Abstract
The impact of tumor focality on prostate cancer (PCa) prognosis has been addressed in several studies with conflicting results. Tumor foci from multifocal (MF) PCa can show highly heterogeneous molecular features. Our aim was to analyze the protein expression of PTEN, SPOP, SLC45A3, ETV1, ERG and the “triple hit” (ERG overexpression, PTEN plus SLC45A3 loss) in unifocal (UF) and MF PCa, to evaluate their value as prognostic markers according to focality, and the role of tumor heterogeneity in MF disease. PTEN, SPOP, SLC45A3, ETV1 and ERG immunohistochemical expression was evaluated in 185 PCa from 9 TMAs, 51 UF and 134 MF. In a subset of 69 MF cases, the dominant and secondary foci (DF and SF) were compared. Heterogeneity was considered when both tumor foci presented different expression patterns. Relationship with clinicopathological features was also analyzed. MF PCa was diagnosed in significantly younger patients when compared to UF ones (p = 0.007). ETV1 overexpression was associated with UF disease (p = 0.028). A shorter time to PSA recurrence was related to SLC45A3 wt expression in UF PCa (p = 0.052), and to SPOP expression loss (p = 0.043) or “triple hit” phenotype in MF PCa (p = 0.041). In MF cases, PTEN loss, SLC45A3 loss and “triple hit” phenotype were associated with the DF and had significant heterogeneity. In conclusion, our results indicate that UF and MF PCa have relevant and consistent molecular differences. The analysis of an immunohistochemical panel, composed by PTEN, SPOP, SLC45A3, ETV1 and ERG, could be useful to predict outcome in MF cases.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Prostate Cancer (PCa) is a highly heterogeneous disease from the clinical, anatomical and molecular points of view [1, 2]. Its clinical course differs vastly and it often presents as a multifocal (MF) malignancy [3]. Some studies have reported multiple tumor foci in 60% to 90% of cases [1, 4,5,6,7,8,9,10]. Molecular features can be so variable among tumor areas in a given MF PCa as they can be among unifocal (UF) PCa from different patients [1, 5]. Several studies have shown that low- and high-grade tumor foci in MF PCa exhibit different molecular signatures, including tumor mutational burden, copy number alterations, gene expression profiles, weighted genome instability index, proteomics and clonal evolutionary processes [11,12,13,14,15]. Different theories have tried to explain the origin of heterogeneity in MF disease. In cases with multiclonal origin, each focus would evolve independently through the accumulation of different alterations. Alternatively, an initially uniclonal tumor could give rise to multiple tumor foci by intraglandular dissemination plus accumulation of alterations over time [6, 16, 17]. In fact, both models are possible, and they can even be found combined in some cases [16, 18].
The Cancer Genome Atlas reported that, based on molecular alterations, 74% of clinically-localized PCa can be classified in two major carcinogenic pathways: one related to ETS fusions and another related to somatic mutations [19]. In both pathways, frequent copy number alterations have been considered potential drivers of progression [20]. In previous studies, we analyzed the protein expression of relevant genes from the two major PCa pathways, mainly PTEN, SPOP, SLC45A3, ETV1 and ERG, as well as the “triple hit” combination (ERG overexpression, PTEN plus SLC45A3 loss), and we reported relevant associations between changes in these molecules and the clinicopathological features of the tumors [21,22,23].
In the present study, we aimed to understand the role of PTEN, SPOP, SLC45A3, ETV1 and ERG alterations in UF and MF PCa etiopathogenesis, to assess their value as prognostic markers, and the heterogeneity in the expression of these proteins in MF disease. With this purpose, we analyzed their immunohistochemical expression, individually or as components of the “triple hit” combination, in a well-defined series of UF and MF localized PCa.
Materials and methods
Patients and tumor samples
One hundred and eighty-five patients with PCa who underwent radical prostatectomy were retrospectively selected from the files of the Hospital del MAR Biobank (MARBiobanc, Barcelona, Spain). Grade Group (GG) or ISUP/WHO grade at diagnosis, age at diagnosis, pre-operative PSA levels, tumor stage, biochemical recurrence, perineural infiltration, extra-prostatic extension, seminal vesicle invasion, resection margin status, and tumor focality information was retrieved from the patients’ clinical history records, and it is shown in Table 1. Grade Group at diagnosis refers to the GG of the single tumor focus in UF cases, and the GG of the dominant focus in MF cases (the highest GG). Perineural infiltration was defined as the invasion of the extra-prostatic neurovascular bundles by the tumor. Tumor focality was classified as UF vs MF. Multifocality was considered when at least two tumor foci were identified in the prostatectomy specimens, without any overlap between them in the axial and sagittal planes of consecutive prostate sections.
Immunohistochemistry
We selected 185 cases with complete PTEN, SPOP, SLC45A3, ETV1 and ERG immunostaining from a previous series of 230 PCa. From them, 51 were UF and 134 MF. Formalin-fixed paraffin-embedded PCa samples were included in 9 tissue microarrays (TMAs) [21,22,23]. All the cases were re-reviewed by two expert pathologists to confirm the grade of the PCa foci. In the UF cases, the single tumor focus was included. In 65 of the MF cases, only the dominant focus (DF, the one with the highest GG) was included. From the 69 remaining MF cases, at least two tumor foci were included in the TMAs. In the latter, two tumor foci have been considered, both the DF plus a secondary focus (SF, a focus with a lower GG than the DF).
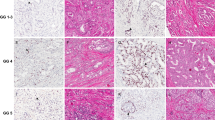
As it has been previously described, PTEN, SPOP and SLC45A3 nuclear and cytoplasmic loss were assessed using a semi-quantitative scoring system considering two categories: wt or loss of expression. Adjacent normal tissue staining was used as an internal reference for PTEN and SLC45A3, and smooth muscle staining for SPOP [21, 23]. For ERG, wt or nuclear overexpression were considered [21]. Finally, ETV1 cytoplasmic expression was graded quantitatively by a histoscore system ([1 × (%1 + cells)] + [2 × (%2 + cells)] + [3 × (%3 + cells)]), and subsequently wt (0–99) and overexpression (≥ 100) categories were established [22]. For both ETV1 and ERG, endothelial cells were used as a positive internal control. Examples of altered and wt immunostainings are shown in Supplementary Fig. 1.
Statistical analysis
Categorical variables were presented as frequencies and percentages, and quantitative variables as average and ranges. Pearson Chi-Square, Fisher’s Exact or Wilcoxon Mann Whitney tests were used. Nominal p-values < 0.05 were considered statistically significant (not corrected for multiple testing). The McNemar test was used for the heterogeneity analysis, in which a p-value < 0.05 indicated significant heterogeneity between foci.
The relationship with time to PSA recurrence was analyzed by Cox proportional hazards regression and visualized using Kaplan–Meier curves. Log-Rank test was applied to compare the survival probability between groups in 179 patients (6 cases in this series were lost for follow-up). Multivariate Cox’s proportional hazards models were used to estimate adjusted hazard ratios (HR). Patients were followed at regular intervals of 3 months for one year and every 6 months for the subsequent years, and a PSA test was performed before every follow-up visit. None of the patients received pre- or post-operative radiation, nor adjuvant hormone therapy. Recurrence was defined as an increase in serum PSA > 0.2 ng/ml at the time of the last clinical follow-up appointment (i.e., two consecutive increases). Patients’ follow-up ranged from 5 to 274 months, with an average value of 92.8 months and a median of 96 months. In the PSA recurrence analysis, a p-value < 0.05 was considered statistically significant. Statistical analyses were performed using R programming language version 4.3.0 (R Foundation, Vienna, Austria).
Results
Clinicopathological characteristics of unifocal and multifocal prostate cancer
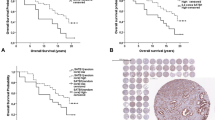
Tumors were classified as UF or MF, and no association was detected between most of the clinicopathological characteristics and tumor focality (Table 2a). However, MF cases were diagnosed in younger patients, as the average age at diagnosis was 64.2 years for men with MF and 66.9 years for men with UF tumors (p = 0.002). In addition, a different distribution of cases according to the PCa GG was observed in UF vs MF disease, especially in GG2 (29.4% vs 40.3%) and GG3 (23.5% vs 6.7%) groups (p = 0.025). Survival analysis was performed to compare the time to PSA recurrence in UF vs MF PCa, but no differences were detected (HR 1.24, p = 0.499) (Fig. 1a).
Immunohistochemical expression in unifocal and multifocal prostate cancer
Protein expression of PTEN, SPOP, SLC45A3, ETV1, ERG, and the so called “triple hit” [21, 24], were evaluated. Expression was considered as altered when loss or overexpression was present in the single focus from UF cases or at least in one of the tumor foci from MF ones. Expression alterations in UF and MF PCa were compared, and the results are shown in Table 2b. UF and MF tumors showed similar percentages of SPOP and SLC45A3 expression loss, ERG overexpression and “triple hit” phenotype. PTEN loss of expression was more frequent in MF cases, but without statistical significance. Interestingly, ETV1 overexpression was associated with UF disease (p = 0.028).
Immunohistochemical expression and relationship with clinicopathological features according to focality
The relationship between immunohistochemical expression in UF and MF PCa and different clinicopathological features, such as age at diagnosis, GG at diagnosis, tumor stage, perineural infiltration, extra-prostatic extension, seminal vesicle invasion, and resection margin status was analyzed. In UF PCa, ETV1 overexpression was related to pT3 tumor stage (p = 0.031), and ERG overexpression was associated with perineural infiltration (p = 0.044), the latter also showing a trend to be related to younger patients (p = 0.058) (Supplementary Table 1).
In MF PCa, several clinicopathological features were strongly related to different protein expression alterations (Table 3). Thus, patients with MF PCa overexpressing ERG were younger (p = 0.069). Low GG was associated with wt expression of SPOP, SLC45A3 and ETV1 (p = 0.022; p = 0.025 and p = 0.010), as well as with the lack of the “triple hit” phenotype (p = 0.002), and showed a trend to be associated with PTEN wt (P = 0.059). Tumor stage pT3 was statistically related to PTEN and SPOP loss of expression (p = 0.012 and p = 0.011), as well as to ERG overexpression and the “triple hit” phenotype (p = 0.051 and p < 0.001). Perineural infiltration was also associated with PTEN loss (p = 0.002), ERG overexpression (p = 0.047) and the “triple hit” phenotype (p < 0.001). Finally, SPOP loss (P = 0.060) and the “triple hit” phenotype (p = 0.021) were detected in a high proportion of MF PCa with seminal vesicle invasion when compared to MF cases without this feature.
The impact of these alterations on PSA recurrence according to PCa focality was analyzed. In the subgroup of UF tumors, only SLC45A3 wt expression showed an association with a shorter time to PSA recurrence (HR 0.13, p = 0.052) (Fig. 1b). In the subgroup of MF tumors, SPOP loss (HR 2.26, p = 0.043) (Fig. 1c) and the “triple hit” phenotype (HR 2.78, p = 0.041) (Fig. 1d) also showed an association with a shorter time to PSA recurrence. Moreover, a multivariate COX proportional hazard regression analysis was performed to elucidate if the association between expression alterations and a shorter time to PSA recurrence in UF or MF PCa was maintained when other clinicopathological characteristics were considered (Supplementary Table 2). The results indicated that the correlation between PSA recurrence and SLC45A3 wt in UF cases was close to be preserved (HR 7.83, p = 0.067). By contrast, the relationship between PSA recurrence and SPOP loss (HR 1.55, p = 0.344) or the “triple hit” phenotype (HR 1.34, p = 0.647) in MF tumors was lost.
Immunohistochemical expression in tumor foci with different aggressiveness
In a subgroup of 69 MF PCa, the immunohistochemical expression was analyzed in both the dominant focus (DF, with the highest GG) and a secondary focus (SF, a second focus with lower GG) (Fig. 2a). Considering PTEN status in the overall number of foci (69 DF and 69 SF), expression loss was statistically associated with the DF, as it was detected in 43.4% of DF but in only 26.1% of SF (p = 0.032). There was a statistical association between SLC45A3 expression loss and the DF, as it was found in 36.2% of DF but in only 7.2% of SF (p < 0.001). Regarding SPOP, expression loss was more frequently detected in DF (43.5%) rather than in SF (31.9%), but there were no significant differences. For ETV1 and ERG, both foci showed similar percentages of overexpression. Finally, the “triple hit” phenotype was detected in 13% of the DF, but in none of the SF (p = 0.003).
Tumor foci heterogeneity in multifocal prostate cancer
The heterogeneity between paired DF and SF from the same 69 MF PCa was assessed (Fig. 2b). Homogeneity or heterogeneity were established when both foci showed concordant or discordant immunostaining patterns, respectively. Comparing the paired foci one by one, PTEN expression showed significant heterogeneity (p = 0.019), and the DF were more frequently altered than the SF (24.6% vs 7.3%). In 18.8% of cases both foci displayed homogeneous PTEN loss. SPOP homogeneous loss was detected in 17.4% of cases, while loss exclusively in the DF or in the SF was found in 26.1% and in 14.5%, respectively (p = 0.186). Data for SLC45A3 indicated high heterogeneity (p < 0.001), as homogeneous loss in both foci was very uncommon (4.3%). Among the cases with heterogeneous loss, most of the alterations were found in the DF (31.9%), and only 2.9% exhibited loss exclusively in the SF. ETV1 overexpression was homogeneously found in 10.1% of cases, while exclusively detected in the DF from 18.8%, and in the SF from 11.6% cases (p = 0.383). On the contrary, ERG expression showed the highest homogeneity (p = 1), given that 37.7% of cases overexpressed ERG in both foci, and also 37.7% were absolutely wt. No focus predominated among the discordant cases (11.6% vs 13%). Finally, all cases with the “triple hit” phenotype showed a heterogeneous pattern, as it was exclusively detected in their DF (p = 0.007).
Discussion
The finding of multiple tumor foci in different anatomical zones of the prostate is particularly frequent [1, 5,6,7,8, 10], but multifocality is not an exclusive feature of PCa [25,26,27]. The impact of tumor multifocality in PCa prognosis has been addressed in several studies with conflicting results [5, 16]. Some authors have reported that MF and UF prostatic carcinomas may be biologically different, with the former being more aggressive, less differentiated and showing higher stage and shorter time to PSA recurrence [28], and even that some of the secondary foci in a patient with MF disease may have clinical significance [4, 6,7,8, 29, 30]. Nevertheless, in another study patients with UF tumors showed higher rates of positive surgical margins, high Gleason score and shorter time to PSA recurrence [31]. Other studies have failed to demonstrate any relationship between tumor focality and PCa clinicopathological features [7, 32, 33].
Our data from a series of PCa distributed in UF and MF cases, suggested a lack of association between tumor focality and most of the clinicopathological characteristics assessed, including tumor stage, PSA recurrence, perineural infiltration, extra-prostatic extension, seminal vesicle invasion, and resection margin status. Our findings clearly indicated that patients with MF PCa are diagnosed earlier than the ones with UF disease. Despite observing a significantly different case distribution in UF and MF PCa according to the GG, there was no consistent trend towards more aggressiveness for neither of them.
Numerous studies have described alterations in genes belonging to the two major carcinogenic pathways for PCa [19, 34,35,36], such as PTEN, SPOP, SLC45A3, ETV1 and ERG. In this regard, our group reported significant associations between alterations in the expression of these proteins and prognosis [21,22,23,24, 37]. Nevertheless, the role of alterations in these genes on tumor focality and inter-foci heterogeneity in MF disease is still an area deserving further research. In the present study, immunostaining for PTEN, SPOP, SLC45A3, ETV1 and ERG was evaluated in a large and well characterized cohort of UF and MF PCa. Interestingly, only ETV1 overexpression was associated with PCa focality, as it was more frequently altered in UF cases. This is in agreement with our previous research in a different cohort of PCa already pointing at this association [22]. Despite PTEN loss was found at a higher incidence in MF cases, the association was not statistically significant. UF and MF tumors showed similar SPOP, SLC45A3, ERG and “triple hit” expression patterns.
Regarding clinicopathological characteristics and tumor focality, ERG overexpression showed a trend to be associated with a younger age in both MF and UF cases. This finding is in concordance with other studies suggesting a relationship between ERG overexpression and young-age patients, but they did not take into account PCa focality [38,39,40]. Lack of alterations in SLC45A3, SPOP, ETV1 or in the “triple hit”, were related to low GG tumors exclusively in MF cases. In line with these results, alterations in the expression of PTEN, SPOP, ERG or the “triple hit” phenotype were associated with adverse clinicopathological features in MF PCa, including high tumor stage, perineural infiltration, seminal vesicle invasion or PSA recurrence. By contrast, in UF PCa, only the overexpression of ETV1 and ERG were related to high tumor stage and perineural infiltration, respectively. It is worth noting that SLC45A3 wt was associated with PSA recurrence. Therefore, alternative molecular driver alterations may be characterizing this subset of SLC45A3 wt UF PCa.
In a subgroup of MF PCa, immunohistochemical expression was analyzed in both the dominant and the secondary foci. Taking into account the results from the overall number of foci, PTEN and SLC45A3 expression loss, as well as the “triple hit” phenotype, were statistically associated with the DF. Previous studies already suggested that low- and high-grade PCa foci may exhibit distinct expression signatures, with an enrichment of alterations in high GG foci [13, 15]. The results from the McNemar test, comparing the paired foci from MF cases, confirmed that PTEN and SLC45A3 immunostaining patterns, as well as the “triple hit” phenotype, were highly heterogeneous in our series. On the contrary, ERG showed highly concordant immunohistochemical expression in both foci. Altogether, these data agree with previous studies reporting that ERG overexpression is less heterogeneous in MF disease, while PTEN loss consistently exhibits variable expression patterns [41, 42]. In this regard, TMPRSS2-ERG fusion has been considered as an initial event in PCa [24, 37, 43,44,45], and this could be the reason for the high homogeneity in ERG immunostaining. Conversely, PTEN loss and SLC45A3-ERG fusion have been defined as more advanced and secondary events that take place after TMPRSS2-ERG fusion [21, 24, 41, 46,47,48]. Our finding of a heterogeneous expression pattern for PTEN and SLC45A3 would support this hypothesis.
In conclusion, our data support the hypothesis that UF and MF PCa may be different molecular entities. The study of an immunohistochemical panel, composed by PTEN, SPOP, SLC45A3, ETV1 and ERG, could provide prognostic information about the outcome of MF cases. Our findings will require prospective validation in a larger cohort of patients with MF PCa, and more research is needed to identify molecular alterations with prognostic value in the UF subgroup.
Data availability
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Tolkach Y, Kristiansen G (2018) The heterogeneity of prostate cancer: A practical approach. Pathobiology 85:108–116. https://doi.org/10.1159/000477852
Haffner MC, Zwart W, Roudier MP, True LD, Nelson WG, Epstein JI, Marzo AMD, Nelson PS, Yegnasubramanian S (2021) Genomic and phenotypic heterogeneity in prostate cancer. Nat Rev Urol 18:79–92. https://doi.org/10.1038/s41585-020-00400-w
Sathianathen NJ, Konety BR, Crook J, Saad F, Lawrentschuk N (2018) Landmarks in prostate cancer. Nat Rev Urol 15:627–642. https://doi.org/10.1038/s41585-018-0060-7
Huang CC, Deng FM, Kong MX, Ren Q, Melamed J, Zhou M (2014) Re-evaluating the concept of ‘dominant/index tumor nodule’ in multifocal prostate cancer. Virchows Arch 464:589–594. https://doi.org/10.1007/s00428-014-1557-y
Ibeawuchi C, Schmidt H, Voss R, Titze U, Abbas M, Neumann J, Eltze E, Hoogland AM, Jenster G, Brandt B, Semjonow A (2013) Genome-wide investigation of multifocal and unifocal prostate cancer-are they genetically different? Int J Mol Sci 14:11816–11829. https://doi.org/10.3390/ijms140611816
Karavitakis M, Ahmed HU, Abel PD, Hazell S, Winkler MH (2011) Tumor focality in prostate cancer: Implications for focal therapy. Nat Rev Clin Oncol 8:48–55. https://doi.org/10.1038/nrclinonc.2010.190
Stamatiou KN, Dilernia GC, Ilias GK, Daskalopoulos GK, Koutelekos IK, Marianou SN, Sofras FA (2009) The phenomenon of multifocality does not affect the biologic behavior of histologic prostate carcinoma. Med Sci Monit 15:61–63
Choi YH, Yu JW, Jeong BC, Seo SI, Jeon SS, Lee HM, Jeon HG (2019) Histological characteristics of the largest and secondary tumors in radical prostatectomy specimens and implications for focal therapy. Diagn Pathol 14:1–6. https://doi.org/10.1186/s13000-019-0782-8
Zhang W, Wang T, Wang Y, Zhu F, Shi H, Zhang J, Wang Z, Qu M, Zhang H, Wang T, Qian Y, Yang J, Gao X, Li J (2022) Intratumor heterogeneity and clonal evolution revealed in castration-resistant prostate cancer by longitudinal genomic analysis. Transl Oncol 16:101311. https://doi.org/10.1016/j.tranon.2021.101311
Xu J, Isaacs WB, Mamawala M et al (2021) Association of prostate cancer polygenic risk score with number and laterality of tumor cores in active surveillance patients. Prostate 81:703–709. https://doi.org/10.1002/pros.24140
Wu B, Lu X, Shen H, Yuan X, Wang X, Yin N, Sun L, Shen P, Hu C, Jian H, Wang D (2020) Intratumoral heterogeneity and genetic characteristics of prostate cancer. Int J Cancer 146:3369–3378. https://doi.org/10.1002/ijc.32961
De Vargas RL, Jacobs A, Rueschoff JH et al (2022) Single-cell proteomics defines the cellular heterogeneity of localized prostate cancer. Cell Reports Med 3:100604. https://doi.org/10.1016/j.xcrm.2022.100604
Løvf M, Zhao S, Axcrona U et al (2019) Multifocal Primary Prostate Cancer Exhibits High Degree of Genomic Heterogeneity. Eur Urol 75:498–505. https://doi.org/10.1016/j.eururo.2018.08.009
Boutros PC, Fraser M, Harding NJ et al (2015) Spatial genomic heterogeneity within localized, multifocal prostate cancer. Nat Genet 47:736–745. https://doi.org/10.1038/ng.3315
Salami SS, Hovelson DH, Kaplan JB et al (2018) Transcriptomic heterogeneity in multifocal prostate cancer. JCI insight 3:e123468. https://doi.org/10.1172/jci.insight.123468
Andreoiu M, Cheng L (2010) Multifocal prostate cancer: biologic, prognostic, and therapeutic implications. Hum Pathol 41:781–793. https://doi.org/10.1016/j.humpath.2010.02.011
Vendramin R, Litchfield K, Swanton C (2021) Cancer evolution: Darwin and beyond. EMBO J 40:e108389. https://doi.org/10.15252/embj.2021108389
Erickson A, Hayes A, Rajakumar T, Verrill C, Bryant RJ, Hamdy FC, Wedge DC, Woodcock DJ, Mills IG, Lamb AD (2021) A Systematic Review of Prostate Cancer Heterogeneity: Understanding the Clonal Ancestry of Multifocal Disease. Eur Urol Oncol 4:358–369. https://doi.org/10.1016/j.euo.2021.02.008
Abeshouse A, Ahn J, Akbani R et al (2015) The Molecular Taxonomy of Primary Prostate Cancer. Cell 163:1011–1025. https://doi.org/10.1016/j.cell.2015.10.025
Wang X, Grasso CS, Jordahl KM et al (2020) Copy number alterations are associated with metastatic-lethal progression in prostate cancer. Prostate Cancer Prostatic Dis 23:494–506. https://doi.org/10.1038/s41391-020-0212-8
Hernández-Llodrà S, Juanpere N, de Muga S et al (2017) ERG overexpression plus SLC45A3 (prostein) and PTEN expression loss : Strong association of the triple hit phenotype with an aggressive pathway of prostate cancer progression. Oncotarget 8:74106–74118. https://doi.org/10.18632/oncotarget.18266
Segalés L, Juanpere N, Lorenzo M, Albero-González R, Fumadó L, Cecchini L, Bellmunt J, Lloreta-Trull J, Hernández-Llodrà S (2019) Strong cytoplasmic ETV1 expression has a negative impact on prostate cancer outcome. Virchows Arch 475:457–466. https://doi.org/10.1007/s00428-019-02573-1
Hernández-Llodrà S, Segalés L, Juanpere N, Lorenzo M, Salido M, Nonell L, López D, Rodríguez-Vida A, Bellmunt J, Fumadó L, Cecchini L (2021) Lloreta-Trull J (2021) SPOP and CHD1 alterations in prostate cancer: Relationship with PTEN loss, tumor grade, perineural infiltration, and PSA recurrence. Prostate 81:1267–1277. https://doi.org/10.1002/pros.24218
Hernández S, Font-Tello A, Juanpere N, de Muga S, Lorenzo M, Salido M, Fumadó L, Serrano L, Cecchini L, Serrano S, Lloreta J (2016) Concurrent TMPRSS2-ERG and SLC45A3-ERG rearrangements plus PTEN loss are not found in low grade prostate cancer and define an aggressive tumor subset. Prostate 76:854–865. https://doi.org/10.1002/pros.23176
Salgado R, Aftimos P, Sotiriou C, Desmedt C (2015) Evolving paradigms in multifocal breast cancer. Semin Cancer Biol 31:111–118. https://doi.org/10.1016/j.semcancer.2014.07.002
Simmer F, van der Linden RLA, Ligtenberg MJL et al (2021) Multifocal Colorectal Cancer—Do Intraluminal Metastases Occur? Gastroenterology 160:1853–1855. https://doi.org/10.1053/j.gastro.2020.12.010
Leventakos K, Peikert T, Midthun DE et al (2017) Management of Multifocal Lung Cancer: Results of a Survey. J Thorac Oncol 12:1398–1402. https://doi.org/10.1016/j.jtho.2017.05.013
Karavitakis M, Ahmed HU, Abel PD, Hazell S, Winkler MH (2012) Anatomically versus biologically unifocal prostate cancer: A pathological evaluation in the context of focal therapy. Ther Adv Urol 4:155–160. https://doi.org/10.1177/1756287212447092
Tourinho-Barbosa R, Srougi V, Nunes-Silva I, Baghdadi M, Rembeyo G, Eiffel SS, Barret E, Rozet F, Galiano M, Cathelineau X, Sanchez-Salas R (2018) Biochemical recurrence after radical prostatectomy : what does it mean? Int Braz J urol 44:14–21. https://doi.org/10.1590/S1677-5538.IBJU.2016.0656
Kneppers J, Krijgsman O, Melis M, de Jong J, Peeper DS, Bekers E, van der Poel HG, Zwart W, Bergman AM (2019) Frequent clonal relations between metastases and non-index prostate cancer lesions. JCI Insight 4:e124756. https://doi.org/10.1172/jci.insight.124756
Rice KR, Furusato B, Chen Y, McLeod DG, Sesterhenn IA, Brassell SA (2009) Clinicopathological Behavior of Single Focus Prostate Adenocarcinoma. J Urol 182:2689–2694. https://doi.org/10.1016/j.juro.2009.08.055
Masterson TA, Cheng L, Mehan RM, Koch MO (2011) Tumor focality does not predict biochemical recurrence after radical prostatectomy in men with clinically localized prostate cancer. J Urol 186:506–510. https://doi.org/10.1016/j.juro.2011.03.106
Iremashvili V, Pelaez L, Manoharan M, Acosta K, Rosenberg DL, Soloway MS (2012) Tumor focality is not associated with biochemical outcome after radical prostatectomy. Prostate 72:762–768. https://doi.org/10.1002/pros.21481
Arora K, Barbieri C (2018) Molecular Subtypes of Prostate Cancer. Curr Oncol Rep 20:55–58. https://doi.org/10.1007/s11912-018-0707-9
Light A, Ahmed A, Dasgupta P, Elhage O (2020) The genetic landscapes of urological cancers and their clinical implications in the era of high-throughput genome analysis. BJU Int 126:26–54. https://doi.org/10.1111/bju.15084
Compérat E, Wasinger G, Oswald A, Kain R, Cancel-Tassin G, Cussenot O (2020) The genetic complexity of prostate cancer. Genes (Basel) 11:1396. https://doi.org/10.3390/genes11121396
Font-Tello A, Juanpere N, de Muga S, Lorenzo M, Lorente JA, Fumadó L, Serrano L, Serrano S, Lloreta J, Hernández S (2015) Association of ERG and TMPRSS2-ERG with grade, stage, and prognosis of prostate cancer is dependent on their expression levels. Prostate 75:1216–1226. https://doi.org/10.1002/pros.23004
Schaefer G, Mosquera JM, Ramoner R, Park K, Romanel A, Steiner E, Horninger W, Bektic J, Ladurner-Rennau M, Rubin MA, Demichelis F, Klocker H (2013) Distinct ERG rearrangement prevalence in prostate cancer: Higher frequency in young age and in low PSA prostate cancer. Prostate Cancer Prostatic Dis 16:132–138. https://doi.org/10.1038/pcan.2013.4
Song C, Chen H (2018) Predictive significance of TMRPSS2-ERG fusion in prostate cancer: A meta-analysis. Cancer Cell Int 18:1–12. https://doi.org/10.1186/s12935-018-0672-2
Lu Z, Williamson SR, Carskadon S, Arachchige PD, Dhamdhere G, Schultz DS, Stricher H, Peabody JO, Jeong W, Chitale DA, Bismar TA, Rogers CG, Menon M, Gupta NS, Palanisamy N (2020) Clonal evaluation of early onset prostate cancer by expression profiling of ERG, SPINK1, ETV1, and ETV4 on whole-mount radical prostatectomy tissue. Prostate 80:38–50. https://doi.org/10.1002/pros.23914
Yoshimoto M, Ding K, Sweet JM, Ludkovski O, Trottier G, Song KS, Joshua AM, Fleshner NE, Squire JA, Evans AJ (2013) PTEN losses exhibit heterogeneity in multifocal prostatic adenocarcinoma and are associated with higher Gleason grade. Mod Pathol 26:435–447. https://doi.org/10.1038/modpathol.2012.162
Yun JW, Lee S, Ryu D, Park S, Park WY, Joung JG, Jeong J (2019) Biomarkers Associated with Tumor Heterogeneity in Prostate Cancer. Transl Oncol 12:43–48. https://doi.org/10.1016/j.tranon.2018.09.003
Clark JP, Cooper CS (2009) ETS gene fusions and prostate cancer. Nat Rev Urol 6:429–439. https://doi.org/10.1038/nrurol.2009.127
Perner S, Mosquera JM, Demichelis F, Hofer MD, Paris PL, Simko J, Collins C, Bismar TA, Chinnaiyan AM, de Marzo AM, Rubin MA (2007) TMPRSS2-ERG Fusion Prostate Cancer: An Early Molecular Event Associated With Invasion. Am J Surg Pathol 31:882–888. https://doi.org/10.1097/01.pas.0000213424.38503.aa
Carver BS, Tran J, Gopalan A, Chen Z, Shaikh S, Carracedo A, Alimonti A, Nardella C, Varmeh S, Scardino PT, Cordon-Cardo C, Gerald W, Pandolfi PP (2009) Aberrant ERG expression cooperates with loss of PTEN to promote cancer progression in the prostate. Nat Genet 41:619–624. https://doi.org/10.1038/ng.370
Liu D, Augello MA, Grbesa I, Prandi D, Liu Y, Shoag JE, Karnes RJ, Trock BJ, Klein EA, Den RB, Demichelis F, Davicioni E, Sboner A, Barbieri CE (2021) Tumor subtype defines distinct pathways of molecular and clinical progression in primary prostate cancer. J Clin Invest 131:e147878. https://doi.org/10.1172/JCI147878
Liu R, Zhou J, Xia S, Li T (2019) The impact of PTEN deletion and ERG rearrangement on recurrence after treatment for prostate cancer: a systematic review and meta-analysis. Clin Transl Oncol 22:694–702. https://doi.org/10.1007/s12094-019-02170-3
Jamaspishvili T, Berman DM, Ross AE, Scher HI, de Marzo AM, Squire JA, Lotan TL (2018) Clinical implications of PTEN loss in prostate cancer. Nat Rev Urol 15:222–234. https://doi.org/10.1038/nrurol.2018.9
Acknowledgements
The authors are grateful to Jorge Domínguez from the MARGenomics’ Bioinformatics Unit, to Xavier Duran from the Consulting Service on Methodology for Biomedical Research, and to the Hospital del MAR Biobank (MARBiobanc) (Hospital del Mar Research Institute, Barcelona, Spain).
Funding
Open Access funding provided thanks to the CRUE-CSIC agreement with Springer Nature. The study has been funded with the grant FIS/Instituto Carlos III/FEDER PI15/00452 from the Spanish Ministry of Health.
Author information
Authors and Affiliations
Contributions
SHLl and JLl designed and supervised the research study. NJ, LF, LC, JB and ARV managed patients and acquired samples and data. SHLl and LS organized data and constructed databases. ML and DL constructed TMAs and performed immunohistochemical staining. NJ, JLl, SHLl, LS and NG evaluated immunohistochemical staining. JPB, SHLl and LS performed the analysis and interpretation of data. LS and SHLl wrote the manuscript. All the authors reviewed the manuscript.
Corresponding author
Ethics declarations
Ethics approval
CEIC Parc de Salut Mar approval status, project number 2015/3211/I.
Conflict of interest disclosure
The authors declare no conflicts of interest related to the present study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Segalés, L., Juanpere, N., Gallarín, N. et al. Immunohistochemical markers as predictors of prognosis in multifocal prostate cancer. Virchows Arch 485, 281–290 (2024). https://doi.org/10.1007/s00428-023-03699-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-023-03699-z