Abstract
Immunotherapeutic strategies are increasingly used in the treatment of a number of malignancies including high-grade urothelial carcinoma (HGUC) of the bladder. Because of this, detailed and accurate assessment of the tumor immune microenvironment is paramount. In this study, we aimed to correlate the composition of the tumor immune microenvironment with oncologic outcome and the expression of two cancer testis antigens (CTAs), CT10 and PRAME, potential cancer vaccine targets, as well as major histocompatibility complex I (MHC I), a molecule associated with tumor immune escape and resistance to immunotherapy. Triplicate tissue microarrays (TMAs) were constructed using 207 cases of HGUC of the bladder. Oncologic outcome data was gathered for each case. Consecutive sections from the TMA blocks were stained with CD3, CD4, CD8, FOXP3, PD1, PD-L1, CT10, PRAME, and MHC I. Twenty-one percent and 15% of cases expressed CT10 and PRAME, respectively. Eighty-eight percent of cases showed absent or decreased MHC I expression. CT10-expressing tumors showed a significantly worse disease specific survival (p = 0.007, hazard ratio 2.245, confidence interval 1.223–4.122). CT10, PRAME, and MHC I expression significantly correlated with other some immune parameters. CT10 and PRAME are expressed in a subset of HGUC and CTA and MHC I expression correlate with a number of important immune parameters. Together, these findings highlight the potential for exploring novel immune therapeutic strategies in HGUC. Additional studies evaluating the clinical relevance of these findings are warranted.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
High-grade urothelial carcinoma (HGUC) of the bladder (Fig. 1) is a common and aggressive tumor type which is increasingly being studied with regard to its tumor immune microenvironment. Our group and others have previously shown that the composition and degree of inflammation surrounding and within a tumoral focus of HGUC may provide information about the tumor’s underlying biology [1] and likely prognosis [2,3,4,5,6].
In addition to therapy targeting programmed cell death 1 (PD-1)/programmed cell death ligand 1 (PD-L1) which is being used in the treatment of a number of malignancies including HGUC [7,8,9,10,11,12], cancer vaccines are another immunotherapy treatment strategy which is being studied in a number of tumor types. Cancer vaccines function to activate a patient’s immune system in order to mount an immune response specifically targeting cancer cells [13, 14] and importantly, some cancer vaccines have shown promising benefit in recent clinical trials [15]. One specific target for cancer vaccines is the cancer testis antigens (CTAs), a group of molecules found to be expressed in different cancer types and in normal tissues restricted to immune privileged site such as testicular germ cells [16]. Interestingly, in addition to potentially serving as targets for cancer vaccines, CTAs may play a role in modulating tumoral gene expression [17] and in supporting tumor cell growth and survival [18]. The expression of a number of CTAs in HGUC has been studied by some groups [19,20,21] including Sharma et al. who showed that 77% of the studied cases expressed at least one of nine CTAs while 61% of cases expressed two or more CTAs [22]. Preferentially Expressed Antigen in Melanoma (PRAME) is another CTA which has been shown to be highly upregulated in a number of different neoplasms [17]; interestingly, it has been suggested that the PRAME may antagonize retinoic acid receptor signaling, thereby conferring proliferative advantages to tumor cells [23].
Integral to the proper functioning of the normal immune system is Major Histocompatibility Complex I (MHC I), a cell surface protein complex present on all nucleated cells. MHC I normally functions to present “non-self” cytosolic peptide antigens to CD8+ T cells, eventually leading to propagation of the cytotoxic pathway leading to cell death of malignant or virally infected cells. Because of its importance in the establishment of a cytotoxic response, lack of MHC I expression on tumor cells has not surprisingly been proposed as an important mechanism in tumor immune escape and resistance to immunotherapy [15, 24,25,26].
The aim of this work was to expand our understanding of the tumor immune contexture in HGUC of the bladder using an immunohistochemical approach. In light of the ongoing developments in the study of MHC I, CTAs, and cancer vaccines, we chose to assess the association of CTAs with immune markers and impact on oncologic outcomes.
Materials and methods
This project was approved by the Research Ethics Board at Sunnybrook Health Sciences Centre in Toronto, Ontario, Canada (REB 187-2016).
Case selection and review
All cases were identified through a retrospective search of the laboratory information system, Sunquest CoPath (n = 235), as previously described [27]. Briefly, the search criteria included cases of HGUC of the bladder treated by cystectomy between 1999 and 2015. Exclusion criteria included cases that were non-invasive, non-urothelial histology, presence of a neuroendocrine carcinoma component, and divergent differentiation (squamous, glandular, sarcomatoid) encompassing > 50% of the tumor. Original hematoxylin and eosin (H&E) slides for each case were retrieved from our departmental archive and reviewed by a pathologist with subspecialty training in genitourinary pathology (MRD) who confirmed the tumor histology and grade.
The following demographic and clinicopathologic data was recorded for each case: age at diagnosis, patient sex, smoking status, tumor size, tumor focality, presence/absence of carcinoma in situ, presence/absence of lymphovascular invasion, margin status, presence/absence of lymph node metastases, history/type of neoadjuvant therapy (if applicable), pT stage, AJCC stage, date of last known follow-up, date of disease relapse (if applicable), and date of death (if applicable). Evidence of disease relapse was based on operative and/or radiologic findings. Only deaths that occurred within the hospital or under the palliative care service were accessible.
Tissue microarray construction
Triplicate 1-mm core tissue microarrays (TMAs) were constructed from 207 of the available cases; 28 cases were excluded from the TMAs due to lack of tissue or when a suitable tumor block for punching was not available. Technical details regarding construction of the TMAs have been previously described [27]. Four-micrometer thick unstained sections were prepared from the TMA blocks and were stained with CD4, CD8, FOXP3, PD-1, and PD-L1 (SP263) in a sequential fashion; CT10, PRAME, and MHC I staining was completed on a different date on unstained sections from the same TMA blocks.
Immunohistochemistry and scoring
Immunohistochemical expression was assessed in immune cells (ICs) and/or tumor cells (TCs), depending on the marker being evaluated. The identification of TCs and ICs was based on morphologic features alone with assistance of H&E slides. All scoring was done by a single reviewer who is subspecialist genitourinary pathologist (MRD).
CD4, CD8, FOXP3, and PD-1 expression was assessed in ICs (CD4, CD8, PD-1—membranous, FOXP3—nuclear). For each marker, the absolute number of positive cells was evaluated in one hot spot/core (i.e., 1 representative 40× field/core), with the results averaged across all cores per case. PD-1-positive cases were dichotomized into high and low groups based on the median value of positive cells/40× field. PD-L1 expression (SP263 clone, Ventana Medical Systems, AZ, USA) was assessed in ICs (cytoplasmic or membranous staining of any intensity) and TCs (partial or complete membranous staining). The Ventana Benchmark Ultra automated staining platform was utilized according to the manufacturer’s protocol with the Optiview DAB IHC detection kit. TC and IC staining was assessed as present or absent with the estimated percentage of positive TCs and positive ICs recorded for each TMA core. The results were then averaged across the triplicate cores to give one estimate of percentage TCs and percentage ICs for each case. The degree of PD-L1 staining was considered positive when > 25% of TCs or ICs showed expression. MHC I expression (A4 clone, ThermoFisher Scientific, Waltham, MA, USA) was assessed in TCs; the percentage of TCs with cytoplasmic staining was noted and the cases were categorized as follows: positive (> 75% of TCs), decreased expression (25–74% of TCs), or negative (< 25% of TCs). CT10 (CT10#5 clone, provided by the Ludwig Institute for Cancer Research, New York, NY, USA) [28] was assessed in TCs and any immunopositivity was considered positive. The location of staining (cytoplasmic and/or nuclear) and the percentage of CT10-positive TCs were recorded. Like CT10, PRAME (EPR20330 clone, Abcam, Cambridge, MA, USA) immunopositivity was defined as any expression within TCs. The location of staining (cytoplasmic and/or nuclear) and the percentage of PRAME-positive TCs were also recorded.
Statistical analysis
All statistical analyses were performed using the SPSS software 24.0 (IBM Corporation, New York, NY, USA). The prognostic significance of PRAME, CT10, and MHC I was assessed using log rank test with Kaplan-Meier analysis for disease-specific survival (DSS) and progression-free survival (PFS). Hazard ratio and 95% confidence interval were calculated using Cox proportional regression model. The Pearson correlation coefficient was computed among the expression of each antibody. p values less than 0.05 were considered to be statistically significant.
Results
Demographic and clinicopathologic characterization
The mean and median age at diagnosis was 70.3 and 71.0 years of age, respectively (range 33–93 years). Males (n = 150, 73%) were more commonly affected compared with females (n = 57, 27%). Fifty-nine (29%) of the patients had no smoking history, while 38 (18%) and 82 (40%) patients respectively were current or former smokers, respectively. Information regarding smoking status was not available for 28 (13%) patients.
A total of 131 (63%) tumors were more than 3 cm in size while 70 (34%) were less than 3 cm in size; 6 (3%) tumors were not measured. A total of 193 (93%) tumors were unifocal while the remaining were multi-focal (14, 7%). Ninety-one (44%) cases demonstrated the presence of carcinoma in situ while the remaining 116 (56%) did not. One hundred thirty-nine (67%) cases demonstrated the presence of lymphovascular invasion, 4 (2%) cases were indeterminate, and the remaining 64 (31%) did not demonstrate the presence of any lymphovascular invasion. Eighty-three (40%) cases showed lymph node metastases while 115 (56%) did not; the remaining 9 (4%) cases lacked any lymph nodes. Soft tissue margins were positive in 52 (25%) cases. A total of 7 (3%), 30 (15%), 111 (54%), and 59 (28%) cases were staged as pT1-pT4 respectively. Seven (3%), 22 (11%), 95 (46%), and 83 (40%) were staged as AJCC stages 1–4, respectively.
Immune microenvironment characterization
CD4, CD8, FOXP3, PD-1, and PD-L1 expression has been previously characterized in this cohort [1].
CTA and MHC I characterization
CTA and MHC I expression shown in Table 1 and Fig. 2. A total of 201 cases were evaluable for CT10 and PRAME expressions: 42/201 (21%) and 31/201 (15%) of cases expression CT10 and PRAME, respectively. Both CT10 and PRAME expressions were predominantly nuclear in location. There were no cases where the individual scores for triplicate cores were discordant (i.e., no cases where one core was scored as positive and the other ones as negative, or vice versa). Of note, CT10 and PRAME were co-expressed in 9/201 (5%) of cases.
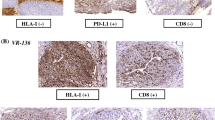
Cancer testis antigen and major histocompatibility complex I expression in high-grade urothelial carcinoma. Expression of CT10 (a) and PRAME (b) in high-grade urothelial carcinoma; note the nuclear expression in tumor cells and lack of expression in background stromal/inflammatory cells. Retained/normal MHC I expression (c), compared with complete lack of MHC I expression (d); note the retained expression in background stromal/inflammatory cells. Scale bar = 300 μm
A total of 202 cases were evaluable for MHC I expression: 25/202 (12%) cases demonstrated normal/retained expression, 18/202 (9%) cases demonstrated decreased/low expression, and the remaining 159/202 (79%) cases showed absent expression. There were no cases where the individual scores for triplicate cores were discordant.
Correlation of immune microenvironment components with each other, neoadjuvant therapy status, and oncologic outcome
The Pearson correlation coefficients between the assessed immune parameters are shown in Table 2. Significant correlations (p < 0.05) were noted for many immune parameter pairs including PRAME and FOXP3, PRAME and CT10, CT10 and PD-L1 in TCs and FOXP3, MHC I and PD-L1 in TCs, PD-L1 in ICs, PD-1, FOXP3, CD4, and CD8, PD-L1 in TCs and PD-L1 in ICs, PD-1, FOXP3, and CD8, PD-L1 in ICs and PD-1, FOXP3, CD4, and CD8, PD-1 and FOXP3, CD4, and CD8, FOXP3 and CD4 and CD8, and finally, CD4 and CD8. Figure 3 shows a graphical representation of some of the most significant correlations among the assessed immune parameters.
A total of 73 patients received some form of neoadjuvant therapy prior to cystectomy (31 with intravesical Bacillus Calmette-Guerin (BCG), 46 with chemotherapy/radiation; 4 patients received both BCG and chemotherapy/radiation). Of the 31 cases which were positive for PRAME, 15 (48%) were pretreated. Of the PRAME-negative cases, 58 (34%) were pretreated (p = 0.156, Fischer’s exact test). Of the 42 cases which were positive for CT10, 15 (36%) were pretreated. Of the CT10-negative cases, 58 (36%) were pretreated (p = 1.000, Fisher’s exact test). Three of the pretreated cases (2 BCG, 1 chemotherapy/radiation) were positive for both PRAME and CT10. None of the cases which had been pretreated with both BCG and chemotherapy/radiation expressed was positive for either PRAME or CT10.
With regard to oncologic outcome, follow-up data was available for 131 cases (median 4 months, range 1–206 months). Tumor recurrence was identified in 76/131 (58%) cases. 21/76 (28%) patients had metastatic disease. 24/131 (19%) patients were in palliative care at the time of last known follow-up. 4/24 patients in palliative care did not suffer from recurrence. 45/131 (34%) patients had died, 2 of which whose deaths were attributed to causes other than HGUC (myeloma/end stage renal disease and lymphoma, respectively).
Figure 4 shows the Kaplan-Meier survival curves (disease-specific survival and progression-free survival) comparing CT10-positive vs. CT10-negative tumors. With regard to DSS, CT10-positive tumors did significantly worse compared with CT10-negative tumors (log rank test p = 0.007, hazard ratio 2.245, 95% confidence interval 1.223–4.122). PFS did not differ significantly between CT10-positive and -negative tumors (log rank test p = 0.252). PRAME and MHC I expression did not significantly affect DSS or PFS (log rank test p > 0.050).
Discussion
The tumor immune microenvironment is dynamic and complex and its evaluation has become increasingly important in a number of malignancies including HGUC of the bladder. Clinical trial data has shown checkpoint inhibition to be effective in the treatment of these variably aggressive malignancies [7,8,9,10] and because of this, there is increasing excitement regarding the possible use of other types of immunotherapies, including cancer vaccines, in the treatment of these and other tumors [11, 29,30,31]. CTAs are potential targets of cancer vaccines given that their expression is normally restricted to immune privileged sites with aberrant expression in cancer cells [17]. In this study, we identified CT10 and PRAME as cancer-associated antigens expressed in a subset of HGUCs. CT10 expression has been previously reported in bladder cancer [22]; however, to the best of our knowledge, PRAME expression has not been previously studied in bladder cancer. Our assessment identified co-expression of CT10 and PRAME in a small proportion (5%) of our muscle-invasive HGUC cohort; the relevance and potential of this combinatory expression are as yet unclear. In a study which evaluated cases from a mixed stage cohort of HGUC, co-expression of CTAs was identified in up to 61% of cases [22]. The discrepancy in the number of co-expressed CTAs is likely explained by the fact that our cohort was evaluated with only two CTAs compared with Sharma et al.’s study which evaluated the expression of nine different CTAs. In addition, we utilized TMAs compared with whole tissue sections and our cohort is composed entirely of muscle invasive tumors whereas tumors ranging from pTa to pT4 were included in the aforementioned study.
Previous studies suggest that CTA expression by malignant cells is a poor prognostic factor, particularly in lung cancer [32,33,34,35,36]; however, there are some discrepant reports in the literature [37,38,39,40]. As we have shown, CT10 expression was associated with significantly worse DSS in HGUC of the bladder. This is in contrast to the previously mentioned study by Sharma et al. (n = 94) which showed that patients with CT10-positive tumors had improved disease free and overall survival compared with patients with CT10-negative tumors [22]. As indicated above, the likely explanation for these differences is the difference in cohort composition, in addition to our cohort containing a much larger number of cases; however, additional studies are needed to further evaluate this relationship.
Importantly, a number of clinical trials are studying cancer vaccines in human malignancies and several have shown promising results in the treatment of bladder cancer [20, 41,42,43]. To the best of our knowledge, the relationship and correlation we have identified between CT10 and PD-L1 in TCs, although weak in this study, are novel. Of course, the clinical and therapeutic significance of this relationship will need to be further explored in future studies.
Of note, we did not identify any heterogeneity among CTA-expressing tumors in that all cases which were found to be positive for CT10 and/or PRAME expressed the antigen relatively equally throughout all evaluated TMA cores (both high and low expressing tumors). In addition, we did not observe any background expression in normal tissue elements (i.e., stromal cells, inflammatory cells). These findings are important, as they indicate that CTA assessment in small samples (i.e., biopsies) is reliable and a viable tool for evaluating CTA expression.
It is well known that antigen presentation by the MHC I-tumor antigen complex is a necessary step in the initiation of anti-tumor CD8+ T cell response [44]. Loss of MHC I expression has been recognized as a mechanism of tumor immune escape and there has been some discussion in the literature regarding possible MHC I recovery approaches, in order to improve tumor response to immunotherapeutic treatments [26, 45]. In our study, we show that MHC I expression correlates with many other cellular components of the immune microenvironment including CD8+ T cells. These relationships are intriguing and deserve additional study to determine their biological significance. Similar to PRAME, MHC I expression levels were not found to be significantly associated with worse DSS or PFS; however, the differences may be significant in a cohort with more MHC-retained cases.
In conclusion, we have identified a number of interesting associations with regard to our characterization of the expanded tumor immune microenvironment in HGUC. Our understanding regarding the dynamics of the tumor immune microenvironment is evolving, and there is still much to be understood regarding the significance of correlations between different immune parameters. Further exploration of different components of the tumor immune microenvironment in a prospective multi-institutional cohort is warranted.
References
Hodgson A, Liu SK, Vesprini D, Xu B, Downes MR (2018) Basal-subtype bladder tumours show a “hot” immunophenotype. Histopathology 73:748–757. https://doi.org/10.1111/his.13696
Hodgson A, Xu B, Satkunasivam R, Downes MR (2018) Tumour front inflammation and necrosis are independent prognostic predictors in high-grade urothelial carcinoma of the bladder. J Clin Pathol 71:154–160. https://doi.org/10.1136/jclinpath-2017-204562
Liu X, Dowell AC, Patel P, Viney RP, Foster MC, Porfiri E, James ND, Bryan RT (2014) Cytokines as effectors and predictors of responses in the treatment of bladder cancer by bacillus Calmette-Guérin. Future Oncol 10:1443–1456. https://doi.org/10.2217/fon.14.79
Zuiverloon TCM, Nieuweboer AJM, Vékony H, Kirkels WJ, Bangma CH, Zwarthoff EC (2012) Markers predicting response to bacillus Calmette-Guérin immunotherapy in high-risk bladder cancer patients: a systematic review. Eur Urol 61:128–145. https://doi.org/10.1016/j.eururo.2011.09.026
Horn T, Laus J, Seitz AK, Maurer T, Schmid SC, Wolf P, Haller B, Winkler M, Retz M, Nawroth R, Gschwend JE, Kübler HR, Slotta-Huspenina J (2016) The prognostic effect of tumour-infiltrating lymphocytic subpopulations in bladder cancer. World J Urol 34:181–187. https://doi.org/10.1007/s00345-015-1615-3
Winerdal ME, Marits P, Winerdal M, Hasan M, Rosenblatt R, Tolf A, Selling K, Sherif A, Winqvist O (2011) FOXP3 and survival in urinary bladder cancer. BJU Int 108:1672–1678. https://doi.org/10.1111/j.1464-410X.2010.10020.x
Powles T, Eder JP, Fine GD, Braiteh FS, Loriot Y, Cruz C, Bellmunt J, Burris HA, Petrylak DP, Teng SL, Shen X, Boyd Z, Hegde PS, Chen DS, Vogelzang NJ (2014) MPDL3280A (anti-PD-L1) treatment leads to clinical activity in metastatic bladder cancer. Nature 515:558–562. https://doi.org/10.1038/nature13904
Rosenberg JE, Hoffman-Censits J, Powles T, van der Heijden MS, Balar AV, Necchi A, Dawson N, O'Donnell PH, Balmanoukian A, Loriot Y, Srinivas S, Retz MM, Grivas P, Joseph RW, Galsky MD, Fleming MT, Petrylak DP, Perez-Gracia JL, Burris HA, Castellano D, Canil C, Bellmunt J, Bajorin D, Nickles D, Bourgon R, Frampton GM, Cui N, Mariathasan S, Abidoye O, Fine GD, Dreicer R (2016) Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet 387:1909–1920. https://doi.org/10.1016/S0140-6736(16)00561-4
Sharma P, Retz M, Siefker-Radtke A, Baron A, Necchi A, Bedke J, Plimack ER, Vaena D, Grimm MO, Bracarda S, Arranz JÁ, Pal S, Ohyama C, Saci A, Qu X, Lambert A, Krishnan S, Azrilevich A, Galsky MD (2017) Nivolumab in metastatic urothelial carcinoma after platinum therapy (CheckMate 275): a multicentre, single-arm, phase 2 trial. Lancet 18:312–322. https://doi.org/10.1016/S1470-2045(17)30065-7
Powles T, O’Donnell PH, Massard C et al (2017) Efficacy and safety of durvalumab in locally advanced or metastatic urothelial carcinoma: updated results from a phase 1/2 open-label study. JAMA Oncol 3:e172411. https://doi.org/10.1001/jamaoncol.2017.2411
Donin NM, Lenis AT, Holden S, Drakaki A, Pantuck A, Belldegrun A, Chamie K (2017) Immunotherapy for the treatment of urothelial carcinoma. J Urol 197:14–22. https://doi.org/10.1016/j.juro.2016.02.3005
Muthigi A, George AK, Brancato SJ, Agarwal PK (2016) Novel immunotherapeutic approaches to the treatment of urothelial carcinoma. Ther Adv Urol 8:203–214. https://doi.org/10.1177/1756287216628784
Guo C, Manjili MH, Subjeck JR et al (2013) Therapeutic cancer vaccines: past, present, and future. Adv Cancer Res 119:421–475. https://doi.org/10.1016/B978-0-12-407190-2.00007-1
Wong KK, Li WA, Mooney DJ, Dranoff G (2016) Advances in therapeutic cancer vaccines. Adv Immunol 130:191–249. https://doi.org/10.1016/bs.ai.2015.12.001
Melero I, Gaudernack G, Gerritsen W, Huber C, Parmiani G, Scholl S, Thatcher N, Wagstaff J, Zielinski C, Faulkner I, Mellstedt H (2014) Therapeutic vaccines for cancer: an overview of clinical trials. Nat Rev Clin Oncol 11:509–524. https://doi.org/10.1038/nrclinonc.2014.111
Boon T, Coulie PG, Van den Eynde B (1997) Tumor antigens recognized by T cells. Immunol Today 18:267–268. https://doi.org/10.1016/S0167-5699(97)80020-5
Whitehurst AW (2014) Cause and consequence of cancer/testis antigen activation in cancer. Annu Rev Pharmacol Toxicol 54:251–272. https://doi.org/10.1146/annurev-pharmtox-011112-140326
Gjerstorff MF, Andersen MH, Ditzel HJ (2015) Oncogenic cancer/testis antigens: prime candidates for immunotherapy. Oncotarget 6:15772–15787. https://doi.org/10.18632/oncotarget.4694
Yin B, Liu G, Wang X-S, Zhang H, Song YS, Wu B (2012) Expression profile of cancer-testis genes in transitional cell carcinoma of the bladder. Urol Oncol 30:886–892. https://doi.org/10.1016/j.urolonc.2010.08.017
Sharma P, Gnjatic S, Jungbluth AA et al (2003) Frequency of NY-ESO-1 and LAGE-1 expression in bladder cancer and evidence of a new NY-ESO-1 T-cell epitope in a patient with bladder cancer. Cancer Immun 3:19
Lausenmeyer EM, Braun K, Breyer J, Gierth M, Denzinger S, Burger M, Voelker HU, Otto W (2018) Strong expression of cancertestis antigens CTAG1B and MAGEA3 is correlated with unfavourable histopathological features and MAGEA3 is associated with worse progression-free survival in urothelial bladder cancer. Urol Int 102:1–6. https://doi.org/10.1159/000493577
Sharma P, Shen Y, Wen S, Bajorin DF, Reuter VE, Old LJ, Jungbluth AA (2006) Cancer-testis antigens: expression and correlation with survival in human urothelial carcinoma. Clin Cancer Res 12:5442–5447. https://doi.org/10.1158/1078-0432.CCR-06-0527
Epping MT, Wang L, Edel MJ, Carlée L, Hernandez M, Bernards R (2005) The human tumor antigen PRAME is a dominant repressor of retinoic acid receptor signaling. Cell 122:835–847. https://doi.org/10.1016/j.cell.2005.07.003
van der Burg SH, Arens R, Ossendorp F, van Hall T, Melief CJM (2016) Vaccines for established cancer: overcoming the challenges posed by immune evasion. Nat Rev Cancer 16:219–233. https://doi.org/10.1038/nrc.2016.16
Tagliamonte M, Petrizzo A, Tornesello ML, Buonaguro FM, Buonaguro L (2014) Antigen-specific vaccines for cancer treatment. Hum Vaccines Immunother 10:3332–3346. https://doi.org/10.4161/21645515.2014.973317
Garrido F, Aptsiauri N, Doorduijn EM, Garcia Lora AM, van Hall T (2016) The urgent need to recover MHC class I in cancers for effective immunotherapy. Curr Opin Immunol 39:44–51. https://doi.org/10.1016/j.coi.2015.12.007
Hodgson A, Xu B, Downes MR (2017) p53 immunohistochemistry in high-grade urothelial carcinoma of the bladder is prognostically significant. Histopathology 71:296–304. https://doi.org/10.1111/his.13225
Zhuang R, Zhu Y, Fang L et al (2006) Generation of monoclonal antibodies to cancer/testis (CT) antigen CT10/MAGE-C2. Cancer Immun 6:7
Chevalier MF, Nardelli-Haefliger D, Domingos-Pereira S, Jichlinski P, Derré L (2014) Immunotherapeutic strategies for bladder cancer. Hum Vaccines Immunother 10:977–981. https://doi.org/10.4161/hv.27621
Gupta M, Kates M, Bivalacqua TJ (2019) Immunotherapy in nonmuscle invasive bladder cancer: current and emerging treatments. Curr Opin Oncol 31:183–187. https://doi.org/10.1097/CCO.0000000000000533
Dutcher GMA, Bilen MA (2018) Therapeutic vaccines for genitourinary malignancies. Vaccines 6:E55. https://doi.org/10.3390/vaccines6030055
Jin S, Cao S, Li J, Meng Q, Wang C, Yao L, Lang Y, Cao J, Shen J, Pan B, Hu J, Yu Y (2018) Cancer/testis antigens (CTAs) expression in resected lung cancer. OncoTargets Ther 11:4491–4499. https://doi.org/10.2147/OTT.S159491
Futawatari N, Fukuyama T, Yamamura R, Shida A, Takahashi Y, Nishi Y, Ichiki Y, Kobayashi N, Yamazaki H, Watanabe M (2017) Early gastric cancer frequently has high expression of KK-LC-1, a cancer-testis antigen. World J Gastroenterol 23:8200–8206. https://doi.org/10.3748/wjg.v23.i46.8200
Grah JJ, Katalinic D, Juretic A, Santek F, Samarzija M (2014) Clinical significance of immunohistochemical expression of cancer/testis tumor-associated antigens (MAGE-A1, MAGE-A3/4, NY-ESO-1) in patients with non-small cell lung cancer. Tumori 100:60–68. https://doi.org/10.1700/1430.15817
Yoshida N, Abe H, Ohkuri T et al (2006) Expression of the MAGE-A4 and NY-ESO-1 cancer-testis antigens and T cell infiltration in non-small cell lung carcinoma and their prognostic significance. Int J Oncol 28:1089–1098
Matković B, Juretić A, Spagnoli GC, Šeparović V, Gamulin M, Šeparović R, Šarić N, Bašić-Koretić M, Novosel I, Krušlin B (2011) Expression of MAGE-A and NY-ESO-1 cancer/testis antigens in medullary breast cancer: retrospective immunohistochemical study. Croat Med J 52:171–177
Chitale DA, Jungbluth AA, Marshall DS, Leitao MM, Hedvat CV, Kolb D, Spagnoli GC, Iversen K, Soslow RA (2005) Expression of cancer-testis antigens in endometrial carcinomas using a tissue microarray. Mod Pathol Off J U S Can Acad Pathol Inc 18:119–126. https://doi.org/10.1038/modpathol.3800232
Prasad ML, Jungbluth AA, Patel SG, Iversen K, Hoshaw-Woodard S, Busam KJ (2004) Expression and significance of cancer testis antigens in primary mucosal melanoma of the head and neck. Head Neck 26:1053–1057. https://doi.org/10.1002/hed.20112
Bandić D, Juretić A, Sarcević B et al (2006) Expression and possible prognostic role of MAGE-A4, NY-ESO-1, and HER-2 antigens in women with relapsing invasive ductal breast cancer: retrospective immunohistochemical study. Croat Med J 47:32–41
Zhang Y, Zhang Y, Zhang L (2019) Expression of cancer-testis antigens in esophageal cancer and their progress in immunotherapy. J Cancer Res Clin Oncol 145:281–291. https://doi.org/10.1007/s00432-019-02840-3
Nishiyama T, Tachibana M, Horiguchi Y et al (2001) Immunotherapy of bladder cancer using autologous dendritic cells pulsed with human lymphocyte antigen-A24-specific MAGE-3 peptide. Clin Cancer Res 7:23–31
Obara W, Ohsawa R, Kanehira M, Takata R, Tsunoda T, Yoshida K, Takeda K, Katagiri T, Nakamura Y, Fujioka T (2012) Cancer peptide vaccine therapy developed from oncoantigens identified through genome-wide expression profile analysis for bladder cancer. Jpn J Clin Oncol 42:591–600. https://doi.org/10.1093/jjco/hys069
Colombel M, Heidenreich A, Martínez-Piñeiro L, Babjuk M, Korneyev I, Surcel C, Yakovlev P, Colombo R, Radziszewski P, Witjes F, Schipper R, Mulders P, Witjes WPJ (2014) Perioperative chemotherapy in muscle-invasive bladder cancer: overview and the unmet clinical need for alternative adjuvant therapy as studied in the MAGNOLIA trial. Eur Urol 65:509–511. https://doi.org/10.1016/j.eururo.2013.10.056
Sabbatino F, Schwab JH, Ferrone S, Ferrone CR (2013) Evolution of studies of HLA class I antigen processing machinery (APM) components in malignant cells. Clin Transpl 453–463
Garrido F, Ruiz-Cabello F, Aptsiauri N (2017) Rejection versus escape: the tumor MHC dilemma. Cancer Immunol Immunother 66:259–271. https://doi.org/10.1007/s00262-016-1947-x
Acknowledgments
CT10, PRAME, and MHC I immunohistochemical staining was performed in the Department of Pathology, Memorial Sloan Kettering Cancer Center in New York, NY, USA. The SP263 antibody was purchased using a Sunnybrook Health Sciences Centre departmental Educational Grant courtesy of Astra Zeneca.
Author information
Authors and Affiliations
Contributions
Drs. Downes and Xu conceived and designed the study. Drs. Hodgson, Xu, and Downes gathered the data. Drs. Hodgson, Katabi, Jungbluth, Xu, and Downes reviewed and interpreted the data. Dr. Xu performed the statistical analysis. Drs. Hodgson, Katabi, Jungbluth, Xu, and Downes contributed to drafting and review of the manuscript as well as approval of the final version. Dr. Downes takes full responsibility for the work as a whole, including the study design, access to data, and the decision to submit and publish the manuscript.
Corresponding author
Ethics declarations
This project was approved by the Research Ethics Board at Sunnybrook Health Sciences Centre in Toronto, Ontario, Canada (REB 187-2016).
Conflict of interest
Dr. Downes has received compensation from Hoffman – La Roche and Astra Zeneca for participating on advisory boards and honoraria from Astra Zeneca.
Dr. Xu has received honoraria from Merck.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hodgson, A., Jungbluth, A.A., Katabi, N. et al. Evaluation of cancer testis antigen (CT10, PRAME) and MHC I expression in high-grade urothelial carcinoma of the bladder. Virchows Arch 476, 535–542 (2020). https://doi.org/10.1007/s00428-019-02661-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-019-02661-2