Abstract
Aim
We aimed to evaluate the effect of concomitant proton pump inhibitors (PPI) use with nivolumab on survival outcomes in metastatic renal cell carcinoma (mRCC) in second-line setting.
Methods
The study was designed as a multicenter and retrospective involving patients with metastatic renal cell carcinoma receiving second-line nivolumab therapy. One hundred and nine patients with mRCC were divided into two groups based on whether they use PPI concomitantly with nivolumab: concomitant PPI users and non-users. Overall survival (OS) and progression-free survival (PFS) were compared between the groups with and without concurrent PPIs.
Results
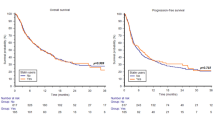
Of 109 patients in our study, 59 were not using PPI concomitantly with nivolumab and 50 were using PPI concomitantly. The median PFS was 6.37 (5.2–7.5) months in the concomitant PPI group and 9.7 (4.5–15) months in the non-users (p = 0.03). The median OS was 14.6 (7.1–22.1) months in patients on PPI concurrently with nivolumab and 29.9 (17.1–42.7) months in the non-users (p = 0.01). Accordingly, PPI use for PFS (Non-use vs. Use = HR: 0.44, 95%Cl 0.28–0.96, p = 0.014) and PPI use for OS (Non-use vs. Use = HR: 0.68, 95%Cl 0.22–0.88, p = 0.01) were found to be as independent risk factors.
Conclusions
Concomitant use of PPIs is associated with worse survival outcomes in patients with mRCC treated with nivolumab. Clinicians should carefully consider the concomitant use of PPIs in such patients.


Similar content being viewed by others
Data availability
Data will be provided by the corresponding author.
References
Ariel H, Cooke JP (2019) Cardiovascular risk of proton pump inhibitors. Methodist Debakey Cardiovasc J 15(3):214
Aybay C, Imir T, Okur H (1995) The effect of omeprazole on human natural killer cell activity. General Pharmacol Vascul Syst 26(6):1413–1418
Çağlayan D, Koçak MZ, Geredeli Ç, Tatli AM, Göksu SS, Eryilmaz MK, Araz M, Artaç M (2022) The effect of concomitant use of proton pump inhibitors with CDK 4/6 inhibitors on survival in metastatic breast cancer. Europ J Clin Pharmacol 79(2):1–6
Chaput N, Lepage P, Coutzac C, Soularue E, Le Roux K, Monot C, Boselli L, Routier E, Cassard L, Collins M (2017) Baseline gut microbiota predicts clinical response and colitis in metastatic melanoma patients treated with ipilimumab. Ann Oncol 28(6):1368–1379
Derosa L, Hellmann M, Spaziano M, Halpenny D, Fidelle M, Rizvi H, Long N, Plodkowski A, Arbour K, Chaft J (2018) Negative association of antibiotics on clinical activity of immune checkpoint inhibitors in patients with advanced renal cell and non-small-cell lung cancer. Ann Oncol 29(6):1437–1444
Desai BV, Qadri MN, Vyas BA (2022) Proton pump inhibitors and osteoporosis risk: exploring the role of TRPM7 channel. Eur J Clin Pharmacol 78:35–41
Gopalakrishnan V, Helmink BA, Spencer CN, Reuben A, Wargo JA (2018) The influence of the gut microbiome on cancer, immunity, and cancer immunotherapy. Cancer Cell 33(4):570–580
Imhann F, Bonder MJ, Vila AV, Fu J, Mujagic Z, Vork L, Tigchelaar EF, Jankipersadsing SA, Cenit MC, Harmsen HJ (2016) Proton pump inhibitors affect the gut microbiome. Gut 65(5):740–748
Jackson MA, Goodrich JK, Maxan M-E, Freedberg DE, Abrams JA, Poole AC, Sutter JL, Welter D, Ley RE, Bell JT (2016) Proton pump inhibitors alter the composition of the gut microbiota. Gut 65(5):749–756
Jeong SH, Molloy L, Ang E, Helsby N (2022) Re-thinking the possible interaction between proton pump inhibitors and capecitabine. Cancer Chemother Pharmacol 90(5):381–388
Kichenadasse G, Miners JO, Mangoni AA, Karapetis CS, Hopkins AM, Sorich MJ (2021) Proton pump inhibitors and survival in patients with colorectal cancer receiving fluoropyrimidine-based chemotherapy. J Natl Compr Canc Netw 19(9):1037–1044
Korus H, Vakil N (2023) Acid suppression and effects on the gastrointestinal microbiome. Esophageal disease and the role of the Microbiomeed. Elsevier, Amsterdam Netherlands, pp 35–46
Laheij RJ, Sturkenboom MC, Hassing R-J, Dieleman J, Stricker BH, Jansen JB (2004) Risk of community-acquired pneumonia and use of gastric acid–suppressive drugs. JAMA 292(16):1955–1960
Li C, Xia Z, Li A, Meng J (2020) The effect of proton pump inhibitor uses on outcomes for cancer patients treated with immune checkpoint inhibitors: a meta-analysis. Annals Trans Med 8(24):1655
Mollica V, Santoni M, Matrana MR, Basso U, De Giorgi U, Rizzo A, Maruzzo M, Marchetti A, Rosellini M, Bleve S (2022) Concomitant proton pump inhibitors and outcome of patients treated with nivolumab alone or plus ipilimumab for advanced renal cell carcinoma. Target Oncol 17(1):61–68
Motzer RJ, Escudier B, McDermott DF, George S, Hammers HJ, Srinivas S, Tykodi SS, Sosman JA, Procopio G, Plimack ER (2015) Nivolumab versus everolimus in advanced renal-cell carcinoma. N Engl J Med 373(19):1803–1813
2023 OSoPCB-MSChweeeedp-io-e-p-iepAJ
Pinato DJ, Howlett S, Ottaviani D, Urus H, Patel A, Mineo T, Brock C, Power D, Hatcher O, Falconer A (2019) Association of prior antibiotic treatment with survival and response to immune checkpoint inhibitor therapy in patients with cancer. JAMA Oncol 5(12):1774–1778
Rassy E, Dalban C, Colomba E, Derosa L, Silva CAC, Negrier S, Chevreau C, Gravis G, Oudard S, Laguerre B (2022) Efficacy and safety of concomitant proton pump inhibitor and nivolumab in renal cell carcinoma: results of the GETUG-AFU 26 NIVOREN multicenter phase II study. Clin Genitourin Cancer 20(5):488–494
Rizzo A, Santoni M, Mollica V, Ricci AD, Calabrò C, Cusmai A, Gadaleta-Caldarola G, Palmiotti G, Massari F (2022) The impact of concomitant proton pump inhibitors on immunotherapy efficacy among patients with urothelial carcinoma: a meta-analysis. J Personal Med 12(5):842
Routy B, Le Chatelier E, Derosa L, Duong CP, Alou MT, Daillère R, Fluckiger A, Messaoudene M, Rauber C, Roberti MP (2018) Gut microbiome influences efficacy of PD-1–based immunotherapy against epithelial tumors. Science 359(6371):91–97
Roy S, Trinchieri G (2017) Microbiota: a key orchestrator of cancer therapy. Nat Rev Cancer 17(5):271–285
Salgia NJ, Bergerot PG, Maia MC, Dizman N, Hsu J, Gillece JD, Folkerts M, Reining L, Trent J, Highlander SK (2020) Stool microbiome profiling of patients with metastatic renal cell carcinoma receiving anti–PD-1 immune checkpoint inhibitors. Eur Urol 78(4):498–502
Seto CT, Jeraldo P, Orenstein R, Chia N, DiBaise JK (2014) Prolonged use of a proton pump inhibitor reduces microbial diversity: implications for Clostridium difficile susceptibility. Microbiome 2(1):1–11
Sharma M, Holmes HM, Mehta HB, Chen H, Aparasu RR, Shih YCT, Giordano SH, Johnson ML (2019) The concomitant use of tyrosine kinase inhibitors and proton pump inhibitors: prevalence, predictors, and impact on survival and discontinuation of therapy in older adults with cancer. Cancer 125(7):1155–1162
Spigel D, Vicente D, Ciuleanu T, Gettinger S, Peters S, Horn L, Audigier-Valette C, Aranda NP, Juan-Vidal O, Cheng Y (2021) Second-line nivolumab in relapsed small-cell lung cancer: CheckMate 331☆. Ann Oncol 32(5):631–641
Srinutta T, Chewcharat A, Takkavatakarn K, Praditpornsilpa K, Eiam-Ong S, Jaber BL, Susantitaphong P (2019) Proton pump inhibitors and hypomagnesemia: a meta-analysis of observational studies. Medicine 98(44):e17788
Trifan A, Stanciu C, Girleanu I, Stoica OC, Singeap AM, Maxim R, Chiriac SA, Ciobica A, Boiculese L (2017) Proton pump inhibitors therapy and risk of Clostridium difficile infection: Systematic review and meta-analysis. World J Gastroenterol 23(35):6500
Wang Y, Jenq RR, Wargo JA, Watowich SS (2023) Microbiome influencers of checkpoint blockade–associated toxicity. J Exp Med 220(3):e20220948
Xia B, Yang M, Nguyen LH, He Q, Zhen J, Yu Y, Di M, Qin X, Lu K, Kuo ZC (2021) Regular use of proton pump inhibitor and the risk of inflammatory bowel disease: pooled analysis of 3 prospective cohorts. Gastroenterology 161(6):1842–1852
Zedtwitz-Liebenstein K, Wenisch C, Patruta S, Parschalk B, Daxböck F, Graninger W (2002) Omeprazole treatment diminishes intra-and extracellular neutrophil reactive oxygen production and bactericidal activity. Crit Care Med 30(5):1118–1122
Zhang Y, Liang M, Sun C, Song EJ, Cheng C, Shi T, Min M, Sun Y (2020) Proton pump inhibitors use and dementia risk: a meta-analysis of cohort studies. Eur J Clin Pharmacol 76:139–147
Funding
None.
Author information
Authors and Affiliations
Contributions
Concept: MU, MZK and MA. Design: MU and MA. Supervision: all authors. Data collection and/or processing: all authors. Analysis and/or interpretation: MZK and MA. Literature search: MME, EH, TBG, MÇ, GD and DÇ. Writing: MU, SU, MZK, and MA. Critical reviews: all authors.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Ethical approval
Ethics approval was obtained from Necmettin Erbakan University Meram Medical Faculty, Konya, Turkey (Ethics Committee Number: 2022/4155).
Informed consent
Because the study was designed retrospectively, no informed consent was obtained from the patients.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Uğraklı, M., Koçak, M.Z., Dinç, G. et al. The effect of concomitant proton pump inhibitor use on survival outcomes of Nivolumab-treated renal cell carcinoma patients: a multicenter study. J Cancer Res Clin Oncol 149, 9183–9189 (2023). https://doi.org/10.1007/s00432-023-04844-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-023-04844-6