1β, TNFα, and IL-6 levels and anti-inflammatory IL-10 compared to LPS control. Our findings indicate the anti-inflammatory potential of T. spiralis muscle larvae excretory-secretory products and propose signaling pathways which might be engaged in the mechanism of how muscle larvae ESP affect human macrophages.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Trichinella spiralis is a zoonotic nematode which causes human disease, trichinellosis. Pathological processes of the disease divide to intestinal and muscle phases (Chen et al. 2013) where all three parasite life stages, infective muscle larvae (ML), adult, and newborn larvae (NBL), develop in one host. The source of infection is raw or undercooked meat of animals infected with this parasite. Infective ML are released in the stomach, molt, and develop into an adult stage in the small intestine. Newborn larvae are released into the circulation and spread throughout the organs and tissues, reaching striated muscles where their maturation into muscle larvae is possible. During the infection, different stages of T. spiralis interact with the host immune system to evade or dampen the immune responses (Bruschi 2002). Intestinal phase induces pathological changes resulting in inflammatory responses (Khan and Collins 2004). This early intestinal immune response is due to the interaction of parasite with intestinal mucosal cells and present immune cells. During this phase, the immune response involves predominantly Th1 and subsequent predominance of a Th2 response in the muscle stage of infection with high expression of IL-4, IL-10, and IL-13 (Yu et al. 2013). Contradictory, other research shows an increase in intestinal mucosal immune cells of infected mice and induced mixed Th1/Th2 phenotypes with a predominance of Th2 already at the early stage of infection (Ding et al. 2017). How does T. spiralis regulate the host immune response remains to be elucidated.
The parasite completes its whole life cycle in one host, affecting the host immune system with surface molecules and excretory-secretory products (ESP) from each life cycle stage. Most research focus on the immunomodulatory properties of ML ESP, and the knowledge of modulation by different stages of the parasite is limited. Through the infection, parasite interacts with immune cells via direct contact or more indirectly through excretory-secretory molecules.
The host immune system needs to recognize and react to pathogen-associated molecular patterns (PAMPs) to initiate and develop an adaptive immune response. This process depends on crucial players—professional antigen-presenting cells (APCs) such as dendritic cells (DCs) and macrophages. APCs recognize and react to PAMPs through pattern recognition receptors (PRRs) such as toll-like receptors (TLRs) and C-type lectin receptors (CLR) (Reis e Sousa 2001). Helminths are targeting these interactions by blocking PRR ligand-dependent activation of DCs and macrophages. TLR signals through MyD88 pathway, leading to the synthesis of pro-inflammatory cytokines induced by nuclear factor kappa B (NFκB). The in vivo infection study shows that each T. spiralis life-cycle stage differently affects TLR expression by having a diverse impact on host immune response (Yu et al. 2013). Excretory-secretory products from different parasite stages can modulate macrophage function in vitro by suppressing pro-inflammatory cytokines (Bai et al. 2012). ML ESP regulate the immune response by its inhibitory effect on DC maturation and T cell polarization (Gruden-Movsesijan et al. 2011; Ilic et al. 2018; Sun et al. 2019).
Our study aimed to investigate the effect of parasite stages (adult worm, ML, and NBL) on the modulation of human THP-1 macrophage activity during direct contact of parasites with the cells. Additionally, we compared the immunomodulatory potential of direct ML and indirect ML ESP interaction with macrophages in normal and inflammatory conditions induced by LPS.
Materials and methods
Experimental animals and ethics statement
C3H/W mice were kept in plastic cages in the animal house facilities of the Institute of Parasitology, PAS. Animals were given ad libitum access to food and water, and natural photoperiod conditions were ensured. All experimental procedures used in the present study had been pre-approved by the 1st Local Ethical Committee for Scientific Experiments on Animals in Warsaw, Poland (resolution nr 020/2016, 23rd of March 2016).
Isolation of different life stages of parasite and preparation of ESP antigens
The nematode T. spiralis, strain ISS003, was maintained by several passages in female BALB/c mice at the Institute of Parasitology, PAS. ESP were obtained from T. spiralis muscle larvae recovered from experimentally infected mice (Kapel and Gamble 2000). Infection of mice was carried out using 500 T. spiralis ML at least 3 months before their euthanasia. The recovery of ML was performed by standard HCl-pepsin digestion of tissue followed by several washes with water through succeeding steps of sedimentation in cylinders. After the final sedimentation, the ML were collected. The larval pellet was extensively washed three times in sterile RPMI supplemented with antibiotics (50 U/ml penicillin, 50 μg/ml streptomycin). ESP were collected from ML cultivated in RPMI 1640 medium (Sigma) supplemented with 25 mM HEPES, 2 mM l-glutamine, antibiotics (50 U/ml penicillin, 50 μg/ml streptomycin) and incubated with 5% CO2 in a 75 cm2 culture flask at 37 °C for up to 18 h, followed by sedimentation in 50 ml conical tubes. Culture fluid containing the ML ESP were filtered through a 0.22-μm filter and concentrated on Amicon® Ultra Centrifugal Filters Ultracel-3K (Millipore). Protein concentration (ESP) was determined in the Bradford protein assay. Samples were stored at − 80 °C until analysis. ML used for direct culture with macrophages were added to the cells directly after washing with sterile RPMI and counting. Adult-stage parasites were collected from the small intestine of BALB/c mice (3–4 months old thirty-five BALB/c mice were infected with 900 ML) 6 days post-infection (dpi). NBL were recovered from adult worms incubated in culture medium for 18 h. The intestines were washed in water and cut into pieces of approximately 20 × 20 mm. The intestinal fragments were incubated for 2 h at 37 °C on a mesh placed on top of a conical dish filled with RPMI medium supplemented with 25 mM HEPES, 2 mM l-glutamine, antibiotics (50 U/ml penicillin, 50 μg/ml streptomycin). The adult worms collected from the bottom of the dish were washed and incubated in RPMI medium supplemented with 25 mM HEPES, 2 mM l-glutamine, antibiotics (50 U/ml penicillin, 50 μg/ml streptomycin), and 1 mM Na-pyruvate for 18 h at 37 °C in a humidified atmosphere of 5% CO2. After cultivation, NBL were separated from adult worms with a sterile, 75-μm sieve (Beiting et al. 2004). Both parasite stages were intensively washed three times in fresh RPMI medium, counted and used for culture with macrophages. Only the viable parasites were used for cell stimulation as judged by their motility in microscopic evaluation.
Cell culture and stimulation
A human monocyte cell line THP-1 was purchased from the American Type Culture Collection (ATCC). Cells were cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum, 2 mM glutamine, 100 U/ml penicillin, and 100 μg/ml streptomycin at 37 °C in a humidified atmosphere of 5% CO2. Cells were plated in 6-well plates at a seeding density of 1 × 106 cells/well in whole volume of 4.8 ml/well. The differentiation of cells into macrophages was performed in the presence of 100 ng/ml phorbol 12-myristate 13-acetate (PMA) for 72 h followed by two washes with fresh media w/o PMA. For each well 50 adults, 100 ML/NBL or 5 μg/ml of ESP were added. The concentration of ESP was chosen based on previously published papers (Aranzamendi et al. 2012; Bai et al. 2012; Du et al. 2014; Langelaar et al. 2009). In LPS-treated cells, LPS (100 ng/ml) was added 1 h before the stimulation with ESP or the parasite. Culture media was collected after 24 h stimulation. Cells were washed with sterile PBS and lysed with lysis buffer from Proteome Profiler kit (R&D) for phosphokinase analysis or treated with fenozol supplied with total RNA kit (A&A Biotechnology) for RNA isolation and stored in − 80 until use.
cDNA synthesis and real-time PCR analysis
Total RNAs were isolated from the cells stimulated with ES products or whole parasite according to the kit manufacturer’s instructions. The parasites were washed out from the cells with additional PBS washing steps. For the first-strand cDNA synthesis, 0.7 μg of total isolated RNA were used according to protocol of Maxima™. First Strand cDNA Synthesis Kit for RT-qPCR (Thermo Scientific). The Luminaris Color HiGreen High ROX qPCR master mix (Thermo Scientific) was used for 10 μl qPCR reactions in StepOne Real-Time PCR System, Applied Biosystems.
Gene-specific primers, presented in Table 1, are intron-spanning and were purchased from Sigma. Primers sequences were designed or taken from the Jaguin M. et al. (2013) paper. In the experiment, two housekeeping genes (β-actin and RPL37A) were used. The selection of housekeeping genes was based on a previously published paper (Maess et al. 2010). All primers pairs were designed to have a melting temperature of about 64 °C. The reactions were performed as follows: 2 min at 50 °C and 10 min at 95 °C followed by 40 cycles of a two-step PCR consisting of a denaturing phase at 95 °C for 15 s and a combined annealing and extension phase at 72 °C for 30 s. The Ct value of β-actin and RPL37A was subtracted from that of the gene of interest to obtain a ΔCt value. The ΔCt value of the least abundant sample at all time points for each gene was subtracted from the ΔCt value of each sample to obtain a ΔΔCt value. The gene expression level relative to the calibrator was expressed as 2-ΔΔCt.
Phospho-kinase arrays
The phospho-antibody array analysis was performed using the Proteome Profiler Human Phospho-Kinase Array Kit from R&D Systems according to the manufacturer’s instructions. After 24 h stimulation macrophages were lysed with Lysis Buffer 6 (R&D Systems) and agitated for 30 min at 4 °C. Cell lysates were clarified by microcentrifugation at 14,000×g for 5 min, and the protein content of the supernatants was determined using Pierce BCA Protein Assay Kit (Thermo Scientific). Pre-blocked nitrocellulose membranes of the Human Phospho-Kinase Array were incubated with ~ 240 μg of cellular extract overnight at 4 °C on a rocking platform. The membranes were washed three times with 1× Wash Buffer (R&D Systems) to remove unbound proteins and then incubated with a mixture of biotinylated detection antibodies and streptavidin-HRP antibodies. Chemiluminescent detection reagents were applied to detect spot densities. Membranes were exposed to X-ray film for 3, 5, and 10 min. Array images were analyzed using the image analysis software (Quantity One, Bio-Rad Laboratories, Inc.). All specific phosphorylation sites tested for each kinase are described in Online resource 1.
ELISAs
Cytokine (TNFα, IL-1β, IL-6, IL-10) concentrations were determined using commercial ELISA kits OptEIATM Set Human (BD Biosciences) in culture supernatants. All experiments were performed independently in triplicate. Optical densities were read at the appropriate wavelength on a microplate reader, and measurements were calculated as means ± SEs.
Statistical analysis
ΔCt values for all genes were normalized to mean Ct of β-actin and RPL37 housekeeping genes. ΔCt values for treated samples and controls (calibrators) were compared by t test for independent samples. Differences at P < 0.05 were considered significant. Analyses were performed using Prism-GraphPad software. Annotations used are as follows: ***P < 0.001, **P < 0.01, and *P < 0.05. The same t test was used for the analysis of ELISA experiments.
Results
Cytokine response in macrophages stimulated with different stages of T. spiralis
To determine whether different stages of T. spiralis adult worms, NBL or ML altered the cytokine response of human macrophages, several key pro-inflammatory and anti-inflammatory cytokines were measured by ELISA. Co-culture of parasite and cells in direct contact without LPS addition had no significant impact on IL-1β levels. At the same time, we could observe considerable induction of TNFα in adult stage culture and slight but notable increase of IL-10 with NBL (Online resource 1).
Interestingly, in settings with the addition of LPS, the levels of IL-1β and TNFα significantly increased in adult parasite co-culture, and the same pattern was observed for NBL but not for ML. No changes in IL-6 secretion were detected for all stages of T. spiralis (Fig. 1).
Cytokine ELISA analysis. Cytokine levels in culture media of THP-1 macrophages stimulated only with LPS (−), LPS and adult, muscle larvae (ML), new-born larvae (NBL), or muscle larvae excretory-secretory products (ESP). Cells (1 × 106/ml) were co-cultured for 24 h with whole parasite or 5 μg/ml ESP with LPS (100 ng/ml) addition. Results are presented as a mean ± SEM of three independent experiments. Statistical analysis was performed in Prism-GraphPad program by t test. A value of P ≤ 0.05 was considered significant (***P < 0.001; **P < 0.01; *P < 0.05)
While we tested ES products to compare its effect with whole ML parasite, we detected an increase of pro-inflammatory cytokines such as IL-1β and TNFα in no LPS settings. The levels of IL-6 in experiments without LPS were undetectable using selected ELISA kit. ML ESP significantly suppressed the inflammatory response judging by IL-1β, TNFα, and IL-6 levels compared with LPS control but also inhibits the anti-inflammatory IL-10 in these conditions (Fig. 1, Online resource 1).
Change in kinase phosphorylation profiles in THP-1 macrophages after ESP and ML stimulation
To find the signaling pathway, which might be involved in ML ESP mechanism to dampen the inflammation, we screened the phosphorylation of kinases crucial in many different paths. Stimulation with ESP and LPS induces substantial fold increase of Hsp60, Hsp27, c-Jun, and STAT3 versus control LPS samples. In this condition, the phosphorylation of β-catenin, ERK1/2, and Akt 1/2/3 S473 decrease comparing with control (Fig. 2). While in ML samples, there was no phosphorylation decrease detected, we could identify an evident increase in phosphorylation of STAT3, eNOS, and similarly to ESP, higher levels of Hsp60 and Hsp70 (Fig. 2). Analysis of stimulation with ESP without LPS revealed higher fold increase of Hsp60, Hsp27 JNK1/2/3, and c-Jun comparably with ESP + LPS, whereas among reduced phosphorylation levels, we could find ERK1/2 and Akt1/2/3 S473. In ML-stimulated cells, degrees of phosphorylation of most tested kinases were higher than control samples (Online resource 2).
Phospho-kinase analysis. Changes in kinase phosphorylation levels in macrophages stimulated with muscle larvae (ML) and muscle larvae ESP and LPS (100 ng/ml) was determined by Proteome Profiler Human Phospho-Kinase Array Kit. Stimulated cells were lysed with kit lysis buffer and frozen in − 80 °C until use. As a result, we present the adjusted mean volume (OD × mm2) fold change of ML or ESP versus LPS control. The average intensity of the pixels in background volume was calculated and subtracted from each pixel in all standard and unknown
T. spiralis ESP impact on THP-1 macrophage gene expression levels
Our findings indicate that ESP from muscle larvae has a significant inhibitory effect on macrophage-originated cytokines. To evaluate the effect of ESP on macrophage activation, we investigated the expression of certain additional factors on mRNA level. Stimulation of macrophages with ESP and LPS significantly reduced TNFα gene expression compared with control, what was also confirmed on protein level in cell culture media analyses (Fig. 3). Additionally, expression levels of chemokines CCL3, CXCL11, CCL22, and intracellular adhesion molecule ICAM-1 were reduced after stimulation with ESP. No effect was noted in the mRNA levels of CCL4, while expression of HLA-A and HLA-DR was higher for ESP stimulated cells as well as CHI3L1, PPARγ, TGFβ, and IL-10 (Fig. 3).
Gene expression levels. Gene expression analysis was determined by qPCR. The results are calculated as 2-ΔΔCt. ΔCt values for all genes were normalized to mean Ct of β-actin and RPL37 housekeeping genes. Results are presented as a mean ± SEM of three independent experiments. Statistical analysis was performed in Prism-GraphPad program by t test. A value of P ≤ 0.05 was considered significant (***P < 0.001; **P < 0.01; *P < 0.05)
Discussion
Helminths can manipulate host immune responses on different levels. Most helminths secrete an array of immune-modulatory products that facilitate survival within their host (Loukas et al. 2016; Maizels et al. 2018); however, the direct contact of parasite’s surface proteins is also crucial. The interaction of Trichinella spiralis and host immune system is unclear, especially within the human host. Trying to elucidate this, we tested how different stages of the parasite will affect human macrophages during direct contact of whole parasites and indirect through ESP of muscle larvae. Research shows the influence of parasitic worms on macrophage polarization (Kelly and O’Neill 2015), although the detailed mechanisms underlying this process are unknown. Our team chose THP-1 human leukemia monocyte cell line to generate macrophages as other researchers and we successfully used it previously to test functional, cytokine, and signaling pathway changes which may occur during helminth infection (Baska et al. 2013; Dlugosz et al. 2019; Johnston et al. 2010; Silva-Alvarez et al. 2016; Zawistowska-Deniziak et al. 2017). We found that co-culture of macrophages with an adult or NBL stage significantly induced the production of inflammatory IL-1β and TNFα in LPS-induced macrophages. Our data are inconsistent with the paper of Bai et al. (2012), where authors show suppression of pro-inflammatory cytokines in mouse macrophages after treatment with ES products from these parasite stages. Although in our experiment parasites in co-culture were alive and secreting ES products, the direct contact of surface molecules with cells might have abolished this effect. Another possible reason is the amount of actively released ES products, which in the end was lower than used by Bai et al. (2012). Macrophages without LPS stimulation were producing more TNFα after adult stage co-culture and more IL-10 but significant only for NBL stage. The effect on IL-10 production in these conditions was the same as Bai team shows for ES products of adult and NBL stage, and TNFα was not tested. The direct interaction of ML with macrophages showed no significant change in expression of tested cytokines.
Interestingly, we demonstrated that ESP of muscle larvae has anti-inflammatory potential by inhibiting IL-1β, TNFα, and IL-6 levels in cells stimulated with LPS what was consistent with previous paper (Bai et al. 2012). Even though we identified higher gene expression of IL-10 and TGFβ, the protein level for IL-10 was reduced compared with LPS control and TGFβ was undetectable. The previous study shows that high levels of mRNA expression do not necessarily reflect the amount of protein secreted by the cell and cells regulate it on a transcriptional and translational level (Vogel and Marcotte 2012). ESP stimulated macrophages had lower expression of CLL3 but not CCL4, major factors produced by macrophages and monocytes after stimulation with bacterial endotoxins (Sherry et al. 1988) and cytokines like IL-1β (Menten et al. 2002). We identified lower mRNA levels for CXCL11—chemotactic for activated T cells (Sauty et al. 2001), and CCL22—inducer of migration of CD4 T cells and enhancer of IFNγ production that may impair immune tolerance (Ushio et al. 2018). Reports show T. spiralis-infected muscles area have a high density of macrophages, lower immune cell infiltration, and the effect is IL-10 dependent (Beiting et al. 2004), but might also be chemokine dependent. Thus, our results suggest that ESP modulate the balance between anti-inflammatory and pro-inflammatory responses not only on cytokine but also on chemokine levels.
Less inflammatory phenotype of macrophages induced with muscle larvae ESP illustrates the higher expression of PPARγ and CHI3L1, one of the characteristic factors of alternative macrophage activation (Chawla 2010). Several studies demonstrate the suppressive effect of muscle larvae ES products on dendritic cell maturation stimulated with LPS and increased levels of immunoregulatory cytokines (Cvetkovic et al. 2014; Ilic et al. 2008; Sun et al. 2019). Our results clearly show that stimulation of human macrophages with muscle larvae ESP inhibits the inflammatory response of macrophages and generates a more anti-inflammatory phenotype. Interestingly, direct co-culture of muscle larvae with macrophages lacks this effect, which may be the consequence of a lower amount of ESP produced by larvae. Also, the presence of tegumental molecules might block the ESP impact.
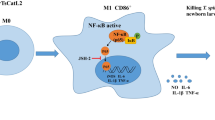
To elucidate the signaling pathways that might be involved in the anti-inflammatory effect of muscle larvae ESP, we compared the levels of kinase phosphorylation after ESP and whole ML cell treatment. The use of the commercial Proteome Profiler Human Phospho-Kinase Array enabled simultaneous analysis of 43 kinase phosphorylation sites. One of the most pronounced (about 20-fold) increase in phosphorylation was observed for heat shock protein 27 (HSP27) present in ESP and ML-treated cells compared with LPS control. Similarly, HSP60 levels increased to the same extent in these two conditions. Heat shock proteins play important roles in antigen presentation, activation of lymphocytes, macrophages, and maturation of dendritic cells (Tsan and Gao 2009).
The most decreased phosphorylation levels specific for ESP treatment was Akt 1/2/3 S473, ERK1/2 and β-catenin when compare with LPS control. Considering the relevance of ERK1/2 in signaling involved in the regulation of immune response (Arthur and Ley 2013), the other researchers have already tested the impact of T. spiralis ES products. While the data presented by Bai et al. (2012) are consistent with ours, the results of Cvetkovic et al. (2014) are contradictory. In our experiments, inhibition of ERK phosphorylation was also present in conditions without LPS. ERK1/2 is a member of the mitogen-activated protein kinase family (MAPK) and its role in LPS-induced inflammation is well known (Shi et al. 2002). The NFκB and MAPK pathways are two important regulators of LPS-stimulated pro-inflammatory response in macrophages. Excretory-secretory products of other helminths inhibit NFκB activation (Puneet et al. 2011) and reduce phosphorylation of ERK 1/2 and p38 in mouse macrophages.
The PI3K-Akt signaling pathway has also been suggested to regulate the responses of macrophages to inflammatory stimuli (Hazeki et al. 2007; Lee et al. 2011) and may act as a safety mechanism to limit the response to pathogens. We observed reduced phosphorylation of Akt1/2/3 on S473. The literature shows that mammalian target of rapamycin complex 2 (mTORC2) controls the phosphorylation of this particular serine (Sarbassov et al. 2005), and it is AMPK which activates the mTORC2 (Kazyken et al. 2019). ESP stimulation also reduced the AMPK phosphorylation levels. Our data indicate that ESP anti-inflammatory effect might be controlled and shaped by the mTORC1-mTORC2 network, which reconfigures cellular metabolism and regulates translation, cytokine responses, antigen presentation, macrophage polarization, and cell migration (Weichhart et al. 2015).
Interestingly, we identified reduced phosphorylation levels for the epidermal growth factor receptor (EGFR). Activation of EGFR reportedly couples to tyrosine kinase-induced auto-phosphorylation, which subsequently activates multiple cellular signaling cascades like Pi3K-AKT and Raf-MAPK-ERK1/2 pathways (Leto and Trusolino 2014; Wieduwilt and Moasser 2008; Zhou et al. 2015). Inhibition of EGFR-tyrosine kinase diminishes neuroinflammation and affects Akt1/2/3 S473 and ERK1/2 phosphorylation (Chen et al. 2019).
Muscle larvae ESP highly reduced β-catenin levels, which implies that Wnt/β-catenin signaling might be involved. The literature shows that this pathway regulates both pro-inflammatory (Blumenthal et al. 2006; Halleskog and Schulte 2013) and anti-inflammatory (Suryawanshi et al. 2016; Swafford and Manicassamy 2015) responses in various cell types. For example, activation of canonical Wnt/β-catenin significantly induced pro-inflammatory cytokine production secretion in murine macrophages (Yu et al. 2014). However, knockdown of β-catenin also increased the pro-inflammatory cytokine IL-6 expression in RAW264.7 macrophages, indicating the anti-inflammatory effects of β-catenin in those cells (Lee et al. 2012). In several tissues, overactivated canonical Wnt/β-catenin pathway downregulates PPARγ, and conversely (Vallee and Lecarpentier 2018). Therefore, β-catenin might play a role in the regulation of ESP anti-inflammatory function what relates to induced PPARγ mRNA levels.
In summary, this study demonstrates that direct contact of the parasite with macrophages might induce a different reaction of macrophages when compared with excretory-secretory products. Like the others, our data show the anti-inflammatory potential of muscle larvae ESP on macrophages. To our knowledge, this is the first study on human macrophages co-cultured with T. spiralis whole parasite at different stages. Our analysis revealed signaling pathways which might be involved in ESP anti-inflammatory function. Further research is essential as ESP are not only a promising approach to understand host-parasite interaction but also hold potential as therapeutics. Adoptive transfer of macrophages obtained from infected mice and ES protein-activated macrophages is effective for prevention and treatment of airway allergy in mice and is a promising therapeutic for treating inflammatory diseases (Kang et al. 2019). Our future research will focus on the identification and characterization of the immunomodulatory potential of single molecules present in ES products mixture. This strategy could be crucial to fully understand the mechanisms of T. spiralis immunomodulation strategy and future use of parasite molecules as potential treatment.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Aranzamendi C et al (2012) Trichinella spiralis-secreted products modulate DC functionality and expand regulatory T cells in vitro parasite. Immunol 34:210–223. https://doi.org/10.1111/j.1365-3024.2012.01353.x
Arthur JS, Ley SC (2013) Mitogen-activated protein kinases in innate immunity. Nat Rev Immunol 13:679–692. https://doi.org/10.1038/nri3495
Bai X et al (2012) Regulation of cytokine expression in murine macrophages stimulated by excretory/secretory products from Trichinella spiralis in vitro. Mol Cell Biochem 360:79–88. https://doi.org/10.1007/s11010-011-1046-4
Baska P, Wisniewski M, Krzyzowska M, Dlugosz E, Zygner W, Gorski P, Wedrychowicz H (2013) Molecular cloning and characterisation of in vitro immune response against astacin-like metalloprotease Ace-MTP-2 from Ancylostoma ceylanicum. Exp Parasitol 133:472–482. https://doi.org/10.1016/j.exppara.2013.01.006
Beiting DP, Bliss SK, Schlafer DH, Roberts VL, Appleton JA (2004) Interleukin-10 limits local and body cavity inflammation during infection with muscle-stage Trichinella spiralis. Infect Immun 72:3129–3137. https://doi.org/10.1128/IAI.72.6.3129-3137.2004
Blumenthal A et al (2006) The wingless homolog WNT5A and its receptor Frizzled-5 regulate inflammatory responses of human mononuclear cells induced by microbial stimulation. Blood 108:965–973. https://doi.org/10.1182/blood-2005-12-5046
Bruschi F (2002) The immune response to the parasitic nematode Trichinella and the ways to escape it. From experimental studies to implications for human infection. Curr Drug Targets Immune Endocr Metabol Disord 2:269–280. https://doi.org/10.2174/1568008023340523
Chawla A (2010) Control of macrophage activation and function by PPARs. Circ Res 106:1559–1569. https://doi.org/10.1161/CIRCRESAHA.110.216523
Chen Y, Huang B, Huang S, Yu X, Li Y, Song W, Lu F (2013) Coinfection with Clonorchis sinensis modulates murine host response against Trichinella spiralis infection. Parasitol Res 112:3167–3179. https://doi.org/10.1007/s00436-013-3493-1
Chen YJ, Hsu CC, Shiao YJ, Wang HT, Lo YL, Lin AMY (2019) Anti-inflammatory effect of afatinib (an EGFR-TKI) on OGD-induced neuroinflammation. Sci Rep 9:2516. https://doi.org/10.1038/s41598-019-38676-7
Cvetkovic J, Ilic N, Sofronic-Milosavljevic L, Gruden-Movsesijan A (2014) Glycans expressed on Trichinella spiralis excretory-secretory antigens are important for anti-inflamatory immune response polarization. Comp Immunol Microbiol Infect Dis 37:355–367. https://doi.org/10.1016/j.cimid.2014.10.004
Ding J, Bai X, Wang X, Shi H, Cai X, Luo X, Liu M, Liu X (2017) Immune cell responses and cytokine profile in intestines of mice infected with Trichinella spiralis. Front Microbiol 8:2069. https://doi.org/10.3389/fmicb.2017.02069
Dlugosz E, Basalaj K, Zawistowska-Deniziak A (2019) Cytokine production and signalling in human THP-1 macrophages is dependent on Toxocara canis glycans. Parasitol Res 118:2925-2933 doi:https://doi.org/10.1007/s00436-019-06405-8
Du L, Liu L, Yu Y, Shan H, Li L (2014) Trichinella spiralis excretory-secretory products protect against polymicrobial sepsis by suppressing MyD88 via mannose receptor. Biomed Res Int 2014:898646. https://doi.org/10.1155/2014/898646
Gruden-Movsesijan A, Ilic N, Colic M, Majstorovic I, Vasilev S, Radovic I, Sofronic-Milosavljevic L (2011) The impact of Trichinella spiralis excretory-secretory products on dendritic cells. Comp Immunol Microbiol Infect Dis 34:429–439. https://doi.org/10.1016/j.cimid.2011.08.004
Halleskog C, Schulte G (2013) WNT-3A and WNT-5A counteract lipopolysaccharide-induced pro-inflammatory changes in mouse primary microglia. J Neurochem 125:803–808. https://doi.org/10.1111/jnc.12250
Hazeki K, Nigorikawa K, Hazeki O (2007) Role of phosphoinositide 3-kinase in innate immunity. Biol Pharm Bull 30:1617–1623. https://doi.org/10.1248/bpb.30.1617
Ilic N, Colic M, Gruden-movsesijan A, Majstorovic I, Vasilev S, Sofronic-Milosavljevic L (2008) Characterization of rat bone marrow dendritic cells initially primed by Trichinella spiralis antigens. Parasite Immunol 30:491–495. https://doi.org/10.1111/j.1365-3024.2008.01049.x
Ilic N et al (2018) Trichinella spiralis excretory-secretory products induce tolerogenic properties in human dendritic cells via toll-like receptors 2 and 4. Front Immunol 9:11. https://doi.org/10.3389/fimmu.2018.00011
Jaguin M, Houlbert N, Fardel O, Lecureur V (2013) Polarization profiles of human M-CSF-generated macrophages and comparison of M1-markers in classically activated macrophages from GM-CSF and M-CSF origin. Cell Immunol 281:51–61. https://doi.org/10.1016/j.cellimm.2013.01.010
Johnston MJ, Wang A, Catarino ME, Ball L, Phan VC, MacDonald JA, McKay DM (2010) Extracts of the rat tapeworm, Hymenolepis diminuta, suppress macrophage activation in vitro and alleviate chemically induced colitis in mice. Infect Immun 78:1364–1375. https://doi.org/10.1128/IAI.01349-08
Kang SA, Park MK, Park SK, Choi JH, Lee DI, Song SM, Yu HS (2019) Adoptive transfer of Trichinella spiralis-activated macrophages can ameliorate both Th1- and Th2-activated inflammation in murine models. Sci Rep 9:6547. https://doi.org/10.1038/s41598-019-43057-1
Kapel CM, Gamble HR (2000) Infectivity, persistence, and antibody response to domestic and sylvatic Trichinella spp. in experimentally infected pigs. Int J Parasitol 30:215–221. https://doi.org/10.1016/s0020-7519(99)00202-7
Kazyken D et al. (2019) AMPK directly activates mTORC2 to promote cell survival during acute energetic stress. Sci Signal 12 https://doi.org/10.1126/scisignal.aav3249
Kelly B, O’Neill LA (2015) Metabolic reprogramming in macrophages and dendritic cells in innate immunity. Cell Res 25:771–784. https://doi.org/10.1038/cr.2015.68
Khan WI, Collins SM (2004) Immune-mediated alteration in gut physiology and its role in host defence in nematode infection. Parasite Immunol 26:319–326. https://doi.org/10.1111/j.0141-9838.2004.00715.x
Langelaar M, Aranzamendi C, Franssen F, Van Der Giessen J, Rutten V, van der Ley P, Pinelli E (2009) Suppression of dendritic cell maturation by Trichinella spiralis excretory/secretory products. Parasite Immunol 31:641–645. https://doi.org/10.1111/j.1365-3024.2009.01136.x
Lee YG, Lee J, Byeon SE, Yoo DS, Kim MH, Lee SY, Cho JY (2011) Functional role of Akt in macrophage-mediated innate immunity. Front Biosci (Landmark Ed) 16:517–530. https://doi.org/10.2741/3702
Lee H, Bae S, Choi BW, Yoon Y (2012) WNT/beta-catenin pathway is modulated in asthma patients and LPS-stimulated RAW264.7 macrophage cell line. Immunopharmacol Immunotoxicol 34:56–65. https://doi.org/10.3109/08923973.2011.574704
Leto SM, Trusolino L (2014) Primary and acquired resistance to EGFR-targeted therapies in colorectal cancer: impact on future treatment strategies. J Mol Med (Berl) 92:709–722. https://doi.org/10.1007/s00109-014-1161-2
Loukas A, Hotez PJ, Diemert D, Yazdanbakhsh M, McCarthy JS, Correa-Oliveira R, Croese J, Bethony JM (2016) Hookworm infection. Nat Rev Dis Primers 2:16088. https://doi.org/10.1038/nrdp.2016.88
Maess MB, Sendelbach S, Lorkowski S (2010) Selection of reliable reference genes during THP-1 monocyte differentiation into macrophages. BMC Mol Biol 11:90. https://doi.org/10.1186/1471-2199-11-90
Maizels RM, Smits HH, McSorley HJ (2018) Modulation of host immunity by helminths: the expanding repertoire of parasite effector molecules. Immunity 49:801–818. https://doi.org/10.1016/j.immuni.2018.10.016
Menten P, Wuyts A, Van Damme J (2002) Macrophage inflammatory protein-1. Cytokine Growth Factor Rev 13:455–481. https://doi.org/10.1016/s1359-6101(02)00045-x
Puneet P et al (2011) The helminth product ES-62 protects against septic shock via toll-like receptor 4-dependent autophagosomal degradation of the adaptor MyD88. Nat Immunol 12:344–351. https://doi.org/10.1038/ni.2004
Reis e Sousa C (2001) Dendritic cells as sensors of infection. Immunity 14:495–498. https://doi.org/10.1016/s1074-7613(01)00136-4
Sarbassov DD, Guertin DA, Ali SM, Sabatini DM (2005) Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307:1098–1101. https://doi.org/10.1126/science.1106148
Sauty A, Colvin RA, Wagner L, Rochat S, Spertini F, Luster AD (2001) CXCR3 internalization following T cell-endothelial cell contact: preferential role of IFN-inducible T cell alpha chemoattractant (CXCL11). J Immunol 167:7084–7093. https://doi.org/10.4049/jimmunol.167.12.7084
Sherry B et al (1988) Resolution of the two components of macrophage inflammatory protein 1, and cloning and characterization of one of those components, macrophage inflammatory protein 1 beta. J Exp Med 168:2251–2259. https://doi.org/10.1084/jem.168.6.2251
Shi L, Kishore R, McMullen MR, Nagy LE (2002) Lipopolysaccharide stimulation of ERK1/2 increases TNF-alpha production via Egr-1. Am J Phys Cell Phys 282:C1205–C1211. https://doi.org/10.1152/ajpcell.00511.2001
Silva-Alvarez V et al (2016) Echinococcus granulosus antigen B binds to monocytes and macrophages modulating cell response to inflammation. Parasit Vectors 9:69. https://doi.org/10.1186/s13071-016-1350-7
Sun XM, Guo K, Hao CY, Zhan B, Huang JJ, Zhu X (2019) Trichinella spiralis excretory–secretory products stimulate host regulatory t cell differentiation through activating dendritic cells. Cells 8:8. https://doi.org/10.3390/cells8111404
Suryawanshi A, Tadagavadi RK, Swafford D, Manicassamy S (2016) Modulation of inflammatory responses by Wnt/β-catenin signaling in dendritic cells: a novel immunotherapy target for autoimmunity and cancer. Front Immunol 7:460. https://doi.org/10.3389/fimmu.2016.00460
Swafford D, Manicassamy S (2015) Wnt signaling in dendritic cells: its role in regulation of immunity and tolerance. Discov Med 19:303–310
Tsan MF, Gao B (2009) Heat shock proteins and immune system. J Leukoc Biol 85:905–910. https://doi.org/10.1189/jlb.0109005
Ushio A et al (2018) CCL22-producing resident macrophages enhance T cell response in Sjogren’s syndrome. Front Immunol 9:2594. https://doi.org/10.3389/fimmu.2018.02594
Vallee A, Lecarpentier Y (2018) Crosstalk between peroxisome proliferator-activated receptor gamma and the canonical WNT/β-catenin pathway in chronic inflammation and oxidative stress during carcinogenesis. Front Immunol 9:745. https://doi.org/10.3389/fimmu.2018.00745
Vogel C, Marcotte EM (2012) Insights into the regulation of protein abundance from proteomic and transcriptomic analyses. Nat Rev Genet 13:227–232. https://doi.org/10.1038/nrg3185
Weichhart T, Hengstschlager M, Linke M (2015) Regulation of innate immune cell function by mTOR. Nat Rev Immunol 15:599–614. https://doi.org/10.1038/nri3901
Wieduwilt MJ, Moasser MM (2008) The epidermal growth factor receptor family: biology driving targeted therapeutics. Cell Mol Life Sci 65:1566–1584. https://doi.org/10.1007/s00018-008-7440-8
Yu YR, Deng MJ, Lu WW, Jia MZ, Wu W, Qi YF (2013) Systemic cytokine profiles and splenic toll-like receptor expression during Trichinella spiralis infection. Exp Parasitol 134:92–101. https://doi.org/10.1016/j.exppara.2013.02.014
Yu CH, Nguyen TT, Irvine KM, Sweet MJ, Frazer IH, Blumenthal A (2014) Recombinant Wnt3a and Wnt5a elicit macrophage cytokine production and tolerization to microbial stimulation via Toll-like receptor 4. Eur J Immunol 44:1480–1490. https://doi.org/10.1002/eji.201343959
Zawistowska-Deniziak A, Basalaj K, Strojny B, Mlocicki D (2017) New data on human macrophages polarization by hymenolepis diminuta tapeworm—an in vitro study. Front Immunol 8:148. https://doi.org/10.3389/fimmu.2017.00148
Zhou J, Du T, Li B, Rong Y, Verkhratsky A, Peng L (2015) Crosstalk between MAPK/ERK and PI3K/AKT signal pathways during brain ischemia/reperfusion. ASN Neuro 7. https://doi.org/10.1177/1759091415602463
Funding
Financial support for this study was provided by the National Science Center Poland (grant UMO-2015/18/E/NZ6/00502).
Author information
Authors and Affiliations
Contributions
AZ-D designed and performed the experiments, JB-K produced different stages of parasite, AZ-D and KB interpreted the data, AZ-D drafted the manuscript, and AZ-D, JB-K, and KB reviewed and approved the final version of the manuscript. The authors agreed to be accountable for the content of the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
All experimental procedures used in the present study had been pre-approved by the 1st Local Ethical Committee for Scientific Experiments on Animals in Warsaw, Poland (resolution nr 020/2016, 23rd of March 2016).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Code availability
Not applicable.
Additional information
Section Editor: Abdul Jabbar
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Online resource 1
Cytokine ELISA analysis. Cytokine levels in culture media of THP-1 macrophages stimulated with PBS (−), adult, muscle larvae (ML), newborn larvae (NBL), or muscle larvae excretory-secretory products (ESP). Cells (1 × 106/ml) were co-cultured for 24 h with whole parasite or 5 μg/ml ESP. Results are presented as a mean ± SEM of three independent experiments. Statistical analysis was performed in Prism-GraphPad program by t test. A value of P ≤ 0.05 was considered significant (***P < 0.001; **P < 0.01; *P < 0.05) (PNG 150 kb)
Online resource 2
Human kinases phosphorylation sites (DOCX 13 kb)
Online resource 3
Phospho-kinase analysis. Changes in kinase phosphorylation levels in macrophages stimulated with muscle larvae (ML) and muscle larvae ESP was determined by Proteom Profiler Human Phospho-Kinase Array Kit. Stimulated cells were lysed with kit lysis buffer and frozen in − 80 °C until use. As a result, we present the adjusted mean volume (OD × mm2) fold change of ML or ESP versus control. The average intensity of the pixels in background volume was calculated and subtracted from each pixel in all standard and unknown (DOCX 208 kb)
Rights and permissions
About this article
Cite this article
Zawistowska-Deniziak, A., Bień-Kalinowska, J. & Basałaj, K. Regulation of human THP-1 macrophage polarization by Trichinella spiralis. Parasitol Res 120, 569–578 (2021). https://doi.org/10.1007/s00436-020-07000-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00436-020-07000-y