Abstract
There are no guidelines for the optimal manner and timing of permanent central catheter removal in the hemodynamically unstable pediatric hemato-oncology patient with suspected catheter-related bloodstream infections (CRBSI). Our goals were to examine current practices of permanent central catheter management and choice of removal in the hemodynamically unstable pediatric patient with suspected CRBSI among practitioners in diverse subspecialties. We performed a literature review on the subject, and conducted a multi-disciplinary survey included pediatric oncologists, pediatric emergency medicine physicians, and pediatric intensive care physicians whom we queried about their choice of permanent central catheter management and removal while treating the hemodynamically unstable pediatric patient with suspected CRBSI. Most of the 78 responders (n = 47, 59%) preferred to utilize the existing permanent central catheter for initial intravenous access rather than an alternative access. There were no significant differences between physician subspecialties (p = 0.29) or training levels (p = 0.14). Significantly more pediatric emergency medicine physicians preferred not to remove the permanent central catheter at any time point compared to the pediatric hemato-oncologists, who preferred to remove it at some point during the acute presentation (44.4% vs. 9.4%, respectively, p = 0.02).
Conclusion: Our study findings reflect the need for uniform guidelines on permanent central catheter use and indications for its removal in the hemodynamically unstable pediatric patient. We suggest that permanent central catheter removal should be urgently considered in a deteriorating patient who failed to be stabilized with medical treatment.
What is Known: • There are no guidelines for the optimal choice and timing of permanent central catheter removal in the hemodynamically unstable pediatric hemato-oncology patient with suspected catheter-related bloodstream infection (CRBSI). | |
What is New: • We found variations in practices among pediatricians from diverse subspecialties and conflicting data in the literature. • There is a need for prospective studies to provide uniform guidelines for optimal management of suspected CRBSI in the hemodynamically unstable pediatric patient. |
Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Avoid common mistakes on your manuscript.
Introduction
Catheter-related bloodstream infection (CRBSI) is a major cause of morbidity and mortality among pediatric hemato-oncology patients [1, 2], particularly during episodes of chemotherapy-induced neutropenia [3], but also among non-neutropenic non-hospitalized patients [4]. Previous studies in pediatric oncology patients reported on an overall CABSIs rate of 2.2–7.4 infections per 1000 central venous catheter (CVC) days [5, 6]. Adler et al. reported 35% prevalence of CRBSI among oncologic pediatric patients [5]. Young age, low body weight, and hematopoietic stem cell transplantation are all well-recognized risk factors for long-term catheter infections [5, 7,8,9,10,11]. According to the Infectious Disease Society of America (IDSA) guidelines, the indications for long-term catheter removal are well-defined when the diagnosis of CRBSI has been confirmed, mainly by microbiological recognition [12].
There are, however, no guidelines on catheter removal when CRSBI is suspected but has not yet been confirmed, posing a dilemma of whether or not the permanent catheter should be removed [7]. This dilemma is even more challenging in the context of unstable patients, who require hemodynamic monitoring, fluid intake, antibiotics, blood products, and vasopressors for their stabilization [7]. The decision whether to retain or to remove their permanent catheter depends upon the perceived balance between potential benefits and risks. While some observational studies found better clinical outcome with an early removal of the permanent catheter [7, 13], it should be borne in mind that its removal leads to the insertion of a new catheter, which may be difficult to perform and potentially cause mechanical, infectious, and anesthetic complications [7].
There are only sparse data on early versus late catheter removal in the setting of unconfirmed CRBSI [7, 13]. The current literature is mainly focused upon neonates, most of the studies are retrospective with conflicting conclusions, and there are no randomized control prospective trials addressing these issues [13,14,15,16,17,18].
Another question that needs to be considered is whether to use a permanent vascular access or an alternative access during acute hemodynamic deterioration. This point is also not well-established in the pediatric guidelines [19]. We aimed to examine the current practices of catheter management and choice of removal in the unstable pediatric patient with suspected but unconfirmed CRBSI among providers in various subspecialties, and to explore the optimal management in those patients.
Materials and methods
We conducted a survey among pediatric hemato-oncologists, pediatric emergency medicine (PEM) physicians, and pediatric intensive care unit (PICU) physicians, including both fellows and attending physicians, who are regularly involved in the acute care of pediatric hemato-oncology patients. The online anonymous survey was distributed via email invitations to members of the Israeli specialty-specific professional societies.
The survey was developed and reported according to guidelines published by the Academy group [20]. This custom-designed survey was based upon the approach taken by similar previously published ones [21]. The clinical scenario presented in the survey was developed by the current study authors who represent the three subspecialties of pediatric emergency medicine, pediatric hematology-oncology, and pediatric intensive care, and the complete questionnaire was evaluated for language clarity and technical feasibility. Physicians who were involved with its preliminary testing did not participate in its final application.
The survey included three parts (Appendix): part 1—a short clinical scenario detailing a brief patient history, history of present illness, vital signs, physical examination findings, and partial recent laboratory findings. Part 2—five questions concerning the preference of vascular access for the clinical scenario in part 1. Participants were asked to detail their choice of initial vascular access (question 1), choice of vascular access for specific interventions if alternatives were available (questions 2–4), and their consideration of urgent removal of an implanted central access device (question 5). Part 3—demographic and practice characteristics of the survey participant (gender, subspecialty, and years of clinical experience). Participants were able to provide free-text feedback and comments at the end of the survey.
Statistical analysis
Data entry and analysis were performed with SPSS Statistics, version 28 (SPSS Inc., Chicago, IL, USA). Categorical variables were described by number (proportion), and continuous variables by the mean and standard deviation (SD) or the median and interquartile range (IQR), as appropriate. The differences between continuous variables between two groups were assessed with Student’s t-test, or one way ANOVA between 3 groups, between categorical variables by the chi-square or Fisher’s exact test, and between medians by the Mann–Whitney U test for independent means. A two-tailed p < 0.05 value was considered statistically significant.
The study and the online survey were approved by the local institutional review board (0753–19-TLV).
Results
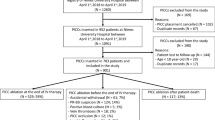
A total of 274 members of the national societies of pediatric hematology-oncology, PEM, and PICU physicians practicing in 20 children’s hospitals across Israel were invited to participate in this survey. Seventy-eight responded (28.4% response rate, 41 [50.6%] males) and completed all sections of the survey, and their data were used in the analysis. They included pediatric hemato-oncologists (n = 32, 39.5%), PICU physicians (n = 28, 34.6%), and PEM physicians (n = 18, 22.2%). Most of the participants identified themselves as attending physicians (n = 62, 76%) with a median of 7.5 years of experience (IQR 4.2–12.0 years), and 16 (19.8%) were fellows.
In the first question, the participants were asked to detail their choice of initial vascular access. Table 1 lists initial IV access preferences according to subspecialty. Most of the study participants (n = 47, 59%) indicated that they would prefer to utilize the existing permanent catheter as the initial choice of vascular access, with the second most common response being the use of a peripheral intravenous (IV) catheter (n = 24, 30%). Only a minority indicated that they would prefer a temporary placed central line, an intraosseous (IO) access device, or not having any preference (n = 3, 3.7%; n = 2, 2.5%, and n = 4, 5%, respectively). A univariate analysis showed no significant difference between physician subspecialty (p = 0.29), training level (p = 0.14), or gender (p = 0.52) in the choice of initial IV access. Table 1 lists initial IV access preferences according to subspecialty.
In the ensuing three questions, the participants were given a choice between the use of a newly established alternative vascular access (either IV or IO) or the existing permanent catheter for the delivery of specific treatments. Table 2 demonstrates the preference of treatment access in specific treatments. In question 2, the participants were asked to detail their choice of initial vascular access for IV fluid resuscitation. Most physicians opted to use an alternative vascular access (n = 51, 64%), and 18 (23%) preferred the use of the existing permanent catheter, while 11 (14%) indicated no preference. More PEM physicians preferred to use the permanent catheter compared to hemato-oncology and PICU physicians, who preferred to use an alternative access, however, these differences did not reach a level of significance (p = 0.08 and p = 0.059, respectively) (Table 2). In question 3, the participants were asked to detail their choice of initial vascular access for empiric antibiotic administration, most physicians elected to utilize the existing permanent catheter (n = 51, 65%), rather than an alternative vascular access (n = 24, 30%), and only a few indicated having no preference (n = 4, 5%). There was no significant difference between physician subspecialties (Table 2). In question 4, the participants were asked to detail their choice of initial vascular access for the administration of vasopressors. Most of the participants preferred using the existing permanent catheter for the administration of vasopressors (n = 40, 50%), followed by an alternative vascular access (n = 27, 34%), and no preference (n = 13, 16%). There was a significant difference between the three subspecialties in their preference of access when administering vasopressors (p = 0.006). A post hoc analysis revealed significantly higher rates of PICU physicians who preferred to use the permanent catheter compared to hemato-oncologists (67.9% vs. 28.1%, respectively, p < 0.001). No significant differences were found in vasopressor access preference between PICU and PEM physicians or between hemato-oncologist and PEM physicians (p = 0.08 and p = 0.1, respectively) (Table 2).
In the final question (question 5) in the clinical scenario, the study participants were asked to indicate the appropriate time to consider removal of the permanent catheter in the hemodynamically unstable pediatric patient. Their responses to the multiple-choice question are presented in Fig. 1: 48 physicians (60%) chose to remove the permanent catheter in case of failure to stabilize the patient with fluid resuscitation, antibiotics, or vasopressors/inotropes. Table 3 demonstrates a comparison of timing of permanent catheter removal according to subspecialty. Significantly more PEM physicians responded that they would not remove the permanent catheter at any time point during the acute situation compared to hemato-oncologists (44.4% vs. 9.4%, respectively, p = 0.02). Most of the hemato-oncologist physicians (29/32, 90.6%) responded that they would remove the permanent catheter at some point during the acute presentation. There were no significant differences in the choice of permanent catheter removal between the PEM and PICU physicians or between the PICU and hemato-oncology physicians (Table 3).
Discussion
When asked about treating an unstable hemato-oncology febrile pediatric patient with suspected but unconfirmed CRBSI, more than one-half of our study participants, regardless of their subspecialty, preferred to utilize the existing permanent central line catheter rather than switch to an alternative one. However, 60% of the participants responded that they would remove the permanent catheter if a change in the management, such as the addition of antibiotics, fluids, and/or vasopressors, failed to stabilize the patient.
The dilemma between using a functional and crucial permanent catheter access during resuscitation as opposed to not using it and even removing it in the unstable pediatric patient with suspected CRBSI is understandable and complex. While the IDSA position is very clear about short-term central line removal in the unstable patient and recommends its removal in any patient with an acute febrile episode who is seriously ill (hypotension/hypoperfusion/end-organ failure), there are no clear guidelines with respect to permanent catheter removal in the unstable pediatric patient with suspected but unconfirmed CRBSI [12]. The current indications for permanent catheter removal refer solely to the time point at which the diagnosis of CRBSI has been established: for example, permanent catheter removal is recommended in a patient with microbiologically confirmed CRBSI who presents with complications, such as severe sepsis [12]. It remains unclear, however, which is the preferred strategy for patients with suspected catheter-related infection that has not yet been confirmed by blood culture results.
In the absence of data from prospective trials, decisions regarding the timing of removal of central permanent catheters in infants with bloodstream infections continue to rely upon the findings of observational retrospective studies [13]. Studies that focused upon catheter removal among neonates showed better clinical outcome with early removal when there was evidence of microbiological clearance (bacteremia or fungemia) and less end-organ involvement and damage due to inflammatory cascades [17, 18, 22,23,24,25,26,27]. However, these findings may be due to confounding factors, such as clinicians electing to delay permanent catheter removal in smaller, less mature, or sicker infants, and they should be interpreted cautiously [13]. It should be borne in mind that neonatal patients and oncology patients are two different risk populations, treated with different CVCs (e.g., percutaneously inserted catheters versus surgically implanted catheters) and having different prevalence of pathogens [12, 13].
Previous study among pediatric oncology patients with CRBSI reported that the permanent catheter was removed in 46% of the cases. Hypotension, persistent bacteremia, previous stem cell transplantation, multiple CABSIs in the same CVC, exit-site infection, inappropriate empiric antibiotic therapy, and Candida infection were all significantly associated with increased risk of catheter removal due to antibiotic treatment failure [28].
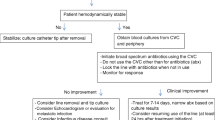
On the other hand, there is a true management challenge, which is well recognized by the IDSA guidelines, in the removal of surgically implantable intravascular devices in children. They suggest validating the presence of a true CRBSI before removal and even attempting CRBSI treatment without catheter removal [12]. Several studies have reported successful CRBSI management among children without catheter removal [29,30,31]. Viscoli et al. [32] reported rates of clinical and microbiologic resolution of 57% without removal of the catheter in children with confirmed CRBSI. However, together with the above permissive approach, the IDSA guidelines emphasize that children that are treated without catheter removal should be closely monitored, and that the device should be removed in the event of clinical deterioration or recurrence of CRBSI [12]. We therefore suggest that an urgent catheter removal should be strongly considered and not delayed in unstable patients who have already deteriorated clinically, especially if they had failed to be stabilized with initial medical treatment. However, a catheter removal should be performed only following the insertion of an alternative access.
Indications for the choice between the use of a permanent catheter versus an alternative vascular access during the acute presentation of an unstable patient with suspected CRBSI are not provided in the current IDSA guidelines [12]. Antibiotic lock therapy is generally recommended as a part of the approach for trying to save the permanent catheter in children with microbiologically confirmed CRBSI, with the recognition that dwell times may be variable, based upon limited venous access and the necessity to use the catheter [12]. However, in the unstable deteriorating child, together with measures taken to save the permanent catheter, it is reasonable to use an alternative access, when possible, rather than a permanent catheter that is assumed to be the source of infection and a possible cause of additional deterioration [18]. When there is no appropriate alternative vascular access, however, the treatment should be administrated through the permanent catheter, especially in an unstable deteriorating pediatric patient who urgently requires the administration of fluids and medications for recovery.
Our study has several limitations that bear mention. A sampling bias may have occurred with regard to the proportion of responders. Hemato-oncology, PICU, and PEM teams are heterogeneous and vary in number and experience from center to center. Despite our surveyal of all members of the Israeli national specialty-specific societies, the response rate was only 28.4%. As a result, the low statistical power has a reduced chance of detecting a true difference in practice.
The variations in practices among pediatricians from diverse subspecialties that were documented in this survey, together with conflicting data from the literature, reflect the ongoing uncertainty and the need for prospective studies and uniform guidelines for optimal management of suspected but unconfirmed CRBSI in the hemodynamically unstable pediatric patient. Future prospective trials may provide evidence-based recommendations and potentially improve clinical care and patient outcome.
Data Availability
All data and original material are available for transparency.
Abbreviations
- CRBSI:
-
Catheter-related bloodstream infections
- IDSA:
-
Infectious Disease Society of America
- IO:
-
Intraosseous
- IQR:
-
Interquartile range
- IV:
-
Intravenous
- PEM:
-
Pediatric emergency medicine
- PICU:
-
Pediatric intensive care unit
- SD:
-
Standard deviation
References
Jarvis WR, Edwards JR, Culver DH et al (1991) Nosocomial infection rates in adult and pediatric intensive care units in the United States. National Nosocomial Infections Surveillance System. Am J Med 91:185S-91S
Hord JD, Lawlor J, Werner E, Billett AL, Bundy DG, Winkle C et al (2016) Central line associated blood stream infections in pediatric hematology/oncology patients with different types of central lines. Pediatr Blood Cancer 63(9):1603–1607
Joo EJ, Kang CI, Ha YE, Park SY, Kang SJ, Joung MK et al (2011) Clinical outcome of catheter salvage in neutropenic cancer patients with catheter-related infection. Scand J Infect Dis
Garaventa A, Castagnola E, Dallorso S et al (1995) Sepsis in children with malignant neoplasia, equipped with a Broviac-type venous catheter. Pediatr Med Chir 17(2):147–150
Adler A, Yaniv I, Steinberg R, Solter E, Samra Z, Stein J, Levy I (2006) Infectious complications of implantable ports and Hickman catheters in paediatric hematology–oncology patients. J Hosp Infect 62:358–365
Celebi S, Sezgin ME, Cakır D et al (2013) Catheter-associated bloodstream infections in pediatric hematology-oncology patients. Pediatr Hematol Oncol 30(3):187–194. https://doi.org/10.3109/08880018.2013.772683
Janum S, Zingg W, Classen V, Afshari A (2013) Bench-to-bedside review: challenges of diagnosis, care and prevention of central catheter-related bloodstream infections in children. Crit Care 17(4):238. https://doi.org/10.1186/cc12730.PMID:24041298;PMCID:PMC4057411
Pinon M, Bezzio S, Tovo PA, Fagioli F, Farinasso L, Calabrese R, Marengo M, Giacchino M (2009) A prospective 7-year survey on central venous catheter-related complications at a single pediatric hospital. Eur J Pediatr 168:1505–1512
Ruebner R, Keren R, Coffi NS, Chu J, Horn D, Zaoutis TE (2006) Complications of central venous catheters used for the treatment of acute hematogenous osteomyelitis. Pediatrics 117:1210–1215
Barrett AM, Imeson J, Leese D, Philpott C, Shaw ND, Pizer BL, Windebank KP (2004) United Kingdom Children’s Cancer Study Group; Paediatric Oncology Nurses Forum of the Royal College of Nursing: Factors influencing early failure of central venous catheters in children with cancer. J Pediatr Surg 39:1520–1523
Garcia-Teresa MA, Casado-Flores J, Delgado Dominguez MA, Roqueta-Mas J, Cambra-Lasaosa F, Concha-Torre A, Fernandez-Perez C (2007) Spanish Central Venous Catheter Pediatric Study Group: Infectious complications of percutaneous central venous catheterization in pediatric patients: a Spanish multicenter study. Intensive Care Med 33:466–476
Mermel LA, Allon M, Bouza E, Craven DE, Flynn P, O'Grady NP, Warren DK (2009) Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 update by the Infectious Diseases Society of America. Clin Infect Dis 49(1):1–45. https://doi.org/10.1086/599376
Vasudevan C, McGuire W (2011) Early removal versus expectant management of central venous catheters in neonates with bloodstream infection. Cochrane Database Syst Rev 8:CD008436
Benjamin DK Jr, Miller W, Garges H, Benjamin DK, McKinney RE Jr, Cotton M, Fisher RG, Alexander KA (2001) Bacteremia, central catheters, and neonates: when to pull the line. Pediatrics 107:1272–1276
Karlowicz MG, Furigay PJ, Croitoru DP, Buescher ES (2002) Central venous catheter removal versus in situ treatment in neonates with coagulase-negative staphylococcal bacteremia. Pediatr Infect Dis J 21:22–27
Greenberg RG, Moran C, Ulshen M, Smith PB, Benjamin DK Jr, CohenWolkowiez M (2010) Outcomes of catheter-associated infections in pediatric patients with short bowel syndrome. J Pediatr Gastroenterol Nutr 50:460–462
Dato VM, Dajani AS (1990) Candidemia in children with central venous catheters: role of catheter removal and amphotericin B therapy. Pediatr Infect Dis J 9:309–314
Chapman RL, Faix RG (2003) Persistent bacteremia and outcome in late onset infection among infants in a neonatal intensive care unit. Ped Infect Dis J 22:17–21
Lehrnbecher T, Robinson P, Fisher B, Alexander S, Ammann RA, Beauchemin M, Carlesse F, Groll AH, Haeusler GM, Santolaya M, Steinbach WJ, Castagnola E, Davis BL, Dupuis LL, Gaur AH, Tissing WJE, Zaoutis T, Phillips R, Sung L (2017) Guideline for the management of fever and neutropenia in children with cancer and hematopoietic stem-cell transplantation recipients: 2017 update. J Clin Oncol 35(18):2082–2094. https://doi.org/10.1200/JCO.2016.71.7017. Epub 2017 May 1 PMID: 28459614
Burns KE, Duffett M, Kho ME, Meade MO, Adhikari NK, Sinuff T, Cook DJ (2008) ACCADEMY Group. A guide for the design and conduct of self-administered surveys of clinicians. CMAJ. 179(3):245–52. https://doi.org/10.1503/cmaj.080372. PMID: 18663204; PMCID: PMC2474876
Artino AR Jr, Durning SJ, Sklar DP (2018) Guidelines for reporting survey-based research submitted to academic medicine. Acad Med 93(3):337–340. https://doi.org/10.1097/ACM.0000000000002094
Eppes SC, Troutman JL, Gutman LT (1989) Outcome of treatment of candidemia in children whose central catheters were removed or retained. Pediatr Infect Dis J 8:99–104
Karlowicz MG, Hashimoto LN, Kelly RE Jr, Buescher ES (2000) Should central venous catheters be removed as soon as candidemia is detected in neonates? Pediatrics 106:E63
Benjamin DK Jr, Miller W, Garges H, Benjamin DK, McKinney RE Jr, Cotton M et al (2001) Bacteremia, central catheters, and neonates: when to pull the line. Pediatrics 107:1272–1276
O’Grady NP, Alexander M, Dellinger EP, Gerberding JL, Heard SO, Maki DG, Masur H, McCormick RD, Mermel LA, Pearson ML, Raad II, Randolph A, Weinstein RA (2002) Guidelines for the prevention of intravascular catheter related infections. The Hospital Infection Control Practices Advisory Committee, Center for Disease Control and Prevention, U.S. Pediatrics
Nazemi KJ, Buescher ES, Kelly RE Jr, Karlowicz MG (2003) Central venous catheter removal versus in situ treatment in neonates with enterobacteriaceae bacteremia. Pediatrics 111:e269–e274
Karlowicz MG, Furigay PJ, Croitoru DP, Buescher ES (2002) Central venous catheter removal versus in situ treatment in neonates with coagulase-negative staphylococcal bacteremia. Pediatr Infect Dis J 21:22–27
Adler A, Yaniv I, Solter E et al (2006) Catheter-associated bloodstream infections in pediatric hematology-oncology patients: factors associated with catheter removal and recurrence. J Pediatr Hematol Oncol 28(1):23–28
Hartman GE, Shochat SJ (1987) Management of septic complications associated with Silastic catheters in childhood malignancy. Pediatr Infect Dis J 6:1042–1047
Flynn PM, Shenep JL, Stokes DC, Barrett FF (1987) In situ management of confirmed central venous catheter-related bacteremia. Pediatr Infect Dis J 6:729–734
Wiener ES (1995) Catheter sepsis: the central venous line Achilles’ heel. Semin Pediatr Surg 4:207–214
Viscoli C, Garaventa A, Boni L et al (1988) Role of Broviac catheters in infections in children with cancer. Pediatr Infect Dis J 7(8):556–560
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation and data collection were performed by Tom Rosenberg, Neta Cohen, Ayelet Rimon, and Shirley Friedman. Data analysis was performed by Neta Cohen and Tom Rosenberg. The first draft of the manuscript was written by Neta Cohen and Tom Rosenberg and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study and the online survey were approved by the local institutional review board (0753–19-TLV). This article does not contain any studies with human participants or animals performed by any of the authors.
Consent to participate
Informed consent was obtained from all individuals participants who accepted the invitation to be included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Tobias Tenenbaum
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendix
Appendix
Case description:
A 6-year-old boy newly diagnosed with pre-B ALL is undergoing the first month of induction, with weekly chemotherapy in addition to systemic steroids (IV prednisolone 60 mg/m2) and omeprazole. Chemotherapy is given via a Port-A-Cath. He had been in good general condition when seen at the clinic during the preceding week. The CBC shows him to be neutropenic (WBC = 1000 cells/µL, absolute neutrophil count = 0 cells/µL). He is brought to the emergency department by his parents who report that he appeared to be weaker and more tired than usual over the past few hours. Inspection reveals a pale, weak-appearing child, who was only partially responsive to verbal stimulation.
Vital signs: temperature 39 °C (102.2 °F), heart rate 170 beats/min, blood pressure 65/40 mmHg, respiratory rate 40 breaths/min, and O2 saturation 93% in room air. Physical examination: extremities cool to touch, peripheral pulses weak, and capillary refill time significantly prolonged. Port site appearance is good, and there are no other abnormal findings.
Question #1:
Which venous access will you use for the initial management of this patient?
-
Insert a peripheral venous catheter
-
Use the existing central line (Port-A-Cath)
-
Insert an intraosseous catheter
-
Insert a temporary central line
-
No preference/no institutional guideline
Question #2:
Assuming an alternative venous access was established (peripheral or intraosseous) in addition to the pre-existing central line, which would you prefer to use for fluid resuscitation?
-
The alternative catheter—peripheral or intraosseous
-
The central line (Port-A-Cath)
-
No preference/no institutional guideline
Question #3:
Assuming an alternative venous access was established (peripheral or intraosseous) in addition to the pre-existing central line, which would you prefer to use for administering empiric antibiotics?
-
The alternative catheter—peripheral or intraosseous
-
The central line (Port-A-Cath)
-
No preference/no institutional guideline
Question #4:
Assuming an alternative venous access was established (peripheral or intraosseous) in addition to the pre-existing central line, which would you prefer to use for administering vasopressors/inotropes?
-
The alternative catheter—peripheral or intraosseous
-
The central line (Port-A-Cath)
-
No preference/no institutional guideline
Question #5:
In the patient who is hemodynamically unstable, at what point in this clinical scenario should an emergent removal of the pre-existing central line be considered?
-
Immediately at presentation of a hemodynamically unstable neutropenic patient, regardless of alternative vascular access
-
At the time of presentation of a hemodynamically unstable neutropenic patient, immediately after obtaining an alternative vascular access
-
After fluid resuscitation and empiric antibiotic treatment if they fail to stabilize the patient
-
After fluid resuscitation, empiric antibiotic treatment, and initiation of inotropes if they fail to stabilize the patient
-
There is no indication for emergent central line removal during this scenario
Demographics:
What is your subspecialty?
-
Pediatric Emergency Medicine
-
Pediatric Intensive Care
-
Pediatric Hematology-Oncology
What is your gender identification?
-
Male
-
Female
-
Prefer not to answer
Are you an attending or a fellow?
-
Attending
-
Fellow
Attending physician—how many years of experience do you have in your field?
Please add any additional comments or questions.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cohen, N., Rosenberg, T., Rimon, A. et al. Early removal of a permanent catheter during the acute management of the unstable pediatric hemato-oncology patient with suspected catheter-related bloodstream infection: a multi-disciplinary survey and review of the literature. Eur J Pediatr 182, 795–802 (2023). https://doi.org/10.1007/s00431-022-04747-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-022-04747-7